Abstract
The underlying spatial and environmental processes shape the freshwater fish assemblage of streams and rivers. Due to dispersal barriers between the river basins, species filtering is associated with longitudinal environmental gradients resulting in distinct assemblages. This study primarily aims to assess the freshwater fish beta diversity profile inhabiting two rivers of the Upper Brahmaputra basin of Eastern Himalayas, namely River Teesta (large-scale) and Murti (small-scale). The beta profile is further disintegrated into three components, i.e., beta turnover, beta nestedness, and local contribution to beta diversity (LCBD). River Teesta has higher beta diversity and beta turnover values, while River Murti has a higher nestedness in community composition. LCBD is found to be higher in altitudinal extremities, and River Murti seems to have higher values. However, turnover in River Teesta is highly correlated (r > 0.5) with 17 environmental factors, while in River Murti, 15 of them seem to be significantly correlated (r > 0.5). Similarly, nestedness in River Teesta is correlated with stream slope while with water velocity and river width in River Murti.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
According to the current living planet index, the diversity of freshwater species worldwide decreases substantially over time (McRae et al. 2017; Barrett et al. 2018). The largest decline is found in the Neotropical, followed by the Indo-Pacific realm (Barrett et al. 2018). In recent times, such a decline of freshwater fishes is mostly due to habitat degradation and overexploitation, followed by changing climate, increasing pollution, and human footprints (Barrett et al. 2018). Fishes are characteristically different from other organisms in terms of their sensitivity to the environment of freshwater reaches (Leprieur et al. 2009, 2011). They are evolved to exploit different freshwater habitats (Keast and Webb 1966; Hynes and Hynes 1970; Lowe-McConnell 1975; Gorman and Karr 1978). Their community structure is modulated by various factors (Zaret and Rand 1971; Gorman and Karr 1978).
Among the three biodiversity hotspots of South-Asia, the Eastern Himalayan biodiversity hotspot is the largest spreading across 524,190 sq. km through central Nepal to northwest Yunnan in China (Allen 2010; Pathak and Mool 2010). It also encompasses Bhutan, the north-eastern states, and northern Bengal hills in India, south-eastern Tibet, and northern Myanmar (Chettri et al. 2010; Pathak and Mool 2010). Many large and numerous small-scale rivers are flowing downstream through the diverse landscapes of Eastern Himalayas (EH) (Chettri et al. 2010). Amongst 1073 freshwater species in total, ichthyofauna dominates with 520 taxa inhabiting these freshwater reaches (Allen 2010). Such ichthyofaunal assemblages are distributed in three drainage basins, among which the Ganga-Brahmaputra drainage basin is the most diversified and so, the most prioritized for conservation (Allen 2010; Bhatt et al. 2012). River Teesta, the most significant in northern Bengal, and its tributaries are flowing down through the Upper Brahmaputra basin of EH into the River Brahmaputra (Galy et al. 2008; Bhatt et al. 2012; Goswami et al. 2012). Evidently, the altitudinal gradient and water discharge are the most influential factors leading to varied local fish assemblages in this riverine system (Bhatt et al. 2012).
Beta diversity accounts for compositional changes in biotic communities between two given places (Davies et al. 2005). It indicates the turnover/replacement structure in species assemblage while indirectly delineating the formation of biotic regions within the context of regional biota (Davies et al. 2005; Legendre and De Cáceres 2013; 2014). Two additive components, i.e., species turnover and species nestedness (Baselga 2010; Baselga and Orme 2012), have been primarily applied to study beta diversity profiles of species assemblages (Koleff et al. 2003; Anderson et al. 2006; Baselga 2010; Astorga et al. 2014; Edge et al. 2017; Zbinden and Matthews 2017; Antiqueira et al. 2018). In freshwater systems, communities have been found to vary along the environmental and spatial gradients (Holyoak et al. 2005; Heino 2011), followed by their dispersal limitation and niche differences (Hubbell 2001; Chase and Leibold 2003; Heino 2011). Therefore, understanding the influence of environmental variability on beta diversity would explain the underlying species sorting process in freshwater ecosystems.
Several influential research (Chakrabarty and Homechaudhuri 2014, 2015; Debnath 2015; Dey et al. 2015a, b; Sarkar and Pal 2018) previously described different freshwater piscine assemblage of EH. However, a detailed analysis of beta diversity and its relation to environmental variability is still lacking. In Himalayan rivers, rheophilic fish species are reported to dominate headwaters. In contrast, cold-eurythermal species tend to inhabit the lower meandering zones depending upon the scale of the freshwater reaches (Sehgal 1999; Chakrabarty and Homechaudhuri 2013). Large-scale Himalayan rivers also differ significantly from other small-scale torrential rivers in their environmental characteristics, resulting in characteristic fish assemblages (Rudra 2018; Chettri et al. 2010; Panja et al. 2020). In this study, an account of piscine beta diversity along the longitudinal gradients has been compared between two rivers, representing large (Teesta) and small (Murti)-scale freshwater reaches of EH. The underlying processes of fish replacement and nestedness related to environmental variability have been further assessed for understanding the ecosystem response of these two characteristic freshwater rivers.
Methodology
Study area
River Teesta and Murti typically characterize large and small-scale torrential rivers, respectively, in the riverine landscape of EH. River Teesta, originating from north Sikkim, traverses through Sikkim and northern Bengal and then enters Bangladesh to finally merge with the River Brahmaputra (Bhatt et al. 2012; Chakrabarty and Homechaudhuri 2014). This river runs through 309 km with a drainage area of 12540 km2 (Chakrabarty and Homechaudhuri 2014, 2015). In contrast, River Murti is comparatively smaller in scale, originating from the Mo forest in the Neora Valley National Park in Darjeeling Himalayas (Chakraborty and Datta 2013; Kar et al. 2014; Kundu et al. 2019; Panja et al. 2020). It traverses 47.5 km before its confluence with a sizeable Himalayan river Jaldhaka near Gorumara National Park (Chakraborty and Datta 2013; Kar et al. 2014; Kundu et al. 2019). Both these rivers are replenished by snow-melt water from the mountains of Sikkim and Bhutan (Rudra 2016).
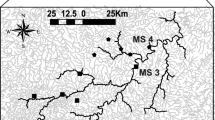
As these rivers are torrential bodies, a longitudinal elevational gradient (Bhatt et al. 2012; Chakrabarty and Homechaudhuri 2014, 2015) in both the rivers was considered for selecting sampling stations. River Teesta being a comparatively large river, a range of 1032–67m elevation gradient was covered. In comparison, a range of 597–100m elevation gradient was studied in River Murti. Therefore, seven sites (considering pool and riffle systems) each along the Teesta (Site 1–7) and the Murti (Site 8–14) river were selected for this study (Fig. 1). However, sampling stations were set up, avoiding those parts of the rivers draining through the protected areas as reserve forests in the region.
Sampling
Sampling for the environmental variables and fish species from the selected sites were conducted from May 2016–April 2018 during pre-monsoon (March–April), monsoon (July–August), and post-monsoon (November–December) seasons. A 90m (45 m upstream and 45 m downstream) stretch was considered at each site. Environmental variables were sampled at three equal distance (30 m) points within this stretch. Fish sampling was performed on the entire 90 m stretch at each sampling site. Freshwater fish abundance was recorded by applying a unified single-pass electrofishing method by anode type Electro Fisher-Fish Machine Shocker connected to a 300V, DC power system. This event was followed by seining through current nets and gill nets (mesh size 2.5 × 2.5 cm). The removal method of fish sampling (Bohlin et al. 1989) was applied and achieved through three consecutive efforts at each 90 m stretch for 1 hour. All the immobilized fishes were identified up to species level following Jhingran and Talwar (1991) and Fish Base (www.fishbase.org) (Froese and Pauly 2011) and released as quickly as possible to the same spot. The observed mortality rate for the captured fishes was 7%.
Seventeen environmental variables addressing five categories, i.e., climate, hydrology, landscape, habitat quality, and anthropogenic pressure, were measured following their relevance to freshwater fish distributions (Edwards and Huryn 1996; Poiani et al. 2000; Hauer and Lamberti 2011; Pettorelli et al. 2011). Under the category of climate, water temperature (°C) (WT), air temperature (°C) (AT), and annual precipitation (mm) (AP) were measured. Five hydrological variables, namely, water velocity (ms-1) (WV), pH (PH), river width (m) (RW), dissolved oxygen (mgl-1) (DO), turbidity (ppm) (TDS) were assessed. The landscape attributes viz. stream order (SO), altitude (m) (AL), topographic wetness index (TWI), and slope (°) (SL) of the sampling sites were computed. For habitat diversity, the normalized difference vegetation index (NDVI), substrate coarseness (SC), and qualitat del bosc de ribera (QBR) index were measured. Furthermore, the basin pressure index (BP) and land surface temperature (°C) (LST) were quantified to address the anthropogenic pressure of the sampling sites. WT, AT, WV, PH, RW, DO, TD, SC, BP, and QBR index were recorded during in-field sampling and averaged to obtain final values.
OAKTON Multiparameter PCSTestr 35 probe was used to record WT, AT, and TDS. pH and DO were recorded following the standard protocol of Water Ecology Kit, Hach Model AL-36B Kit 180202. WV was measured using a propeller-type water current meter (Lawrence and Mayo), while measuring tape (wherever possible, else in GIS) was used to quantify RW. SC of the stream segment was calculated following the Wentworth scale (Wentworth 1922), while QBR Index and BP were calculated following the standard datasheet developed by Colwell (2007) and Hermoso et al. (2009), respectively. However, AP, NDVI, and LST were obtained from secondary sources, i.e., Indian Meteorological Survey (www.imd.gov.in/pages/services_hydromet.php), ISRO Bhuvan Database (https://bhuvan.nrsc.gov.in/bhuvan_links.php), and Climate Engine (http://climateengine.org/), respectively for each of the sampling sites. SO, AL, TWI, and SL were computed in the QGIS platform (Version: QGIS 3.10 A Coruña) using a digital elevation model from the HydroSHEDS database (https://www.hydrosheds.org/downloads).
Data analysis
The presence and absence of fish assemblage were used for data analysis. All the multivariate environmental variables were subjected to a log transformation before analysis.
Multivariate homogeneity among groups
The multivariate homogeneity of groups’ dispersions (Anderson et al. 2006) between River Teesta and Murti was calculated, followed by an analysis of variance (ANOVA). The results were further projected in principle coordinate (PCoA) analysis to represent distances among the rivers in Euclidean space (Anderson et al. 2006).
Computation and partitioning beta diversity
The total beta diversity (BDTotal) as the total variance within each river was calculated along with two other components viz. species contributions to beta diversity (SCBD) and local contributions to beta diversity (LCBD) statistics (Legendre and De Cáceres 2013; Borcard et al. 2018). SCBD values were differentially plotted for these two rivers to find the critical species with a higher contribution in differential community assemblages (Legendre and Legendre 2012). However, LCBD values were mapped for each site to address their comparative evaluation of ecological integrity (Legendre and De Cáceres 2013; Lopes et al. 2014; Szabó et al. 2017). In the next step, total beta diversity was partitioned for each river into two components, i.e., the total spatial turnover (BetaSim) and nestedness (BetaNes) using Sorensen distances (Baselga 2010; Baselga and Orme 2012). Furthermore, the incidence-based pair-wise dissimilarity matrices were calculated for turnover (replacement) and nestedness following their correlation with the environmental distances (Baselga and Orme 2012).
Identification of key environmental parameters
Seventeen environmental variables were used to calculate pair-wise dissimilarity matrices (Clarke and Gorley 2006) for each river. Following dissimilarity, the sites were clustered using group averages for turnover (replacement) and nestedness as well as channeled into non-metric multidimensional plots (nMDS) (Clarke and Gorley 2006). Furthermore, critical variables were identified (Pearson’s correlation value > 0.5) (Clarke and Gorley 2006) and plotted into the same nMDS plot. This analysis would identify the critical environmental factors behind the beta diversity profile of two characteristically different rivers belonging to EH.
All the above analyses were computed in the PRIMER V 6.1.15 platform (Clarke and Gorley 2006) and R platform (Team 2018; 2019) using packages, namely vegan, Betapart and adespatial.
Results
A total of 92 fish species (See Supporting Information) have been found within two river systems. River Teesta and Murti harbor 75 and 41 fish species, respectively, out of a total of 11,700 individuals collected from River Teesta and 3906 individuals from River Murti (see Appendix Tables). The multivariate homogeneity of groups’ dispersions (variances) is prominent and turned out to be significant at a 0.05 level of significance (See Supporting Information). In the PCoA plot (Fig. 2), River Teesta and Murti are distantly plotted with few overlaps. River Teesta has a higher distance from centroid than River Murti (See Supporting Information). The total beta diversity (BDTotal) of the River Teesta is 0.8137, more significant than the small-scale River Murti, i.e., 0.5271.
For River Teesta, higher SCBD values (Appendix A: Table 1) are found for fish species, namely Amblyceps mangois, Olyra kempi, Neolissochilus hexagonolepis, Labeo angra, Schizothorax richardsonii, and Mystus bleekeri. However, in River Murti (Appendix A: Table 2), fish species with higher SCBD values are different. They are Neolissochilus hexagonolepis, Neolissochilus hexastichus, Crossocheilus latius, Devario aequipinnatus, Barilius barila, and Psilorhynchus balitora. Similarly, higher LCBD values (Fig. 3) are observed in sites 1, 2, and 7 for River Teesta. In contrast, sites 8 and 14 in the River Murti, indicating a differential degree of ecological uniqueness within the same water reach.
In additive partitioning of beta diversity, the turnover (BetaSim) component seems to be higher than nestedness (BetaNes) (Fig. 4) in both the rivers, irrespective of their scales. The turnover values are higher (Fig. 4) for River Teesta than Murti. However, River Murti seems to have higher nestedness than the large-scale river, Teesta (Fig. 4). In the nMDS plot, all the 17 environmental factors are positively correlated with the beta turnover pattern in the River Teesta (Fig. 5) and Murti (except TWI and WV) (Fig. 6). However, RW and WV seem to be correlated with the high nestedness pattern observed in the River Murti (Fig. 6). SL seems to be the only factor associated with the observed nested pattern in the large-scale river, Teesta (Fig. 5). Site 4 and 5 have lower turnover distances in River Teesta (Fig. 5), while other sites are distinctly separated due to significant species turnover. In contrast, sites 8–9 and 10–12 are projected with lower turnover distances in River Murti (Fig. 6).
Non-metric multidimensional plot for beta turnover (left) and nestedness (right) of ichthyofaunal assemblage in River Teesta. WT Water temperature, AT air temperature, AP annual precipitation, WV water velocity, PH pH, RW river width, DO dissolved oxygen, TDS turbidity, SO stream order, AL altitude, TWI topographic wetness index, SL slope, NDVI normalized difference vegetation index, SC substrate coarseness, QBR index qualitat del bosc de ribera index, BP basin pressure index, LST land surface temperature
Non-metric multidimensional plot for beta turnover (left) and nestedness (right) of ichthyofaunal assemblage in River Murti. WT Water temperature, AT air temperature, AP annual precipitation, WV water velocity, PH pH, RW river width, DO dissolved oxygen, TDS turbidity, SO stream order, AL altitude, TWI topographic wetness index, SL slope, NDVI normalized difference vegetation index, SC substrate coarseness, QBR index qualitat del bosc de ribera index, BP basin pressure index, LST land surface temperature
Discussion
Detailed analysis of beta diversity provides an understanding of the ecological and evolutionary process of species filtering in natural systems (Davies et al. 2005; Legendre et al. 2005). It is apparent from the study that the transition of regional biota into localized communities is significant (Fig. 2) and differs based on the scale of the water reaches. This inference is in agreement with a previous study indicating the presence of significant variation within-stream community compositions (Al‐Shami et al. 2013) but contrary to a previous finding (Zhou et al. 1999) where beta diversity has been shown to bear no significant differences in three branches of the same river. Since the freshwater basins impose a substantial dispersal barrier (Cottenie 2005; Heino 2011), a significant difference in beta profile between River Teesta and Murti is justified from their difference in diversity scale, freshwater connectivity, stream order, and longitudinal gradients. As is known, the elevation is strongly associated with spatial patterns of species assemblage (Ali et al. 2010; Bhatt et al. 2012). Since both the courses of River Teesta and Murti have varied elevational gradients (Bhatt et al. 2012; Rudra 2018; Panja et al. 2020), their catchment dynamics must have been influenced by the hierarchical intertwined processes (Ali et al. 2010). Therefore, watercourse distance is significant for stream fishes representing successive levels of scale hierarchy (Ali et al. 2010), which filters the localized species assemblages in the system. This study supports the relative contribution of both the spatial process and environmental gradients contingent upon the underlying process of species sorting (Heino et al. 2015).
The total beta diversity value is higher in River Teesta, indicating differential assemblage of fishes following the drainage basin (Heino 2011; Al‐Shami et al. 2013; Zbinden and Matthews 2017). An inference has been drawn from previous studies that altitudinal gradients and water discharge are the significant factors to modulate species richness in these waters (Bhatt et al. 2012). Habitat characteristics have also been assessed by computing a surrogate metric, LCBD, which is indicated to contribute towards resultant beta diversity (Legendre 2014; Lopes et al. 2014; Szabó et al. 2017). High LCBD values generally put a site away from the mean of the sites in terms of species composition because it might contain unusual or rare species within (Legendre 2014). In this study, sites with elevational extremities have comparatively high LCBD values in both the rivers indicating marked differences in community structure. It also confirms the presence of rare or unique species at two elevational levels, which differ from the other parts of the same water reaches. River Murti has comparatively higher LCBD in sites 8 and 14 (Fig. 3), which call for priority consideration for ecological restoration to conserve the unique and rare assemblage of piscine communities (Legendre and De Cáceres 2013).
According to an earlier study (Goswami et al. 2012), fish species belonging to the genus Neolissochilus, Garra, Psilorhynchus, Barilius are highly adaptive species inhabiting freshwater reaches of EH. However, these species have been found to inhibit both these rivers but are assigned higher SCBD values for River Murti. Among them, Neolissochilus hexagonolepis is categorized as red-listed by the International Union for Conservation of Nature (IUCN) (IUCN 2020; Ramirez-Chaves et al. 2015). Therefore, they seem to have delimited distributions and subsequently become habitat specialists in River Murti.
In agreement with the previous studies (Heino 2011; Al‐Shami et al. 2013; Zbinden and Matthews 2017), the present study indicates the spatial turnover component to be significant for the existing beta diversity profile along the longitudinal gradient of these water reaches. The results support the view of a significant association of beta turnover with turbidity, stream order, substrate types, altitude, NDVI, and pH in accordance with the previous studies (Al‐Shami et al. 2013; Zbinden and Matthews 2017). As the headwaters are characterized by varied attitudinal courses followed by merging of anastomosing streams and shift of terrains, the resultant habitat heterogeneity might modulate the turnover of fishes in these torrential waters (Jackson et al. 2001; Heino 2011; 2015; Zbinden and Matthews 2017). As dispersal is limited between freshwater drainage basins (Cottenie 2005; Heino 2011; Laskar et al. 2013), such a turnover may directly correspond with the species sorting following resource availability and environmental heterogeneity (Jackson et al. 2001; Heino et al. 2015).
Our findings establish that the nestedness component of beta diversity is weaker than turnover, similar to previous studies on freshwater organisms (Heino 2011; Zbinden and Matthews 2017). Since the replacement is high in River Teesta, a clear nested structure is untenable and indicates a strong influence of spatial processes. The slope is associated with the observed nestedness, which acts as a proxy for stream dissolved oxygen levels, water flow direction, and accumulation (Austin 2007; Kuemmerlen et al. 2014). Therefore, the observed nestedness pattern (Fig. 5) might arise based on the adaptability of fishes comprehending the stream slope in River Teesta. However, in River Murti, a prominent correlation of water velocity and river width with beta nestedness has been indicated. Such a higher nestedness might arise due to its smaller scale compared to the large river, Teesta.
Characteristic hill-scapes, undulating valleys, climatic conditions of EH has ensued numerous torrential streams and rivers (Kar et al. 2006, 2010; Allen 2010; Chettri et al. 2010; Tse-ring et al. 2010) with rich piscine diversity (Goswami et al. 2012; Vishwanath 2017a; b). Bhatt et al. (2012) inferred the differential impact of drivers modulating richness gradients in terrestrial and aquatic ecosystems of EH. Several relevant studies (Fu et al. 2004; Bhatt et al. 2012, 2016) agree in unison that freshwater fish diversity decreases gradually with elevation while, on the contrary, the endemic and rare species would show an atypical response. In a longitudinal gradient riverscape, the lateral and vertical ecological attributes for freshwater fish assemblages are also relevant (Panja et al. 2020). This study synthesizes and explains beta diversity with its potential additive components and ecological variability following such observations. With the understanding of species sorting, this study could formulate a better prioritization schedule and conservation planning of these freshwater reaches (Leprieur et al. 2009, 2011; Astorga et al. 2014; Edge et al. 2017). The detailed comparison indicates that a fish assemblage of small-scale upland reaches differs from large-scale rivers within the same ecoregion. Small-scale reaches may harbor a significant assemblage of characteristics fish species with habitat specialization, prominent nestedness, and delimited distributions. In contrast, large-scale rivers are more diverse, with beta diversity composition being markedly distinctive as spatial turnover prominently determines the species assemblage.
Conclusion
Hydrological and landscape parameters are contingent on providing a strong filter resulting in a high beta turnover in the fish assemblages of these water reaches. Due to strong spatial structures, large-scale rivers exhibit more heterogeneity through courses and harbors more ecologically fragile sites with unique species assemblages. Therefore, it can be inferred that fishes of torrential waters of EH might exhibit a varied assemblage scale (local to intermediate and regional). As the stream fishes respond to the environmental changes and are highly constrained by dispersal limitation, they are distributed and adapted following the environmental gradients supported by the environmental filtering hypothesis.
Data availability statement
The raw data is not being presently submitted at this moment, so that it cannot be reproduced in any other form before publication of the manuscript. However, it may be shared in the review/revision stage for better analytical clarity during review/ revision.
References
Ali GA, Roy AG, Legendre P (2010) Spatial relationships between soil moisture patterns and topographic variables at multiple scales in a humid temperate forested catchment. Water Resour Res. https://doi.org/10.1029/2009WR008804
Allen DJ (2010) The status and distribution of freshwater biodiversity in the Eastern Himalaya. IUCN
Al-Shami S, Heino J, Che-Salmah M, Abu-Hassan A, Suhaila A, Madrus M (2013) Drivers of beta diversity of macroinvertebrate communities in tropical forest streams. Freshw Biol 58(6):1126–1137
Anderson MJ, Ellingsen KE, McArdle BH (2006) Multivariate dispersion as a measure of beta diversity. Ecol Lett 9(6):683–693
Antiqueira PAP, Petchey OL, dos Santos VP, de Oliveira VM, Romero GQ (2018) Environmental change and predator diversity drive alpha and beta diversity in freshwater macro and microorganisms. Glob Change Biol 24(8):3715–3728
Astorga A, Death R, Death F, Paavola R, Chakraborty M, Muotka T (2014) Habitat heterogeneity drives the geographical distribution of beta diversity: the case of New Zealand stream invertebrates. Ecol Evol 4(13):2693–2702
Austin M (2007) Species distribution models and ecological theory: a critical assessment and some possible new approaches. Ecol Model 200(1–2):1–19
Barrett M, Belward A, Bladen S, Breeze T, Burgess N, Butchart S, Clewclow H, Cornell S, Cottam A, Croft S (2018) Living planet report 2018: aiming higher. WWF, Gland, Switzerland
Baselga A (2010) Partitioning the turnover and nestedness components of beta diversity. Glob Ecol Biogeogr 19(1):134–143
Baselga A, Orme CDL (2012) betapart: an R package for the study of beta diversity. Methods Ecol Evol 3(5):808–812
Bhatt JP, Manish K, Pandit MK (2012) Elevational gradients in fish diversity in the Himalaya: water discharge is the key driver of distribution patterns. PLoS One 7(9):e46237
Bhatt JP, Manish K, Mehta R, Pandit MK (2016) Assessing potential conservation and restoration areas of freshwater fish fauna in the Indian river basins. Environ Manage 57(5):1098–1111
Bohlin T, Hamrin S, Heggberget TG, Rasmussen G, Saltveit SJ (1989) Electrofishing—theory and practice with special emphasis on salmonids. Hydrobiologia 173(1):9–43
Borcard D, Gillet F, Legendre P (2018) Numerical ecology with R. Springer, New York
Chakrabarty M, Homechaudhuri S (2013) Fish guild structure along a longitudinally-determined ecological zonation of Teesta, an eastern Himalayan river in West Bengal India. Arxius de Miscel·lània Zoològica 11:196–213
Chakrabarty M, Homechaudhuri S (2014) Analysis of trophic gradient through environ-mental filter influencing fish assemblage structure of the river Teesta in Eastern Himalayas. J Biodiv Environ Sci 4(4):218–232
Chakrabarty M, Homechaudhuri S (2015) Resource partitioning as determining factor in structuring fish diversity pattern along ecological gradient of River Teesta in Eastern Himalaya. Int J Fish Aquat Stud 2(4):74–80
Chakraborty S, Datta K (2013) Causes and consequences of channel changes–a spatio-temporal analysis using remote sensing and GIS—Jaldhaka-Diana River System (Lower Course), Jalpaiguri (Duars), West Bengal India. J Geogr Nat Disast 3(1):1–13
Chase JM, Leibold MA (2003) Ecological niches: linking classical and contemporary approaches. University of Chicago Press, Chicago
Chettri N, Sharma E, Shakya B, Thapa R, Bajracharya B, Uddin K, Choudhury D and Oli KP (2010) Biodiversity in the Eastern Himalayas: status, trends and vulnerability to climate change. Climate Change Impact and Vulnerability in the Eastern Himalayas, Technical Report-2, International Centre for Integrated Mountain Development (ICIMOD), Kathmandu, Nepal
Clarke K, Gorley R (2006) PRIMER v6: User Manual PRIMER-E. Plymouth, UK.
Colwell S (2007) The application of the QBR Index to the riparian forests of central Ohio streams. The Ohio State University
Cottenie K (2005) Integrating environmental and spatial processes in ecological community dynamics. Ecol Lett 8(11):1175–1182
Davies KF, Chesson P, Harrison S, Inouye BD, Melbourne BA, Rice KJ (2005) Spatial heterogeneity explains the scale dependence of the native–exotic diversity relationship. Ecology 86(6):1602–1610
Debnath S (2015) Present status of ichthyofaunal diversity of Gadadhar River at Cooch Behar District, West Bengal India. Int J Pure App Biosci 3(5):42–49
Dey A, Nur R, Sarkar D, Barat S (2015a) Ichthyofauna Diversity of River Kaljani in Cooch Behar District of West Bengal India. Int J Pure App Biosci 3(1):247–256
Dey A, Sarkar K, Barat S (2015b) Evaluation of fish biodiversity in rivers of three districts of eastern Himalayan region for conservation and sustainability. IJAR 1(9):424–435
Edge CB, Fortin M-J, Jackson DA, Lawrie D, Stanfield L, Shrestha N (2017) Habitat alteration and habitat fragmentation differentially affect beta diversity of stream fish communities. Landsc Ecol 32(3):647–662
Edwards ED, Huryn AD (1996) Effect of riparian land use on contributions of terrestrial invertebrates to streams. Hydrobiologia 337(1–3):151–159
Froese R, Pauly D (2011) FishBase. 2011. World Wide Web electronic publication Available at: http://www.fishbase. org (Accessed 22 February 2011).
Fu C, Wu J, Wang X, Lei G, Chen J (2004) Patterns of diversity, altitudinal range and body size among freshwater fishes in the Yangtze River basin China. Glob Ecol Biogeogr 13(6):543–552
Galy V, France-Lanord C, Lartiges B (2008) Loading and fate of particulate organic carbon from the Himalaya to the Ganga-Brahmaputra delta. Geochim Cosmochim Acta 72(7):1767–1787
Gorman OT, Karr JR (1978) Habitat structure and stream fish communities. Ecology 59(3):507–515
Goswami UC, Basistha SK, Bora D, Shyamkumar K, Saikia B, Changsan K (2012) Fish diversity of North East India, inclusive of the Himalayan and Indo Burma biodiversity hotspots zones: a checklist on their taxonomic status, economic importance, geographical distribution, present status and prevailing threats. Int J Biodiv Conserv 4(15):592–613
Hauer FR, Lamberti G (2011) Methods in stream ecology. Academic Press, Burlington
Heino J (2011) A macroecological perspective of diversity patterns in the freshwater realm. Freshw Biol 56(9):1703–1722
Heino J, Melo AS, Bini LM, Altermatt F, Al-Shami SA, Angeler DG, Bonada N, Brand C, Callisto M, Cottenie K (2015) A comparative analysis reveals weak relationships between ecological factors and beta diversity of stream insect metacommunities at two spatial levels. Ecol Evol 5(6):1235–1248
Hermoso V, Linke S, Prenda J (2009) Identifying priority sites for the conservation of freshwater fish biodiversity in a Mediterranean basin with a high degree of threatened endemics. Hydrobiologia 623(1):127–140
Holyoak M, Leibold MA, Holt RD (2005) Metacommunities: spatial dynamics and ecological communities. University of Chicago Press, Chicago
Hubbell SP (2001) The unified neutral theory of biodiversity and biogeography (MPB-32). Princeton University Press, Princeton and Oxford
Hynes HBN, Hynes H (1970) The ecology of running waters. Liverpool University Press, Liverpool
IUCN (2020) The IUCN Red List of Threatened Species. Version 2020–2. International Union for Conservation of Nature and Natural Resources, Gland, Switzerland.
Jackson DA, Peres-Neto PR, Olden JD (2001) What controls who is where in freshwater fish communities the roles of biotic, abiotic, and spatial factors. Can J Fish Aquat Sci 58(1):157–170
Kar D, Nagarathna A, Ramachandra T, Dey S (2006) Fish diversity and conservation aspects in an aquatic ecosystem in Northeastern India. Zoos Print J 21(7):2308–2315
Kar D, Barbhuiya AH, Das B (2010) Fish diversity, habitat parameters and fish health in wetlands and rivers of North-East India. Wetlands, Biodiv Clim Change 1–68
Kar R, Chakraborty T, Chakraborty C, Ghosh P, Tyagi AK, Singhvi AK (2014) Morpho-sedimentary characteristics of the Quaternary Matiali fan and associated river terraces, Jalpaiguri, India: implications for climatic controls. Geomorphology 227:137–152
Keast A, Webb D (1966) Mouth and body form relative to feeding ecology in the fish fauna of a small lake, Lake Opinicon, Ontario. J Fish Board Can 23(12):1845–1874
Koleff P, Gaston KJ, Lennon JJ (2003) Measuring beta diversity for presence–absence data. J Anim Ecol 72(3):367–382
Kuemmerlen M, Schmalz B, Guse B, Cai Q, Fohrer N, Jähnig SC (2014) Integrating catchment properties in small scale species distribution models of stream macroinvertebrates. Ecol Model 277:77–86
Kundu S, Rath S, Laishram K, Pakrashi A, Das U, Tyagi K, Kumar V, Chandra K (2019) DNA barcoding identified selected ornamental fishes in Murti river of East India. Mitochondrial DNA Part B 4(1):594–598
Laskar BA, Bhattacharjee MJ, Dhar B, Mahadani P, Kundu S, Ghosh SK (2013) The species dilemma of northeast Indian mahseer (Actinopterygii: Cyprinidae): DNA barcoding in clarifying the riddle. PloS one 8(1):e53704
Legendre P (2014) Interpreting the replacement and richness difference components of beta diversity. Glob Ecol Biogeogr 23(11):1324–1334
Legendre P, De Cáceres M (2013) Beta diversity as the variance of community data: dissimilarity coefficients and partitioning. Ecol Lett 16(8):951–963
Legendre P, Legendre L (2012) Numerical ecology. Springer, New York
Legendre P, Borcard D, Peres-Neto PR (2005) Analyzing beta diversity: partitioning the spatial variation of community composition data. Ecol Monogr 75(4):435–450
Leprieur F, Olden JD, Lek S, Brosse S (2009) Contrasting patterns and mechanisms of spatial turnover for native and exotic freshwater fish in Europe. J Biogeogr 36(10):1899–1912
Leprieur F, Tedesco PA, Hugueny B, Beauchard O, Dürr HH, Brosse S, Oberdorff T (2011) Partitioning global patterns of freshwater fish beta diversity reveals contrasting signatures of past climate changes. Ecol Lett 14(4):325–334
Lopes PM, Bini LM, Declerck SA, Farjalla VF, Vieira LC, Bonecker CC, Lansac-Toha FA, Esteves FA, Bozelli RL (2014) Correlates of zooplankton beta diversity in tropical lake systems. PloS One. https://doi.org/10.1371/journal.pone.0109581
Lowe-McConnell RH (1975) Fish communities in tropical freshwaters. Longman
McRae L, Deinet S, Freeman R (2017) The diversity-weighted living planet index: controlling for taxonomic bias in a global biodiversity indicator. PloS One 12(1):e0169156
Panja S, Podder A, Homechaudhuri S (2020) Evaluation of Aquatic Ecological Systems through dynamics of Ichthyofaunal diversity in a Himalayan torrential river, Murti. Limnologica. https://doi.org/10.1016/j.limno.2020.125779
Pathak D, Mool P (2010) Climate change impacts on hazards in the Eastern Himalayas. Climate Change Impact and Vulnerability in the Eastern Himalayas International Centre for Integrated Mountain Development (ICIMOD)., Kathmandu, Nepal
Pettorelli N, Ryan S, Mueller T, Bunnefeld N, Jędrzejewska B, Lima M, Kausrud K (2011) The Normalized Difference Vegetation Index (NDVI): unforeseen successes in animal ecology. Climate Res 46(1):15–27
Poiani KA, Richter BD, Anderson MG, Richter HE (2000) Biodiversity conservation at multiple scales: functional sites, landscapes, and networks. AIBS Bull 50(2):133–146
Ramirez-Chaves H, Tavares V, Torres-Martinez M (2015) Vampyressa melissa. The IUCN red list of threatened species.
Rudra DK (2016) State of India’s river. In: India river week South Asia network on dams, Rivers and People, Delhi, India, pp 1–24
Rudra K (2018) Rivers of the tarai-Doors and barind tract. Rivers of the Ganga-Brahmaputra-Meghna Delta. Springer, Cham, pp 27–47
Sarkar T, Pal J (2018) Diversity and Conservation Status of Ichthyofauna in the river Jaldhaka, West Bengal. Int J Fish Aquat Stud 6(2):339–345
Sehgal K (1999) Coldwater fish and fisheries in the Indian Himalayas: rivers and streams. Fish and fisheries at higher altitudes: Asia. Food Agric Organ UN Tech Pap 385:41–63
Szabó B, Lengyel E, Stenger-Kovács C, Padisák J (2017) Beta-diversity and structuring forces of diatom communities in small lakes of the Carpathian Basin. Acta Biol Plant Agrien 5(1):44–44
Talwar P, Jhingran A (1991) Inland fishes of India and adjacent countries. Oxford & IBH Publishing Co., New Delhi, Bombay, Calcutta
Team R (2019) RStudio: integrated development for R. (42), Boston, MA, http://www.rstudio.com.
Team RC (2018) R: A language and environment for statistical computing. https://cran.r-project.org/.
Tse-ring K, Sharma E, Chettri N, Shrestha AB (2010) Climate change vulnerability of mountain ecosystems in the Eastern Himalayas. In: International centre for integrated mountain development (ICIMOD), Kathmandu, Nepal
Vishwanath W (2017a) Conservation of biodiversity in aquatic ecosystems, specially the fish genetic resources of the Eastern Himalayas. Souvenir 39
Vishwanath W (2017b) Diversity and conservation status of freshwater fishes of the major rivers of northeast India. Aquat Ecosyst Health Manage 20(1–2):86–101
Wentworth CK (1922) A scale of grade and class terms for clastic sediments. J Geol 30(5):377–392
Zaret TM, Rand AS (1971) Competition in tropical stream fishes: support for the competitive exclusion principle. Ecology 52(2):336–342
Zbinden ZD, Matthews WJ (2017) Beta diversity of stream fish assemblages: partitioning variation between spatial and environmental factors. Freshw Biol 62(8):1460–1471
Zhou W, Liu J, Ye X (1999) A comparison of fish beta diversity among three branches of Yuanjiang River System. Yunnan Zool Res 20(2):111–117
Acknowledgements
The authors of this study sincerely acknowledge DST INSPIRE-AORC for the financial support of this research (Sanction No. DST/INSPIRE Fellowship/2016/IF160059). We would also like to acknowledge the Department of Zoology, the University of Calcutta, for providing the facilities and frameworks and sincere gratitude to those locals and anglers of Sikkim, northern Bengal, Dooars ecoregion for their concern, help, and immense supports. We highly appreciate the reviewers for giving valuable inputs, which has improved the content and readability of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that no conflict of interest has been raised during the study and published until this report.
Ethical approval statement
This study has been conducted by following the ethical guidelines endorsed by the University of Calcutta, University Grant Commission, and Govt. of India. No vertebrate animals have been sampled, which are already forbidden to be captured from the wild. No surveys and sampling procedures were extended to the protected areas and the water bodies within. The authors are with this declaring fulfillment of all ethical commitments subjected to this research work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Appendix A
Appendix A
Rights and permissions
About this article
Cite this article
Panja, S., Chakrabarty, M., Podder, A. et al. Comparative assessment of piscine beta diversity profile and key determinant environmental factors in two freshwater rivers of variable spatial scale in Dooars, West Bengal, India. Trop Ecol 62, 589–599 (2021). https://doi.org/10.1007/s42965-021-00171-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42965-021-00171-4