Abstract
Variation in bird community composition across habitats may be reflected by changes in species’ ecological characteristics. By their comparison between habitats, we can learn information about the factors underlying these changes. With this purpose, we used data from a nation-wide breeding bird monitoring scheme surveying birds in 15 habitat types sorted into four broad categories (forests, open, urban, and humid habitats) in a central European country, Czechia. We considered life-history strategy, migration distance, climatic niche position, European rarity, and diet niche as species’ ecological characteristics and compared their mean values across the habitat types. Although habitat type explained relatively low proportion of variability in these characteristics indicating that birds widely overlap in their habitat use, we observed significant differences in ecological characteristics between broad habitat categories, as well as between habitat types within a given category. For example, urban habitats hosted species with generally lower degree of insectivory than forest habitats. Within forests, coniferous stands hosted species with colder climatic niche than deciduous stands. The greatest differences were observed among humid habitat types: species recorded in water bodies were rarer in Europe and had slower life-history strategies than species recoded in running water. Within the open habitat category, mining areas were the most specific habitat with long-migrating and warm-dwelling species. The observed patterns can be driven by various factors including habitat-specific selection pressures, biogeographic constraints and human-induced habitat changes. On their basis, we discuss our findings.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The concept of habitat plays an essential role in ecological research (Chapman & Reich, 2007; Stirnemann et al., 2014). It facilitates our understanding of the division of space and energy among species (Storch & Okie, 2019) and helps to explain spatial variability in species abundances (Brown et al., 1995). According to this concept, species differ in their habitat associations resulting in variation of community composition across habitats (Crosby et al., 2019). At the same time, ecological factors acting in different habitats select for specific values of ecological traits carried by the species sharing the same habitat preference (Cody, 1981). These species-specific trait values can be averaged across species present in a given habitat to reveal the traits characterizing the ecology of local communities (e.g., Hořák et al., 2015). Whereas the variation of community-level metrics across habitats has frequently been studied in terms of various diversity indices (e.g., Korňan, 2004) and facets such as functional and phylogenetic diversity (Morelli et al., 2018), the species’ ecological characteristics were less frequently explored in different habitats (but see, e.g., Barbaro & Van Helder, 2009; Barnagaud et al., 2013; Montaño‐Centellas et al., 2021). Since the ecological characteristics reflect the effects of different environmental filters (Webb et al., 2010), knowledge of their habitat-specific mean values can elucidate the contributions of different drivers to the community assembly.
Previous studies exploring spatial variation of bird community structure in central European landscape revealed that four habitat categories, namely forest, open habitat, urban habitat, and wetland, account for the largest part of variability in bird community composition in a central European country, Czechia (Reif et al., 2008; Storch & Kotecký, 1999). Here we recognize 15 habitat types within these broad categories and investigate the changes in bird community composition across different habitats in relation to the characteristics that are strongly linked to bird species’ ecology: life-history strategy, migration distance, climatic niche position, European rarity, and diet niche.
According to the life-history strategy, species can be clustered along several axes, but the strongest gradient sorts the species from those with slow strategies—investing into their survival, having long generation time, low number of offspring, and long parental care (so-called K-selected species), to those with fast strategies—investing into their reproduction, having short generations, high number of offspring, and short parental care (so-called r-selected species) (Reznick et al., 2016). Migration is one of the most conspicuous phenomena in bird ecology being connected with species’ flexibility in responses to phenological changes—the longer the migration distance, the less flexible species (Koleček et al., 2020); and can be lost or gained in response to large changes in environmental pressures—loss of migration due to winter climate amelioration, gain of migration due to colonization of new areas at the leading range edges (Berthold, 2001). Climatic niche position mirrors the climatic conditions prevailing in species geographic ranges sorting the species along a temperature gradient from the cold-adapted ones breeding in polar regions or mountains to warm-adapted species breeding in the southern regions or lowlands (Jiguet et al., 2010). European rarity is based on species’ breeding population size in European continent which is underpinned by species’ dispersal ability, niche breadth, and resource availability when species adapted to the common resources are more common (Gregory & Gaston, 2000). Diet is the only kind of energy input birds accept, and food availability is thus essential for their existence from individuals to populations (Gill, 2006). At the same time, food resources are diverse and different species adapted to their various types resulting in multidimensional nature of birds’ diet niche (Pigot et al., 2020).
Although previous studies described relationships of some these characteristics to bird-habitat preferences, such as the association with forest habitat for cold-dwelling species (Barnagaud et al., 2013) or faster life-history strategy of birds breeding in more seasonal habitats (Hořák et al., 2015), we are not aware of any studies focusing simultaneously on a wider set of ecological characteristics across many different habitats at the national level. To fill this knowledge gap, we perform here an exploratory analyses using data from mapping of bird occurrences in 15 habitat types collected within a nationwide citizen science bird monitoring project in Czechia. Specifically, we aim to explore how values of different characteristics vary across respective habitats.
Material and methods
Bird occurrence mapping
Birds were mapped within Breeding Bird Survey Czechia (Liniové sčítání druhů—LSD), a national breeding bird monitoring scheme launched in 2018. Within LSD, skilled voluntary fieldworkers record positions of individual birds into a map using a custom-designed application for smartphones and tablets. Fieldworkers must be able to recognize all regionally occurring bird species by both acoustic and visual detections. Localities included in LSD are selected by a stratified random sampling with a stratum corresponding to the observers’ availability. In this respect, a square of 2.8 km × 3 km is chosen randomly within a radius of 10 km around a point indicated by the observer. Within the randomly selected square, the observer establishes two linear 1-km long transects situated at least 0.5 km from each other and 0.25 km from the square boundaries. Birds are mapped twice per breeding season: from 15th April to 10th May and from 15th May to 10th June to record both early and late breeders; the mapping dates correspond to the phenology patterns reported for most of the species in national literature (Kloubec & Čapek, 2012). During each early morning visit (from sunrise to 10 a.m.), the observer walks along the transects and records positions of all birds into an aerial photograph recognizing the type of detection (visual or acoustic) and the type of behavior (territorial or other) for each bird. Specific registrations are devoted to birds flying over the transect, flocks, breeding colonies, nests, and families with fledglings. The sampling effort is set to exactly one hour for one visit at one transect.
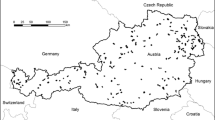
For purposes of this study, we used all records up to 100 m of the perpendicular distance from the transects (i.e., within a 200-m wide belt), but excluded birds flying over the transects. We used the data from 2018 supplemented by the data from the transects established in 2019 (n = 206 transects in total, Fig. 1). For further analysis, we had dataset of 151 species (Table S1).
Location of study sites and the distribution of the four main habitat categories in Czechia. Blue triangles—study sites used for bird counts (each represented by a square of 2.8 km × 3 km containing two 1-km transects along that the birds were counted); green areas—forests, brown areas—open habitats, red areas—urban habitats, blue areas—humid habitats
Species’ ecological characteristics
For each species, we collected literature information on the following five ecological characteristics (Table S1). Life-history strategy was taken as a species’ position along the main ordination axis obtained by PCA on six life-history traits (egg mass, clutch size, laying date, number of broods per season, incubation time, and body mass) ran by Koleček and Reif (2011). The axis sorted the species from the slow, so-called K-selected species (large eggs, large body mass, long incubation time, and small clutches), to the fast, so-called r-selected species (small eggs, small body mass, short incubation time, and large clutches). Migration distance was taken from Hanzelka et al. (2019) who measured the distance between centroids of species’ breeding and non-breeding ranges based on maps taken from BirdLife International and Nature Serve (2014). Climatic niche position was extracted from Reif et al. (2016) as a mean temperature in species’ European breeding range in the peak breeding season (April–June). European rarity was quantified as the minus logarithm of the species’ relative European breeding population size. Relative European breeding population size of a given species was calculated as ratio of species’ breeding abundance in Europe to the total European breeding population size of all focal bird species based on the data from European Red List of Birds (BirdLife International, 2015). Diet niche was expressed using two composite variables obtained by running a principal component analysis (PCA) on nine diet types recognized by Storchová and Hořák (2018) for European bird species. PCA revealed two most important gradients (Fig. S1) explaining together 61.0% of variability in species’ diet: pc1 describing increasing insectivory, and pc2 depicting a gradient from carnivorous to granivorous diet. Positions of the species along these two axes (Table S1) were taken for further analysis.
Habitat data
Habitat data come from the consolidated layer of ecosystems (CLE). CLE is a compilation of datasets on vegetation mapping, land cover and topography originating from a nationwide habitat mapping conducted between 2001 and 2005 with regular updates until 2018 (Hönigová & Chobot, 2014). CLE has a complete coverage of the country’s territory into 39 non-overlapping habitat types at the scale of 1:10,000. For purposes of this study, we merged these habitat types into 15 habitat types that can be sorted into four broader habitat classes: forests—(1) coniferous forest, (2) mixed forest (forest stand containing both coniferous and deciduous trees), (3) deciduous forest; open habitats—(4) shrubland, (5) vegetation along roads, (6) cropland (arable field with annual crops), (7) grassland (meadow, pasture), (8) mining area (opencast mine, unreclaimed slag heap), (9) rock; urban habitats—(10) urban green area (park, orchard, garden), (11) part of human settlement with sparse built-up area (city margin, village), (12) part of human settlement with continuous built-up area (city center, housing estate); humid habitats—(13) water body (fishpond, water reservoir), (14) running water (stream, river), (15) wetland (reedbed, swamp, peat bog). By that means, we created a map of non-overlapping polygons of these 15 habitats covering the whole country.
We overlaid LSD bird records and the habitat information based on CLE by restricting both datasets to the same 100 m perpendicular distance from the LSD transects obtaining the non-overlapping habitat polygons with bird records. For further analysis, we only considered polygons hosting at least two species and being larger than 100 m2 at the same time because smaller areas hold only small fragments of bird territories and are thus not appropriate for studying bird communities. As a result, we obtained 2215 polygons in total sorted into focal habitat types and categories (Table 1).
For each polygon, we expressed the list of detected bird species (Table S2) and habitat identity (Table S3). Each polygon-specific species list was considered being a bird community. Then we calculated mean values for each of the ecological characteristics across the species in the bird community of a given polygon using the species-specific trait values (see above). The calculation followed the approach of Devictor et al. (2008), who obtained the community-level average by weighting the species-specific values of a given ecological characteristic by the relative abundance of each species within a given bird community. As a result, we obtained the values of respective characteristics for every habitat polygon (Table S3).
Statistical analysis
The analyses were performed at two different levels—species level with species as replicates, and community level where the statistical units were individual habitat polygons.
For the species-level analysis, we first quantified relationships of each species to respective habitats using canonical correspondence analysis (CCA). CCA is a direct gradient analysis technique that relates species to environmental variables producing the independent gradients in species composition with respect to changes in values of environmental variables. Counts of bird species in respective habitat polygons represented the response variables (Table S2) and the habitat type (15 habitats) of polygons (Table S3) was the explanatory variable. We considered four most important habitat gradients represented by the first four canonical axes (cca1–cca4) explaining the decisive part of variability in bird–habitat relationships. Positions of individual species along these gradients were taken for further analysis. In the next step, we related species’ characteristics as response variables (each variable was included in one model) to species’ positions along cca1-cca4 as explanatory variables (all four axes were included together in every model) using linear models (Gaussian distribution, identity link).
At the community level, we related the ecological characteristics as respective response variables to the habitat type as a factorial explanatory variable using linear mixed models (Gaussian distribution, identity link). We also included species richness of individual habitat polygons as an additional covariate. To account for possible non-independence of the habitat polygons within the same transects, we used the square (each containing two 1-km transects, see above) as a random effect. The square was nested within the large square (12 × 11.1 km) to control for a possible large-scale spatial structure in data. Model performance was expressed using pseudo-R-squared. We checked the model assumptions by plotting residuals vs. fitted values for every model (Figs. S2, S3). Moreover, we tested for the presence of spatial autocorrelation in residuals of the models used in the community-level analysis (Fig. S4). No indication of spatial autocorrelation was observed in model residuals; only the smallest distances showed slightly negative values in some models (Fig. S4).
Statistical analysis was performed in R version 3.4.3 (R Core Team, 2017) using the packages ‘nlme’ (Pinheiro, 2021) for running mixed effects models, ‘MuMIn’ (Bartoń, 2020) for producing the pseudo-R-squared values for individual mixed effects models, and ‘ncf’ (Bjornstad & Cai, 2020) for testing the spatial autocorrelation. Multivariate analyses (PCA, CCA) were ran in Canoco for Windows 4.5 (ter Braak & Šmilauer, 2002).
Results
Species level
CCA sorted the species along four most important habitat gradients together explaining 86.2% of variability in species occurrence in respect of the 15 habitat types considered (Fig. 2, Table 1, Supplementary Table S1). First axis (explaining 42.8% of variability in relationships of species to the focal habitats (Fig. 2A)) was a gradient from forest (with species such as Crested Tit Lophophanes cristatus, Coal Tit Periparus ater, Eurasian Treecreeper Certhia familiaris) to urban habitats (House Sparrow Passer domesticus, House Martin Delichon urbicum, Collared Dove Streptopelia decaocto). Second axis (28.7%, Fig. 2A) represented a gradient from urban habitats with the species mentioned above to open habitats (cropland, grassland, mining areas and rocks) with typical species including Northern Wheatear (Oenanthe oenanthe), Yellow Wagtail (Motacilla flava), Eurasian Skylark (Alauda arvensis), and Meadow Pipit (Anthus pratensis). Third axis (9.9%, Fig. 2B) was a gradient of increasing wetness represented by water body, wetland, and (to lesser extend) by running water with typical species including Common Tern (Sterna hirundo), Common Pochard (Aythya ferina), and Red-crested Pochard (Netta rufina). Fourth axis (4.8%, Fig. 2B) was a gradient from grassland to mining areas, rocks, and coniferous forest. Species associations with this axis were modest as also indicated by low proportion of variability explained, but Tawny Pipit (Anthus campestris), Sand Martin (Riparia riparia), and Little Bittern (Ixobrychus minutus) showed conspicuous positive correlations (Fig. 2B).
Results of the canonical correspondence analysis (CCA) expressing relationships of bird species to 15 habitat types using the first four most important canonical axes (A: cca1 and cca2, B: cca3 and cca4) representing the main habitat gradients. Note that only the habitats (red arrows and labels) and species (blue triangles, black abbreviated names) showing the strongest relationships to respective axes are depicted. See Table 1 for full names of habitat types and their loadings, see Table S1 for full names of species and their scores along respective axes
By relating species-specific values of the ecological characteristics to the positions of individual species along these gradients (Table 2) we revealed that species with slower life-history strategies occurred in more open (cca2, Fig. 3A) and in wetter habitats (ccc3. Figure 3B). Moreover, a relationship to cca4 (Fig. 3C) indicates that species with slower life histories occurred in rocks, mining areas, and coniferous forests, while species with faster strategies were recorded in grasslands. Migration for longer distances was positively related to cca2 (Fig. 3D), indicating that species of open habitats migrate for longer distances, where species of urban habitats migrate shorter. Migration for longer distance was also characteristic for species with preference for wetter habitat (cca3, Fig. 3E). Considering European rarity, rarer species occurred in forests and commoner species in urban habitats (cca1, Fig. 3F). Moreover, increasing rarity was also associated with more open habitats (cca2, Fig. 3G), wetter habitats (cca3, Fig. 3H), and grassland (cca4, Fig. 3I). Climatic niche and descriptors of species’ diet niche did not indicate significant relationships to their habitat preferences (Table 2).
Significant relationships between species-specific values of ecological characteristics and positions of individual species along habitat gradients (cca1–cca4, see Fig. 2) estimated by linear models (see Table 2 for overall model fit): A life-history strategy and cca2 (t = − 4.50, p < 0.001), B life-history strategy and cca3 (t = − 5.79, p < 0.001), C life-history strategy and cca4 (t = 4.30, p < 0.001), D migration distance and cca2 (t = 3.18, p = 0.002), E migration distance and cca3 (t = 2.07, p = 0.040), F European rarity and cca1 (t = − 2.83, p = 0.005), G European rarity and cca2 (t = 4.86, p < 0.001), H European rarity and cca3 (t = 6.91, p < 0.001), I European rarity and cca4 (t = − 3.08, p = 0.002). Relationships are plotted as residual plots controlling for the effects of other variables included in respective models
Community level
Explanatory variables explained small (5–18%), but significant part of variability in values of ecological characteristics across polygons (Table 3). Whereas the polygon habitat type was significant in all models, species richness of individual polygons was only related to life-history strategy and European rarity (Table 3).
Species with faster life-history strategies occupied polygons dominated by woody plant vegetation (Fig. 4A): all three forest types, shrubland, and vegetation along roads. Moreover, these species were in parts of human settlements with sparse built-up areas. Slower life-history was characteristic in polygons with cropland, rocks, urban green areas, running water, grassland, mining areas, and parts of human settlements with continuous built-up areas, although the difference was not significant in the last three habitat types (Fig. 4A). Species with much slower life-history strategies were recorded in wetland and the slowest strategies were observed in water bodies (Fig. 4A).
Predicted values of respective species’ ecological characteristics in individual habitat types estimated by linear mixed models (see Table 3): A life-history strategy, B migration distance, C climatic niche position, D European rarity, E diet pc1, F diet pc2. Significant differences between habitats are marked by different letters
Bird communities containing the species with the longest migration distances were in polygons of mining areas, water bodies, rocks, and wetlands, but the migration distance of the species in the latter two habitats did not differ significantly from the other habitat types (Fig. 4B). In contrast, species occupying coniferous forest, deciduous forest, vegetation along roads, parts of human settlements with continuous built-up areas, and running water had the shortest migration distances (Fig. 4B). Bird communities of the other habitat types can be considered as those with migration distance being intermediate between the above-mentioned habitat groups (Fig. 4B), but the differences were insignificant with the exception of grassland that showed the intermediate position significantly (Fig. 4B).
In respect of the climatic niche position, species breeding in the coldest climate of Europe were those recorded in coniferous forest (Fig. 4C), whereas the species recorded in mining areas were those breeding in the warmest climate. Preference for warm climate had also species recorded in all types of urban areas, whereas relatively colder-dwelling species were observed in grassland (Fig. 4C). Remaining habitat types showed considerable overlap in respect of climatic niche values of bird communities (Fig. 4C).
Wetlands and water bodies were occupied by species being the rarest in Europe (Fig. 2D). Species with relatively high European rarity values (but being significantly lower than in the above-mentioned habitats) were recorded in cropland, mining, and rock areas and in running water (Fig. 4D). On the other hand, all types of forests, polygons situated in urban areas, along roads, in shrubland, and in grassland were characterized by bird communities with the lowest rarity values (Fig. 4D). However, within this group of habitats, shrubland, grassland, and parts of human settlements with sparse built-up areas hosted relatively rarer species (Fig. 4D).
Diet expressed as pc1 reflecting the degree of insectivory showed that this trait is characteristic for bird communities of all forest types, shrubland, cropland, grassland, and wetland (Fig. 4E). In contrast, birds in vegetation along roads, all urban habitat types, running water polygons, and water bodies were less insectivorous (Fig. 4E). Diet expressed as pc2 (carnivory vs. granivory) did not show much differences among habitats (Fig. 4F). Continuous built-up urban areas and urban green areas hosted more granivorous species, whereas coniferous and deciduous forest and mining areas are more carnivorous species (Fig. 4F).
Discussion
Our dataset was based on bird occurrences in habitat polygons represented by 15 habitat types from four broad habitat categories (forests, open habitats, urban habitats, and humid habitats). Across these habitats, we compared various ecological characteristics of birds (life-history strategy, migration distance, climatic niche position, European rarity, and diet niche) by the analyses performed both at the species and the community level. Habitat type explained relatively low proportion of variability in these characteristics because birds are relatively large and mobile organisms that often use a wider spectrum of habitats in contrast to more specialized (e.g., insects) or sedentary (e.g., plants) taxa (Roth et al., 2014; Thomas, 1995). Despite their relative habitat generalism, we found that birds clustered along several main habitat gradients that corresponded to the bird–habitat associations previously reported from Central Europe (Reif et al., 2008; Storch & Kotecký, 1999) and to our a priori defined broad habitat categories. According to the species-level analysis, species’ positions along these gradients were related to life-history strategy, migration distance, and European rarity, but unrelated to climatic niche position and diet. Community-level analysis provided additional insights by detecting differences in several characteristics across the 15 habitat types both within and between the broad habitat categories. Below we interpret the main findings.
The most conspicuous pattern in respect of species’ life-history strategy was the association of slow strategies with humid habitats which particularly concerned water bodies and wetlands. The pattern was driven by frequent occurrence of numerous large-bodied water birds such as waterfowl (e.g., Mute Swan Cygnus olor), herons (e.g., Gray Heron Ardea cinerea), and gulls (e.g., Caspian Gull Larus cachinans) with these habitats. In the case of water bodies that were typically represented by fishponds in our dataset, this association was facilitated by high stocks of Carp (Cyprus carpio) (Pechar, 2000) providing superabundant food resource for the large-bodied fish eaters (gulls, herons, Great Cormorant Phalacrocorax carbo). At the opposite end of life-history continuum, we found an interesting association of fast life-history strategies with habitats dominated by wood plants. They were represented not only by various forest types, but also by shrubland and vegetation along roads. Faster life-history strategy of species in those habitats may be partly driven by cavity nesting passerines that have large clutches (Jetz et al., 2008) due to investment in the current breeding attempt after finding a suitable nest site which may be scarce (Martin & Li, 1992). Another possibility may be small body size as an adaptation to maneuverability in dense woody vegetation (Norberg, 1979, 1995).
Species-level analysis showed an association of long-distance migration with open and humid habitats, but the community-level analysis uncovered that these patterns were driven by particularly long migration distances in species recorded in mining areas and water bodies. Mining areas were occupied mainly by species such as Tawny Pipit, Sand Martin, and Northern Wheatear that all winter in sub-Saharan Africa. Their adaptation to long-distance migration likely results from their insectivorous diet that is absent in their breeding habitat during winter. This explanation is less obvious for birds recorded in water bodies that show higher interspecific variability in diet and the winter food limitation is undoubtedly less strict in herbivores and fish eaters. However, we should bear in mind that these species often breed in northern Europe and migrate further south in non-breeding season (Keller et al., 2020) which makes the mean migration distance across the species recorded in water bodies relatively long even though many of them do not migrate to sub-Saharan Africa. Short migration distances were found in species recorded in various different habitats including built-up areas of human settlements and forests. The interpretation of these results is less clear. In the former case, it is well known that birds may benefit from the availability of food resources throughout the year in human settlements represented by various garbage the birds can feed on (Bonnet-Lebrun et al., 2020) as well as by targeted feeding by humans (Robb et al., 2008) even though important exceptions, such as urbanized aerial feeders, exist.
The differences in climatic niche position among habitats were modest which may raise concerns about biological importance of these statistically significant results. However, it is widely documented that even small changes in climatic niches have serious ecological consequences (e.g., Devictor et al., 2012) and the patterns we report here have genuine interpretations indeed. The lowest climatic niche position had the species recorded in coniferous forest. This forest type prevails in boreal zone of Europe and in higher altitudes, and it is thus not surprising that those species prefer colder climate (Barnagaud et al., 2013). Relatively northerly breeding ranges may also explain lower climatic niche positions in species of mixed forest and water bodies or wetlands (Keller et al., 2020). Community-level analysis uncovered a higher position of climatic niche in species recorded in urban areas. It may be explained by the southern origin of urbanization in some of that species such as European Serin (Serinus serinus), Black Redstart (Phoenicurs ochruros), or Eurasian Collared Dover (Streptopelia decaocto) which subsequently spread northward (Kinzelbach, 2004; Rocha-Camarero & De Trucios, 2002). In addition, urban habitat may act as a heat island (Taha, 1997) and thus may attract such warm-adapted species. The highest climatic niche position was found in species recorded in mining areas. They resemble xeric steppe or semi-desert habitats of southern Europe, so they host several species with a warmer climatic preference such as Tawny Pipit, Corn Bunting (Emberiza calandra), or European Stonechat (Saxicola rubicola).
European rarity was the characteristic showing considerable variation across habitats. The rarest species were recorded in water bodies and wetlands. This may be connected with reduced regional availability of these habitats because they account only for 1.5% of area in Czechia and 7.3% in Europe (Čížková et al., 2013). As a consequence, species associated with rare habitats can be regionally less common than species associated with more widespread habitats in Europe (Gregory & Gaston, 2000). In addition, large body size of water birds can also contribute to European rarity of species recorded in water bodies and wetlands since abundance is inversely linked with body mass following the metabolic scaling laws (Nee et al., 1991). On the other hand, species recorded in forest habitats and various types of human settlements were those more common in Europe. The pattern found for forests is more difficult to interpret, but we suggest that it may be linked to generally lower habitat specialization of European forest birds (Reif et al., 2016) when less specialized species are more common at the same time (Gaston et al., 1997). On the other hand, open habitats showed generally higher rarity values, but interesting differences were observed between different open habitat types. Specifically, species recorded in mining areas and cropland were more rare than species recorded in shrubland and grassland. As noted above, mining areas resemble steppe and semi-desert habitats that are confined to southern Europe and extend further east to Asia. Due to these biogeographic constrains their bird assemblage is composed by species being relatively rare in the whole-European context (Blondel, 1997). A similar explanation may apply to cropland, but in this case we can also expect an influence of habitat deterioration. Widely reported negative impacts of agricultural intensification decrease the quality of this habitat for birds (Stoate et al., 2009). At the same time, these limitations are most likely less pronounced in shrubland and grassland.
In contrast to European rarity, changes in bird community composition across the focal habitats were only weakly related to species’ diet niche showing no significant relationships in the species-level analysis and only a few patterns at the community level. We found lower degree of insectivory in both running water and water bodies, all types of urban habitats and in vegetation along roads. In the former case, lower degree of insectivory may be driven by occurrence of numerous fish eaters in both types of water habitats. In addition to the large-bodied waterbirds mentioned above, it is for example Common Kingfisher (Alcedo atthis) which breedings along rivers and small stream. Interestingly, wetlands showed higher degree of insectivory than water bodies, probably due to occurrence of various warbler species (Acrocephalus sp., Locustella sp.) and shorebirds (e.g., Northern Lapwing, Vanellus vanellus) in this habitat. Lower degree of insectivory of species recorded in human-modified habitats, most notably the urban ones, may result from their flexibility in resource use (Ducatez et al., 2015) than is linked to bird occurrence in human settlements (Evans et al., 2011; Møller, 2009). At the same time, our results indicate that urban continuous built-up areas and green areas were characterized by a higher degree of granivory. In those areas, see-eaters may benefit from feeding at diary and poultry farms (Havlíček et al., 2021) or bird feeders (Robb et al., 2008).
In conclusion, the patterns revealed in our data are ecologically meaningful and provide further insights into the factors that govern bird community composition across habitats in Central European landscape. In would be interesting to repeat these analyses using datasets from different European regions, for instance, boreal zone or the Mediterranean region, to show to what extend the patterns we report here hold in different landscape and biogeographical contexts.
Data availability
Supplementary Tables S1–S3.
Code availability
Supplementary file S5.
References
Barbaro, L., & van Halder, I. (2009). Linking bird, carabid beetle and butterfly life-history traits to habitat fragmentation in mosaic landscapes. Ecography, 32, 321–333.
Barnagaud, J. Y., Barbaro, L., Hampe, A., Jiguet, F., & Archaux, F. (2013). Species’ thermal preferences affect forest bird communities along landscape and local scale habitat gradients. Ecography, 36, 1218–1226.
Bartoń, K. (2020). MuMIn: Multi-model inference. R package version 1.43.17, https://CRAN.R-project.org/package=MuMIn
Berthold, P. (2001). Bird migration. Oxford Univ Press.
BirdLife International. (2015). European Red List of Birds. Office for Official Publications of the European Communities.
BirdLife International and Nature Serve. (2014). Bird species distribution maps of the world. BirdLife International and NatureServe.
Bjornstad, O. N., Cai, J. (2020). ncf: Spatial covariance functions. R package version 1.2-9. https://CRAN.R-project.org/package=ncf
Blondel, J. (1997). Evolution and history of the European bird fauna. In W. J. M. Hagemeier & M. J. Blair (Eds.), The EBCC atlas of European breeding birds (p. cxxiii–cxxvi). T & AD Poyser.
Bonnet-Lebrun, A.-S., Manica, A., & Rodrigues, A. S. L. (2020). Effects of urbanization on bird migration. Biological Conservation, 244, 108423.
Brown, J. H., Mehlman, D. W., & Stevens, G. C. (1995). Spatial variation in abundance. Ecology, 76, 2028–2043.
Chapman, K. A., & Reich, P. B. (2007). Land use and habitat gradients determine bird community diversity and abundance in suburban, rural and reserve landscapes of Minnesota, USA. Biological Conservation, 135, 527–541.
Čížková, H., Květ, J., Comin, F. A., Laiho, R., Pokorný, J., & Pithart, D. (2013). Actual state of European wetlands and their possible future in the context of global climate change. Aquatic Sciences, 75, 3–26.
Cody, M. L. (1981). Habitat selection in birds: The roles of vegetation structure, competitors, and productivity. BioScience, 31, 107–113.
Crosby, A. D., Bayne, E. M., Cumming, S. G., Schmiegelow, F. K. A., Denes, F. V., & Tremblay, J. A. (2019). Differential habitat selection in boreal songbirds influences estimates of population size and distribution. Diversity and Distributions, 25, 1941–1953.
Devictor, V., Julliard, R., Clavel, J., Jiguet, F., Lee, A., & Couvet, D. (2008). Functional biotic homogenization of bird communities in disturbed landscapes. Global Ecology and Biogeography, 17, 252–261.
Devictor, V., van Swaay, C., Brereton, T., Brotons, L., Chamberlain, D., Heliola, J., Herrando, S., Julliard, R., Kuussaari, M., Lindström, A., Reif, J., Roy, D. B., Schweiger, O., Settele, J., Stefanescu, C., van Strien, A., van Turnhout, C., Vermouzek, Z., WallisDeVries, M., … Jiguet, F. (2012). Differences in the climatic debts of birds and butterflies at a continental scale. Nature Climate Change, 2, 121–124.
Ducatez, S., Clavel, J., & Lefebvre, L. (2015). Ecological generalism and behavioural innovation in birds: Technical intelligence or the simple incorporation of new foods? Journal of Animal Ecology, 84, 79–89.
Evans, K. L., Chamberlain, D. E., Hatchwell, B. J., Gregory, R. D., & Gaston, K. J. (2011). What makes an urban bird? Global Change Biology, 17, 32–44.
Gaston, K. J., Blackburn, T. M., & Lawton, J. H. (1997). Interspecific abundance–range size relationships: An appraisal of mechanisms. Journal of Animal Ecology, 66, 579–601.
Gill, F. B. (2006). Ornithology (3rd ed.). WH Freeman.
Gregory, R. D., & Gaston, K. J. (2000). Explanations of commonness and rarity in British breeding birds: Separating resource use and resource availability. Oikos, 88, 515–526.
Hanzelka, J., Horká, P., & Reif, J. (2019). Spatial gradients in country-level population trends of European birds. Diversity and Distributions, 25, 1527–1536.
Havlíček, J., Riegert, J., Bandhauerová, J., Fuchs, R., & Šálek, M. (2021). Species-specific breeding habitat association of declining farmland birds within urban environments: Conservation implications. Urban Ecosystem, 24, 1259–1270.
Hönigová, I., & Chobot, K. (2014). Jemné předivo české krajiny v GIS: Konsolidovaná vrstva ekosystémů. Ochrana Přírody, 10, 27–30.
Hořák, D., Toszogyová, A., & Storch, D. (2015). Relative food limitation drives geographical clutch size variation in South African passerines: A large–scale test of Ashmole’s seasonality hypothesis. Global Ecology and Biogeography, 24, 437–447.
Jetz, W., Sekercioglu, C. H., & Böhning-Gaese, K. (2008). The worldwide variation in avian clutch size across species and space. PLoS Biology, 6, 2650–2657.
Jiguet, F., Gregory, R. D., Devictor, V., Green, R. E., Vorisek, P., Van Strien, A., & Couvet, D. (2010). Population trends of European common birds are predicted by characteristics of their climatic niche. Global Change Biology, 16, 497–505.
Keller, V., Herrando, S., Voříšek, P., Franch, M., Kipson, M., Milanesi, P., Martí, D., Anton, M., Klvaňová, A., Kalyakin, M. V., Bauer, H. G., & Foppen, R. P. B. (2020). European breeding bird atlas 2: Distribution abundance and change. European Bird Census Council & Lynx Edicions.
Kinzelbach, R. K. (2004). The distribution of the serin (Serinus serinus L., 1766) in the 16th century. Journal of Ornithology, 145, 177–187.
Kloubec, B., & Čapek, M. (2012). Seasonal and diel patterns of vocal activity in birds: Methodological aspects of field studies. Sylvia, 38, 74–101.
Koleček, J., Adamík, P., & Reif, J. (2020). Shifts in migration phenology under climate change: Temperature vs. abundance effects in birds. Climate Change, 159, 177–194.
Koleček, J., & Reif, J. (2011). Differences between the predictors of abundance, trend and distribution as three measures of avian population change. Acta Ornithologica, 46, 143–153.
Korňan, M. (2004). Structure of the breeding bird assemblage of a primaeval beech–fir forest in the Sramkova National Nature Reserve, the Mala Fatra Mts. Biologia, 59, 219–231.
Martin, T. E., & Li, P. J. (1992). Life-history traits in open-nesting vs cavity-nesting birds. Ecology, 73, 579–592.
Møller, A. P. (2009). Successful city dwellers: A comparative study of the ecological characteristics of urban birds in the Western Palearctic. Oecologia, 159, 849–858.
Montaño-Centellas, F. A., Loiselle, B. A., & Tingley, M. W. (2021). Ecological drivers of avian community assembly along a tropical elevation gradient. Ecography, 44, 574–588.
Morelli, F., Benedetti, Y., Perna, P., & Santolini, R. (2018). Associations among taxonomic diversity, functional diversity and evolutionary distinctiveness vary among environments. Ecological Indicators, 88, 8–16.
Nee, S., Read, A. F., Greenwood, J. J. D., & Harvey, P. H. (1991). The relationship between abundance and body size in British birds. Nature, 351, 312–313.
Norberg, U. M. (1979). Morphology of the wings, legs and tail of three coniferous forest tits, the goldcrest, and the treecreeper in relation to locomotor pattern and feeding station selection. Philosophical Transactions of the Royal Society B, 287, 131–165.
Norberg, U. M. (1995). How a long tail and changes in mass and wing shape affect the cost for flight in animals. Functional Ecology, 9, 48–54.
Pechar, L. (2000). Impacts of long-term changes in fishery management on the trophic level water quality in Czech fish ponds. Fisheries Management and Ecology, 7, 23–31.
Pigot, A. L., Sheard, C., Miller, E. T., Bregman, T. P., Freeman, B. G., Roll, U., Seddon, N., Trisos, C. H., Weeks, B. C., & Tobias, J. A. (2020). Macroevolutionary convergence connects morphological form to ecological function in birds. Nature Ecology & Evolution, 4, 230–239.
Pinheiro, J. (2021). nlme: Linear and nonlinear mixed effects models. R package version 3.1-153. https://CRAN.R-project.org/package=nlme
R Core Team. (2017). R: A language and environment for statistical computing. R Foundation for Statistical Computing.
Reif, J., Hořák, D., Krištín, A., Kopsova, L., & Devictor, V. (2016). Linking habitat specialization with species’ traits in European birds. Oikos, 125, 405–413.
Reif, J., Storch, D., & Šímová, I. (2008). Scale-dependent habitat gradients structure bird assemblages: A case study from the Czech Republic. Acta Ornithologica, 43, 197–206.
Reznick, D., Bryant, M. J., & Bashey, F. (2016). r- and K-Selection revisited: The role of population regulation in life-history evolution. Ecology, 83, 1509–1520.
Robb, G. N., McDonald, R. A., Chamberlain, D. E., & Bearhop, S. (2008). Food for thought: Supplementary feeding as a driver of ecological change in avian populations. Frontiers in Ecology and the Environment, 6, 476–484.
Rocha-Camarero, G., & De Trucios, S. J. H. (2002). The spread of the Collared Dove Streptopelia decaocto in Europe: Colonization patterns in the west of the Iberian Peninsula. Bird Study, 49, 11–16.
Roth, T., Plattner, M., & Amrhein, V. (2014). Plants, birds and butterflies: Short-term responses of species communities to climate warming vary by taxon and with altitude. PLoS ONE, 9, e82490.
Stirnemann, I. A., Ikin, K., Gibbons, P., Blanchard, W., & Lindenmayer, D. B. (2014). Measuring habitat heterogeneity reveals new insights into bird community composition. Oecologia, 177, 733–746.
Stoate, C., Baldi, A., Beja, P., Boatman, N. D., Herzon, I., van Doorn, A., de Snoo, G. R., Rakosy, L., & Ramwell, C. (2009). Ecological impacts of early 21st century agricultural change in Europe—A review. Journal of Environmental Management, 91, 22–46.
Storch, D., & Kotecký, V. (1999). Structure of bird communities in the Czech Republic: The effect of area, census technique and habitat type. Folia Zoologica, 48, 265–277.
Storch, D., & Okie, J. G. (2019). The carrying capacity for species richness. Global Ecology and Biogeography, 28, 1519–1532.
Storchová, L., & Hořák, D. (2018). Life-history characteristics of European birds. Global Ecology and Biogeography, 27(4), 400–406. https://doi.org/10.1111/geb.12709
Taha, H. (1997). Urban climates and heat islands: Albedo, evapotranspiration, and anthropogenic heat. Energy and Buildings, 25, 99–103.
ter Braak, C. J. F. & Šmilauer, P. (2002). CANOCO Windows: software for canonical community ordination (version 4.5). Microcomputer Power.
Thomas, J. A. (1995). Why small cold-blooded insects pose different conservation problems to birds in modern landscapes. Ibis, 137, S112–S119.
Webb, C. T., Hoeting, J. A., Ames, G. M., Pyne, M. I., & Poff, N. L. (2010). A structured and dynamic framework to advance traits–based theory and prediction in ecology. Ecology Letters, 13, 267–283.
Acknowledgements
We are grateful to all voluntary observers for their fieldwork and to Tomáš Telenský for his help with designing the monitoring scheme. The Consolidated Layer of Ecosystems was supplied by the Nature Conservation Agency of the Czech Republic (TD010066). JR was supported by Charles University (PRIMUS/17/SCI/16).
Funding
This study was funded by Czech Science Foundation (18-16738S) and Charles University, Prague (PRIMUS/17/SCI/16).
Author information
Authors and Affiliations
Contributions
JR conceived the idea; ZV, PV, and JR designed the research; ZV, PV, and JR collected data; JR, DR, and FM analyzed data; JR drafted the manuscript with inputs from all authors.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare they have no conflict of interests.
Supplementary Information
Below is the link to the electronic supplementary material.
42974_2022_89_MOESM1_ESM.docx
Fig. S1: Gradients in species’ diet composition revealed by the principal component analysis on nine diet types recognized by Storchová and Hořák (2018). (DOCX 82 kb)
42974_2022_89_MOESM2_ESM.docx
Fig. S2: Residuals vs fitted values for respective linear models relating species’ ecological characteristics to their positions along the most important habitat gradients revealed by canonical correspondence analysis (cca1-cca4): (A) life history strategy, (B) migration distance, (C) climatic niche position, (D) European rarity, (E) diet pc1 and (F) diet pc 2. (DOCX 176 kb)
42974_2022_89_MOESM3_ESM.docx
Fig. S3: Residuals vs fitted values for respective linear mixed models relating mean values of species’ ecological characteristics in habitat polygons to the habitat types: (A) life history strategy, (B) migration distance, (C) climatic niche position, (D) European rarity, (E) diet pc1 and (F) diet pc 2. (DOCX 589 kb)
42974_2022_89_MOESM4_ESM.docx
Fig. S4: Spatial autocorrelation in residuals of respective linear mixed models relating mean values of species’ ecological characteristics in habitat polygons to the habitat types: (A) life history strategy, (B) migration distance, (C) climatic niche position, (D) European rarity, (E) diet pc1 and (F) diet pc 2. (DOCX 151 kb)
42974_2022_89_MOESM6_ESM.xlsx
Table S1: List of bird species and their characteristics. Abbreviation—abbreviated species name used in Fig. 2; Life history strategy—a gradient from species with slow strategies to species with fast strategies expressed using scores from a principle component analysis on six life-history traits; Migration distance—distance between centroids of species’ breeding and non-breeding ranges in km; Climatic niche position—mean temperature in species’ European breeding range in the peak breeding season (April–June); European rarity—species’ proportional breeding occupancy in Czechia; Diet pc1 and pc2—positions of species along first two axes obtained by a principal component analysis on nine diet types; cca1-4—positions of species along first four canonical axes representing the most important habitat obtained by a canonical correspondence analysis on species occurrences in 15 habitat types. (XLSX 30 kb)
42974_2022_89_MOESM8_ESM.xlsx
Table S3: List of habitat polygons and their characteristics. Polygon_ID—polygon identifier; Square—location of a given polygon; Large square—location of a given square; Habitat type—habitat type of a given polygon; Latitude and Longitude—geographic position of polygon’s centriod; Species richness—number of species recorded in respective habitat polygons. Life history strategy, Migration distance, Climatic niche position, European rarity, Diet pc1 and pc2—mean values of respective ecological characteristics across species recorded in individual habitat polygons. (XLSX 255 kb)
Rights and permissions
About this article
Cite this article
Reif, J., Vermouzek, Z., Voříšek, P. et al. Birds’ ecological characteristics differ among habitats: an analysis based on national citizen science data. COMMUNITY ECOLOGY 23, 173–186 (2022). https://doi.org/10.1007/s42974-022-00089-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42974-022-00089-4