Abstract
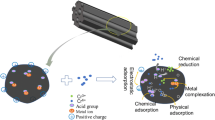
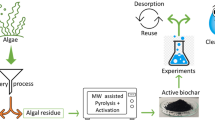
One-pot chemical activation and pyrolysis process was developed for biochar production from red macroalgae residue of Gelidium sesquipedale. The macroalgae residue was activated by various catalysts (KOH, NaOH, H3PO4, and CH4ON2) with the two concentrations (2.5 wt% and 5 wt%) using a pulverization system followed by slow pyrolysis at 500 °C. The activated biochars showed a porous morphology with an increase of water holding capacity compared to the unactivated one. The properties of activated biochar observed by further characterization (i.e., FTIR, SEM, TGA) revealed their feasibility to be used as an adsorbent. The results of adsorption experiment confirmed that adsorption was dependent not only on the surface area but also on the surface charge, and functional groups. The sorption performance of activated biochars (AcBC), in terms of the adsorption of methylene blue, was comparable to commercial activated charcoal (Norit®). NaOH (2.5 wt%)-activated biochar had the removal efficiency of 87% versus 97% for commercial activated charcoal.





Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.References
Aboulkas A, Hammani H, El Achaby M et al (2017) Valorization of algal waste via pyrolysis in a fixed-bed reactor: production and characterization of bio-oil and bio-char. Bioresour Technol 243:400–408. https://doi.org/10.1016/j.biortech.2017.06.098
Aghababaei A, Ncibi MC, Sillanpää M (2017) Optimized removal of oxytetracycline and cadmium from contaminated waters using chemically-activated and pyrolyzed biochars from forest and wood-processing residues. Bioresour Technol 239:28–36. https://doi.org/10.1016/j.biortech.2017.04.119
Ainane T, M’hammed C (2011) Valorisation de la biomasse algale du Maroc : Potentialités pharmacologiques et Ap- plications environnementales, cas des algues brunes Cystoseira tamariscifolia et Bifurcaria bifurcata. Autre. Faculté des Sciences Ben M’sik Université Hassan II Casablanca, 2011. Français. NNT : 213/11. tel-00637002
Alahabadi A, Hosseini-Bandegharaei A, Moussavi G et al (2017) Comparing adsorption properties of NH4Cl-modified activated carbon towards chlortetracycline antibiotic with those of commercial activated carbon. J Mol Liq 232:367–381. https://doi.org/10.1016/j.molliq.2017.02.077
Aluigi A, Rombaldoni F, Tonetti C, Jannoke L (2014) Study of Methylene Blue adsorption on keratin nanofibrous membranes. J Hazard Mater 268:156–165. https://doi.org/10.1016/j.jhazmat.2014.01.012
Bahng MK, Mukarakate C, Robichaud DJ, Nimlos MR (2009) Current technologies for analysis of biomass thermochemical processing: a review. Anal Chim Acta 651:117–138. https://doi.org/10.1016/j.aca.2009.08.016
Barakat A, Monlau F, Solhy A, Carrere H (2015) Mechanical dissociation and fragmentation of lignocellulosic biomass: effect of initial moisture, biochemical and structural proprieties on energy requirement. Appl Energy 142:240–246. https://doi.org/10.1016/j.apenergy.2014.12.076
Carpenter D, Westover TL, Czernik S, Jablonski W (2014) Biomass feedstocks for renewable fuel production: a review of the impacts of feedstock and pretreatment on the yield and product distribution of fast pyrolysis bio-oils and vapors. Green Chem 16:384–406. https://doi.org/10.1039/C3GC41631C
Chen X, Chen G, Chen L et al (2011) Adsorption of copper and zinc by biochars produced from pyrolysis of hardwood and corn straw in aqueous solution. Bioresour Technol. https://doi.org/10.1016/j.biortech.2011.06.078
Dehkhoda AM, Gyenge E, Ellis N (2016) A novel method to tailor the porous structure of KOH-activated biochar and its application in capacitive deionization and energy storage. Biomass Bioenerg. https://doi.org/10.1016/j.biombioe.2016.02.023
Dong X, Ma LQ, Li Y (2011) Characteristics and mechanisms of hexavalent chromium removal by biochar from sugar beet tailing. J Hazard Mater 190:909–915. https://doi.org/10.1016/j.jhazmat.2011.04.008
Fayoud N, Tahiri S, Alami Younssi S et al (2016) Kinetic, isotherm and thermodynamic studies of the adsorption of methylene blue dye onto agro-based cellulosic materials. Desalin Water Treat 57:16611–16625. https://doi.org/10.1080/19443994.2015.1079249
Francavilla M, Manara P, Kamaterou P et al (2014) Cascade approach of red macroalgae Gracilaria gracilis sustainable valorization by extraction of phycobiliproteins and pyrolysis of residue. Bioresour Technol 184:305–313. https://doi.org/10.1016/j.biortech.2014.10.147
González ME, Cea M, Sangaletti N et al (2013) Biochar derived from agricultural and forestry residual biomass: characterization and potential application for enzymes immobilization. J Biobased Mater Bioenergy 7:724–732. https://doi.org/10.1166/jbmb.2013.1373
Inyang M, Dickenson E (2015) The potential role of biochar in the removal of organic and microbial contaminants from potable and reuse water: a review. Chemosphere 134:232–240. https://doi.org/10.1016/j.chemosphere.2015.03.072
Inyang M, Gao B, Zimmerman A et al (2014) Synthesis, characterization, and dye sorption ability of carbon nanotube-biochar nanocomposites. Chem Eng J 236:39–46. https://doi.org/10.1016/j.cej.2013.09.074
Kalderis D, Kayan B, Akay S et al (2017) Adsorption of 2,4-dichlorophenol on paper sludge/wheat husk biochar: process optimization and comparison with biochars prepared from wood chips, sewage sludge and HOG fuel/demolition waste. J Environ Chem Eng. https://doi.org/10.1016/j.jece.2017.04.039
Kim WK, Shim T, Kim YS et al (2013) Characterization of cadmium removal from aqueous solution by biochar produced from a giant Miscanthus at different pyrolytic temperatures. Bioresour Technol 138:266–270. https://doi.org/10.1016/j.biortech.2013.03.186
Lee Y, Park J, Ryu C et al (2013) Comparison of biochar properties from biomass residues produced by slow pyrolysis at 500°C. Bioresour Technol 148:196–201. https://doi.org/10.1016/j.biortech.2013.08.135
Leng L, Yuan X, Zeng G et al (2015) Surface characterization of rice husk bio-char produced by liquefaction and application for cationic dye (Malachite green) adsorption. Fuel 155:77–85. https://doi.org/10.1016/j.fuel.2015.04.019
Li Y, Shao J, Wang X et al (2014) Characterization of modified biochars derived from bamboo pyrolysis and their utilization for target component (furfural) adsorption. Energy Fuels 28:5119–5127. https://doi.org/10.1021/ef500725c
Li G, Zhu W, Zhang C et al (2016) Effect of a magnetic field on the adsorptive removal of methylene blue onto wheat straw biochar. Bioresour Technol 206:16–22. https://doi.org/10.1016/j.biortech.2015.12.087
Li B, Yang L, Wang C et al (2017) Adsorption of Cd(II) from aqueous solutions by rape straw biochar derived from different modification processes. Chemosphere. https://doi.org/10.1016/j.chemosphere.2017.02.061
Li XY, Han D, Xie JF et al (2018) Hierarchical porous activated biochar derived from marine macroalgae wastes (: enteromorpha prolifera): Facile synthesis and its application on Methylene Blue removal. RSC Adv 8:29237–29247. https://doi.org/10.1039/c8ra04929g
Liu SB, Tan XF, Liu YG et al (2016) Production of biochars from Ca impregnated ramie biomass (Boehmeria nivea (L.) Gaud.) and their phosphate removal potential. RSC Adv 6:5871–5880. https://doi.org/10.1039/C5RA22142K
Lonappan L, Rouissi T, Das RK et al (2016) Adsorption of methylene blue on biochar microparticles derived from different waste materials. Waste Manag 49:537–544. https://doi.org/10.1016/j.wasman.2016.01.015
Lyu H, Gao B, He F et al (2018) Experimental and modeling investigations of ball-milled biochar for the removal of aqueous methylene blue. Chem Eng J 335:110–119. https://doi.org/10.1016/j.cej.2017.10.130
Mahmoud DK, Salleh MAM, Karim WAWA et al (2012) Batch adsorption of basic dye using acid treated kenaf fibre char: equilibrium, kinetic and thermodynamic studies. Chem Eng J 181–182:449–457. https://doi.org/10.1016/j.cej.2011.11.116
Manyà JJ, Laguarta S, Ortigosa MA (2013) Study on the biochar yield and heat required during pyrolysis of two-phase olive mill waste. Energy Fuels 27:5931–5939. https://doi.org/10.1021/ef4012388
Mecozzi M, Pietroletti M, Tornambé A (2011) Molecular and structural characteristics in toxic algae cultures of Ostreopsis ovata and Ostreopsis spp. evidenced by FTIR and FTNIR spectroscopy. Spectrochim Acta Part A Mol Biomol Spectrosc 78:1572–1580. https://doi.org/10.1016/j.saa.2011.02.002
Monlau F, Sambusiti C, Antoniou N et al (2015) Pyrochars from bioenergy residue as novel bio-adsorbents for lignocellulosic hydrolysate detoxification. Bioresour Technol 187:379–386. https://doi.org/10.1016/j.biortech.2015.03.137
Oliveira FR, Patel AK, Jaisi DP et al (2017) Environmental application of biochar: current status and perspectives. Bioresour Technol 246:110–122. https://doi.org/10.1016/j.biortech.2017.08.122
Pandey KK (1999) A study of chemical structure of soft and hardwood and wood polymers by FTIR spectroscopy. J Appl Polym Sci 71:1969–1975. https://doi.org/10.1002/(SICI)1097-4628(19990321)71:12%3c1969:AID-APP6%3e3.3.CO;2-4
Putro JN, Soetaredjo FE, Lin S-YY et al (2016) Pretreatment and conversion of lignocellulose biomass into valuable chemicals. RSC Adv 6:46834–46852. https://doi.org/10.1039/C6RA09851G
Puziy AM, Poddubnaya OI, Martínez-Alonso A et al (2002) Synthetic carbons activated with phosphoric—acid I. Surface chemistry and ion binding properties. Carbon N Y 40:1493–1505. https://doi.org/10.1016/S0008-6223(01)00317-7
Sluiter A, Hames B, Ruiz CS R, et al (2008) Determination of structural carbohydrates and lignin in biomass: laboratory analytical procedure (LAP); issue date: April 2008; revision date: July 2011 (version 07-08-2011)—42618.pdf. Tech Rep NREL/TP-510-42618 1–15. NREL/TP-510-42618
Song W, Guo M (2012) Quality variations of poultry litter biochar generated at different pyrolysis temperatures. J Anal Appl Pyrolysis 94:138–145. https://doi.org/10.1016/j.jaap.2011.11.018
Sun L, Wan S, Luo W (2013) Biochars prepared from anaerobic digestion residue, palm bark, and eucalyptus for adsorption of cationic methylene blue dye: characterization, equilibrium, and kinetic studies. Bioresour Technol 140:406–413. https://doi.org/10.1016/j.biortech.2013.04.116
Sun L, Chen D, Shungang Wan ZY (2015) Performance, kinetic, and equilibrium of methylene blue adsorption on biochar derived from eucalyptus saw dust modified with citric, tartaric, and acetic acids. Bioresour Technol 198:300–308
Tan IAW, Ahmad AL, Hameed BH (2008) Adsorption of basic dye on high-surface-area activated carbon prepared from coconut husk: equilibrium, kinetic and thermodynamic studies. J Hazard Mater 154:337–346. https://doi.org/10.1016/j.jhazmat.2007.10.031
Thompson KA, Shimabuku KK, Kearns JP et al (2016) Environmental comparison of biochar and activated carbon for tertiary wastewater treatment. Environ Sci Technol 50:11253–11262. https://doi.org/10.1021/acs.est.6b03239
Xu RK, Xiao SC, Yuan JH, Zhao AZ (2011) Adsorption of methyl violet from aqueous solutions by the biochars derived from crop residues. Bioresour Technol 102:10293. https://doi.org/10.1016/j.biortech.2011.08.089
Yaman S (2004) Pyrolysis of biomass to produce fuels and chemical feedstocks. Energy Convers Manag 45:651–671. https://doi.org/10.1016/S0196-8904(03)00177-8
Yang H, Yan R, Chen H et al (2007) Characteristics of hemicellulose, cellulose and lignin pyrolysis. Fuel 86:1781–1788. https://doi.org/10.1016/j.fuel.2006.12.013
Yao Y, Gao B, Inyang M et al (2011) Biochar derived from anaerobically digested sugar beet tailings: characterization and phosphate removal potential. Bioresour Technol 102:6273–6278. https://doi.org/10.1016/j.biortech.2011.03.006
Yuan JH, Xu RK, Zhang H (2011) The forms of alkalis in the biochar produced from crop residues at different temperatures. Bioresour Technol 102:3488–3497. https://doi.org/10.1016/j.biortech.2010.11.018
Zhang YJ, Xing ZJ, Duan ZK et al (2014) Effects of steam activation on the pore structure and surface chemistry of activated carbon derived from bamboo waste. Appl Surf Sci 315:279–286. https://doi.org/10.1016/j.apsusc.2014.07.126
Zhang Y, Zhu C, Liu F et al (2019) Science of the total environment effects of ionic strength on removal of toxic pollutants from aqueous media with multifarious adsorbents: a review. Sci Total Environ 646:265–279. https://doi.org/10.1016/j.scitotenv.2018.07.279
Zheng W, Guo M, Chow T et al (2010) Sorption properties of greenwaste biochar for two triazine pesticides. J Hazard Mater 181:121–126. https://doi.org/10.1016/j.jhazmat.2010.04.103
Zhu L, Zhao N, Tong L, Lv Y (2018) Structural and adsorption characteristics of potassium carbonate activated biochar. RSC Adv 8:21012–21019. https://doi.org/10.1039/c8ra03335h
Acknowledgements
The authors would like to thank the SETEXAM Company in Morocco for providing the algae residues. This work was based on a formal collaboration between the INRA Montpellier, Mohammed VI Polytechnic University (UM6P), OCP group and the APESA-Pole Valorization (ATLASS Project).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tayibi, S., Monlau, F., Fayoud, Ne. et al. One-pot activation and pyrolysis of Moroccan Gelidium sesquipedale red macroalgae residue: production of an efficient adsorbent biochar. Biochar 1, 401–412 (2019). https://doi.org/10.1007/s42773-019-00033-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42773-019-00033-2