Abstract
Cultural landscapes, shaped by centuries of traditional low-intensity agricultural practices, have been declining all over the world. In Transylvania (Romania), traditionally mown and extensively grazed grasslands have been part of a diverse landscape mosaic for centuries. In contrast to Western European countries, these species-rich grasslands are still present in Transylvania, but have declined in recent years, due to the pressure of intensification or abandonment, and inconsistent land use policies. The quality of these grasslands could easily be assessed using plants and butterflies, which are considered among the best indicators for habitat quality because of their characteristic response to land use change. Our study aimed to assess butterfly and plant community assemblages, richness and diversity in Transylvania’s most typical grassland type, by using transect and relevé methods. We investigated the drivers of change in butterfly and plant community assemblages, and decline in biodiversity of basiphilous dry grasslands. We found that present and past land use practices, geographic distance between sites and soil physical–chemical characteristics have major impacts on different aspects of plant and butterfly diversity (like species composition and richness). Grasslands that were converted to croplands in the past have lost their typical species assemblages and have not completely recovered in the last 30 years. Mown grasslands supported the most diverse communities of butterflies and plants, and diversity declined with land use change to extensive and then intensively grazed pastures. For the conservation of basiphilous dry grasslands in Transylvania, we recommend a case-by-case analysis of grasslands, with emphasis on prioritizing long-term traditional mowing when applying agro-environmental schemes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Land use is a major driver of biodiversity in grasslands. Some of Europe’s most biodiverse habitats are found in agricultural landscapes (Billeter et al., 2008; Oppermann et al., 2012). In contrast to the more homogenous natural landscapes, cultural landscapes consist of a mosaic of natural, semi-natural and artificial habitat types, shaped by traditional low-intensity agricultural practices (Loos et al., 2014; Oppermann et al., 2012; Rákosy, 2011; Turtureanu et al., 2014; van Elsen, 2000). The decline of biodiversity throughout Europe is strongly connected with the abandonment of traditional agricultural practices in favour of intensification or complete abandonment (Krebs et al., 1999; Robinson & Sutherland, 2002).
Traditional mowing and grazing are among the low-intensity agricultural practices that have contributed for centuries to the species-rich cultural landscapes of Transylvania (Rákosy, 2011). However, in a similar manner to European trends, in the last 50 years, major social-economic changes have influenced agricultural practices both directly and indirectly (Loos et al., 2014; Paulini et al., 2011; Schmitt & Rákosy, 2007; Stoate et al., 2009). Political, social and economic perturbations and institutional instability in the past century, particularly after 1989, have favoured land abandonment, intensification and revival of extensive subsistence agricultural practices (Hartel et al., 2016). Romania’s 2007 accession to the EU brought forth policies that have favoured the intensification of agriculture in some areas, but also some extensive, biodiversity-friendly farming practices through agro-environmental payments. In recent years, the profitability of farming depends on subsidies (Muru et al., 2014). One of these agro-environmental regulations in Romania aims specifically to reduce land abandonment (European Parliament 2013—EC regulation 1307/2013, “direct payment per agricultural surface”). However, by encouraging any type of land use, this measure involuntarily encourages those practices, which are less labour-intensive and are more profitable, e.g. intensive sheep grazing (over 0.7 heavy livestock units ha−1). Intensive grazing is extremely widespread all over Romania in recent years. One of the most representative example in Romania of the spreading of intensive grazing in the last decade is the Natura 2000 site ROSCI0295 (Directia Sanitar Veterinara și pentru Siguranța Alimentelor Cluj, 2016). Romania is among the world’s top 10 countries that export lambs, and it holds over ten million sheep heads (http://www.worldstopexports.com/top-sheep-and-lamb-meat-exports/).
The number of sheep flocks of large farmers increased in size (INSSE, 2017) over the past 3 years. The overall number of sheep is not necessary a problem in itself; however, the random distribution of very large herds on available grasslands and the grazing pressure throughout the year leads to a patterned distribution of alternating very low (abandoned areas) and very high grazing pressure (many of them within Natura 2000 sites) (INSSE 2018). The profitability of intensive grazing results from the multiple subsidy income that land users are able to access. Apart from the monetary value of sheep meat (http://www.fao.org/faostat/en/#home), direct payments are available both for agricultural land ownership (if land is cleared and then used for agricultural purposes) and per-capita (MADR 2015).
Meadows (mown grasslands), historically widespread in low- and highlands of Transylvania, have become less profitable in recent years (Paulini et al., 2011). Intensive livestock farms increasingly use concentrated, enriched fodder and nutritional supplements instead of hay, or more frequently, are grazing in winter too. Even though villages have specifically defined areas for pastures and meadows, respectively, some land owners lease their meadows to shepherds to be grazed, for various reasons (e.g. owners are too old to mow themselves, or owners live away in the city and prefer to get periodically dairy products from shepherds rather than mow themselves or contract someone else to mow).
The life cycle of butterflies is tightly linked to certain plant species as food sources, organisms serving as hosts during their life cycle (including also host ants important for some Lycaenidae butterflies) or entire plant communities that offer shelter and opportunities for basking, foraging and patrolling for butterflies (Dennis et al., 2006). Their high habitat specificity, short generation time and relatively low mobility are traits that show a strong connection to certain plant communities and a quick reaction to habitat changes (Kuussaari et al., 2007; Thomas, 2005). Therefore, butterflies and plants are interconnected in many ways, and they have been considered to be useful indicators for grassland quality and biodiversity by many studies (e.g. Erhardt, 1985; Erhardt & Thomas, 1991; Loos et al., 2015; Thomas, 2005; Van Swaay & Warren, 2012).
Despite grassland butterfly and plant diversity being a well-studied topic, models and assumptions previously developed in Western Europe cannot be applied fully to the current social-economic and political context of these ecosystems in Transylvania. Due to the continuously changing terms of land use and ambiguous legal status of grassland land use in particular, it is increasingly important to build up reliable expertise about how land use influences grassland diversity in Transylvania. In this study, we aimed to investigate whether plant and butterfly richness, diversity and community composition have already responded to land use changes, or whether there are other major factors that influence the community assembly and diversity of these two taxonomic groups. We hypothesized that the traditional practices still in use today would yield the highest diversity parameters in these groups and that intensity of land use will play a crucial role.
Materials and methods
Study area
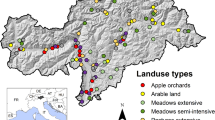
We assessed butterfly and plant communities in 23 plots of 1 ha each (Fig. 1) between April and September in 2015. Plots were located within (N = 21) and in the surroundings (N = 2) of the Natura 2000 site ROSCI0295 “Dealurile Clujului de Est”, approximately 30 km from Cluj-Napoca (Transylvania, Romania, 46°48′–46°58′N, 23°34′–23°44′E). The two plots outside the Natura 2000 site were located in the close proximity of it and were selected to fulfil the criteria of site selection, i.e. slope aspect, inclination and land use. Plots were placed at a distance of 0.2–11 km from each other. Some plots were more grouped together, while others were more scattered (Fig. 1); therefore, we considered two geographic parameters in our analyses (locality and geographic distance—see variables Loc and GD further on). All the plots were located in basiphilous dry grasslands belonging to the Cirsio-Brachypodion pinnati Hadač & Klika in Klika & Hadač 1944 alliance (Dengler et al., 2012). This alliance comprises those plant communities from Transylvania, which have been found to be extremely species rich on a small spatial scale, holding world records in this respect (Dengler et al., 2012; Wilson et al., 2012). Most of the plots were situated on north–north-west, north or north–north-east-facing slopes and had similar slope inclinations (2°–15°). Climatic variables (www.worldclim.org/bioclim) had similar values for all plots. In the study area, the climate is temperate continental with a mean annual temperature of 8.68 °C, mean annual precipitation of 567.9 mm and maximum precipitation occurring during the summer months (based on data between 1923 and 2014, Cluj-Napoca Meteorological Station; http://www.ecad.eu). The following variables: GPS coordinates (geographic distance — GD), nearest locality (Loc), elevation (Elv), slope aspect (Exp) and inclination (Incl), habitat openness (percentage of shrub cover — OSC) and average vegetation height (cm; measured in 16 random points within each plot - Veg_hgh) were recorded in each plot. These variables were assigned to the “geographic variables” category. Percentage cover of bare soil surfaces (Osa) and of litter (Litt.cov) were estimated in four randomly selected relevés of 4 m × 4 m within a 50 m × 50 m square in each plot. Measured values were subsequently averaged. Litter thickness (cm - Litt_ths) was measured in 16 random points within each plot. Variables Osa, Litt.cov, Litt_ths were assigned to the “habitat variables” category.
Additionally, we measured 11 soil variables (assigned to the “soil variables” category) grouped as soil physical–chemical parameters: humus content (“organic layer” horizon) (%) (Hum), pH at 3 and 30 cm soil depth (pH), redox potential (mV - ROP), electro-conductivity (μS/m - EC), humidity (% - U), porosity (por25), structure (Str25) and texture (Tex25), all three at a depth of 25 cm, soil ammonium (mg/l - NH4+), nitrate (mg/l - NO3−) and phosphate (mg/l) (PO4−) content.
The plots were selected to cover the following present land use (PVLU) types: abandoned grassland, occasionally grazed (PZLUab, n = 4 plots), pastures (PZLUgr, n = 8), mown grasslands (which we considered equivalent to mown meadows) (PZLUmo, n = 11). The intensity of the present land use types was classified as following (Int): (1) abandoned grasslands, (2) traditionally mown grasslands and extensively grazed pastures, (3) grasslands mown with light machinery, (4) grasslands mown with tractors, (5) intensively grazed pastures. We also recorded the previous land use (i.e. the land was mown, grazed or cropped in the past) (PVLU), the age of present land use (AGLU), the number of land use changes in the last 30 years (NLUCH30) (both PVLU and NLUCH30 estimated from cadastral maps, Corine Land Cover data and topographic maps) and the distance to the nearest sheepfold (Google earth maps and field observations) (Dist). Variables PZLU, Int, PVLU, AGLU, NLUCH30 and Dist were assigned to the “land use variable” category. Regarding the present land use and its intensity, we considered abandoned grassland as a successional stage of former pastures that have been abandoned for the last 6–10 years. Extensive pastures are considered to have a low stocking rate (a maximum of 0.7 livestock unit ha−1) of sheep or cattle periodically grazing throughout the year. Intensively grazed pastures have a high stocking rate of sheep grazing constantly throughout the year for the last 2–3 years after shrubs have been cleared as a result of applying agricultural recommendations (European Parliament 2013, EC regulation 1307/2013, “direct payment per agricultural surface”). All mown grasslands are part of the grassland landscape of a region and are not cultivated. They are mown once per year between July 25th and September 15th. Traditionally grasslands are mown by scythe. Light mowing machines (provided by Brielmaier company—www.brielmaier.com) were used for one year prior to the start of the survey and in the following year, at the same time when neighbouring grasslands (meadows) were mown by hand. Traditional practices in this region allow sheep to graze and to pass through mown grasslands occasionally in fall after mowing and in spring before the 1st of May, a common tradition in many European semi-natural grasslands (Crofts & Jefferson, 1999; Pedersen & Widgren, 2011).
Plant and butterfly survey
Butterflies were surveyed within each plot on a square of 50 × 50 m where we applied a modified transect method of patrolling in a serpentine pattern throughout the entire square area (Cremene et al., 2005; Douwes, 1976; Hall, 1981 have used the same adapted method). Squares were surveyed once every other week between 8 May and 15 September 2015 by the same two persons. Transects were surveyed only under good weather conditions (sunny, temperature ≥ 18 °C and wind speed ≤ 16 km/h [Beaufort scale 3]) between 10.00 and 17.00 h. We recorded all species and individuals of Papilionoidea. Multiple counting of the same individuals could not be completely avoided. However, this error may not influence our interpretation, because we performed a comparison of plot communities; thus, the error would be the same for each plot. The sequence in which transects were surveyed varied in order to avoid systematic effects of time of the day.
We recorded species richness and individual abundance of butterflies for each study plot. We assigned conservation statuses (IUCN categories—CR, EN, VU, NT, LC and DD) and ecological character (HF—hygrophilous, MHF—meso-hygrophilous, MF—mesophilous, U—ubiquitous, MXF—mesoxerophilous, XF—xerophilous and MI—migrant) to each species. Conservation status, ecological character, as well as species identification followed Rákosy (2013).
Plant communities were sampled in four randomly selected relevés of 4 m × 4 m within a 50 m × 50 m square in each plot (overlapped with the butterfly survey square), in May and June 2015. Percentage cover was visually estimated for each vascular plant species rooted in, or in the case of dwarf shrubs or shrubs, overhanging each square. Cover measurements can add up to more than 100% in squares with multiple layers. Percentage cover was averaged for each square. High conservation value plant species were considered based on Boșcaiu et al. (1994), Dihoru and Dihoru (1994) and Oltean and Negrean (1994).
Data analysis
Firstly, we performed a Mantel test to examine whether the similarities of the relative abundance pattern of butterfly species and plant species, respectively, between the communities (butterfly matrix and plant matrix, respectively) were determined by the similarities of the cumulative environmental factors (environmental factors matrix; see variables listed in the Study area Methods section and S3) and by geographic distances (distance matrix). Since the results of Mantel test showed significant correlations, we included geographic distance in our multivariate analyses and generalized linear mixed models to account for spatial autocorrelation.
Then, we described the species composition and its relation with constrained variables (S3) by applying a constrained correspondence analysis (CCA) to each butterfly and plant species abundance matrix. CCA can be used to model the multivariate response of a species assemblage to a matrix of environmental variables (Borcard et al., 1992; Legendre & Legendre, 1998). The species abundance matrices were ln (x + 1) transformed to maintain normal distribution and to avoid the “arch effect” in CCA (Ter Braak, 1986). All numerical environmental variables were scaled to mean zero and unit variance. The CCA was performed using vegan package (Oksanen et al., 2006), and to select variables of environmental matrix that best explain the species matrix, i.e. constraints, we performed a forward stepwise model using "ordistep" function in the package. We calculated variance inflation factors (VIF) for each of the constraints and contrast in factor constrains from environmental matrix using function “vif.cca”. Values over 10 indicate redundant constraints. All the selected variables using stepwise model were independent from each other (VIF < 10). The permutation procedure (based on 9999 cycles) was used to test the significance of the CCA model, of the explanatory variables in CCA and explained variance by the CCA axes (Oksanen et al., 2006).
The abundance of species in different ecological and conservation categories was compared between present land use types with the Kruskal–Wallis test.
Species richness and abundance for butterflies and plants from the field surveys were used to compute Shannon—Wiener (Shannon’s H) diversity indices for each plot. Furthermore, in order to correct for small sample size bias, we used several asymptotic richness estimators based on species accumulation curves for both butterfly and plant richness: Chao, first- and second-order jackknife (jackknives 1 and 2), and bootstrap methods.
Generalized linear mixed models (GLMM) of species richness and Shannon–Wiener diversity index (H) were performed for both butterfly and plant data set using the lme4 package (Bates & Maechler, 2010; Bates et al., 2014). We designed a set of 48 candidate models. The first 36 candidate models included a single predictor. We also considered models that included two and three and four predictors, with no collinearity problems, of habitat, soil physical–chemical parameters and land use characteristics. To detect collinearity between two or more predictor variables, we used VIF. A VIF > 3 is a signal of collinearity. We excluded highly collinear variables through “vifsptep” in “usdm” package. “vifstep” calculates VIF for all variables, excludes the one with highest VIF and repeats the procedure until no variables with VIF greater than three remains. We assumed a Poisson error distribution and a logarithmic link function for the number of species and a Gaussian error and identity link function for H. Locality was included in the models as a random effect, and to account for overdispersion (i.e. non-independent (aggregated) data for plots that are close to each other), we also added an observation random effect.
In order to assess the relative performance of these models, we used a selection technique based on Akaike’s information criterion corrected for sample size (AICc: Burnham & Anderson, 2002; Johnson & Omland, 2004) using aictab function in AICcmodavg package (Mazerolle, 2015). We ranked the models according to their AICc, and the one with the lowest AICc was used as the reference for calculating the AICc difference (∆i). The relative evidence of each model is given by Akaike weights (wi), and these are interpreted as the likelihood of a model given the data and the model. Models within two AICc units of the AICcmin were considered competitive and more plausible than others (Burnham & Anderson, 2002). We used the most parsimonious model for obtaining parameter estimates and predictions of species richness and diversity because the effects of other environmental covariates then included in the best models were weak (Table 1) and model averaging, which might be used to account for model selection uncertainty, would only shrink those effects (Burnham & Anderson, 1998). We checked for correlations between species richness and diversities of butterflies and plants by first checking for normal distribution of data, using the Shapiro–Wilk test and subsequently calculating the Pearson’s correlation coefficient for species richness values and Spearman’s rank correlation coefficient for Shannon’s H indices.
All the statistical procedures were implemented in R 3.2.3 (R Development Core Team, 2015).
Results
Community analyses
We recorded 9599 butterflies belonging to 65 taxa including one species previously not recorded in the site (Brintesia circe). Of these, according to Rákosy (2013), two (3%) taxa are endangered (EN—Maculinea nausithous, M. teleius), seven (11%) are considered vulnerable (VU—Lycaena dispar, L. alciphron, Maculinea alcon cruciate ecotype, Cupido osiris, Brenthis daphne, B. hecate, Iphiclides podalirius), nine (14%) species are near threatened (NT), one (1%) is considered under the category “data deficient” (DD), and the majority of species (46 = 71%) is classified under the “least-concern” category (LC). All communities were dominated by mesophilous species which accounted for 62–97% of the total individuals found in each plot (median = 80%, Q1 = 73.7%, Q3 = 84.1%). 2–22% of the communities were represented by xerophilous species (median = 7.0%, Q1 = 5.7%, Q3 = 11.5%). Mesoxerophilous represented 0.4–21% of all communities (median = 7.6%, Q1 = 3.9%, Q3 = 8.8%). Ubiquitous species represented 0–7% of all communities (median = 2.6%, Q1 = 1.8%, Q3 = 3.5%). The other ecological categories make up less than 4% in each community (median < 1%). For a more detailed view, the figure in S2 summarizes the most abundant species from each present land use type with frequencies over 1% of the individuals found in each present land use type.
We recorded 260 plant species in all study sites. We found in total six red-listed and rare plant species in the study sites: Centaurea trinervia, C. triumfettii, Crambe tataria, Dianthus collinus, Orchis coriophora subsp. fragrans and Serratula radiata, and two species of community interest (Council Directive 92/43/EEC 1992): Echium russicum and Crambe tataria.
The Mantel test results showed that relative abundance pattern of butterflies was strongly influenced both by the cumulative environmental factors (r = 0.377, p = 0.002, 10,000 permutations) and the geographic distance (r = 0.255, p = 0.006, 10,000 permutations) while the relative abundance pattern of plants was strongly influenced by the geographic distance (r = 0.390, p < 0.001, 10,000 permutations) but not by the cumulative environmental factors (r = 0.175, p = 0.089, 10,000 permutations).
For butterflies, the global CCA model, which selected locality, previous land use and habitat openness as meaningful explanatory variables, was significant (P < 0.001). A total of 43.25% of the overall variation in butterfly species composition was attributed to these explanatory variables, of which the majority was explained by the first and second axes (Fig. 2) which accounted for 28.44% and 14.81% of the total variation, respectively. In Fig. 2, we highlighted 12 species that were significantly correlated with one or both first two CCA axes. Among these, Boloria dia (Bdia), was strongly influenced by grazed previous land use, while Colias crocea (Ccroc) was strongly correlated with mown previous land use. Species (Brintesia circe—Bcirc, Argynnis adippe—Aadip, Argynnis aglaja—Aagla, Argynnis paphia—Apaph) from the upper left quadrant were strongly associated with cropped previous land use and the locality of Badesti (LocBad) while Coenonympha pamphilus (Cpamp) was correlated with low value of habitat openness.
Biplots of the CCA model of the butterfly species abundance matrix in relation to environmental variables (shown in grey); PVLU—previous land use (cr—cropped; gr—grazed; mo—mown); Loc—locality (Bad-; Bor-; Ciu-; Dab-; Fan-; Fei-); OSC—habitat openness. Species names were abbreviated using the first letter of the genus and four letters of species name, respectively (shown in black). Abbreviations for the species are shown in text
For plant communities, the global CCA model selected locality, present land use, geographic distance and soil NO3− and PO4− content as meaningful (significant, P < 0.001) explanatory variables (Fig. 3). A total of 33.15% of the overall variation in species composition was attributed to these explanatory variables, out of which the first and second axes accounted for 19.74% and 13.41%, respectively. Figure 3 shows that Ajuga genevensis (Ajuggene) was strongly correlated with high soil NO3− concentrations, while Anagallis arvensis (Anagarve) was strongly correlated with high soil PO4− concentrations. Agrimonia eupatoria (Agrieupa) and Achillea collina (Achicoll) were most strongly associated with grazing as present land use. Agrostis tenuis (Agrotenu) was strongly associated with abandonment as present land use, while Asperula cynanchica (Aspecyna), Agropyron intermedium (Agrointe), Arrhenatherum elatius (Arrhelat) and Astragalus cicer (Astrcice) were strongly associated with mowing as present land use. The species in the lower left quadrant were strongly correlated with geographic distance.
Biplots of the CCA model of the plants species abundance matrix in relation to environmental variables (shown in grey); Loc—locality (Bad-; Bor-; Ciu-; Dab-; Fan-; Fei-); PZLU—present land use (ab—abandoned; gr—grazed; mo—mown); soil NO3− and PO4− content; GD geographic distance. Species names were abbreviated using the first four letters of the genus and species name, respectively (shown in black). Abbreviations are shown in text
The proportion of mesophilous, xerophilous, mesoxerophilous and ubiquitous butterfly species did not differ between present land use types (Kruskal–Wallis χ2 test for proportion of the communities represented by mesophilous species: χ2 = 2.212, df = 2, P = 0.331; Kruskal–Wallis χ2 test for proportion of the communities represented by xerophilous species: χ2 = 5.117, df = 2, P = 0.077; Kruskal–Wallis χ2 test for proportion of the communities represented by mesoxerophilous species: χ2 = 2.280, df = 2, P = 0.320; Kruskal–Wallis χ2 test for proportion of the communities represented by ubiquitous species: χ2 = 5.809, df = 2, P = 0.055). When considering conservation status, endangered butterfly species were found only in mown grasslands and were absent from grazed and abandoned ones (Kruskal–Wallis χ2 test: χ2 = 14.249, df = 2, P = 0.0009, followed by pairwise Mann–Whitney U tests: P < 0.05 in both comparisons). Other conservation categories did not differ between present land use types (Kruskal–Wallis χ2 test: P > 0.1 in all comparisons).
Diversity analyses
Species accumulation curves of Chao, jackknife 1, jackknife 2 and bootstrap species richness estimators are presented in Fig S4 for butterfly and Fig S5 for plants, in the Supplementary material.
The model selection indicated that for species richness only one model was well supported by the butterfly data and two models by the plant data (ΔAICc < 2) (Table 1). The best model for species richness for butterfly data included intensity of land use, while for plants the best model included soil NO3−, PO4− and NH4 content. For species diversity, model selection indicated that only one model was well supported both for butterfly and plants data (ΔAICc < 2) (Table 1). The best model for species diversity for butterfly data included previous land use, while for plants the best model included soil pH (Table 1).
Parameter estimates of species richness from the most parsimonious model showed that the butterfly richness was different among different intensities of land use (χ2 = 19.78, df = 2, P < 0.001 and Table 2). Butterfly richness was significantly lower in the highest intensity of land use than in all other categories (Fig. 4). Plant species richness increased significantly with the increase in soil NO3− content (χ2 = 21.767, df = 1, P < 0.001, Fig. 5a) but not with soil PO4− content (χ2 = 2.484, df = 1, P = 0.115, Fig. 5b), while decreased significantly with the increase in soil NH4 content (χ2 = 20.810, df = 1, P < 0.001, Fig. 5c) (F[1,2] = 20.761, P < 0.001).
Relationship between land use intensity (1-abandoned grasslands, 2-traditionally mown grasslands and extensively grazed pastures, 3-grasslands mown with light machinery, 4-grasslands mown with tractors, 5-intensively grazed pastures) and the predicted richness for butterflies. The bars represent the 95% confidence intervals
Parameter estimates of species diversity from the most parsimonious model showed that the butterfly diversity was different among previous land use types (F[1,2] = 20.761, P < 0.001). Butterfly diversity was lowest in the case of former croplands and the highest in the case of former hayfields (Fig. 6). Plant species diversity increased significantly with the increased of soil pH (F[1,16] = 10.210, P = 0.006, Fig. 7).
Species richness of plants and butterflies showed a significant positive correlation (r = 0.60, P = 0.002), while their diversities did not show a significant correlation (rS = − 0.20, P = 0.371).
Discussion
Geographic distance and locality effects
The Mantel test and the CCA models showed that geographic distances and locality play important roles in determining the species assemblages of both butterflies and plants. These findings are in line with the meta-analysis of Soininen et al., (2007), which found community similarity to significantly decline with distance particularly in terrestrial ecosystems, depending on the scale of the study and the organisms’ traits. Additional traits like dispersal ability and trophic position additionally explain the distance-decay similarity of communities, supporting the species-sorting hypothesis (Leibold et al., 2004). It is not surprising that the similarity in plant community composition was highly influenced by spatial proximity since local floras are filtered by local abiotic conditions and dispersal abilities.
At the same time, the assemblages of butterfly species are expected to be strongly connected with local landscape features (Bergman et al., 2004; Schmitt & Rákosy, 2007). Several previous studies showed that landscape structure affects butterfly diversity and species assemblages in some habitat types (e.g. woodland matrix on calcareous grasslands and orchards—Ernst et al., 2017, woody habitats and croplands around grasslands—Aviron et al., 2007), but most of them emphasized the fact that habitat type and management are more important than landscape-scale features. We agree with this point of view, even though in our study sites, locality and geographic distance seem to be as important as habitat type and land use. In our study sites, a different mechanism may have affected our butterfly communities, which is connected rather to present and past land use. Due to the regional character of land use, past actions are now being reflected in the strong effect of locality and geographic distance. This effect will be detailed in the next section referring to land use effect.
Land use effect
Overall, 100 butterfly species are known to occur in the Natura 2000 site “Dealurile Clujului de Est” (Muntean et al., 2015, Sitar C. unpublished data). In the present study, we recorded 65 taxa, estimated by asymptotic accumulation curves at around 70 (Fig.S4). The largest part of the investigated butterfly communities was represented by mesophilous, xerophilous and mesoxerophilous species. As expected, these grasslands harbour also several rare and protected butterfly and plant species, since most of our plots belonged to cultural landscapes shaped by traditional land use practices (Oppermann et al. 2012; Turtureanu et al., 2014; van Elsen, 2000). However, endangered butterfly taxa (Maculinea nausithous and M. teleius) were found only in mown grasslands, and vulnerable taxa (e.g. Brenthis hecate, B. daphne, Cupido osiris, etc.—S1) had over 70% of their abundances in mown grasslands. In comparison with mown grasslands, abandoned grasslands showed smaller proportions of vulnerable taxa, but the near-threatened category (IUCN—NT) was relatively well represented through species typical for shrub areas (e.g. Aporia crataegi, Satyrium pruni, S. spini, etc.). The largest proportion of species in abandoned grasslands and pastures were represented by polyphagous, mesophilous and least-concern species (47% of the butterflies recorded in abandoned grasslands belonged to the species M. jurtina, C. pamphilus, P. icarus, and Plebejus argus; 56% of butterflies recorded in pastures belonged to the species Polyommatus icarus, Maniola jurtina and Coenonympha pamphilus). These findings indicate the high value of traditional mowing practices in the study area. Generally, intensive mowing practices are considered detrimental to butterfly communities by directly killing larvae and reducing abruptly both hostplant and nectar resource cover (Dover et al., 2010; Humbert et al., 2010). On the other hand, traditional extensive mowing (e.g. once per year, as in our study region), occurring in late August or September and with difficult-to-mow patches kept unmown, has a beneficial effect on butterfly communities (Bruppacher et al., 2016; Kühne et al., 2015).
Plant community assemblages were best explained, besides locality and geographic distance also by the present land use and by soil NO3− and PO4− content. Several long-term experimental studies have shown that land use shapes plant community composition (e.g. Kahmen et al., 2002; Köhler et al., 2005; Moog et al., 2002). In its first stage, grassland abandonment leads to litter accumulation and often to the dominance of a few highly productive herbaceous species (e.g. Kahmen et al., 2002; Köhler et al., 2005; Moog et al., 2002; Rudmann-Maurer et al., 2008), while in the second phase there is a shrub encroachment (Bakker et al., 2002; Petersen & Drewa, 2014). In our study, grassland abandonment led to the increase in abundance of Agrostis tenuis, a short stature bunchgrass species, while mowing was linked to the increased abundance of some typical hay meadow species with high mowing tolerance like Arrhenatherum elatius. Land use, and especially grazing, may also influence the composition and species richness of plant communities through the modification of soil chemical properties (Augustine & Frank, 2001). Dung and urine deposition may increase soil nitrogen content, which was probably the case in our study system (Fig. 3, the actual land use grazing and soil NO3− content almost overlap in their effect on plant community composition). Soil chemical properties had a strong effect on plant species richness and species diversity as well. We found that soil NO3− and PO4− content positively influenced the plant species richness, while soil NH4+ content had a negative effect. Apart from grazing, a major source for soil nutrient enrichment in grassland is the fertilizers used in the surrounding croplands, which infiltrate into grassland soils. Previous studies showed that the grassland flora was very sensitive to the increase in soil nutrient content and soil acidity (e.g. Janssens et al., 1998; Roem & Berendse, 2000). Moreover, the symbiotic fixation of atmospheric nitrogen by legumes may contribute to the nitrogen accumulated in the grassland soils. Some of the grasslands in our study system had a quite high number of legume species (especially Dorycnium pentaphyllum subsp. herbaceum, Coronilla varia, Trifolium pretense and Medicago falcata), which might be linked indirectly to increased species richness. Both plant and butterfly community species composition in our study was influenced by the locality, soil parameters and local habitat factors, respectively. The increase in the content of nutrients in soil increased the plant species richness (Fig. 5), which in turn attracted more butterfly species. In the same way that more common or ruderal plant species prevailed in the communities of nutrient-rich grasslands (like Ajuga genevensis and Anagallis arvensis, Fig. 3), more common butterfly species (like Coenonympha pamphilus and Boloria dia, Fig. 2) replaced the typical nutrient-poor grassland species (like Cupido osiris and Argynnis niobe, Fig. 2) in the grazed and abandoned sites. Similar results were found by Öckinger et al. (2006b) in a long-term study in Sweden, which indicated that the soil nutrient enrichment determined the grasslands’ loss of typical nutrient-poor butterfly species. The butterfly communities investigated here responded to the habitat openness, which is a short-term proxy of habitat change (Bolliger et al., 2007). As land abandonment progresses towards more advanced successional stages, open-land species tend to be replaced by those more shadow-loving (Cremene et al., 2005; Dirnböck et al., 2003; Ernst et al., 2017; Labaune & Magnin, 2002; Laiolo et al., 2004).
Factors affecting plant communities indirectly influence butterfly communities since the two groups are closely interconnected through herbivory. Butterflies are generally considered good indicator species due to their capacity to detect short-term land use changes (Ellingsen et al., 1998; Erhardt, 1985). Several studies (e.g. Boch et al., 2016; Börschig et al., 2013; Kleijn et al., 2009; Kruess & Tscharntke, 2002) indicated that present land use intensity has a major influence on arthropod communities including butterflies. Present land use intensity in our study influenced butterfly richness but did not play a major role in shaping butterfly communities. The most remarkable effect of land use intensity was a clear decline in species richness of butterflies in intensively grazed pastures, while there were no significant differences among other present land use types and intensities. Some land use practices (e.g. intensive grazing) simplify the plant diversity, vegetation height and architecture, and as a consequence, specialized and rare butterfly species will decline shortly after, quickly altering butterfly richness, abundance and ecological profile of their community (Kruess & Tscharntke, 2002; Kuussaari et al., 2007). Our results are in line with those of other studies (Koch et al., 2015; Öckinger et al., 2006a) showing no significant difference between the butterfly species richness in the abandoned grasslands (in this study the lowest intensity of land use practice) compared to that in low-intensity traditional hay meadows and extensive pastures. This is most likely the result of increased habitat complexity (Rosenzweig, 1995). Nevertheless, land abandonment favoured the decline of several rare and representative grassland species (like Maculinea teleius, M. alcon cruciata ecotype, Cyaniris semiargus, Plebejus agryrognomon, Lysandra coridon, Glaucopsyche alexis, Brenthis hecate, Melitaea britomartis) that were replaced by more ubiquitous ones. Since land use intensity in the Transylvanian farms and grasslands is highly unpredictable on larger time scales, and the landscape is still composed of a small-scale mosaic of agricultural practices (both traditional and modern), patches of low-intensity management (delayed mowing, temporary abandonment, etc.) represent temporary shelters for butterfly populations (Bruppacher et al., 2016). The low-intensity management practices, however, lost their profitability in the last decade, allowing for the widespread adoption of intensive practices, like intensive sheep grazing (Baur et al., 2007; Kizos et al., 2013). If this tendency persists, the mosaic of land use practices and intensities will soon be changed into a vast intensive pasture favoured by the incentives of the Romanian agro-environment scheme (MADR 2015). Plant communities also responded to the present land use type, showing changes between mown and grazed grasslands. The spreading of intensive grazing will favour the simplification of landscape, soil erosion, accumulation of soil ammonium and lower soil pH (Briemle et al., 1993). These will lead to a loss of plant and butterfly (Blab, 1993) richness and diversity.
Besides present land use, previous land use type and habitat openness were important factors in shaping the species assemblages of butterflies in our study. In the same way, butterfly diversity responded strongly to the previous land use type, with previous croplands having the lowest diversity. Similarly, a declining pattern in bee trait diversity was found by Le Provost et al. (2021) in their study on legacy effects of the conversion of grasslands to croplands in France. Our results contradicted partially the findings of Culbert et al. (2017)’s study, which showed that plant and butterfly communities from Southern Transylvania were not affected by land use history.
Another aspect of historical land use effect was reflected in the strong influence of the locality (see previous section of the Discussion) on butterfly communities. The surroundings of certain localities have suffered more dramatic habitat alterations than others during the past 30 years, with many grasslands converted into croplands (e.g. Bădești). This conversion was very popular in the 1970s in Romania, and, among others, the disappearance of the species B. circe from Transylvania, around this time, illustrates the extension of cropland up to forest edges. The species reappeared slowly during the 1990s in Transylvania. After 35–40 years of abandonment of cropland, the habitat around Bădești appears to be suitable again for this species (Fig. 2). As a result of historical land use changes from grassland to cropland, other local extinctions of butterfly species connected to grassland habitats may have occurred. In support of this explanation, several studies showed that a past reduction in suitable habitat (here grassland) up to a certain threshold produced local extinctions of some populations, resulting in altered communities (Fahrig, 2001; Hanski & Ovaskainen, 2000; Hill & Caswell, 1999; Kareiva & Wennergren, 1995; Öckinger et al., 2006b). Our results support the suggestions made by Collinge et al. (2003), Aviron et al. (2007) and Bergman et al. (2008) that landscape history plays an important role in shaping present plant and butterfly communities.
Both diversity of butterflies and their community assemblages were influenced by the previous land use type. Where large-scale habitat alterations occurred in the past—like the ploughing of large crop fields—butterfly communities and their diversity have remained altered for the long term. At the same time, mown grasslands, which were long-term hay meadows, were the most butterfly rich and diverse, harboured the most protected species, and had typical mesophilous and mesoxerophilous butterfly communities. The meta-analysis of Tälle et al. (2016) and the study of Bonari et al. (2017) similarly indicate that in most European studies mowing has a more beneficial effect on biodiversity than sheep grazing and that the traditional management of mowing with aftermath grazing has an even more positive effect. However, Tälle et al. (2016) conclude that overall moderate grazing has a more positive effect when compared to annual mowing when managing for grassland conservation. Compared to Western Europe’s climate that is influenced by the Atlantic, Romania has a temperate continental climate, with a tendency towards aridification. Therefore, we see here a lesser capacity of grasslands and consequently of butterfly communities to regenerate. Another difference to most studies in Western and Central Europe comes when considering the purpose of management in our study area, which is mostly for providing fodder for animals and not for grassland conservation. Thereafter, higher yields are aimed for in all circumstances in Romania, making extensive grazing a disappearing practice. Agro-environmental schemes aimed at butterfly conservation are, at the moment, the only ones to keep the sheep grazing pressure low, while other measures, such as per-capita payments, increase the same pressure indirectly.
Conclusions
In conclusion, our results indicated that the long-term persistence of a mosaic of alternating land use practices and intensities may represent the key to preserving an overall high diversity of butterflies and plants and characteristic communities at landscape level in Transylvanian basiphilous dry grasslands. Intensive grazing with sheep favours habitat changes towards lower pH and higher ammonium content, followed by simplification of plant communities and a decrease in butterfly richness. Among land use types, maintaining traditional mowing of grasslands is of primary importance for the conservation of protected or locally rare butterfly and plant species, particularly if the intensity of mowing is kept at a low level.
Therefore, we advise against a simplified approach in predicting biodiversity and applying agro-environmental regulations, where present land use is the only considered criterion. In the context of historically different management trends, traditionally small-scaled mosaic grassland management and presently unpredictable land use practices, a case-by-case analysis including both indicator species and soil parameters will always reveal much more about the conservation value of the grasslands in Transylvania. Traditional mosaic-like low-intensity agricultural practices would help maintain the heterogeneous landscape structure and the high biodiversity of Transylvanian basiphilous dry grasslands.
Availability of data and material
Not applicable.
Code availability
Not applicable.
References
Augustine, D. J., & Frank, D. A. (2001). Effects of migratory grazers on spatial heterogeneity of soil nitrogen properties in a grassland ecosystem. Ecology, 82, 3149–3162. https://doi.org/10.1890/0012-9658(2001)082[3149:EOMGOS]2.0.CO;2
Aviron, S., Jeanneret, P., Schüpbach, B., & Herzog, F. (2007). Effects of agri-environmental measures, site and landscape conditions on butterfly diversity of Swiss grassland. Agriculture, Ecosystems and Environment, 122, 295–304. https://doi.org/10.1016/j.agee.2006.12.035
Bakker, J. P., Elzinga, J. A., & de Vries, Y. (2002). Effects of long-term cutting in a grassland system: perspectives for restoration of plant communities on nutrient-poor soils. Applied Vegetation Science, 5, 107–120.
Bates, D., & Maechler, M. (2010) lme4: Linear mixed-effects models using S4 classes. R package version 099937-5.
Bates, D., Maechler, M., Bolker, B, & Walker, S. (2014). lme4: Linear mixed-effects models using Eigen and S4. R Package Version 1.1-7. http://CRAN.R-project.org/package=lme4. Last accessed 30 February 2017.
Baur, B., Cremene, C., Groza, Gh., Schileyko, A. A., Baur, A., & Erhardt, A. (2007). Intensified grazing affects endemic plant and gastropod diversity in alpine grasslands of the Southern Carpathian Mountains (Romania). Biologia, 62(4), 438–445. https://doi.org/10.2478/s11756-007-0086-4
Bergman, K. O., Ask, L., Askling, J., Ignell, H., Wahlman, H., & Milberg, P. (2008). Importance of boreal grasslands in Sweden for butterfly diversity and effects of local and landscape habitat factors. Biodiversity and Conservation, 17, 139–153. https://doi.org/10.1007/s10531-007-9235-x
Bergman, K. O., Askling, J., Eckberg, O., Ignell, H., Wahlman, H., & Milberg, P. (2004). Landscape effects on butterfly assemblages in an agricultural region. Ecography, 27, 619–628. https://doi.org/10.1111/j.0906-7590.2004.03906.x
Billeter, R., Liira, J., Bailey, D., Bugter, R., Arens, P., Augenstein, I., & Edwards, P. J. (2008). Indicators for biodiversity in agricultural landscapes: A pan-European study. Journal of Applied Ecology, 45, 141–150. https://doi.org/10.1111/j.1365-2664.2007.01393.x
Blab J (1993) Grundlagen des Biotopschutzes für Tiere. 4. Auflage. Kilda, Bonn.
Boch, S., Allan, E., Birkhofer, K., Bossdorf, O., Blüthgen, N., Christ-Breulmann, S., Diekötter, T., Dormann, C. F., Gossner, M. M., & Hallmann, C., et al. (2016) Extensive und jährlich wechselnde Nutzungsintensität fördert den Artenreichtum im Grünland [Extensive and alternative land use intensity promotes species richness in grasslands]. ANLiegen Natur 38(1): online 10 pp. https://www.zobodat.at/pdf/AnliegenNatur_38_1_2016_0086-0095.pdf. Accessed 04.05.2021.
Bolliger, J., Kienast, F., Soliva, R., & Rutherford, G. (2007). Spatial sensitivity of species habitat patterns to scenarios of land use change (Switzerland). Landscape Ecology, 22, 773–789. https://doi.org/10.1007/s10980-007-9077-7
Bonari, G., Fajmon, K., Malenovský, I., Zelený, D., Holuša, J., Jongepierová, I., Kočárek, P., Konvička, O., Uřičář, J., & Chytrý, M. (2017). Management of semi-natural grasslands benefiting both plant and insect diversity: The importance of heterogeneity and tradition. Agriculture, Ecosystems and Environment, 246, 243–252. https://doi.org/10.1016/j.agee.2017.06.010
Borcard, D., Legendre, P., & Drapeau, P. (1992). Partialling out the spatial component of ecological variation. Ecology, 73, 1045–1055. https://doi.org/10.2307/1940179
Börschig, C., Klein, A.-M., von Wehrden, H., & Krauss, J. (2013). Traits of butterfly communities change from specialist to generalist characteristics with increasing land use intensity. Basic and Applied Ecology, 14(7), 547–554. https://doi.org/10.1016/j.baae.2013.09.002
Boşcaiu, N., Coldea, G., & Horeanu, C. (1994). Lista roşie a plantelor vasculare dispărute, periclitate, vulnerabile şi rare din flora României [Red list of extinct, threatened, vulnerable and rare vascular plant species of Romania]. Ocrotirea Naturii Şi a Mediului Înconjurător, 38, 45–56.
Briemle, G., Fink, C., & Hutter, C. P. (1993). Wiese. Weitbrecht Verlag.
Bruppacher, L., Pellet, J., Arlettaz, R., & Humbert, J.-Y. (2016). Simple modifications of mowing regime promote butterflies in extensively managed meadows: Evidence from field-scale experiments. Biological Conservation, 196, 196–202. https://doi.org/10.1016/j.biocon.2016.02.018
Burnham, K. P., & Anderson, D. R. (1998). Model selection and inference. Springer.
Burnham, K. P., & Anderson, D. R. (2002). Model selection and multimodel interference. A practical information—Theoretic approach (2nd ed.). Springer.
Collinge, S. K., Prudic, K. L., & Oliver, J. C. (2003). Effects of local habitat characteristics and landscape context on grassland butterfly diversity. Conservation Biology, 17, 178–187.
Council Directive 92/43/EEC (1992) Council Directive on the conservation of natural habitats and of wild fauna and flora. Official Journal L. 206, 22/07/1992 P. 0007–0050.
Cremene, C., Groza, Gh., Rákosy, L., Schileyko, A. A., Baur, A., Erhardt, A., & Baur, B. (2005). Alterations of steppe-like grasslands in Eastern Europe: A threat to regional biodiversity hotspots. Conservation Biology, 19, 1606–1618.
Crofts, A., & Jefferson, R. G. (Eds.). (1999). The lowland grassland management handbook. English Nature/The Wildlife Trusts.
Culbert, P. D., Dorresteijn, I., Loos, J., Clayton, M. K., Fischer, J., & Kuemmerle, T. (2017). Legacy effects of past land use on current biodiversity in a low-intensity farming landscape in Transylvania (Romania). Landscape Ecology, 32, 429–444. https://doi.org/10.1007/s10980-016-0441-3
Dengler, J., Becker, T., Ruprecht, E., Szabó, A., Becker, U., Beldean, M., & Uğurlu, E. (2012). Festuco-Brometea communities of the Transylvanian Plateau (Romania)—a preliminary overview on syntaxonomy, ecology, and biodiversity. Tuexenia, 32, 319–359.
Dennis, R. L. H., Shreeve, T. G., & Van Dyck, H. (2006). Habitats and resources: The need for a resource-based definition to conserve butterflies. Biodiversity and Conservation, 15, 1943–1966. https://doi.org/10.1007/s10531-005-4314-3
Dihoru, G., & Dihoru, A. (1994) Plante rare, periclitate şi endemice în flora României—Lista Roşie [Rare, threatened and endemic plant species of Romania—the Red List]. Acta Botanica Horti Bucurestiensis 173–199.
Directia Sanitar Veterinara și pentru Siguranța Alimentelor Cluj (2016) Răspuns la solicitarea Societății Lepidopterologice Române privind situația efectivelor de ovine și caprine începând din 2007, din unitățile administrativ teritoriale menționate [Response letter to the Romanian Lepidopterological Society inquiry on the ovine effectives evolution since 2007 in the territorial units of the Natura 2000 site ”Dealurile Clujului de Est”].
Dirnböck, T., Dullinger, S., & Grabherr, G. (2003). A regional impact assessment of climate and land use change on alpine vegetation. Journal of Biogeography, 30, 401–417. https://doi.org/10.1046/j.1365-2699.2003.00839.x
Douwes, P. (1976). An area census method for estimating butterfly population numbers. J Res Lepidoptera, 15, 146–152.
Dover, J. W., Rescia, A., Fungariño, S., et al. (2010). Can hay harvesting detrimentally affect adult butterfly abundance? Journal of Insect Conservation, 14, 413–418. https://doi.org/10.1007/s10841-010-9267-5
Ellingsen, H., Beinlich, B., & Plachter, H. (1998). Large-scale grazing systems and species protection in the eastern Carpathians of Ukraine. La Canada, 9, 10–12.
Erhardt, A. (1985). Diurnal lepidoptera: Sensitive indicators of cultivated and abandoned grassland. Journal of Applied Ecology, 22, 849–861.
Erhardt, A., & Thomas, J. A. (1991). Lepidoptera as indicators of change in the seminatural grasslands of lowland and upland Europe. In N. M. Collins & J. A. Thomas (Eds.), The conservation of insects and their habitats (pp. 213–236). Academic Press.
Ernst, L. M., Tscharntke, T., & Batáry, P. (2017). Grassland management in agricultural vs. forested landscapes drives butterfly and bird diversity. Biological Conservation, 216, 51–59. https://doi.org/10.1016/j.biocon.2017.09.027
Fahrig, L. (2001). How much habitat is enough? Biological Conservation, 100, 65–74. https://doi.org/10.1016/S0006-3207(00)00208-1
Hall, M. L. (1981). Butterfly monitoring scheme: Instructions for independent recorders. Institute of Terrestrial Ecology.
Hanski, I., & Ovaskainen, O. (2000). The metapopulation capacity of a fragmented landscape. Nature, 404, 755.
Hartel, T., Réti, K.-O., Craioveanu, C., Gallé, R., Popa, R., Ioniţă, A., Demeter, L., Rákosy, L., & Czúcz, B. (2016). Rural social–ecological systems navigating institutional transitions: Case study from Transylvania (Romania). Ecosystem Health and Sustainability, 2, 1–12. https://doi.org/10.1002/ehs2.1206
Hill, M. F., & Caswell, H. (1999). Habitat fragmentation and extinction thresholds on fractal landscapes. Ecology Letters, 2, 121–127.
Humbert, J. Y., Ghazoul, J., Sauter, G. J., & Walter, T. (2010). Impact of different meadow mowing techniques on field invertebrates. Journal of Applied Entomology, 134, 592–599.
INSSE (Romanian National Institute for Statistics). (2017). Database AGR201C—Animale ce revin la 100 ha teren, pe principalele categorii de animale, forme de proprietate, macroregiuni, regiuni de dezvoltare si judete, la sfarsitul anului [Animals per 100 ha land, divided by the animal categories, ownership forms, development macro regions and counties at the end of the year]. Free online available database at: http://statistici.insse.ro:8077/tempo-online/#/pages/tables/insse-table. Accessed 23 March 2020.
INSSE (Romanian National Institute for Statistics). (2018). Efective de animale existente la 1 decembrie 2017 [Animal numbers at 1 December 2017]. Press release no. 123/15 May 2018.
Janssens, F., Peeters, A., Tallowin, J. R. B., Bakker, J. P., Bekker, R. M., Fillat, F., & Oomes, M. J. M. (1998). Relationship between soil chemical factors and grassland diversity. Plant and Soil, 202, 69–78.
Johnson, J. B., & Omland, K. S. (2004). Model selection in ecology and evolution. Trends in Ecology and Evolution, 19, 101–108.
Kahmen, S., Poschlod, P., & Schreiber, K.-F. (2002). Conservation management of calcareous grasslands. Changes in plant species composition and response of functional traits during 25 years. Biological Conservation, 104, 319–328.
Kareiva, P., & Wennergren, U. (1995). Connecting landscape patterns to ecosystem and population processes. Nature, 373, 299–302.
Kizos, T., Plieninger, T., & Schaich, H. (2013). “Instead of 40 sheep there are 400”: Traditional grazing practices and landscape change in Western Lesvos, Greece. Landscape Research, 38, 476–498. https://doi.org/10.1080/01426397.2013.783905
Kleijn, D., Kohler, F., Báldi, A., Batáry, P., Concepción, E. D., Clough, Y., Díaz, M., Gabriel, D., Holzschuh, A., Knop, E., et al. (2009). On the relationship between farmland biodiversity and land use intensity in Europe. Proceedings of the Royal Society b: Biological Sciences, 276, 903–909. https://doi.org/10.1098/rspb.2008.1509
Koch, B., Edwards, P. J., Blanckenhorn, W. U., Walter, T., & Hofer, G. (2015). Shrub encroachment affects the diversity of plants, butterflies, and grasshoppers on two swiss subalpine pastures. Arctic, Antarctic, and Alpine Research, 47, 345–357. https://doi.org/10.1657/AAAR0013-093
Köhler, B., Gigon, A., Edwards, P. J., Krüsi, B., Langenauer, R., Lüscher, A., & Ryser, P. (2005). Changes in the species composition and conservation value of limestone grasslands in Northern Switzerland after 22 years of contrasting managements. Perspectives in Plant Ecology, 7, 51–67.
Krebs, J. R., Wilson, J. D., Bradbury, R. B., & Siriwardena, G. M. (1999). The second silent spring? Nature, 400, 611–612.
Kruess, A., & Tscharntke, T. (2002). Contrasting responses of plant and insect diversity to variation in grazing intensity. Biological Conservation, 106, 293–302.
Kühne, I., Arlettaz, R., Pellet, J., Bruppacher, L., & Humbert, J. Y. (2015). Leaving an uncut grass refuge promotes butterfly abundance in extensively managed lowland hay meadows in Switzerland. Conservation Evidence, 12, 25–27.
Kuussaari, M., Heliölä, J., Pöyry, J., & Saarinen, K. (2007). Contrasting trends of butterfly species preferring semi-natural grasslands, field margins and forest edges in northern Europe. Journal of Insect Conservation, 11, 351–366. https://doi.org/10.1007/s10841-006-9052-7
Labaune, C., & Magnin, F. (2002). Pastoral management vs. land abandonment in Mediterranean uplands: Impact on land snail communities. Global Ecol Biogeogr, 11, 237–245.
Laiolo, P., Dondero, F., Ciliento, E., & Rolando, A. (2004). Consequences of pastoral abandonment for the structure and diversity of the alpine avifauna. Journal of Applied Ecology, 41, 294–304.
Le Provost, G., Badenhausser, I., Violle, C., Requier, F., D’Ottavio, M., Roncoroni, M., Gross, L., & Gross, N. (2021). Grassland-to-crop conversion in agricultural landscapes has lasting impact on the trait diversity of bees. Landscape Ecology, 36, 281–295. https://doi.org/10.1007/s10980-020-01141-2
Legendre, P., & Legendre, L. (1998). Numerical ecology. Developments in environmental modelling 20. Elsevier.
Leibold, M. A., Holyoak, M., Mouquet, N., Amarasekare, P., Chase, J. M., Hoopes, M. F., Holt, R. D., Shurin, J. B., Law, R., Tilman, D., Loreau, M., & Gonzalez, A. (2004). The metacommunity concept: A framework for multi-scale community ecology. Ecology Letters, 7, 601–613. https://doi.org/10.1111/j.1461-0248.2004.00608.x
Loos, J., Dorresteijn, I., Hanspach, J., Fust, P., Rákosy, L., & Fischer, J. (2015). Low-intensity agricultural landscapes in transylvania support high butterfly diversity: Implications for conservation. PLoS ONE, 9, e103256. https://doi.org/10.1371/journal.pone.0103256
Loos, J., Turtureanu, P. D., von Wehrden, H., Hanspach, J., Dorresteijn, I., Frink, J. P., & Fischer, J. (2014). Plant diversity in a changing agricultural landscape mosaic in Southern Transylvania (Romania). Agriculture, Ecosystems and Environment, 99, 350–357. https://doi.org/10.1016/j.agee.2014.10.013
Mazerolle, M. J. (2015). AICcmodavg: Model selection and multimodel inference based on (Q)AIC(c). R package version 2.0-3. http://CRAN.R-project.org/package=AICcmodavg.
Moog, D., Poschlod, P., Kahmen, S., & Schreiber, K.-F. (2002). Comparison of species composition between different grassland management treatments after 25 years. Applied Vegetation Science, 5, 99–106. https://doi.org/10.1111/j.1654-109X.2002.tb00539.x
Muntean I, Sitar C, Craioveanu C, Rákosy L (2015) The effect of traditional land use of diurnal lepidoptera from Nature 2000 site “Dealurile Clujului Est”. Studia Universitatis Babeș-Bolyai Biologia 60: 95–105á.
Muru, D., Falqui, A., Schirru, M., Congiatu, P., Virdis, M. R., & Deplano G (2014) Rural lands in protected areas: a methodological approach to the analysis of land use changes. In Proceedings at slow food international congress ed. Cagliari Slow Food, 29–30 May. Cagliari.
Öckinger, E., Eriksson, A. K., & Smith, H. G. (2006a). Effects of grassland abandonment, restoration and management on butterflies and vascular plants. Biological Conservation, 133, 291–300. https://doi.org/10.1016/j.biocon.2006.06.009
Öckinger, E., Hammarstedt, O., Nilsson, S. G., & Smith, H. G. (2006b). The relationship between local extinctions of grassland butterflies and increased soil nitrogen levels. Biological Conservation, 128, 564–573. https://doi.org/10.1016/j.biocon.2005.10.024
Oksanen, J., Blanchet, F. G., Kindt, R., Legendre, L., Minchin, P. R., O’Hara, R. B., Simpson, G. L., Solymos, P., Stevens, M. H. H., & Wagner, H. (2006). Community Ecology Package. R package version 2.0. [Internet] [October 2015]. https://cran.r-project.org/web/packages/vegan/index.html
Oltean, M., & Negrean, G. (1994). Lista roşie a plantelor superioare din România [Red list of vascular plants from Romania]. Studii, Sinteze, Documentaţii De Ecologie, 1, 1–52.
Oppermann, R., Beaufoy, G., & Jones, G. (Eds.). (2012). High Nature Value Farming in Europe. 35 European countries—experiences and perspectives. Verlag Regionalkultur, Ubstadt-Weiher.
Paulini, I., Bărbos, M., Crişan, A., Jitea, I. M., Mihai, V., Moldovan, A., Negoiţă, R., Poledna, R, Rákosy, L., Troc, M., & Schumacher, W. (2011). Grassland conservation through CAP instruments—A Transylvanian case study. 2010 and 2011 summary report of the Mozaic Project. http://www.mozaic-romania.org/media/35626/mozaic%20report_efncp_2010_2011.pdf
Pedersen, E. A., & Widgren, M. (2011). Agriculture in Sweden 800 bc–ad 1000. In J. Myrdal & M. Morell (Eds.), The Agrarian history of Sweden 4000 BC to AD 2000 (pp. 47–71). Nordic Academic Press.
Petersen, S. M., & Drewa, P. B. (2014). Effects of biennial fire and clipping in woody and herbaceous ground layer vegetation: Implications for restoration and management of oak barren ecosystems. Restoration Ecology, 22, 525–533. https://doi.org/10.1111/rec.12102
R Development Core Team (2015) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna; Accessed 15 August 2019. Available from: http://www.R-project.org/
Rákosy, L. (2011). Originea și geneza landșaftului natural-cultural din Transilvania [Origins and genesis of the natural-cultural landscape from Transylvania].In: Rákosy L, Momeu L (Eds.) Volum comemorativ- Bogdan Stugren [Commemorative volume – Bogdan Stugren]. Presa Universitară Clujeană, Cluj-Napoca (pp 25–36).
Rákosy, L. (2013). Fluturii diurni din România. Cunoaștere, protecție, conservare [Butterflies of Romania. Knowledge, protection, conservation]. Editura Mega, Cluj-Napoca.
Regulation (EU) No 1307/2013 of the European Parliament and of the Council of 17 December 2013 establishing rules for direct payments to farmers under support schemes within the framework of the common agricultural policy and repealing Council Regulation (EC) No 637/2008 and Council Regulation (EC) No 73/2009.
Robinson, R., & Sutherland, W. J. (2002). Post-war changes in arable farming and biodiversity in Great Britain. Journal of Applied Ecology, 39, 157–176. https://doi.org/10.1046/j.1365-2664.2002.00695.x
Roem, W. J., & Berendse, F. (2000). Soil acidity and nutrient supply ratio as possible factors determining changes in plant species diversity in grassland and heathland communities. Biological Conservation, 92, 151–161. https://doi.org/10.1016/S0006-3207(99)00049-X
Rosenzweig, M. L. (1995). Species diversity in space and time (1st ed.). Cambridge University Press.
Rudmann-Maurer, K., Weyand, A., Fischer, M., & Stöcklin, J. (2008). The role of landuse and natural determinants for grassland vegetation composition in the Swiss Alps. Basic and Applied Ecology, 9, 494–503. https://doi.org/10.1016/j.baae.2007.08.005
Schmitt, T., & Rákosy, L. (2007). Changes of traditional agrarian landscapes and their conservation implications: A case study of butterflies in Romania. Diversity and Distributions, 13, 855–862. https://doi.org/10.1111/j.1472-4642.2007.00347.x
Soininen, J., McDonald, R., & Hillebrand, H. (2007). The distance decay of similarity in ecological communities. Ecography, 30, 3–12. https://doi.org/10.1111/j.0906-7590.2007.04817.x
Stoate, C., Báldi, A., Beja, P., Boatman, N. D., Herzon, I., van Doorn, A., de Snoo, G. R., Rákosy, L., & Ramwell, C. (2009). Ecological impacts of early 21st century agricultural change in Europe—A review. Journal of Environmental Management, 91, 22–46. https://doi.org/10.1016/j.jenvman.2009.07.005
Tälle, M., Deak, B., Poschlod, P., Valko, O., Westerberg, L., & Milberg, P. (2016). Grazing versus mowing: Meta-analysis of biodiversity benefits for grassland management. Agriculture, Ecosystems and Environment, 222, 200–212. https://doi.org/10.1016/j.agee.2016.02.008
Ter Braak, J. F. C. (1986). Canonical correspondence analysis: A new eigenvector technique for multivariate direct gradient analysis. Ecology, 67, 1167–1179. https://doi.org/10.2307/1938672
Thomas, J. A. (2005). Monitoring change in the abundance and distribution of insects using butterflies and other indicator groups. Philosophical Transactions of the Royal Society b: Biological Sciences, 360, 339–357. https://doi.org/10.1098/rstb.2004.1585
Turtureanu, P. D., Palpurina, S., Becker, T., Dolnik, C., Ruprecht, E., Sutcliffe, L. M. E., Szabó, A., & Dengler, J. (2014). Scale- and taxon-dependent biodiversity patterns of dry grassland vegetation in Transylvania (Romania). Agriculture, Ecosystems and Environment, 182, 15–24. https://doi.org/10.1016/j.agee.2013.10.028
van Elsen, T. (2000). Species diversity as a task for organic agriculture in Europe. Agriculture, Ecosystems and Environment, 77, 101–109. https://doi.org/10.1016/S0167-8809(99)00096-1
Van Swaay, C. A. M., Warren, M. S. (2012). Developing butterflies as indicators in Europe: Current situation and future options. De Vlinderstichting/Dutch Butterfly Conservation, Butterfly Conservation UK, Butterfly Conservation Europe, Wageningen, reportno. VS2012.012.
Wilson, J. B., Peet, R. K., Dengler, J., & Pärtel, M. (2012). Plant species richness: The world records. Journal of Vegetation Science, 23, 796–802. https://doi.org/10.1111/j.1654-1103.2012.01400.x
www.brielmaier.com. Accessed 20 August 2019.
www.ecad.eu. Accessed 20 August 2019.
www.worldclim.org/bioclim. Accessed 20 August 2019.
Acknowledgements
This study was financed through the programme “Partnership in priority areas”—PNII, implemented with the help of the Romanian Ministry of National Education (MEN)—Executive Unit for Financing Higher Education, Research, Development and Innovation (UEFISCDI), Project Number 79/01.07.2014 and through a grant of the Romanian National Authority for Scientific Research and Innovation, CCCDI—UEFISCDI, project number 47/2017, within PNCDI III. During writing the manuscript, ER was supported by the János Bolyai Research Scholarship of the Hungarian Academy of Sciences. We are very grateful to Réka Kiss and Péter Domokos for their invaluable field assistance during the inventory of plant communities. We thank Martyn Davies, Chișu Răzvan and an anonymous referee who brought constructive criticism and helped us with English language corrections.
Funding
This study was financed through the programme “Partnership in priority areas”—PNII, implemented with the help of the Romanian Ministry of National Education (MEN)—Executive Unit for Financing Higher Education, Research, Development and Innovation (UEFISCDI), Project Number 79/01.07.2014 and through a grant of the Romanian National Authority for Scientific Research and Innovation, CCCDI—UEFISCDI, Project Number 47/2017, within PNCDI III. During writing the manuscript, ER was supported by the János Bolyai Research Scholarship of the Hungarian Academy of Sciences.
Author information
Authors and Affiliations
Contributions
CC contributed to conceptualization, methodology, investigation, writing—original draft, writing—review and editing; IM contributed to investigation, writing—original draft, data curation; ER contributed to conceptualization, methodology, investigation, writing—review and editing, supervision; BR-I contributed to methodology, software, formal analysis, writing—review and editing, visualization; AC contributed to methodology, investigation, data curation, writing—review and editing; LR contributed to conceptualization, methodology, writing—review and editing, supervision, funding acquisition.
Corresponding author
Ethics declarations
Conflicts of interest
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Ethical approval
Not applicable.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Craioveanu, C., Muntean, I., Ruprecht, E. et al. Factors affecting butterfly and plant diversity in basiphilous dry grasslands of Transylvania, Romania. COMMUNITY ECOLOGY 22, 295–308 (2021). https://doi.org/10.1007/s42974-021-00055-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42974-021-00055-6