Abstract
Urbanization results in rapid land transformations impacting bio-diversity and ecosystem services. With urban expansions population transition will predominantly occur in the peri-urban areas of the world’s metropolitan cities like India, China, and Nigeria, which are often located in naturally species-rich regions and highly vulnerable to plant invasions. Thus it is increasingly important to monitor the changes occurring in the floral composition in such areas. The present study was therefore conducted across peri-urban habitats of Greater Kolkata, in order to establish a baseline data on plant species richness, invasive species co-occurrence, invader dominance and understand the bio-diversity pattern. Results showed a total of 62 plant species, mostly annual herbs, belonging to 29 families constituting the species richness in the study area. Both native (53%) and alien (47%) species were almost equally distributed and invasive species (32%) represented the majority among alien species. The findings also indicate that the species pool was dominated by sparsely distributed species. The number of invasive species varied from 2 to 9 per site, with 80% of sites supporting 3 or more species. Alternanthera philoxeroides and Mikania micrantha were the most frequently occurring invasive species. Overall, M. micrantha appeared to be the dominant species with 62% of sites with a high cover (> 70%) and was found to be evenly distributed in areas. However, they pose serious threat to local bio-diversity which shows that species-specific management is needed even in the peri-urban habitats.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rapid urbanization and the growth of peri-urban areas is now a global phenomenon and has emerged to be one of the most critical challenges to ecological sustainability in urban areas (Dadashpoor & Ahani, 2021). Peri-urban areas are the peripheral areas or the transition zone in cities where landscape features are subject to rapid modifications induced by human activities and as cities develop, much of their growth is located in such areas (Narayani & Nagalakshmi, 2023). The patterns of peri-urban transformation differ across countries due to significant variation in geography, climate, socioeconomic characteristics and governance frameworks (Chen et al., 2020). While peri-urban growth in developed countries is considered an indicator of urban welfare and well-being, its pace and scale in developing nations is often negatively considered to be a planning challenge (Follmann, 2022). Unplanned urbanization and sprawl of large cities in developing nations result in rapid land transformations which encroach on and degrade the vegetation in peri-urban zones limiting ecosystem services that are critical to human health and well-being (Ellis, 2021; Rozas-Vasquez et al., 2022; Chen et al., 2023). Thus, in an era of climate change and threat of natural hazards, peri-urbanization is an important challenge, especially for developing nations, particularly in terms of environmental management and achieving global targets such as the Sustainable Development Goal (UN-SDG 11) on Sustainable Cities and Communities (Ferreira et al., 2021; Mortoja & Yigitcanlar, 2022).
A rapidly growing human population accelerates economic development and international trade which are factors that promote the introduction of alien and invasive plant species (Early et al., 2016; Seebens et al., 2018). Plant introductions and human disturbance, combined with primary and secondary urbanization processes, have wide-ranging implications for bio-diversity (de Barros Ruas et al., 2022; Jaureguiberry et al., 2022). Global assessments show that urban expansion has caused about 50% loss of local within-site species richness and 38% loss of total abundance of species in intensively used urbanized areas as compared to a naturally un-impacted baseline (Newbold et al., 2015). Moreover, global climate change also aggravates the risk of plant invasions and expands their range (Weiskopf et al., 2020) which may present a growing threat to ecosystems, potentially compromising their function, structure, and service provision (Pyšek et al., 2020a). Alien and invasive plants exert stronger direct and indirect effects on native flora (Allen et al., 2020) be it their ability to survive in stressful urban environments with higher temperatures (urban heat island effect), increased drought and widespread pollutants (Calfapietra et al., 2015; English & Wright, 2021; Jha et al., 2020); or their vegetative and reproductive traits such as high specific leaf area and smaller seeds which help them to rapidly take up available resources, have high intrinsic growth rates allowing them to grow and extend their range rapidly (Zhang et al., 2019); or their multiple modes of reproduction (Wang et al., 2019); or production of allelochemicals that suppress germination and growth of neighboring plants (Thiébaut et al., 2019; Yuan et al., 2021); or through soil amendments (Fahey & Flory, 2022); or trophic imbalance and decreased species resilience which can greatly reduce the biomass, coverage and diversity of native communities which have high habitat specialization (Banerjee & Dewanji, 2017a; Liu et al., 2017; Weiskopf et al., 2020). City centers are reported to be hubs for the spread of invasive alien species (Kühn et al., 2017; Appalasamy & Ramdhani, 2020) making peri-urban areas vulnerable to their spread through transportation corridors.
Although urbanization is known to influence the distribution of plant diversity (Aronson et al., 2015) and may favor alien species over native species (McKinney, 2008; Avolio et al., 2020), the patterns of the plant diversity distribution vary significantly from study to study. Satellite-based remote sensing studies has been widely used to monitor vegetation growth over large areas (Guan et al., 2019; Sharma et al., 2022) but there is limited availability of ground-based observations on vegetation through field surveys (Zhang et al., 2022), especially in tropical cities of developing countries, where trees are the most reported vegetation type studied (Alue et al., 2022; Gopal et al., 2019; Jha et al., 2019). Moreover, many urban bio-diversity studies have been conducted in highly managed city parks and green spaces with relatively few studies occurring in unmanaged, remnant locations such as peri-urban areas (Avolio et al., 2020; Knight et al., 2021). Future urban expansion, with increases in the size of the world's urban population, is projected to be at an alarming rate under different climate change and development pathway (Chen et al., 2020; Gao & O’Neill, 2020). This population transition will predominantly occur in the peri-urban areas of the world’s metropolitan cities of Asia and Africa like India, China, and Nigeria (United Nations, 2018), which are often located in naturally species-rich regions highly vulnerable to plant invasions. Current policies in most of these countries are under-equipped to address emerging threats from invasive alien species both in terms of the current extent of the problem and control efforts for existing species (Early et al., 2016).
India, one of the fastest developing urban systems in the world (Sun et al., 2020), is also undergoing rapid transformation of its existing peri-urban areas which are under tremendous stress due to pressures created by urbanization, including biotic invasions (Aijaz, 2019). Although many Indian databases regarding alien and invasive plant species have come up during the last few years (Inderjit et al., 2018; Pant et al., 2021; Sankaran et al., 2021), but there have been no efforts directed toward prioritization of invasive species for location specific monitoring or management (Mungi et al., 2019) or toward comprehensive appraisals of the economic costs of invasive alien species in India (Bang et al., 2022). Furthermore, due to limited availability of studies in peri-urban areas (Agrawal & Narayan, 2017; Gupta & Narayan, 2006) prone to alien plant invasion, projected to increase in the next three decades (Seebens et al., 2021), there is a primary need to record the presence of species in these transitional areas so that future monitoring or management strategies can be established. Using a rapidly urbanizing city in India as a case study, the present ecological study involving an in situ vegetation survey was therefore carried out across peri-urban habitats within Greater Kolkata in order to establish a baseline data on (i) the herbaceous plant species richness and composition in terms of their families, life forms and invasion status, (ii) the co-occurrence pattern of invasive plants and their dominance in terms of cover percentage and (iii) the diversity patterns which could aid in understanding future changes in floral compositions in disturbed environments that could aid in developing management actions, wherever necessary.
Materials and methods
Study area and site selection
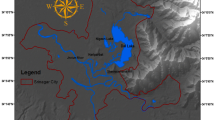
As mentioned, the city of Kolkata, India, was selected as the case study area, in which 21 peri-urban sites in the Greater Kolkata region were surveyed (see ESM1). The Greater Kolkata region is linearly located along the eastern and western banks of the Hooghly river and lies between 88°34′04.91″ E and 22º21′04.07″ N, covering an area of 1886.7 km2. Previous studies using remote sensing data show that the urban expansion has consistently occurred along the length of the Hooghly river in a linear, unidirectional pattern comprising of newly built-up patches from the edge of existing ones (Chakraborty et al., 2021; Sahana et al., 2018). This mostly happens along the transport corridors and the lands that are located adjacent to the city are more prone to this conversion (Sahana et al., 2023). The city has experienced marked expansion in built-up areas at the cost of vegetation, waterbodies and agricultural land (Mazumder et al., 2021; Mondal et al., 2017). The study area and sites (Fig. 1), depicting peri-urban land comprise suburbs and exurbs, are located beyond the urban core or the Kolkata Municipal Corporation (KMC). These peri-urban spaces are experiencing rapid population growth, while stagnant or negative growth rate has been reported from the KMC area (Mondal & Banerjee, 2021). The city has a tropical climate with monthly mean temperatures ranging from 19 to 30 °C (Dasgupta et al., 2013). Summers are extremely hot and humid with maximum temperatures rising to 40 °C while winter lasts for only a few months with seasonal lows dipping to 9 °C (Sharma et al., 2015). The region is also prone to cyclones and has an annual rainfall of 1640 mm (IMD Kolkata 2016).
The site selection included all kinds of spaces well outside the urban core (including suburbs, fringe, exurbs, and other locales) using the pragmatic approach as followed by most studies (Follmann, 2022). Although some studies have tried to spatially demarcate peri-urban zones based on either socioeconomic structures or through land use change analysis using remote sensing (Sahana et al., 2023), there is no standard methodology for the delineation of these complex transitional areas given their irregular and continuously evolving morphology (Mondal & Sen, 2020). Site selection involved a combination of Google earth images and ground-truthing, with the intention of identifying sites which would be representative of a mixture of land use types, typically found in such transitional areas in cities of developing countries. Points were randomly chosen on the Google image laterally along either sides of the Hooghly river and the Google Earth Pro software was used to create a 100 m (diameter) buffer area around each point and generate the study area maps. Urban landscape features such as proximity to roads, industry, high built-up were marked for each point (see ESM2). The 21 sites finally identified for the survey, during ground-truthing, had easy accessibility and involved a combination of 10 sites with atleast one urban element, 6 sites with two urban elements and 5 sites with none (see ESM1).
Plant documentation, collection, and identification
Within each site, a 1 m × 1 m quadrat was randomly placed four times (adjacent to each other) on patches of herbaceous vegetation and all species present in the quadrat were noted for estimating species richness. Cover percent, a surrogate for abundance, was visually recorded for each species per quadrat for better representation of the community dominance at each site. The floristic survey was conducted on ground surface vegetation including road verges. However, plants growing on vertical surfaces such as walls were not considered for the study. The sampling location together with site co-ordinates was recorded during the field visits using a handheld GPS (Garmin eTrex Vista). The names of plant species occurring at each sampling site have been listed along with the site co-ordinates and characteristics in ESM1.
Photographs of live plants were also taken at the time of field visits. Unknown plant samples (with evidence of flowering or fruiting, when available) were collected from quadrats at each site for identification purposes, pressed and herbarium vouchers deposited at the Central National Herbarium, Botanical Survey of India, Shibpur, Howrah for identification and authentication. All scientific names were cross-checked and confirmed using The Plant List (Version 1.1, 2013; accessed January 2018). Family, life forms, and habits were also checked from the literature and internet resources. Alien species were categorized into casual, naturalized, and invasive species following the criterion proposed by Blackburn et al. (2011) and Richardson and Pysek (2012). Species included in the 'invasive' list were only those for which there was strong evidence that they are aliens in India (Reddy et al., 2008; ENVIS). The complete list of species with common names, life forms, habitats and species status is presented in Table 1.
Data analysis
The percent frequency of occurrence (f) was summarized as the number of quadrats with at least one individual divided by the total number of sampled quadrats × 100. When pooled over sites, species presence in quadrats was reported as total numbers recorded in 84 quadrats (21 sites × 4 quadrats). Species were additionally classified into 4 classes in accordance with their percent frequency of presence in sites within the investigated area. They were considered as sparse species, if present in 5% of quadrats, occasional species if present between 6 and 15% of quadrats, common species if present between 16 and 50% of quadrats, and very common species when present in more than 50% of quadrats.
The likelihood ratio test was done between native and alien species groups to check for the difference in their mean value.
The cover data were further classified into four cover classes e.g., negligible species cover (< 10%); low species cover (10– < 30%); moderate species cover (30– < 70%) and high species cover (> 70%) in an attempt to explore the effect of invasive plants on native diversity and to understand the community dominants (Banerjee & Dewanji, 2017b; Bassett et al., 2012; Chatterjee & Dewanji, 2014).
The Q GIS software (version 3.30.0) was used to draw the study area map while graphs were generated using Microsoft Excel 2010.
Calculation of indices
Multiple similarity measure
Using plant species richness data, multiple similarity measure (Diserud & Ødegaard, 2007) was calculated to check for the similar species composition with the study area. Here, the identity of species shared across more than two sites is taken into consideration instead of calculating the average over a set of dependent pairwise similarities. The multiple site similarity measure is in the range of 0 – 1, with 1 indicating complete similarity.
Where
ai is the number of species in Ai.
T = Total number of sites.
ST = Sum of total number of species in site T.
Diversity and evenness study
Using abundance data, Shannon diversity index (H') (Shannon–weaver index 1949) and Pielou’s evenness measure (Pielou’s evenness 1969) was used to check the species bio-diversity of the studied area.
where
s is the number of species.
\(p_{i} \text{ is the proportion of individuals or the abundance of the } {i}{\text{th}}\) species and ln = \({\text {log base n}}\)
Values of diversity index usually lie between 1.5 and 3.5, the higher the value more diverse the site (Kent, 2011; Magurran, 2004).
Evenness measure (\(E\)) (Pielou’s evenness 1969) is given by.
Where
s is the number of species.
pi is the proportion of individuals or the abundance of the ith species expressed as a proportion of total cover.
ln = log base n.
The value of the index usually lies between 0 and 1. The higher the values of E, the more even the species and their distribution within the quadrat (Kent, 2011).
Results and discussion
Composition of plant species in study sites
The distribution of species in peri-urban regions of Greater Kolkata revealed a total of 62 species belonging to 55 genera and 29 different families. The enlisted species along with their botanical and common names, family, life forms, and invasion status are reported in Table 1. Tree species included in the list were those of a few saplings which occasionally appeared in some quadrats. The compositional distribution (including family, life forms, and stages of invasion) of all the species have also been represented in pie charts (Fig. 2). Poaceae and Compositae were the two families that dominated the species pool (Fig. 2a). While native species were mostly represented by Poaceae (Fig. 2b), Compositae, which is the largest dicotyledon family, harbored the highest number of aliens (Fig. 2c). Compositae has been reported to contribute to high numbers of alien species (naturalized and invasive), not only in India (Adhikari et al., 2015; Inderjit et al., 2018; Muzafar et al., 2019) but also worldwide (Pyšek et al., 2017, 2020b).
The purpose of denoting the specific life-forms (herb, shrub, climber, grass, etc.) and habits (annual, perennial, and evergreen) is important as it reflects a plant’s adaptation to its environment, its response toward disturbances (Arnold, 1955) and represents major life-history strategies within plant lineages (Miller et al., 2011). Most of the species (> 50%) growing in peri-urban areas were herbs (Fig. 2d), which are more likely to establish and spread rapidly because of shorter generation times (Inderjit et al., 2018). Among 1747 alien vascular plant species of India, the herbaceous growth form has been reported to be the predominant form (Pant et al., 2021). All plant species were broadly classified into natives and aliens (Fig. 2e). Both native (53%) and invasive (47%) species are almost equally distributed, among aliens the majority were invasive species (32%) as is evident from the figure (Fig. 2e).
Overall and species-specific distribution
The species were further categorized based on their overall frequency % (species numbers in each quadrat) and their distribution is plotted in Fig. 3. On the regional scale, the species pool was dominated by species belonging to the sparse category (~ 43.5%, n = 27/62) which probably indicates variable levels of anthropogenic stress in peri-urban areas. Eight species belonged to the common category while there were only two very common species (e.g., Alternanthera philoxeroides and Mikania micrantha) with > 50% occurrence. Most species assemblages are dominated by relatively sparse species (McGill, 2003) and their numbers vary from place to place because of natural variation in species abundance distribution, or variation in levels of anthropogenic stress (Midolo et al., 2019; Sheil, 2016).
Species-specific insights with respect to the frequency of native and invasive species present in sites, as reflected on a local scale, are provided in Fig. 4. A preponderance of sparse and occasional species is evident for both groups. Most natural and semi-natural communities have a common structure with few very abundant species, and many are only fairly frequent (Magurran, 2004). It is very evident from Fig. 4 that most of the native species (20 out of 33 species) occurred sparsely in sites (< 15%) which possibly indicates the constant shift in native species distribution in areas under the influence of increasing disturbance due to urbanization. Colocasia esculenta was the only species under the very common status (i.e., > 50% frequency), which is probably because of its wide spread cultivation outside the urban city core areas not only for its edibility (both leaves and corms are popularly consumed) but also for its traditional medicinal value (Kapoor et al., 2022; Pawar et al., 2018; Zubair et al., 2023). Among a large number of invasive plants (n = 20) found during the investigation, (Fig. 4), Mikania micrantha (90%) and Alternanthera philoxeroides (71%) were the two very common species, both of which are known to invade disturbed areas (Huang et al., 2015; Pathak et al., 2021).
Site-specific species distribution
Since species exist as communities in the spatial world, accounting for species richness or the number of interacting species in a community is of significance due to its role in maintaining ecosystem functioning (Wagg et al., 2019; Xiong et al., 2021). Site-specific species distribution revealed a substantial variation in total species richness from 4 to 25 (Table 2). The median values, which are more robust to outliers than the mean, were 11, 5, and 5 for total species richness, native species, and alien species numbers respectively. When the sites were judged for richness patterns based on the median values, 50% of sites showed total species richness ≥ 11 indicating a good number of species in the studied area. Suburban neighborhoods can have high heterogeneity between locations fostering different plant assemblages at small scales, consequently supporting many species at a site (de Barros Ruas et al., 2022; McKinney, 2008). Although most of the sites showed a good representation of families, but in the nine sites with species richness < 10, the phylogenetic diversity, in terms of number of families per site, varied from 3 to 7 while sites with higher number of species were represented by higher number of families (8–16) (Table 2). The presence of closely related species that are functionally similar and adapted to disturbances are generally associated with urbanization (Ricotta et al., 2009).
Both the native and alien species were more or less equally distributed in most sites and more than 50% of sites supported a greater number of species than the observed median value of 5. Among the aliens, the number of invasive species varied from 2 to 9 per site, with 80% of sites supporting three or more species, indicating the increasing presence of multiple plant invaders even in peri-urban sites (Table 2). Disturbance in terrestrial ecosystems, especially anthropogenic activities, enhance the diversity and distribution of alien plants; however, the strength of this enhancement relies on the disturbance type (Jauni et al., 2015). The Likelihood ratio test revealed no significant difference between mean numbers of native and alien species richness (s = 0.214; p = 0.643). Site 6, with highest number of species, both for natives (15) and aliens (10), appeared to be a developing area with moderate disturbance intensity (mix of agriculture, residential developments at different stages and remnant natural areas) which often show peaks in regional plant richness (Aronson et al., 2017; del Tredici, 2010; McKinney, 2008). At the local scale, environmental and resource heterogeneity may explain part of the variability in species richness (de Barros Ruas et al., 2022; Pausas & Austin, 2001) as observed in this study. Disturbance in urban cities may create opportunities for alien plant species colonization by removing native plants thereby affecting assembly processes which can play a significant role in the structure of plant communities. Escobedo et al. (2017) indicated how different responses to disturbance in terms of species richness in native and alien species led to a null response of total species richness along a disturbance gradient. Furthermore, it is interesting to note that 62% of the sites, in the investigated area, exhibited greater native species numbers even in presence of multiple invasive species at sites, thereby indicating the contribution of native species toward total species richness. This probably indicates that the independent rate of extinction of native species was not affected by the intensity or type of disturbance or the numbers of invasive species present in many of these peri-urban sites located at a distance from the urban core, and that these sites were still able to support native species possibly as part of remnant natural vegetation.
Co-occurrence of invasive species in the study area
The identity of the twenty invasive species found in the study area, their co-occurrence pattern and cover percent, as a measure of abundance, in sites are shown in Table 3. Species cover data could additionally provide insights into the less visible aspects of a community, such as competition and predation which could help to identify the processes responsible for community structure and dynamics (Bassett et al., 2012). Co-occurrence of M. micrantha and A. philoxeroides, the two most commonly occurring species, was observed in 67% (n = 14) of sites which shows their ability to maintain self-sustaining populations in peri-urban areas. While A. philoxeroides was present in 71% (n = 15) of sites, M. micrantha was the only invasive species to be present in 90% (n = 19) of sites (with nil or negligible presence in 3 sites). Among the two co-occurring invasive species, the contribution of M. micrantha to total invasive cover was maximum in 76% of sites while A. philoxeroides contributed minimally (< 10% of cover) in 90% of sites. The other invasive species did not contribute much to the total invasive cover in sites since they were generally present in amounts < 15%. The only exceptions were Site 1, where P. foetida contributed 42% to a total cover of 66% and Site 4 where A. conyzoides contributed 64% and O. corniculata 17% to the total invasive cover of 81%. M. micrantha, appeared to be the dominant invader, among all invasive species, since it could establish > 50% cover in one-third of the peri-urban sites which shows its potential to proliferate into adjoining areas with an increase in intensity and type of disturbance likely to be created by the continuing urbanization process.
Invasive species like C. odorata, I. cylindrica, L. camara and M. micrantha, reported in this study, have been listed among the 100 top worst weeds around the world (Lowe et al., 2000) while A. philoxeroides, A. conyzoides, C. odorata, L. camara, M. micrantha and P. hysterophorus are some of the plants that have already established themselves on the Indian subcontinent and are well known for their negative impacts (Shah et al., 2020; Shrestha et al., 2022; Thiney et al., 2019). Higher levels of disturbance at sites could probably impact their growth and proliferation, especially in sites with presence of M. micrantha at low or negligible covers. Urban landscapes are highly altered by development and human activities (Kranz et al., 2020; Naylo et al., 2019; Somerville et al., 2020) which is one of the prime reason for the increasing prevalence of alien plant species that are being reported from cities (Stajerova et al., 2017).
Impact of species cover on diversity
The pooled cover values, color coded to differentiate between the four (4) cover classes (negligible, low, moderate and high), for total native and alien (invasive and other) species at each site are reported in Table 4. Since invasive species have notable negative impacts on community structure, of special interest are the total cover values of native and invasive species for sites with moderate and high cover along with the name of the species contributing to maximum cover at a particular site. A large number of sites (81%) had negligible to low cover of native species while moderate to high cover of invasive species were evident in 85% of sites. Moreover, it was the combined cover of invasive species that largely contributed to the abundance of the species pool in most sites. There was only one site, Site 3, with negligible total invasive cover and a high cover (71%) of total native species (with Acmella paniculata contributing 30%, Commelina benghalensis 29% and four other natives contributing the rest). Among the invasive species, the contribution of M. micrantha to total invasive cover was maximum in sites with moderate and high cover of invasive species. The only exceptions were Sites 1 and 4 where other invasive species such as, Passiflora foetida and Ageratum conyzoides were among the dominant species. Although M. micrantha and A. philoxeroides were found to commonly occur in many sites (the corresponding site numbers shown in bold, Table 3), A. philoxeroides did not contribute much to the total invasive cover, unlike M. micrantha, in any site probably suggesting some competition between the two. The extremely fast growth and ability of both sexual and asexual reproduction helps M. micrantha to rapidly colonize disturbed habitats (Banerjee et al., 2017a, 2018) while competing with native vegetation and retarding their growth (Banerjee & Dewanji, 2017a). Studies also indicate a high climatic suitability for M. micrantha growth in some urban environments in India which adds to the risk of further spread of this plant into new areas (Banerjee et al. 2017b).
Patterns of species richness (number of species) along with indices of diversity and evenness (species distribution using number and abundance) of sites, can reflect the plant community composition which can provide important information on bio-diversity maintenance (Holl et al., 2022; Rolls et al., 2023). The multiple similarity index, a comprehensive single index for multiple sites, of 0.76 denotes overall similarity of the biotic community of the study area which points to some abundant species at sites reflecting the increasing cover of M. micrantha, the dominant invader, in different sites (Table 4). The Shannon diversity index values (in bold figures, Table 4) show 33% sites (7 out of 21) to be still diverse as per the bio-diversity range of values from 1.5 to 3.5 (Kent, 2011). The evenness index value revealed only 29% of the sites having ≥ 0.70 index value (Table 4) which clearly indicates the maximum peri-urban plots are still heterogeneous in the investigated areas of Greater Kolkata under the influence of urbanization. It is also interesting to note that all sites with a high total cover of invasive species (mostly > 60%) had low evenness values (E < 0.5, in bold figures, Table 4) which indicate that only a few invasive species dominate these sites, with maximum contribution of cover by M. micrantha. Evenness (E) values vary between 0 and 1.0, with 1.0 representing a situation in which all species are equally abundant (Magurran, 2004). Site 6, which supported the highest number of species (n = 25, Table 2) had a fairly high evenness value (E = 0.72) and was the most diverse as per the Shannon diversity index (H’ = 2.35) as well.
Conclusions
The high presence of M. micrantha and A. philoxeroides in peri-urban areas is clearly indicative of their increasing adaptability to cope with frequently disturbed habitats which directs attention to the need for regular monitoring so that site-specific management strategies could be initiated in areas where containment to restrict rapid growth may be necessary. Among the two, M. micratha was the dominant invader with a great potential to spread in view of its ability to maintain high cover in presence of other invasive species. Since cities harbor multiple invaders, as has also been observed in this study, the presence of more than one species with known negative impacts at a particular site could possibly create additional concerns, if not managed properly. This field study provides useful insights with potential implications for bio-diversity management by identifying a notorious invasive plant, Mikania micratha, for its potential to be one of the most likely successful invader in the near future, thereby prioritizing it accordingly for different management strategies in new and heterogeneous environments.
References
Adhikari, D., Tiwary, R., & Barik, S. K. (2015). Modelling hotspots for invasive alien plants in India. PLoS ONE, 10(7), e0134665.
Agrawal, S., & Narayan, R. (2017). Spatio-temporal organization and biomass dynamics of plant communities in a dry tropical peri-urban region: deterministic role of alien flora in anthropo-ecosystems. Current Science, 113, 53–62.
Aijaz, R. (2019). India’s peri-urban regions: The need for policy and the challenges of Governance. Observer Research Foundation, pp. 12
Allen, W. J., Wainer, R., Tylianakis, J. M., Barratt, B. I., Shadbolt, M. R., Waller, L. P., & Dickie, I. A. (2020). Community-level direct and indirect impacts of an invasive plant favour exotic over native species. Journal of Ecology, 108(6), 2499–2510.
Alue, B. A., Salleh Hudin, N., Mohamed, F., Mat Said, Z., & Ismail, K. (2022). Plant diversity along an urbanization gradient of a tropical city. Diversity, 14(12), 1024.
Appalasamy, M., & Ramdhani, S. (2020). Aliens in the city: Towards identifying non-indigenous floristic hotspots within an urban matrix. Flora, 269, 151631.
Arnold, J. F. (1955). Plant life-form classification and its use in evaluating range conditions and trend. Rangeland Ecology & Management/journal of Range Management Archives, 8(4), 176–181.
Aronson, M. F., Handel, S. N., La Puma, I. P., & Clemants, S. E. (2015). Urbanization promotes non-native woody species and diverse plant assemblages in the New York metropolitan region. Urban Ecosystems, 18, 31–45.
Aronson, M. F., Lepczyk, C. A., Evans, K. L., Goddard, M. A., Lerman, S. B., MacIvor, J. S., Nilon, C. H., & Vargo, T. (2017). Biodiversity in the city: Key challenges for urban green space management. Frontiers in Ecology and the Environment, 15(4), 189–196.
Avolio, M., Pataki, D. E., Jenerette, G. D., Pincetl, S., Clarke, L. W., Cavender-Bares, J., Gillespie, T. W., Hobbie, S. E., Larson, K. L., McCarthy, H. R., & Trammell, T. L. (2020). Urban plant diversity in Los Angeles, California: Species and functional type turnover in cultivated landscapes. Plants, People, Planet, 2(2), 144–156.
Banerjee, A. K., & Dewanji, A. (2017a). Role of intraspecific trait plasticity in Mikania micrantha Kunth growth and impact of its abundance on community composition. Journal of Asia-Pacific Biodiversity, 10(2), 237–249.
Banerjee, A. K., & Dewanji, A. (2017b). Native exotic relationships in plant communities: The role of exotic dominance in framing community composition. Ecological Research, 32(5), 653–665.
Banerjee, A. K., Ghosh, S., & Dewanji, A. (2017a). Do plants have a choice of traits to be modulated? Evidence from an invasive plant Mikania micrantha Kunth in different urban environments. American Journal of Plant Sciences, 8(4), 835–855.
Banerjee, A. K., Medda, C., Bhattacharya, S., & Dewanji, A. (2018). What matters most-role of environment, arrival order and population fitness in invaded community assembly. Acta Oecologica, 93, 56–64.
Banerjee, A. K., Mukherjee, A., & Dewanji, A. (2017b). Potential distribution of Mikania micrantha Kunth in India—evidence of climatic niche and biome shifts. Flora, 234, 215–223.
Bang, A., Cuthbert, R. N., Haubrock, P. J., Fernandez, R. D., Moodley, D., Diagne, C., Turbelin, A. J., Renault, D., Dalu, T., & Courchamp, F. (2022). Massive economic costs of biological invasions despite widespread knowledge gaps: A dual setback for India. Biological Invasions, 24(7), 2017–2039.
Bassett, I., Paynter, Q., Hankin, R., & Beggs, J. R. (2012). Characterising alligator weed (Alternanthera philoxeroides; Amaranthaceae) invasion at a northern New Zealand lake. New Zealand Journal of Ecology, 36, 216–222.
Blackburn, T. M., Pyšek, P., Bacher, S., Carlton, J. T., Duncan, R. P., Jarošík, V., Wilson, J. R., & Richardson, D. M. (2011). A proposed unified framework for biological invasions. Trends in Ecology & Evolution, 26(7), 333–339.
Calfapietra, C., Peñuelas, J., & Niinemets, Ü. (2015). Urban plant physiology: Adaptation-mitigation strategies under permanent stress. Trends in Plant Science, 20(2), 72–75.
Chakraborty, S., Maity, I., Patel, P. P., Dadashpoor, H., Pramanik, S., Follmann, A., Novotný, J., & Roy, U. (2021). Spatio-temporal patterns of urbanization in the Kolkata Urban Agglomeration: A dynamic spatial territory-based approach. Sustainable Cities and Society, 67, 102715.
Chatterjee, A., & Dewanji, A. (2014). Effect of varying Alternanthera philoxeroides (alligator weed) cover on the macrophyte species diversity of pond ecosystems: A quadrat-based study. Aquatic Invasions, 9(3), 343–355.
Chen, T. L., Chiu, H. W., & Lin, Y. F. (2020). How do east and southeast Asian cities differ from western cities? A systematic review of the urban form characteristics. Sustainability, 12(6), 2423.
Chen, W., Wang, G., & Zeng, J. (2023). Impact of urbanization on ecosystem health in Chinese urban agglomerations. Environmental Impact Assessment Review, 98, 106964.
Dadashpoor, H., & Ahani, S. (2021). Explaining objective forces, driving forces, and causal mechanisms affecting the formation and expansion of the peri-urban areas: A critical realism approach. Land Use Policy, 102, 105232.
Dasgupta, S., Gosain, A. K., Rao, S., Roy, S., & Sarraf, M. (2013). A megacity in a changing climate: The case of Kolkata. Climatic Change, 116(3), 747–766.
de Barros Ruas, R., Costa, L. M. S., & Bered, F. (2022). Urbanization driving changes in plant species and communities—A global view. Global Ecology and Conservation, 38, e02243.
Del Tredici, P. (2010). Spontaneous urban vegetation: Reflections of change in a globalized world. Nature and Culture, 5(3), 299–315.
Diserud, O. H., & Ødegaard, F. (2007). A multiple-site similarity measure. Biology Letters, 3(1), 20–22.
Early, R., Bradley, B. A., Dukes, J. S., Lawler, J. J., Olden, J. D., Blumenthal, D. M., Gonzalez, P., Grosholz, E. D., Ibañez, I., Miller, L. P., Sorte, C. J., & Tatem, A. J. (2016). Global threats from invasive alien species in the twenty-first century and national response capacities. Nature Communications, 7(1), 1–9.
Ellis, E. C. (2021). Land use and ecological change: A 12,000-year history. Annual Review of Environment and Resources, 46, 1–33.
English, J., & Wright, A. J. (2021). The effect of urban temperature gradients on grassland microclimate amelioration in Los Angeles, USA. Applied Vegetation Science, 24(1), e12556.
ENVIS Centre on Floral Diversity (http://bsienvis.nic.in/Database/Invasive_Alien_species_15896.aspx)
Escobedo, V. M., Rios, R. S., Salgado-Luarte, C., Stotz, G. C., & Gianoli, E. (2017). Disturbance by an endemic rodent in an arid shrubland is a habitat filter: Effects on plant invasion and taxonomical, functional and phylogenetic community structure. Annals of Botany, 119(4), 659–670.
Fahey, C., & Flory, S. L. (2022). Soil microbes alter competition between native and invasive plants. Journal of Ecology, 110(2), 404–414.
Ferreira, C., Kalantari, Z., & Pereira, P. (2021). Liveable cities: Current environmental challenges and paths to urban sustainability. Journal of Environmental Management, 277, 111458.
Follmann, A. (2022). Geographies of peri-urbanization in the global south. Geography Compass, 16(7), e12650.
Gao, J., & O’Neill, B. C. (2020). Mapping global urban land for the 21st century with data-driven simulations and shared socioeconomic pathways. Nature Communications, 11(1), 2302.
Gopal, D., von der Lippe, M., & Kowarik, I. (2019). Sacred sites, biodiversity and urbanization in an Indian megacity. Urban Ecosystems, 22(1), 161–172.
Guan, X., Shen, H., Li, X., Gan, W., & Zhang, L. (2019). A long-term and comprehensive assessment of the urbanization-induced impacts on vegetation net primary productivity. Science of the Total Environment, 669, 342–352.
Gupta, S., & Narayan, R. (2006). Species diversity in four contrasting sites in a peri-urban area in Indian dry tropics. Tropical Ecology, 47(2), 229–242.
Holl, K. D., Luong, J. C., & Brancalion, P. H. (2022). Overcoming biotic homogenization in ecological restoration. Trends in Ecology & Evolution. https://doi.org/10.1016/j.tree.2022.05.002
Huang, Q., Shen, Y., Li, X., Zhang, G., Huang, D., & Fan, Z. (2015). Regeneration capacity of the small clonal fragments of the invasive Mikania micrantha H.B.K.: Effects of the stolon thickness, internode length and presence of leaves. Weed Biology and Management, 15(2), 70–77.
Inderjit, Pergl, J., van Kleunen, M., Hejda, M., Babu, C. R., Majumdar, S., Singh P, Singh SP, Salamma S, Rao BR, & Pyšek, P. (2018). Naturalized alien flora of the Indian states: biogeographic patterns, taxonomic structure and drivers of species richness. Biological Invasions, 20, 1625–1638.
Jauni, M., Gripenberg, S., & Ramula, S. (2015). Non‐native plant species benefit from disturbance: a meta‐analysis. Oikos, 124(2), 122–129.
Jaureguiberry, P., Titeux, N., Wiemers, M., Bowler, D. E., Coscieme, L., Golden, A. S., Guerra, C. A., Jacob, U., Takahashi, Y., Settele, J., Díaz, S., & Purvis, A. (2022). The direct drivers of recent global anthropogenic biodiversity loss. Science Advances, 8(45), eabm9982.
Jha, P., Banerjee, S., Bhuyan, P., Sudarshan, M., & Dewanji, A. (2020). Elemental distribution in urban sediments of small waterbodies and its implications: A case study from Kolkata, India. Environmental Geochemistry and Health, 42, 461–482.
Jha, R. K., Nölke, N., Diwakara, B. N., Tewari, V. P., & Kleinn, C. (2019). Differences in tree species diversity along the rural-urban gradient in Bengaluru, India. Urban Forestry & Urban Greening, 46, 126464.
Kapoor, B., Singh, S., & Kumar, P. (2022). Taro (Colocasia esculenta): Zero wastage orphan food crop for food and nutritional security. South African Journal of Botany, 145, 157–169.
Kent, M. (2011). Vegetation description and data analysis: A practical approach. John Wiley & Sons.
Knight, T., Price, S., Bowler, D., Hookway, A., King, S., Konno, K., & Richter, R. L. (2021). How effective is ‘greening’of urban areas in reducing human exposure to ground-level ozone concentrations, UV exposure and the ‘urban heat island effect’? An updated systematic review. Environmental Evidence, 10(1), 1–38.
Kolkata Municipal Corporation https://www.kmcgov.in/KMCPortal/jsp/KolkataStatistics.jsp. Retrieved May 2016
Kranz, C. N., McLaughlin, R. A., Johnson, A., Miller, G., & Heitman, J. L. (2020). The effects of compost incorporation on soil physical properties in urban soils–A concise review. Journal of Environmental Management, 261, 110209.
Kuhn, I., Wolf, J., & Schneider, A. (2017). Is there an urban effect in alien plant invasions? Biological Invasions, 19(12), 3505–3513.
Liu, Y., Oduor, A. M., Zhang, Z., Manea, A., Tooth, I. M., Leishman, M. R., Xu, X., & Van Kleunen, M. (2017). Do invasive alien plants benefit more from global environmental change than native plants? Global Change Biology, 23(8), 3363–3370.
Lowe, S., Browne, M., Boudjelas, S., & De Poorter, M. (2000). 100 of the world’s worst invasive alien species: A selection from the global invasive species database (Vol. 12). Invasive Species Specialist Group.
Magurran, A. E. (2004). Measuring biological diversity. Blackwell Publishing.
Mazumder, S., Saha, J., Nandi, G., Naskar, M., Gayen, J., & Datta, D. (2021). Long-term monitoring of cropland transformation in Kolkata Metropolitan Area, India using open-source geospatial technologies. SN Applied Sciences, 3, 1–19.
McGill, B. J. (2003). Does mother nature really prefer rare species or are log-left-skewed SADs a sampling artefact? Ecology Letters, 6(8), 766–773.
McKinney, M. L. (2008). Effects of urbanization on species richness: A review of plants and animals. Urban Ecosystems, 11(2), 161–176.
Midolo, G., Alkemade, R., Schipper, A. M., Benítez-López, A., Perring, M. P., & De Vries, W. (2019). Impacts of nitrogen addition on plant species richness and abundance: A global meta-analysis. Global Ecology and Biogeography, 28(3), 398–413.
Miller, A. D., Roxburgh, S. H., & Shea, K. (2011). How frequency and intensity shape diversity–disturbance relationships. Proceedings of the National Academy of Sciences, 108(14), 5643–5648.
Mondal, B., Dolui, G., Pramanik, M., Maity, S., Biswas, S. S., & Pal, R. (2017). Urban expansion and wetland shrinkage estimation using a GIS-based model in the East Kolkata Wetland, India. Ecological Indicators, 83, 62–73.
Mondal, D., & Banerjee, A. (2021). Exploring peri-urban dynamism in India: Evidence from Kolkata metropolis. Journal of Urban Management, 10(4), 382–392.
Mondal, D., & Sen, S. (2020). Methodological dimensions of delineating peri-urban areas: The case of Kolkata metropolis. Environment and Urbanization ASIA, 11(2), 183–194.
Mortoja, M. G., & Yigitcanlar, T. (2022). Why is determining peri-urban area boundaries critical for sustainable urban development? Journal of Environmental Planning and Management, 66(1), 67–96.
Mungi, N. A., Kaushik, M., Mohanty, N. P., Rastogi, R., Antony Johnson, J., & Qureshi, Q. (2019). Identifying knowledge gaps in the research and management of invasive species in India. Biologia, 74, 623–629.
Muzafar, I., Khuroo, A. A., Mehraj, G., Hamid, M., Rashid, I., & Malik, A. H. (2019). Floristic diversity along the roadsides of an urban biodiversity hotspot in Indian Himalayas. Plant Biosystems-an International Journal Dealing with All Aspects of Plant Biology, 153(2), 222–230.
Narayani, A. R., & Nagalakshmi, R. (2023). Understanding urban sprawl trends in peri urban regions across global cities-survey of case studies. Cities & Health, 7(3), 492–504.
Nations, U. (2018). Revision of world urbanization prospects. United Nations: New York
Naylo, A., Almeida Pereira, S. I., Benidire, L., El Khalil, H., Castro, P. M., Ouvrard, S., Schwartz, C., & Boularbah, A. (2019). Trace and major element contents, microbial communities, and enzymatic activities of urban soils of Marrakech city along an anthropization gradient. Journal of Soils and Sediments, 19, 2153–2165.
Newbold, T., Hudson, L. N., Hill, S. L., Contu, S., Lysenko, I., Senior, R. A., Börger, L., Bennett, D. J., Choimes, A., Collen, B., Day, J., & Purvis, A. (2015). Global effects of land use on local terrestrial biodiversity. Nature, 520(7545), 45–50.
Pant, V., Patwardhan, C., Patil, K., Bhowmick, A. R., Mukherjee, A., & Banerjee, A. K. (2021). ILORA: A database of alien vascular flora of India. Ecological Solutions and Evidence, 2(4), e312105.
Pathak, H. N., Shrestha, B. B., Bhuju, D. R., & Subedi, D. S. (2021). Spatial distribution of invasive alien plants in Pokhara valley. Nepal. Pakistan Journal of Botany, 53(3), 1015–1024.
Pausas, J. G., & Austin, M. P. (2001). Patterns of plant species richness in relation to different environments: An appraisal. Journal of Vegetation Science, 12(2), 153–166.
Pawar, H. A., Choudhary, P. D., & Kamat, S. R. (2018). An overview of traditionally used herb, Colocasia esculenta, as a phytomedicine. Medicinal & Aromatic Plants, 7(02), 1–7.
Pyšek, P., Hejda, M., Čuda, J., Zambatis, G., Pyšková, K., MacFadyen, S., Storch, D., Tropek, R., & Foxcroft, L. C. (2020b). Into the great wide open: Do alien plants spread from rivers to dry savanna in the Kruger National Park? NeoBiota, 60, 61–77.
Pyšek, P., Hulme, P. E., Simberloff, D., Bacher, S., Blackburn, T. M., Carlton, J. T., Dawson, W., Essl, F., Foxcroft, L. C., Genovesi, P., Jeschke, J. M., & Richardson, D. M. (2020a). Scientists’ warning on invasive alien species. Biological Reviews, 95(6), 1511–1534.
Pyšek, P., Pergl, J., Essl, F., Lenzner, B., Dawson, W., Kreft, H., Weigelt, P., Winter, M., Kartesz, J., Nishino, M., Antonova, L. A., & Kleunen, M. V. (2017). Naturalized alien flora of the world. Preslia, 89(3), 203–274.
Reddy, C.S., Bagyanarayana, G., Reddy, K.N., & Raju, V.S. (2008). Invasive alien flora of India.
Richardson, D. M., & Pyšek, P. (2012). Naturalization of introduced plants: Ecological drivers of biogeographical patterns. New Phytologist, 196(2), 383–396.
Richardson, D. M., Pyšek, P., Rejmanek, M., Barbour, M. G., Panetta, F. D., & West, C. J. (2000). Naturalization and invasion of alien plants: concepts and definitions. Diversity and distributions, 6(2), 93–107.
Ricotta, C., La Sorte, F. A., Pyšek, P., Rapson, G. L., Celesti-Grapow, L., & Thompson, K. (2009). Phyloecology of urban alien floras. Journal of Ecology, 97(6), 1243–1251.
Rolls, R. J., Deane, D. C., Johnson, S. E., Heino, J., Anderson, M. J., & Ellingsen, K. E. (2023). Biotic homogenisation and differentiation as directional change in beta diversity: Synthesising driver–response relationships to develop conceptual models across ecosystems. Biological Reviews, 98, 388–1423.
Rozas-Vásquez, D., Spyra, M., Jorquera, F., Molina, S., & Caló, N. C. (2022). Ecosystem services supply from peri-urban landscapes and their contribution to the sustainable development goals: A global perspective. Land, 11(11), 2006.
Sahana, M., Hong, H., & Sajjad, H. (2018). Analyzing urban spatial patterns and trend of urban growth using urban sprawl matrix: A study on Kolkata urban agglomeration, India. Science of the Total Environment, 628, 1557–1566.
Sahana, M., Ravetz, J., Patel, P. P., Dadashpoor, H., & Follmann, A. (2023). Where Is the peri-urban? a systematic review of peri-urban research and approaches for its identification and demarcation worldwide. Remote Sensing, 15(5), 1316.
Sankaran, K. V., Khuroo, A. A., Raghavan, R., Molur, S., Kumar, B., Wong, L. J., & Pagad, S. (2021). Global register of introduced and invasive species—India. Version 1.5. Invasive Species Specialist Group ISSG. Checklist dataset. https://doi.org/10.15468/uvnf8m.Retrieved 24 Aug 2023
Seebens, H., Bacher, S., Blackburn, T. M., Capinha, C., Dawson, W., Dullinger, S., Genovesi, P., Hulme, P. E., van Kleunen, M., Kühn, I., Jeschke, J. M., & Essl, F. (2021). Projecting the continental accumulation of alien species through to 2050. Global Change Biology, 27(5), 970–982.
Seebens, H., Blackburn, T. M., Dyer, E. E., Genovesi, P., Hulme, P. E., Jeschke, J. M., Pagad, S., Pyšek, P., van Kleunen, M., Winter, M., Ansong, M., & Essl, F. (2018). Global rise in emerging alien species results from increased accessibility of new source pools. Proceedings of the National Academy of Sciences, 115(10), E2264–E2273.
Shah, K. K., Tiwari, I., Tripathi, S., Subedi, S., & Shrestha, J. (2020). Invasive alien plant species: A threat to biodiversity and agriculture in Nepal. Agriways, 8(1), 62–73.
Sharma, R., Chakraborty, A., & Joshi, P. K. (2015). Geospatial quantification and analysis of environmental changes in urbanizing city of Kolkata (India). Environmental Monitoring and Assessment, 187(1), 1–12.
Sharma, S., Joshi, P. K., & Fürst, C. (2022). Unravelling net primary productivity dynamics under urbanization and climate change in the western Himalaya. Ecological Indicators, 144, 109508.
Sheil, D. (2016). Disturbance and distributions: Avoiding exclusion in a warming world. Ecology and Society, 21(1), 10.
Shrestha, B. B., Witt, A. B., Shen, S., Khuroo, A. A., Shrestha, U. B., & Naqinezhad, A. (2022). Plant invasions in Asia. In Global plant invasions, pp. 89–127. Springer International Publishing.
Somerville, P. D., Farrell, C., May, P. B., & Livesley, S. J. (2020). Biochar and compost equally improve urban soil physical and biological properties and tree growth, with no added benefit in combination. Science of the Total Environment, 706, 135736.
Stajerova, K., Šmilauer, P., Brůna, J., & Pyšek, P. (2017). Distribution of invasive plants in urban environment is strongly spatially structured. Landscape Ecology, 32(3), 681–692.
Sun, L., Chen, J., Li, Q., & Huang, D. (2020). Dramatic uneven urbanization of large cities throughout the world in recent decades. Nature Communications, 11(1), 5366.
Thiébaut, G., Tarayre, M., & Rodríguez-Pérez, H. (2019). Allelopathic effects of native versus invasive plants on one major invader. Frontiers in Plant Science, 10, 854.
Thiney, U., Banterng, P., Gonkhamdee, S., & Katawatin, R. (2019). Distributions of alien invasive weeds under climate change scenarios in mountainous Bhutan. Agronomy, 9(8), 442.
Wagg, C., Schlaeppi, K., Banerjee, S., Kuramae, E. E., & van der Heijden, M. G. (2019). Fungal-bacterial diversity and microbiome complexity predict ecosystem functioning. Nature Communications, 10(1), 4841.
Wang, Y. J., Chen, D., Yan, R., Yu, F. H., & van Kleunen, M. (2019). Invasive alien clonal plants are competitively superior over co-occurring native clonal plants. Perspectives in Plant Ecology, Evolution and Systematics, 40, 125484.
We conserve PA www.conservationtools.org, Retrieved accessed on April 01, 2022
Weiskopf, S. R., Rubenstein, M. A., Crozier, L. G., Gaichas, S., Griffis, R., Halofsky, J. E., Hyde, K. J., Morelli, T. L., Morisette, J. T., Muñoz, R. C., Pershing, A. J., & Whyte, K. P. (2020). Climate change effects on biodiversity, ecosystems, ecosystem services, and natural resource management in the United States. Science of the Total Environment, 733, 137782.
Xiong, C., He, J. Z., Singh, B. K., Zhu, Y. G., Wang, J. T., Li, P. P., Zhang, Q. B., Han, L. L., Shen, J. P., Ge, A. H., Wu, C. F., & Zhang, L. M. (2021). Rare taxa maintain the stability of crop mycobiomes and ecosystem functions. Environmental Microbiology, 23(4), 1907–1924.
Yuan, L., Li, J. M., Yu, F. H., Oduor, A. M., & van Kleunen, M. (2021). Allelopathic and competitive interactions between native and alien plants. Biological Invasions, 23(10), 3077–3090.
Zhang, L., Yang, L., Zohner, C. M., Crowther, T. W., Li, M., Shen, F., Guo, M., Qin, J., Yao, L., & Zhou, C. (2022). Direct and indirect impacts of urbanization on vegetation growth across the world’s cities. Science Advances, 8(27), eabo0095.
Zhang, Z., Zhou, F., Pan, X., van Kleunen, M., Liu, M., & Li, B. (2019). Evolution of increased intraspecific competitive ability following introduction: The importance of relatedness among genotypes. Journal of Ecology, 107(1), 387–395.
Zubair, M. W., Imran, A., Islam, F., Afzaal, M., Saeed, F., Zahra, S. M., Akhtar, M. N., Noman, M., Ateeq, H., Aslam, M. A., Mehta, S., & Awuchi, C. G. (2023). Functional profile and encapsulating properties of Colocasia esculenta (Taro). Food Science & Nutrition, 11, 2440–2449.
Acknowledgements
The authors want to thank the Indian Statistical Institute (ISI), Kolkata, India for funding this research; Central National Herbarium, Botanical Survey of India, Shibpur, Howrah for identification and authentication of the plant species. We are grateful to Prof. Anup Dewanji, Applied Statistics Unit, Indian Statistical Institute, for his valuable statistical suggestions. We are also thankful to Mrs. Arpita Ganguly for helping us in making the route maps of the Greater Kolkata field. Mr. Sandip Chatterjee and Mr. Susant Mahankur, AERU, ISI are acknowledged for technical assistance in the field.
Funding
Indian Statistical Institute (ISI), Kolkata, India for funding this research.
Author information
Authors and Affiliations
Contributions
AD and SC developed the idea and designed the study. SC collected the data, identified and analyze. AD and SC both wrote the paper and gave final approval for publication.
Corresponding author
Ethics declarations
Conflict of interests
The authors declare that there is no conflict of interests.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chatterjee, S., Dewanji, A. The distribution of invasive alien plant species in peri-urban areas: a case study from the city of Kolkata. COMMUNITY ECOLOGY 25, 29–44 (2024). https://doi.org/10.1007/s42974-023-00169-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42974-023-00169-z