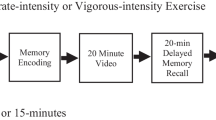
Abstract
The purpose of this meta-analysis was to evaluate the effects of acute exercise on short-term memory improvement. The computerized literature searches using electronic databases and examinations of reference lists from relevant studies yielded six studies meeting our inclusionary criteria. In a total of six studies, 16 standardized regression coefficient effect sizes (ESs) were calculated to be meta-analyzed. The meta-analyses showed a statistically significant increase in short-term memory improvement across both the exercise and non-exercise control groups in trials 1–5 [ES = 0.96, 95% CI (0.95, 0.97), P < 0.001]. However, there was no significant subgroup difference between exercise and control groups (Qb = 0.40, df = 1, P = 0.53). Our meta-analytic review provides suggestive evidence that an acute bout of exercise prior to learning does not result in short-term memory improvement to a greater extent than a non-exercise control. Additional research is needed to further evaluate whether acute exercise enhances long-term memory via enhanced learning and/or post-learning mechanisms.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The retrospective recall of past information involves several critical phases, namely acquisition (encoding and consolidation), storage, and retrieval. Encoding involves the development of the memory trace (engram; a population of neurons that represents the memory) from the exposure of the stimuli. In general, deeper levels of encoding (e.g., utilizing mnemonics) help strengthen the memory trace. After the development of the memory trace, the engram, over time, goes through a consolidation period where it either becomes stabilized or destabilized. Lastly, effective retrieval cues can help facilitate the retrieval of the memory.
Learning is the acquisition of a skill or content, whereas memory is the expression of what has been previously learned. In order for a long-term memory to be successfully retrieved, effective encoding and consolidation need to be occurred. Adequate learning of the material helps create an opportunity to successfully retrieve a memory via optimal encoding and consolidation. Adequate learning may facilitate memory retention by increasing an extension of the experience and reducing the resistance of impeding aspects that conflict with that experience [54].
Both previous experimental research [6, 31, 49] and meta-analytic reviews [5, 32, 36, 47] have demonstrated that acute exercise (i.e., a single bout of exercise) can enhance long-term memory function. Further, the exercise-memory relationship may vary depending on the distinctive types of memory (e.g., episodic memory and working memory) as well as the intensity of exercise (e.g., low-intensity vs. moderate-intensity vs. high-intensity). For example, while acute exercise at lower intensity may play a key role in enhancing working memory, acute exercise at higher intensity may have a facilitative effect on episodic memory [34]. As discussed elsewhere, this effect may, in part, be attributed to exercise-induced (1) increases in neuronal excitability, (2) changes in dopamine, epinephrine, and norepinephrine levels, and (3) increases in BDNF (brain-derived neurotrophic factor) expression [22, 38, 56]. However, what is lacking in the literature are studies specifically evaluating the effect of acute exercise on learning improvement; that is, whether acute exercise facilitates the acquisition (encoding and consolidation) of the stimuli, which may ultimately help induce long-term storage and retrieval of the information. Winter et al. [56], in a sample of 27 young adults, evaluated the effects of acute exercise on learning improvement, i.e., memory performance scores across five successive learning blocks (trials). They demonstrated that a single bout of high-intensity exercise was associated with greater learning improvement across the five learning blocks. They also showed that BDNF, dopamine, and epinephrine uniquely contributed to improved learning, short-term, as well as long-term memory. In other experiments on this topic, researchers evaluated whether there was an intensity-specific impact of acute exercise on learning. This plausibility of this intensity-specific effect [34] can be justified by previous publications demonstrating that acute moderate-intensity exercise promotes memory function as well as learning processes [25, 51]. In contrast to these findings, Frith et al. [19] did not observe facilitative effects of vigorous-intensity acute exercise on learning when compared to a control group. Given these mixed findings, an empirical review that evaluates the effects of acute exercise on learning improvement is needed.
To the best of our knowledge, it is uncertain as to whether acute exercise promotes long-term memory via enhanced learning, or rather, facilitates post-learning mechanisms, such as augmenting storage and retrieval (see El-Sayes et al. [14], Loprinzi et al. [38], Loprinzi et al. [41], Moore and Loprinzi [44] for previous reviews detailing the mechanisms through which acute exercise may enhance memory). Herein, we focus specifically on cognitive-based learning as evaluated by 5 trials of the verbal memory test, which extends previous work that has focused on motor learning [48, 50, 52]. Interestingly, a recent systematic review with meta-analysis by Wanner et al. [53] demonstrated that acute cardiovascular exercise helps to facilitate the consolidation of acquired motor memory, but not motor memory encoding. In particular, exercise before motor skill practice may be favorable for short-term consolidation, whereas post-practice exercise may be effective in improving long-term consolidation. Therefore, the purpose of this meta-analysis is to evaluate the effects of acute exercise on cognitive-based short-term memory improvement. Such an investigation may help to address the mixed findings as well as shed additional insights into future mechanistic studies on this topic. Notably, although “learning” is tacit across the five learning trials, hereafter we refer to this as “short-term memory improvement” instead of learning, as the evaluated studies included a recall assessment after each of the five trials. As such, it is not possible to disentangle whether the improvement in memory across the trials was from learning or recall.
Methods
Data Sources and Search Strategy
The present review was presented in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. We performed a computerized systematic literature search using electronic databases, such as PubMed, PsycINFO, Google Scholar, and Medline. All documents were retrieved from the inception of the study to March 20th, 2021. The final search terms, including their combinations, were: acute exercise, exercise, California Verbal Learning Test (CVLT), Rey Auditory Verbal Learning Test (RAVLT), learning, list learning, and memory. In order to reduce the likelihood of errors during literature searches, we utilized database-appropriate syntax for each database, in combination with the above-noted key words, based on systematic search strategies [2]. Table 1 indicates the suitable syntax components for each database.
Study Selection
Two separate authors (MJ and JB) independently conducted the computerized searches to determine the number of eligible studies. Each of the searches in each respective database was imported into Excel, and then, duplicate references were deleted in Excel. An agreement on eligible studies was reached from the two authors, and a third author was invited to reach a consensus through discussion in the case of any disagreement and/or conflict between the two reviewers. In addition to the above-mentioned database search, two reviewers checked a list of references during full-text screening in order to identify additional eligible articles. All studies appearing to meet our eligible criteria were screened and cross-checked at the full-text level. Similar to the previous stage, a third author was involved in the discussion in case any disagreement occurred during the full-text review.
Eligibility Criteria
Studies were included if they: (1) utilized an experimental design with a comparison to a non-exercising control group, (2) employed a human model, (3) evaluated an acute bout of exercise (defined as a single bout of exercise) as a predictor, (4) performed acute exercise prior to learning, (5) assessed RAVLT or CVLT as the main outcome variable, (6) recalled a recording of 15 listed words for RAVLT (in the case of CVLT, a 16 word-list) five times in a row (trials 1–5), (7) provided sufficient information (e.g., mean, standard deviation [SD], and sample size) to calculate an effect size (ES) estimate, and (8) were published in English.
Methodological Quality Assessment
Using the modified version of the Downs and Black checklist [11], two independent authors evaluated the methodological quality of the included studies. This checklist was developed to measure the methodological quality of randomized and non-randomized studies, which was based on 27 criteria across 4 domains, including reporting, external validity, internal validity, and power, with a total maximum score of 28 (1 point per question except for question five [2 points]). All disagreements in quality scorings between the two original authors were addressed by inviting a third author to reach consensus via discussion. To avoid confusion of power calculation for users, the last question was changed from a 5-point to a 1-point rating, where 1 was scored if a power or sample size computation was reported, and 0 was scored when there was no power computation or indication of whether the number of subjects was appropriate for the study design. Of the 27 questions, 3 questions, “Have the characteristics of patients lost to follow-up been described?” “In trials and cohort studies, do the analyses for different lengths of follow-up of patients, or in case–control studies, is the time period between the intervention and outcome the same for cases and controls?” and “Were losses of patients to follow-up taken into account?” were deleted from the checklist because they did not fit the inclusion criteria in the present meta-analysis. Consequently, we used the revised Downs and Black checklist consisting of 24 questions. The scoring of this checklist ranged from 0 to 25, with a higher score indicating a higher level of methodological quality.
Data Extraction
Two separate authors extracted and coded detailed information from each of the evaluated studies as follows: (1) authors, publication year, and language of publication, (2) participant’s characteristics (e.g., age, gender, and sample size), (3) experimental design (exercise vs. control), (4) exercise protocol (e.g., exercise temporality, exercise intensity, and exercise duration), (5) memory task (e.g., RAVLT and CVLT), and (6) main study findings.
Outcome Measures
As previously described, cognitive-based learning was assessed using RAVLT or CVLT. Both of the two tasks are multiple trials, word-list episodic memory tasks. The standardized RAVLT consists of an immediate memory assessment and a delayed free-recall test [9]. This task requires participants to listen to and immediately recall a recording of a list of 15 words (List A) five times in a row (trials 1–5). Immediately following List A, participants are then asked to listen to and immediately recall a list of 15 new words; this step is represented by trial 6. Similar to the RAVLT, the standardized CVLT provides participants with five learning trials that include a list of 16 words (List A) as well as six learning trials with 16 listed new words [15]. After each trial, participants are asked to immediately recall as many words as they can in any order. Performance on trial 1 is indicative of short-term memory. And the change in performance across trials 1 to 5 is considered a measure of learning; as stated, herein we refer to this as “short-term memory improvement” given that a recall assessment occurred after each trial. Since a central focus of this meta-analysis is the potential effects of acute exercise on learning, we focused on the memory scores obtained from trials 1 to 5 across exercise and non-exercise groups.
Statistical Analyses
To carry out the meta-analysis of repeated measures studies, we employed the trend meta-analysis, which enables the measurement of trends over time at the study specific level of interest (for instance, this study is interested in investigating acute exercise effects [vs. control] on learning). In this approach, means and variances at each time-point within the primary study are required to calculate efficient estimates of the regression slope [45]. Regression is utilized to model the outcome over time and provides a summary effect of the slope estimate. As implemented in JASP (version 0.11.1.0.), linear regression was employed to estimate a standardized regression coefficient (i.e., X-axis = trials and Y-axis = learning scores) from each individual study by each group (exercise vs. control groups). Both JASP and Comprehensive Meta-Analysis software (version 3, Biostat, New Jersey, USA, www.meta-analysis.com) were used to calculate the standardized regression coefficient’s ESs. As we anticipated heterogeneity among the studies, a random-effects model was employed in the meta-analysis. The degree of heterogeneity of the ESs was calculated using Cochran’s Q-statistic (Q) and the variation in ES was calculated with I2, tau (T) (i.e., SD of true effects), and T2 index (i.e., variance of true effects). Q statistics evaluate the null hypothesis that all individual ES estimate the same population ES [33]. I2 indicates the proportion of the variance in observed effects reflecting variance in true effects, rather than sampling error. I2 values of 25%, 50%, and 75% indicate low, moderate, and high levels of heterogeneity, respectively [26]. The subgroup analysis was performed to test how the observed ES differs depending on exercise engagement (exercise vs. non-exercise control). Subgroup analyses typically provide two results: one is to compare the mean ESs across groups/levels, and the other is to compute the mean ES separately for each subgroup of studies [8]. Finally, the risk of bias across studies was assessed with Egger’s test of the regression intercept. This test was conducted to examine the association between the observed ESs and their standard errors. If the regression intercept is statistically significant, publication bias may be present [13]. Accordingly, if there is evidence of publication bias, it is customary to conduct the Duval and Tweedie’s trim and fill analysis [12] to provide an estimate of the number of missing studies and an adjusted effect for the publication bias. Statistical significance was set at an alpha level of 0.05.
Results
Retrieved Articles
Figure 1 depicts the flowchart of the literature search process. The computerized searches from the four databases yielded 1082 articles. Moreover, six additional articles were identified through lists of references on acute exercise and memory through manual searching. Among the 1088 articles, 123 duplicates were removed and 965 articles were screened. After an initial screening of 965 titles and abstracts, 19 potentially relevant articles were carefully reviewed in full. Among these 19 articles, 12 were ineligible because they did not meet our inclusion criteria [e.g., not directly comparing a non-exercise control group to an exercise group [18] and not providing sufficient data to compute an ES (Dougherty et al. [10]). Thus, seven studies met the inclusion criteria for the systematic review through the computerized searches. Lastly, only one (Etnier et al. [17]) of the seven studies was conducted with a sample of children; given that children and adults have different memory capacities, we decided to remove Etnier et al.’s [17] study and exclusively focus on adult studies. Consequently, a total of six studies were selected for the quantitative meta-analysis.
Flowchart describing exclusions of potential studies and final number of included studies in a meta-analysis [43]
Study Characteristics
Detailed information on the study characteristics is displayed in Table 2. Sample sizes ranged from 20 to 88, with all studies testing young adults (18–30 years). Among the six studies, three (50%) employed a within-subject design, and four (67%) utilized an acute bout of moderate-intensity exercise. Further, in all of the 6 studies, learning was assessed using the standard RAVLT.
Study Quality
Based on the modified Downs and Black checklist, the methodological quality of the evaluated studies was good (20.33 ± 1.37, mean ± SD), ranging from 19 to 22 [27]. All studies were scored within an acceptable range of methodological quality, and thus, were included in the meta-analysis.
Quantitative Analysis
The meta-analytic results are shown in Fig. 2. Among these six studies, 16 standardized regression coefficient ESs were calculated: Loprinzi et al. [39] produced six ESs (three each for exercise and non-exercise control groups) and the five other studies [1, 19, 25, 29, 51] each produced two ESs (one for exercise and another for non-exercise control groups). As illustrated by Fig. 2, there was a statistically significant positive relationship between the trials and word recall [ES = 0.96, 95% CI (0.95, 0.97), Z-value = 25.37, P < 0.001] (for more information on Z-values and P-values for each study, see Supplementary Table 1). Further, there was evidence of heterogeneity of ESs [Q(15) = 32.46, P = 0.006]. We rejected the null hypothesis that the true ES was identical across all studies. The I2 statistic was 54%, which means that 54% of the variance in the observed effects reflected variance of the true effects, rather than sampling error. The variance of true effects (T2) was 0.05 and the SD of true effects (T) was 0.22. Table 3 shows the subgroup analysis results. There was no statistically significant subgroup difference between exercise and non-exercise control groups (Qb = 0.40, df = 1, P = 0.53). By further examining the ES and 95% CI of each subgroup, we found a positive relationship between the task trials and memory performance for both the exercise [ES = 0.96, 95% (CI 0.94, 0.98), P < 0.001] and non-exercise control groups [ES = 0.95, 95% CI (0.94, 0.97), P < 0.001]. The Egger’s test of regression intercept was not statistically significant (intercept = 0.51, P = 0.77), indicating that there was no evidence of publication bias across studies.
Forest plot of the effect sizes for the evaluated studies. Only results from an independent exercise group were included in the meta-analysis. The boxes represent the point estimates from the single studies. The diamond represents the pooled result. The horizontal lines represent the length of the 95% confidence intervals of the study result, with each end of the line representing the boundaries of the confidence intervals. RE random effect
Discussion
The purpose of the present meta-analysis was to evaluate the effects of acute exercise on short-term memory improvement compared to a control group (no exercise). Accumulated research demonstrates that an acute bout of exercise may help to enhance both short- and long-term memory function [7, 19, 25, 30, 40], but to date, current data examining the quantitative impacts of acute exercise on short-term memory improvement are inconsistent. This is a major direction for future work due to the limited understanding as to whether acute exercise facilitates long-term memory via enhanced learning, or via post-learning mechanisms. Taken together, across the six studies evaluated in our current meta-analytic review, we provide evidence that acute exercise, when compared to a non-exercise seated control, does not influence short-term memory improvement when evaluated via a list-learning paradigm.
Our meta-analytic findings demonstrated that there was a significant increase in short-term memory improvement across both the exercise and control groups in trials 1 to 5, which aligns with previous experimental studies [29, 51]. Expectedly, repeated exposure to learning stimuli may help to facilitate memory retention because performing memory recall (trials 1–5) may act as a form of studying [46]. The null exercise effects may be explained by several reasons. First, moderate-intensity acute exercise implemented across four studies may be insufficient to influence the short-term memory improvement effect. As thoroughly discussed elsewhere [34], acute moderate-intensity exercise prior to memory encoding may favor executive function, whereas acute high-intensity exercise may be beneficial for episodic memory. Elevated release of catecholamines in the brain, via high-intensity exercise, can enhance synaptic plasticity in the hippocampus [21, 42]. Further, exercise-induced increases in neuronal excitability may help promote long-term potentiation of the engram related to the learning stimuli [35]. Additionally, given the nature of the RAVLT (i.e., episodic memory task), higher intensity acute exercise may be better to enhance learning [56]. Second, five out of six studies employed treadmill walking or jogging-based exercise protocols, which may be the potential cause of the null exercise effects. It is expected that more complex movement patterns have greater effects on regional cerebral blood flow and cortical excitability, which can subserve episodic memory function [4, 24, 55]. It would be interesting for future work to consider the effects of different exercise modalities (e.g., open-skilled exercise) on learning [3, 23]. Lastly, considering that most of the evaluated studies utilized the combined effect of short duration exercise followed by a relatively short recovery period, null effects are not surprising. As demonstrated thoroughly elsewhere [7], the post-exercise recovery period may play a key role in modulating the acute effects of exercise on memory function. Recent work from Loprinzi et al. [39] suggests that a short (e.g., 5-min) recovery period with high-intensity resistance exercise may compromise performance on word-list episodic memory task, but a longer duration recovery period (e.g., 10–20 min) after a single bout of high-intensity resistance exercise may improve word-list episodic memory. In contrast to these findings, Crush et al. [7] suggests that a 5 min resting period with acute moderate-intensity treadmill exercise may have a favorable effect on memory functioning. Altogether, it is possible that the length of the post-exercise recovery period may play a moderating role in the exercise-learning relationship. Due to the limited number of studies included, we were unable to consider this factor as a potential moderator; thus, future meta-analytic review should evaluate whether the post-exercise recovery period moderates the acute exercise effects on short-term memory improvement.
This meta-analysis suggests that repetitive learning may have beneficial effects on memory function, regardless of exercise engagement. A major strength of this study is that this is the first meta-analytic review to investigate the effects of acute exercise on short-term memory improvement. In spite of these strengths, there are some limitations worth considering. Firstly, the present meta-analysis included a relatively small number of experimental studies, all of which came from the laboratory of the authors of this paper. As such, future work, including from other laboratories, is needed. Another limitation is that all six studies utilized a sample of healthy young adults. Future work is needed to establish the dynamic associations between the optimal exercise parameters (e.g., duration, timing, intensity, and type) and learning among a more diverse population, such as children, the elderly, or individuals with mild cognitive impairment. Use of diverse populations may be particularly useful; in the present meta-analysis of college-aged students, the short-term memory improvement curve across the 5 trials was strong, and thus, may have been insensitive to behaviors, such as low-to-moderate intensity exercise.
Secondly, as stated earlier, we initially intended to explore the impact of other potential moderators (e.g., sample characteristics, exercise intensity, and exercise duration); however, we were unable to conduct additional moderator analyses due to the lack of sufficient data. According to Fu et al. [20], each subgroup should have a minimum of four ESs. Small or imbalanced number of studies may weaken statistical power of subgroup analysis tests and warrant careful interpretation of the results from subgroup analyses [28]. Accordingly, future work should consider evaluating whether other moderating variables have differential effects on short-term memory improvement. Lastly, it would also be worthwhile for future work to examine whether other types of learning/memory tasks moderate the effects of acute exercise on short-term memory improvement. Such suggestions will be critical for subsequent research that aims to demonstrate under what conditions this acute exercise-induced effect may occur. For example, in the standard RAVLT learning task, after each trial, participants recall as many words as possible. This retrieval component may have attenuated a potential exercise-induced effect on learning. Therefore, future work should consider revaluating this model, but then, in addition to including conditions where recall is performed after each trial, compare it to conditions when recall is only performed after the encoding of the fifth learning trial; notably, recent research demonstrates reliable effects of acute exercise on improving learning when recall only occurs after the fifth trial [37].
At this point, future work is needed to confirm whether acute exercise can enhance learning. If such work is in alignment with our current meta-analytic findings, then this will suggest that the observed effects of acute exercise on long-term memory may occur from post-learning mechanisms (i.e., hypothesis that acute exercise-induced enhancements in long-term memory may occur less from encoding, but more from consolidation-based mechanisms). As an example, a recent meta-analysis by Loprinzi et al. [36] demonstrated that the largest effect (Cohen’s d = 0.19, P < 0.05) for enhancing long-term episodic memory occurs when the acute bout of exercise occurs shortly after learning. They have previously discussed the potential mechanisms of this effect that acute exercise may upregulate molecular pathways (e.g., BDNF, functional connectivity, and long-term potentiation), which may, in turn, help to facilitate long-term memory [16, 38, 41]. But, this is an area in need of future mechanistic research in human models as the neurophysiological and molecular mechanisms related to this effect remain uncertain at the moment. This is an exciting area of inquiry that will require future experimentation to determine the extent to which exercise enhances long-term memory, and whether this occurs via enhanced learning and/or memory consolidation mechanisms.
References
Austin M, Loprinzi PD. Acute exercise and mindfulness meditation on learning and memory: randomized controlled intervention. Health Promot Perspect. 2019; 9(4):314–18.
Bramer WM, de Jonge GB, Rethlefsen ML, Mast F, Kleijnen J. A systematic approach to searching: an efficient and complete method to develop literature searches. J Med Library Assoc JMLA. 2018;106(4):531–41.
Cantrelle J, Burnett G, Loprinzi PD. Acute exercise on memory function: open vs closed skilled exercise. Health Promot Perspect. 2020;10(2):123–8.
Carey JR, Bhatt E, Nagpal A. Neuroplasticity promoted by task complexity. Exerc Sport Sci Rev. 2005;33(1):24–31.
Chang YK, Labban JD, Gapin JI, Etnier JL. The effects of acute exercise on cognitive performance: a meta-analysis. Brain Res. 2012;1453:87–101.
Coles K, Tomporowski PD. Effects of acute exercise on executive processing, short-term and long-term memory. J Sports Sci. 2008;26(3):333–44.
Crush EA, Loprinzi PD. Dose-response effects of exercise duration and recovery on cognitive functioning. Percept Mot Skills. 2017;124(6):1164–93.
Deeks JJ, Higgins JPT, Altman DG (editors). Chapter 10: Analysing data and undertaking meta-analyses. In: Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA (editors). Cochrane Handbook for Systematic Reviews of Interventions. 2020. https://training.cochrane.org/handbook/current/chapter-10#section-10-10-2. Accessed 10 May 2020.
de Sousa Magalhães S, Fernandes Malloy-Diniz L, Cavalheiro Hamdan A. Validity convergent and reliability test-retest of the rey auditory verbal learning test. Clin Neuropsychiatry. 2012;9(3):129–37.
Dougherty RJ, Schultz SA, Boots EA, Ellingson LD, Meyer JD, Van Riper S, Cook DB. Relationships between cardiorespiratory fitness, hippocampal volume, and episodic memory in a population at risk for Alzheimer’s disease. Brain Behav. 2017;7(3):1–12.
Downs SH, Black N. The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. J Epidemiol Community Health. 1998;52(6):377–84.
Duval S, Tweedie R. Trim and fill: a simple funnel-plot–based method of testing and adjusting for publication bias in meta-analysis. Biometrics. 2000;56(2):455–63.
Egger M, Smith GD, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315(7109):629–34.
El-Sayes J, Harasym D, Turco CV, Locke MB, Nelson AJ. Exercise-induced neuroplasticity: a mechanistic model and prospects for promoting plasticity. Neuroscientist. 2019;25(1):65–85.
Elwood RW. The California Verbal Learning Test: psychometric characteristics and clinical application. Neuropsychol Rev. 1995;5(3):173–201.
Erickson KI, Voss MW, Prakash RS, Basak C, Szabo A, Chaddock L, Kramer AF. Exercise training increases size of hippocampus and improves memory. Proc Natl Acad Sci. 2011;108(7):3017–22.
Etnier J, Labban JD, Piepmeier A, Davis ME, Henning DA. Effects of an acute bout of exercise on memory in 6th grade children. Pediatr Exerc Sci. 2014;26(3):250–8.
Etnier JL, Wideman L, Labban JD, Piepmeier AT, Pendleton DM, Dvorak KK, Becofsky K. The effects of acute exercise on memory and brain-derived neurotrophic factor (BDNF). J Sport Exerc Psychol. 2016;38(4):331–40.
Frith E, Sng E, Loprinzi PD. Randomized controlled trial evaluating the temporal effects of high‐intensity exercise on learning, short‐term and long‐term memory, and prospective memory. Eur J Neurosci. 2017;46(10):2557–564.
Fu R, Gartlehner G, Grant M, Shamliyan T, Sedrakyan A, Wilt TJ, Trikalinos TA. Conducting quantitative synthesis when comparing medical interventions: AHRQ and the Effective Health Care Program. J Clin Epidemiol. 2011;64(11):1187–97.
Gagnon SA, Wagner AD. Acute stress and episodic memory retrieval: neurobiological mechanisms and behavioral consequences. Ann N Y Acad Sci. 2016;1369(1):55–75.
Griffin ÉW, Mullally S, Foley C, Warmington SA, O’Mara SM, Kelly ÁM. Aerobic exercise improves hippocampal function and increases BDNF in the serum of young adult males. Physiol Behav. 2011;104(5):934–41.
Gu Q, Zou L, Loprinzi PD, Quan M, Huang T. Effects of open versus closed skill exercise on cognitive function: a systematic review. Front Psychol. 2019;10:1707.
Gur RC, Jaggi JL, Ragland JD, Resnick SM, Shtasel D, Muenz L, Gur RE. Effects of memory processing on regional brain activation: cerebral blood flow in normal subjects. Int J Neurosci. 1993;72(1–2):31–44.
Haynes IV JT, Frith E, Sng E, Loprinzi PD. Experimental effects of acute exercise on episodic memory function: considerations for the timing of exercise. Psychol Reports. 2019;122(5):1744–754.
Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327(7414):557–60.
Hooper P, Jutai JW, Strong G, Russell-Minda E. Age-related macular degeneration and low-vision rehabilitation: a systematic review. Can J Ophthalmol. 2008;43(2):180–7.
Jackson D, Turner R. Power analysis for random-effects meta-analysis. Res Synth Methods. 2017;8(3):290–302.
Johnson L, Loprinzi PD. The effects of acute exercise on episodic memory function among young University students: Moderation considerations by biological sex. Health Promot Perspect. 2019;9(2):99–104.
Jung M, Zou L, Yu JJ, Ryu S, Kong Z, Yang L, Loprinzi PD. Does Exercise Have a Protective Effect on Cognitive Function under Hypoxia? A Systematic Review with Meta-Analysis. J Sport Health Sci. 2020;9(6):562–77.
Labban JD, Etnier JL. Effects of acute exercise on long-term memory. Res Q Exerc Sport. 2011;82(4):712–21.
Lambourne K, Tomporowski P. The effect of exercise-induced arousal on cognitive task performance: a meta-regression analysis. Brain Res. 2010;1341:12–24.
Lipsey MW, Wilson DB. Practical meta-analysis. Thousand Oaks: Sage; 2001.
Loprinzi PD. Intensity-specific effects of acute exercise on human memory function: Considerations for the timing of exercise and the type of memory. Health Promot Perspect. 2018;8(4):255–62.
Loprinzi PD. The effects of exercise on long-term potentiation: a candidate mechanism of the exercise-memory relationship. OBM Neurobiol. 2019;3(2):13.
Loprinzi PD, Blough J, Crawford L, Ryu S, Zou L, Li H. The temporal effects of acute exercise on episodic memory function: Systematic review with meta-analysis. Brain Sci. 2019;9(4):87.
Loprinzi PD, Day S, Hendry R, Hoffman S, Love A, Marable S, Gilliland B. The effects of acute exercise on short-and long-term memory: considerations for the timing of exercise and phases of memory. Eur J Psychol. 2021;17(1):85–103.
Loprinzi PD, Edwards MK, Frith E. Potential avenues for exercise to activate episodic memory-related pathways: a narrative review. Eur J Neurosci. 2017;46(5):2067–77.
Loprinzi PD, Green D, Wages S, Cheke LG, Jones T. Experimental effects of acute high-intensity resistance exercise on episodic memory function: consideration for post-exercise recovery period. J Lifestyle Med. 2020;10(1):7–20.
Loprinzi PD, Kane CJ. Exercise and cognitive function: a randomized controlled trial examining acute exercise and free-living physical activity and sedentary effects. Mayo Clin Proc. 2015;90(4):450–60.
Loprinzi PD, Ponce P, Frith E. Hypothesized mechanisms through which acute exercise influences episodic memory. Physiol Int. 2018;105(4):285–97.
McMorris T. Developing the catecholamines hypothesis for the acute exercise-cognition interaction in humans: Lessons from animal studies. Physiol Behav. 2016;165:291–9.
Moher D, Liberati A, Tetzlaff J, Altman DG, Prisma Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6(7):e1000097.
Moore D, Loprinzi PD. Exercise influences episodic memory via changes in hippocampal neurocircuitry and long-term potentiation. Eur J Neurosci. 2020. https://doi.org/10.1111/EJN.14728.
Peters JL, Mengersen KL. Meta-analysis of repeated measures study designs. J Eval Clin Pract. 2008;14(5):941–50.
Roediger HL III, Karpicke JD. Test-enhanced learning: Taking memory tests improves long-term retention. Psychol Sci. 2006;17(3):249–55.
Roig M, Nordbrandt S, Geertsen SS, Nielsen JB. The effects of cardiovascular exercise on human memory: a review with meta-analysis. Neurosci Biobehav Rev. 2013;37(8):1645–66.
Roig M, Skriver K, Lundbye-Jensen J, Kiens B, Nielsen JB. A single bout of exercise improves motor memory. PLoS ONE. 2012;7(9):E44594. https://doi.org/10.1371/journal.pone.0044594.
Siddiqui A, Loprinzi PD. Experimental investigation of the time course effects of acute exercise on false episodic memory. J Clin Med. 2018;7(7):157.
Skriver K, Roig M, Lundbye-Jensen J, Pingel J, Helge JW, Kiens B, Nielsen JB. Acute exercise improves motor memory: exploring potential biomarkers. Neurobiol Learn Mem. 2014;116:46–58.
Sng E, Frith E, Loprinzi PD. Temporal effects of acute walking exercise on learning and memory function. Am J Health Promot. 2018;32(7):1518–525.
Snow NJ, Mang CS, Roig M, McDonnell MN, Campbell KL, Boyd LA. The effect of an acute bout of moderate-intensity aerobic exercise on motor learning of a continuous tracking task. PLoS ONE. 2016;11(2):e0150039. https://doi.org/10.1371/journal.pone.0150039.
Wanner P, Cheng FH, Steib S. Effects of acute cardiovascular exercise on motor memory encoding and consolidation: A systematic review with meta-analysis. Neurosci Biobehav Rev. 2020;116:365–81.
Washburne JN. The definition of learning. J Educ Psychol. 1936;27(8):603–11.
Winstein CJ, Grafton ST, Pohl PS. Motor task difficulty and brain activity: investigation of goal-directed reciprocal aiming using positron emission tomography. J Neurophysiol. 1997;77(3):1581–94.
Winter B, Breitenstein C, Mooren FC, Voelker K, Fobker M, Lechtermann A, Knecht S. High impact running improves learning. Neurobiol Learn Mem. 2007;87(4):597–609.
Acknowledgements
We have no conflicts of interest and no funding was used to prepare this manuscript.
Author information
Authors and Affiliations
Corresponding author
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jung, M., Kang, M., Blough, J. et al. Experimental Effects of Acute Exercise on Cognitive-Based Short-Term Memory Improvement: A Meta-analysis of Repeated Measures Studies. J. of SCI. IN SPORT AND EXERCISE 4, 12–20 (2022). https://doi.org/10.1007/s42978-021-00121-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42978-021-00121-5