Abstract
The mangrove forests are among the most carbon-rich ecosystems on earth and vital to climate change mitigation. Understanding the relationship of forest structural attributes with carbon stock is essential to achieve sustainable conservation of forest ecosystems and their carbon storage. However, little is known about their inter-relationship in the mangrove forest ecosystems. This study was aimed to quantify primary structural attributes such as density, abundance, frequency, basal area, height, and dbh (diameter at breast height); derived structural attributes such as Importance Value Index (IVI), complexity index (Ic), niche width, and diversity indices (i.e., species richness, β- diversity, Simpson’s index, Shannon-Weiner index, Pielou evenness index); and vegetation biomass, i.e., above ground biomass (AGB) and below ground biomass (BGB) using the allometric equations for mangroves of Bhitarkanika National Park (BNP), India. To achieve the result, twenty-five sample plots (20 m × 20 m) were laid down in BNP which were distributed in six conserve forest sites. The study recorded 27 species, with the highest IVI of 112.19 for Excoecaria agallocha. The Ic value ranged between 12.18 to 283.87 across sites. The diversity indices showed BNP as a mangrove rich ecosystem. The mean stand biomass was 436.89 ± 59.75 t ha−1 (AGB = 318.79 ± 44.42 and BGB = 118.10 ± 15.39). The total forest biomass, biomass carbon stock, and its CO2 equivalents of whole BNP were 6.3 Mt, 2.96 Mt, and 10.87 Mt, respectively. A positive correlation for biomass with the basal area (r = 0.96), stand density (r = 0.81), IVI (r = 0.80) and Ic (r = 0.91) were observed, whereas it showed a negative correlation with species richness (r = − 0.59) and Shannon-Weiner index (r = − 0.08). This study recommends necessity of site specific conservation approach to increase the structural complexity of degraded forest sites and the carbon storage potential of BNP as a large variation in forest attributes and stand biomass exists across sites.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
‘Mangrove forests’ are well recognized as a potential ecosystem for climate change mitigation and stabilizing greenhouse gas emissions. They account for only ~ 0.7% of the tropical forest, but store significantly large amount of atmospheric carbon (called ‘blue carbon’) mostly in the form of tree biomass and sedimentary stock than temperate and tropical ecosystems (Alongi 2008; Donato et al. 2011; Kauffman et al. 2018; Ragavan et al. 2021). Mangrove biomass has two pools of carbon (C), viz., ‘C’ of above ground biomass (AGB) and below ground biomass (BGB) (Walker et al. 2011; Bal and Banerjee 2019; Ragavan et al. 2021). Accurate quantification and understanding of the spatial distribution of these blue carbon stocks are vital to recognize their true potential for climate mitigation and also in strengthening the conservation practice across mangrove-lined nations (Ragavan et al. 2021). The global AGB and BGB have been predicted as 2.83 Pg and 1.11 Pg with a total biomass (TB) of 3.95 Pg (Hutchison et al. 2014). However, Ouyang and Lee (2020) estimated the total carbon stock (living and dead biomass, sediment OC and inorganic carbon) of global mangrove ecosystem at 3.7–6.2 Pg. Mangroves have the highest mean whole ecosystem carbon stock (956 t C ha−1) compared to rainforests (241 t C ha−1), peat swamps (408 t C ha−1), salt marshes (593 t C ha−1), and seagrasses (142.2 t C ha−1) (Alongi 2014; Ouyang and Lee 2020). The Southeast Asian region hosts the highest mangrove diversity, and they have also been recognized as the blue carbon hotspot of the world as they together store at least 1.9 billion tonnes of carbon which is 45% of the world’s total (Suratman 2008; Hamilton and Friess 2018).
In India, mangroves occupy ~ 4992 sq. km (i.e., 2.7% of global cover & 0.15% of the nation’s geographical area) (FSI 2021). Despite of this small area, they have immense contributions in safeguarding the coastal belt, providing many livelihood opportunities and also in storing significantly high ecosystem carbon stock (Sahu et al. 2016; Suresh et al. 2017; Banerjee et al. 2020; Harishma et al. 2020; FSI 2021; Ragavan et al. 2021). However, understanding the forest structure and carbon storage potential of Indian mangrove ecosystem are far from clear and needs immediate evaluation of their potential contribution to national climate change mitigation policies like, Reducing Emissions from Deforestation and forest Degradation in developing countries (REDD +), Nationally Determined Contributions (NDCs),and other blue carbon projects.
Forest community attributes such as floristic composition, structure measurements and stand biomass is essential in mangrove forest management. Generally, the forest complexity is estimated using quantitative attributes, such as tree diversity, density, dominance and plant height (Holdridge et al. 1971). These forest attributes have a significant effect on various biogeochemical processes of the ecosystem and help to understand forest productivity and atmospheric carbon sequestration. Understanding the relationship between forest structural attributes with carbon stock is essential for the sustainable conservation and management of forest carbon stock (Pragasan 2022). Key factors such as basal area, species richness, stand density, species diversity, etc., affect the forest ecosystem carbon stocks besides the environmental and edaphic factors (Aryal et al. 2018; Chaturvedi et al. 2011). However, in mangrove forests, this type of study is very rare. Despite being recognized as the mangrove paradise of the world, the forest structural attributes and tree biomass of BNP, India haven’t been quantified as of other Indian and global mangrove forests. The objectives of this particular study were to quantify the standing biomass and carbon stock of BNP in relation to its structural attributes. The key hypotheses/questions of this study were:
-
a)
What is the forest structure and composition of mangrove stands in BNP?
-
b)
How much vegetation biomass and carbon is stored in BNP?
-
c)
How the contribution of biomass varies among species?
-
d)
Is there any correlation between stand biomass and forest structural attributes?
Materials and methods
Description of the study area
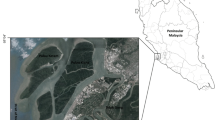
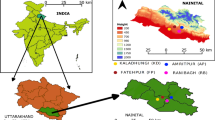
The ‘BNP’ is situated in the Kendrapara district of Odisha state, along the eastern coast of India, between 20◦33’30”N to 20◦50’9”N and 86◦46’15”E to 87◦05’56”E (Fig. 1). This mangrove delta has been formed by rich alluvial deposits of Brahmani, and Baitarani river system. The study site experiences a typical tropical monsoon climate. The mean atmospheric temperature reaches the highest (41.1 °C) and lowest (15.6 °C) in April and December, respectively; whereas the highest monthly rainfall (556.7 mm) is achieved in August and the relative humidity of the site ranges between 54 to 86%.
Data collection and analysis
Forest structure and diversity
To achieve the result, we fixed twenty-five sample plots (size: 20 m × 20 m; area sampled: 10,000 m2) during the period between 2016–2018 that covered six protected conserved mangrove forest sites (Dangmal, Bhitarkanika, Satbhaya, Kalibhanjadiya, Khola, and Habelikhati) of BNP. The sites were selected based on features like feasibility to enter into the forest and habitat diversity (i.e., closeness to river and sea, sites having human interventions, core forest and island forests) (Fig. 1). Plants present within the plots were marked with aluminum tag (Fig. 1). The phytosociological data such as species richness, plant count of each species (excluding the saplings), plant height (h), and circumference at breast height (cbh) were measured at 1.3 m above the ground (for trees and shrubs) by using laser hypsometer and measuring tapes, respectively. The diameter at breast height (dbh) of trees and shrubs was derived using the cbh value (i.e., dbh = cbh/Pi). The existing plants were represented based on dbh and height class. The collected field data were used to quantify the primary structural attributes like plant density (d), abundance (A), frequency (F), basal area (BA); derived structural attributes such as IVI using relative values of density-frequency-dominance as suggested by Curtis and Mclntosh (1950), Curtis (1959), Cintron and Schaffer-Novelli (1984), forest complexity index (Ic) was estimated as suggested by Holdridge (1967) as follows:
where, s = No of species; d = Total stand density; b = Basal area (in m2); h = stand mean height.
The measure of species diversity and distribution is essential to complement the structural attributes of forest communities. Diversity indices are a measure of the number of species in an area and the relative distribution of individuals among those species. We evaluated mean species richness (α-diversity); β-diversity (Whittaker 1972); Simpson’s index (DS) (Simpson 1949); Shannon-Weiner index (H’) (Shannon and Weiner 1963); Pielou evenness index (E) (Pielou 1975); and Niche width (Levins 1968) to understand species evenness, distribution and resource utilization potential of mangroves in BNP. The equations used are as follows:
where, S is the total number of species and α is the mean species richness.
where, \(Pi=\frac{Number\,\, of \,\,individual\,\, of \,\,one\,\, species}{Total \,\,number\,\, of\,\, all\,\, individuals}\)
where, H’ is the Shannon-Weiner index of the community and S is the total number of species in the community.
where, Nij is the density value of species i on stand j.
Estimation of stand biomass and carbon
Total biomass (TB) and total carbon (TC) of a mangrove species are the sum total of above-ground biomass (AGB) with below-ground biomass (BGB) and above ground carbon (AGC) with below-ground carbon (BGC), respectively. To convert dbh to above-ground biomass requires cutting down a sample of trees over a range of sizes, weighing them, and establishing the exponential relationship between dbh and dry weight or biomass. There are also limitations to the accuracy of this method. Each species will have a different relationship between biomass and dbh (Komiyama et al. 2008). To avoid this destructive process, we used the common allometric equations of Komiyama et al. (2005) to estimate the stand and species wise biomass (AGB and BGB) of mangroves in BNP (equations shown below). The required wood specific density values of mangroves were accessed from World Agroforestry Database (Source: http://db.worldagroforestry.org/). Plant dbh values were taken from our plot measurements. We used a conversion factor of 0.47 to convert biomass to carbon (Kauffman and Donato 2012). We quantified the total biomass carbon (AGBC + BGBC) of BNP by extrapolating with average biomass value with the total mangrove forest area, i.e., 14,425 ha (Kripa et al. 2021). The total carbon stock was converted into CO2 equivalents by multiplying with the factor of 3.67 (Kauffman and Donato 2012). The used allometric equations were:
where, D (dbh); and ρ (wood specific density in g cm−3).
Statistical analysis
To understand the interrelation of forest attributes, stand biomass, and carbon stock, we used statistical analysis of these parameters using the software XLSTAT and SPSS 17.0. Mean ± standard error was presented. Pearson’s correlation study was carried out using the results of stand attributes and biomass of mangroves across the sites of BNP to know the existing inter-relation. A regression equation was also developed to represent the relation between IVI and TB.
Results and discussion
Forest structure and species diversity
Altogether, 27 mangrove species from 16 families and 22 genera were recorded from which 22 were woody (Table 1 and Supplementary Table 1). As per the IUCN red list, four species that are categorized as threatened species were Ceriops decandra, Aegialitis rotundifolia, and Phoenix paludosa in the state of near threatened and Heritiera fomes in the endangered category.
The average plant density estimated for overall BNP was 6857 ha−1 and 5159 ha−1 based on the total count and plants with dbh>2.5 cm, respectively (Table 1). Among woody species (i.e., tree and shrub), the highest plant density was recorded for Excoecaria agallocha followed by Heritiera fomes for overall BNP with a site specific variations. The highest relative dominance (RD) was obtained for Excoecaria agallocha (i.e., RD-51.22). However, remarkable variation in stand density (i.e., dbh>2.5 cm) was observed across sites and the value ranged between 908 ha−1 to 7070 ha−1 (Table 1). The basal area (BA) of mangrove stands ranged from 13.96 m2 ha−1 to 69.61 m2 ha−1 across sites (Fig. 2). Four sites such as Dangmal, Bhitarkanika, Kalibhanjadiya, and Habalikhati had pristine mangroves, viz., BA>25 m2 ha−1; Satbhaya had secondary mangrove formation, viz., BA~15 m2 ha−1; and Khola region had disturbed forests, viz., BA < 10 m2 ha−1 (Fig. 2). For BNP, highest IVI was recorded for Excoecaria agallocha (i.e., 112.19), which indicates its dominance (Table 1). Together, the IVI contribution of threatened species was low (~22.33%), viz., Heritiera fomes (10.40%), Ceriops decandra (7.38), Aegialitis rotundifolia (0.52%), and Phoenix paludosa (4.03%). Whereas, 20 species had < 5% IVI, which indicates their rarity in BNP (Table 1). A major portion of mangroves was from a low height and diameter class (Fig. 3a and Fig. 3b), as ~ 25% of plants as a whole were with dbh < 2.5 cm, whereas the value ranged from 2% (Habelikhati) to 84% (Khola) across sites (Fig. 4). Among the 22 woody species, 12 species had all individuals with dbh>2.5 cm, whereas 10 species showed a range between 4 to 18% of their individuals with dbh < 2.5 cm. Only, 52% of mangroves of BNP were with dbh>10 cm, whereas only four species (i.e., Avicennia officinalis, Sonneratia apetala, Avicennia marina, and Lumnitzera racemosa) had dbh>30 cm. Categorization of plants based on height showed that 53% fell within the 5 m class, 31% between 5-10 m, and only 15% had a height > 10 m (Fig. 3b). In total, only 21 plants had a height > 20 m which were from only three species, i.e., Avicennia officinalis, Sonneratia apetala, and Lumnitzera racemosa.
Our estimated d and BA of BNP mangroves was higher than other Indian mangrove forests such as Sundarbans (Joshi and Ghose 2003), Kerala (George et al. 2018; Sreelekshmi et al. 2018; Harishma et al. 2020), Andaman Islands (Kiruba-Sankar et al. 2018; Sreelekshmi et al. 2020), Coringa mangrove estuary, Andhra Pradesh (Satyanarayana et al. 2002), Kachchh, Gujarat (Sawale and Thivakaran 2013), and even to the previous estimation for BNP (Upadhyay and Mishra 2014). In India, the majority of mangrove forests are dominated by species from two genera, ‘Rhizophora’ and Avicennia, whereas Excoecaria agallocha and Heritiera fomes dominate the BNP. Comparing species dominance in terms of IVI of Indian mangrove forests showed that four genera, i.e., Excoecaria, Heritiera, Avicennia, and Ceriops together constitute 70% of the IVI in BNP (this study), whereas three genera viz., Rhizophora, Bruguiera and Ceriops together constituted 67% of the IVI in Andaman Islands (Kiruba-Sankar et al. 2018) and four genera viz., Acanthus, Excoecaria, Avicennia, and Bruguiera together constituted 64% of the IVI in Kerala (Sreelekshmi et al. 2018). Bal and Banerjee (2019) reported Excoecaria agallocha, Heritiera fomes and Avicennia officnalis as major species in BNP, however, they had overestimated the total IVI (i.e., > 300). The IC value is used to describe the structural complexity of forests (Holdridge 1967; Kamruzzaman et al. 2018). Our estimated IC value ranged from 12.18 to 283.87 across sites with a mean value of 132.02 ± 106.14 for BNP (Table 2). This estimation was higher than most of the world’s mangrove forests except Brunei Bay of East Malaysia (Behara et al. 2018), and the Andaman Islands (Kiruba-Sankar et al. 2018). BNP hosts the natural stand with a well-developed forest structure which could be reflected through its high Ic value. The lower value at the Khola region (IC: 12.18) is due to operation of multiple forest loss factors such as stand degradation, development of human pressure and invasion of Acanthus illicifolius. Higher IC value at other sites and altogether for BNP is the result of having pristine mangrove stands with a high value of forest attributes, i.e., d, h, and BA (Table 2; Fig. 2).
In BNP, the diversity indices varied significantly across the sites (Table 2). The overall species richness (or α-diversity) and β-diversity were 5.68 and 4.75 for BNP, respectively (Table 2). Among the sites, maximum β-diversity was recorded from Kalibhanjadiya Island. The high value of β -diversity (i.e., value > 1) resulted from all sites indicated the prevalence of high habitat heterogeneity in the study area. The diversity indices such as H’ (1.91) and DS (0.74) indicated high floral diversity in BNP compared to other mangrove wetlands of India (George et al. 2018; Kiruba-Sankar et al. 2018; Sreelekshmi et al. 2018). However, E (0.58) was lower than other Indian mangrove forests which showed an irregular distribution of mangrove flora inside BNP. We recorded highest niche width for Excoecaria agallocha (12.19) followed by Heritiera fomes (7.64), Avicennia officnalis (7.62), Ceriops decandra (5.78), and Phoenix paludosa (4.81) revealing their better adaptability in the prevailing environment in BNP (Table 1). However, Xylocarpus granatum, Cerbera odollam, Acrostichum aureum, Acrostichum speciosum, Aegiceras corniculatum, Aegialitis rotundifolia, Lumnitzera racemosa, Avicennia marina, Excoecaria indica, and Avicennia alba showed narrow niche width and restricted distribution.
Stand biomass and carbon
Biomass measurement is essential to estimate standing forest carbon and CO2 sequestration potential (IPCC 2013). In BNP, the mean total biomass (TB = AGB + BGB) and mean biomass carbon (BC) stock were 436.89 ± 59.75 t ha−1 and 205.34 ± 28.08 t C ha−1, respectively, whereas the value ranged from 124.46 ± 45.77 t ha−1 to 740 ± 148.54 t ha−1 and 58.48 ± 21.51 t C ha−1 to 347.87 ± 69.82 t C ha−1 across the sites, respectively (Table 2). The mean AGB and BGB for overall BNP were 318.79 ± 44.42 t ha−1 and 118.10 ± 15.39 t ha−1, respectively. The AGB and BGB comprised about 73% and 27% of the total biomass. Our estimated mean TB and BC for BNP was higher compared to many global and Indian mangrove forests indicating its high potential to store atmospheric carbon (Supplementary Table 2). Being a tropical coastal forest, the mangroves of BNP experience high air temperature and rainfall which helped to have higher tree biomass. In BNP, the mangrove stands were represented at different stage of development and the older stands present in Dangmal, Habelikhati, and Bhitarkanika forest sites contributed more to the total biomass. The mangrove biomass and carbon stock of BNP are comparable with Andaman Island mangroves of India (Ragavan et al. 2021). A comparison of species wise contribution to total biomass showed six species (i.e., Excoecaria agallocha, Avicennia officinalis, Heritiera fomes, Ceriops decandra, Lumnitzera racemosa, and Avicennia marina) together contributed 90% of forest biomass (Table 1). Approximately, 36% of C stocks only contributed by Excoecaria agallocha followed by Avicennia officinalis (28.3%) and Heritiera fomes (10.8%) (Table 1; Fig. 5a). This study concluded that the mangrove stands which had higher BA also had high values of TB (Kamruzzaman et al. 2017). Less biogenic disturbances, high structural complexity, mature and older stand, and high precipitation contributes to high vegetation carbon stocks in mangrove forests (Ragavan et al. 2021). The dominance of a given species alone is not a strong determinant of vegetation carbon storage (Harishma et al. 2020). Extrapolating our average biomass value with the total mangrove forested area (i.e., 14,425 ha) resulted in total stand biomass and carbon stock of BNP as 6.3 Mt and 2.96 Mt, respectively (Table 2). The CO2 (or CO2 equivalents) storage value in the form of biomass in BNP was 10.87 Mt. Thus, one hectare loss of natural mangrove forest will lead to the release of 753.59 t CO2 to the atmosphere from BNP. Our estimated value was less than that reported by Banerjee et al. (2020) as they had overestimated the CO2 equivalents value by using the whole sanctuary area of Bhitarkanika, i.e.,672 km2 instead of considering the actual mangrove forested area, i.e., 14,425 ha−1 (Kripa et al. 2021). In India, ‘Odisha state’ recorded to have the highest gain in mangrove cover in 2021 with respect to estimation in 2019 (FSI 2021). Continuation of natural process of mangrove regeneration and plantation along the new intertidal areas and mudflats will help to further increase the blue carbon storage potential of the Odisha state and the BNP.
Correlation study
The correlation relation study revealed that the stand density (r = 0.81), and tree height (r=0.21) were positively correlated to TB (Table 3). BA (r = 0.96) and Ic (r = 0.91) were significantly correlated to TB; stand density (r = 0.88) and BA (r = 0.96) also showed a significant positive correlation with Ic across sites (Table 3). The H’ (r = 0.90) and E had significant positive relation, but H’ (r = − 0.08) was negatively correlated with TB (Table 3). However, this study recorded a negative correlation between species diversity (H’) and biomass. Generally, positive relation is found for forest attributes such as basal area, tree density, maximum diameter, species richness, and species diversity on biomass carbon stock (Kaushal and Baishya 2021). Further, the linear regression study revealed a positive correlation (R2 = 0.803) between IVI and total biomass (Fig. 5b) that indicates species with higher IVI have greater biomass. Similarly, a positive correlation between IVI and AGB has been observed by Alimbon and Manseguiao (2021) for mangrove stands of Panabo Mangrove Park, Philippines.
The major drivers of the changes in the mangrove ecosystem of BNP are population growth, tourism, deforestation, aquaculture, and fertilizer usage (Dhyani et al. 2023). Thus, effective management of these practices, especially regulating aquaculture development, fuel wood collection, and preventing the establishment of new settlements close to mangrove areas not only enhance the natural recruitment and regeneration of mangroves but it will also increase the overall forest structural complexity and carbon storage value.
Conclusion
The present forest structure and biomass carbon stock analysis revealed the existence of high forest complexity and biomass carbon with spatial variation across the forest sites in BNP. Stand structural attributes showed a significant correlation with biomass. The existence of high biomass carbon in BNP indicated as a potential site under the climate action programs, such as NDC and REDD+ for India to reduce carbon emissions from forests. Despite the status of a national park, Bhitarkanika mangrove forests were also experiencing some extent of anthropogenic disturbances which was critically reducing the forest structural complexity and biomass carbon storage capacity. Thus, our assessment of forest attributes, biomass, and carbon pool across sites would help in the implementation of managerial interventions to prevent further loss of forest carbon in BNP.
Data availability
The data generated for the study are included in this article.
References
Alimbon JA, Manseguiao MRS (2021) Species composition, stand characteristics, aboveground biomass, and carbon stock of mangroves in Panabo Mangrove Park, Philippines. Biodiversitas 22:3130–3137
Alongi DM (2008) Mangrove forests: resilience, protection from tsunamis, and responses to global climate change. Estuar Coast Shelf Sci 76:1–13. https://doi.org/10.1016/j.ecss.2007.08.024
Alongi DM (2014) Carbon cycling and storage in mangrove forests. Annu Rev Mar Sci 6:195–219
Aryal S, Shrestha S, Maraseni T et al (2018) Carbon stock and its relationshipswith tree diversity and density in community forests in Nepal. Int for Rev 20(3):263–273
Bal G, Banerjee K (2019) Carbon storage potential of tropical wetland forests of South Asia: a case study from Bhitarkanika Wildlife Sanctuary. India Environ Monit Ass 191:795
Banerjee K, Sahoo CK, Bal G, Mallik K, Paul R, Mitra A (2020) High blue carbon stock in mangrove forests of Eastern India. Trop Ecol 61:150–167. https://doi.org/10.1007/s42965-020-00072-y
Behara S, Aidy MM, Nurul AIH et al (2018) Status of the undisturbed mangroves at Brunei Bay, East Malaysia: a preliminary assessment based on remote sensing and ground-truth observations. Peer J. 6:4397. https://doi.org/10.7717/peerj.4397
Chaturvedi RK, Raghubanshi AS, Singh JS (2011) Carbon density and accumulation in woody species of tropical dry forest in India. For Ecol Manage 262(8):1576–1588. https://doi.org/10.1016/j.foreco.2011.07.006
Cintron G, Schaffer-Novelli YS (1984) Methods for studying mangrove structure. In: Samuel CS, Jane GS (eds) The mangrove ecosystem: research methods. UNESCO publication, Paris, pp 91–113
Curtis JT (1959) The vegetation of Wisconsin. University of Wisconsin Press, Wisconsin, An ordination of plant communities
Curtis JT, Mclntosh RP (1950) The interrelationships of certain analytic and synthetic Phyto-sociological characters. Ecology 31:434–455. https://doi.org/10.2307/1931497
Dhyani S, Shukla J, Kadaverugu R et al (2023) Participatory stakeholder assessment for drivers of mangrove loss to prioritize evidence-based conservation and restoration in bhitarkanika and mahanadi delta. India Sustainabil 15(2):963. https://doi.org/10.3390/su15020963
Donato DC, Kauffman JB, Murdiyarso D et al (2011) Mangroves among the most carbon-rich forests in the tropics. Nat Geosci 4:293–297
FSI (2021) India State of Forest report 2021. Forest Survey of India, Dehradun, India
George G, Krishnan P, Mini KG et al (2018) Structure and regeneration status of mangrove patches along the estuarine and coastal stretches of Kerala. India J for Res 30:507–518
Hamilton SE, Friess DA (2018) Global carbon stocks and potential emissions due to mangrove deforestation from 2000 to 2012. Nat Clim Chang 8:240–244
Harishma KM, Sandeep S, Sreekumar VB (2020) Biomass and carbon stocks in mangrove ecosystems of Kerala, southwest coast of India. Ecol Process 9:31
Holdridge LR (1967) Life zone ecology. Tropical Science Center, San Jose
Holdridge LR, Grenke WC, Hathaway WH, Liang T, Tosi JA (1971) forest environments in tropical life zones: a pilot study. Pergamon Press, New York
Hutchison J, Manica A, Swetnam R et al (2014) Predicting global patterns in mangrove forest biomass. Conserv Lett 7(3):233–240. https://doi.org/10.1111/conl.12060
IPCC (2013) Supplement to the 2006 IPCC guidelines for national greenhouse gas inventories: Wetlands. IPCC, Switzerland
Joshi HG, Ghose M (2003) Forest structure and species distribution along soil salinity and pH gradient in mangrove swamps of the Sundarbans. Trop Ecol 44:197–206
Kamruzzaman M, Ahmed S, Osawa A (2017) Biomass and net primary productivity of mangrove communities along the Oligohaline zone of Sundarbans. Banglad Forest Ecosyst 4:16. https://doi.org/10.1186/s40663-017-0104-0
Kamruzzaman M et al (2018) Stand structure and carbon storage in the oligohaline zone of the Sundarbans mangrove forest, Bangladesh. For Sci Technol 14(1):23–28. https://doi.org/10.1080/21580103.2017.1417920
Kauffman JB, Donato DC (2012) Protocols for the measurement, monitoring, and reporting of structure, biomass and carbon stocks in mangrove forests. CIFOR, Bogor, Indonesia
Kauffman JB, Bernardino AF, Ferreira TO et al (2018) Carbon stocks of mangroves and salt marshes of the Amazon region. Brazil Biol Let 14:4
Kaushal S, Baishya R (2021) Stand structure and species diversity regulate biomass carbon stock under major Central Himalayan forest types of India. Ecol Process 10:14
Kiruba-Sankar R, Krishnan P, Dam Roy S et al (2018) Structural complexity and tree species composition of mangrove forests of the Andaman Islands, India. J Coast Conserv 22:217–234. https://doi.org/10.1007/s11852-017-0588-3
Komiyama A, Poungparn S, Kato S (2005) Common allometric equations for estimating the tree weight of mangroves. J Trop Ecol 21:471–477. https://doi.org/10.1017/S0266467405002476
Komiyama A, Ong JE, Poungparn S (2008) Allometry, biomass, and productivity of mangrove forests: A review. Aquat Bot 89:128–137
Kripa MK, Lele N, Panda M et al (2021) Biodiversity assessment of Indian mangroves using in situ observations and remotely sensed data. Biodiversity 21(4):198–216
Levins R (1968) Evolution in changing environments. Princeton University Press, Princeton
Ouyang X, Lee SY (2020) Improved estimates on global carbon stock and carbon pools in tidal wetlands. Nat Commun 11:317. https://doi.org/10.1038/s41467-019-14120-2
Pielou EC (1975) Ecological Diversity. John Wiley and Sons, New York
Pragasan LA (2022) Tree carbon stock and its relationship to key factors from a tropical hill forest of Tamil Nadu, India. Geol, Ecol Landscap 6(1):32–39
Ragavan P, Kumar S, Kathiresan K et al (2021) Biomass and vegetation carbon stock in mangrove forests of the Andaman Islands, India. Hydrobiologia 848:4673–4693
Sahu SC, Kumar M, Ravindranath NH (2016) Carbon stocks in natural and planted mangrove forests of Mahanadi Mangrove Wetland. East Coast Ind Cur Sci 110(12):2253–2260
Satyanarayana B, Raman AV, Dehair F et al (2002) Mangrove floristic and zonation patterns of Coringa, Kakinada Bay, East coast of India. Wet Ecol Manage 10:25–39
Sawale A, Thivakaran GA (2013) Structural Characteristics of Mangrove Forest of Kachchh, Gujarat. Journal of Marine Biology Assocation of India 55:5–11
Shannon CE, Wiener W (1963) The mathematical theory of communication. University of Illinois Press, Urbana, USA
Simpson EH (1949) Measurement of diversity. Nature 163:688. https://doi.org/10.1038/163688a0
Sreelekshmi S, Preeth CM, Varghese R et al (2018) Diversity, stand structure, and zonation pattern of mangroves in southwest coast of India. J Asia-Pacific Biodiver 11:573–582
Sreelekshmi S, Nandan SB, Sreejith VK, Harikrishnan M (2020) Floristic structure, diversity and edaphic attributes of mangroves of the andaman islands, India. Thalassas 36:47–60
Suratman MN (2008) Carbon sequestration potential of mangroves in southeast Asia. In: Bravo F et al (eds) Managing Forest Ecosystems: The Challenge of Climate Change. Springer, pp 297–314
Suresh HS, Bhat DM, Ravindranath NH, Sukumar R (2017) Carbon stocks and sequestration potential of Indian Mangroves. Trop Ecol 58(3):547–553
Upadhyay VP, Mishra PK (2014) An ecological analysis of mangroves ecosystem of odisha on the eastern coast of India. Proc Indian Natn Sci Acad 80(3):647–661
Walker W, Baccini A, Nepstad M et al (2011) Field guide for forest biomass and carbon estimation. Woods Hole Research Center, Falmouth, Massachusetts, USA
Whittaker RH (1972) Evolution and measurement of species diversity. Taxon 21:213–251
Acknowledgements
We acknowledge our gratitude to Head, Department of Botany, MSCB University, Takatpur, Baripada, Odisha-757003, for his support to carry out the work. We are thankful to PCCF, (Wildlife), Govt. of Odisha for providing permission to conduct the work. We appreciate and thank our esteemed reviewers for providing valuable suggestions to improve the context of the manuscript.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
MTVR, LN, SCS and PM designed the work. MTVR, LN, SRN, PM conducted the field work. PM, LN, and SCS drafted and revised the manuscript. PAK contributed his guidance towards smooth conductance of the work.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no competing interest.
Ethical approval
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Panda, M., Lele, N., Murthy, T.V.R. et al. Assessment of mangrove stand biomass in relation to forest structural attributes in Bhitarkanika National Park, India. Vegetos 37, 950–960 (2024). https://doi.org/10.1007/s42535-023-00630-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42535-023-00630-4