Abstract
Neurological manifestations related to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection may involve both peripheral and central nervous systems, including acute ischemic stroke and cerebral venous sinus thrombosis (CVST). Hereby, we present an adult patient with post-vaccination breakthrough COVID-19 infection and CVST, treated with venous mechanical thrombectomy (MT). The patient manifested continuous tonic-clonic seizures, after an insidious presentation with headache and left-sided weakness. SARS-CoV-2 testing was positive, despite full vaccination, using two approved mRNA platforms. Factor V Leiden polymorphism was detected. The patient was initially managed with anticoagulation, followed by MT with a positive response. We provided a comparison to similar cases of COVID-19-associated CVST undergoing mechanical thrombectomy. High index of suspicion and prompt diagnosis are extremely important to ensure immediate hospitalization and therapy, since CVST associated with either vaccines or COVID-19 seems to evolve rapidly and with a high mortality rate. Even a breakthrough infection may present severe vascular complications. In addition, evaluation of acquired and hereditary thrombophilia may be beneficial in acute phase, also without a previous history of thrombosis. Clinicians should start early medical treatment and additionally consider the endovascular approach as an optimistic choice in refractory CVST related to SARS-CoV-2 infection.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection seems to trigger a pro-thrombotic condition, as it has been hypothesized in course of several cases of cerebral venous sinus thrombosis (CVST) described in the literature [1]. There is still a lack of clarity about the underlying pathogenetic and the most appropriate therapeutic approach. A breakthrough during the SARS-CoV-2 pandemic has been the appearance of new variants of concern, which present a greater transmissibility, although less severe, in patients reinfected or who had received vaccination before the occurrence of new infection with these new variants [2]. Herein, we describe an unusual case of CVST in a patient infected lately with post-vaccination breakthrough COVID-19 who failed to respond to medical treatment and therefore underwent venous mechanical-thrombectomy (MT), showing significant clinical improvement.
Case Report
A patient in their late 30’s was admitted to our hospital with a two-day headache. Neurological assessment was normal. Concomitant SARS-CoV-2 infection, without respiratory symptoms, was diagnosed by nasal swab, but the patient refused hospitalization. He was previously vaccinated against SARS-CoV-2, using the two approved mRNA platforms: Pfizer/BioNTech-BNT162b2-vaccine for the first two doses, whereas the third dose with Moderna-mRNA-1273 was administered 45 days before symptoms onset. After four days from the headache onset, the patient presented again in ER with left upper limb weakness associated to headache and fever and subsequently developed a refractory status epilepticus requiring transfer to an intensive care unit. His medical history was unremarkable for CVST risk factors, and he did not take any domiciliary therapy. The patient was unresponsive to verbal and deep painful stimuli with Glasgow Coma Scale score 3 and was intubated. The estimated National Institutes of Health Stroke Scale (NIHSS) was 29.
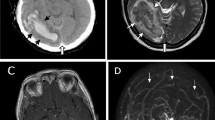
Cerebral computed tomography (CT) and CT angiography (CTA) revealed thrombosis of superior sagittal, transverse, and sigmoid sinus on both sides with bilateral limited venous infarctions in a parietal region. Magnetic resonance angiography (MRA) confirmed severe CVST with bilateral frontoparietal cortical veins and bilateral proximal jugular veins involvement, and more extensive venous infarcts with hemorrhagic transformation (Fig. 1a–c). The laboratory tests showed elevated D-dimer and inflammatory markers. The screening for acquired thrombophilia revealed heterozygosis in polymorphisms of factor V Leiden (FVL), G1691A.
Magnetic resonance imaging (MRI) before and after endovascular treatment (EVT). In the first MRI scan, the axial fluid-attenuated inversion recovery (FLAIR) images (a, b) showed several venous infarctions with hemorrhagic transformation involving the right frontal, parietal and occipital lobes, and the left parietal lobe; the venous MRI phase-contrast angiography (PCA) scans (c) revealed a severe cerebral venous sinus thrombosis (CVST), involving superior sagittal sinus, bilateral sigmoid and transverse sinus, bilateral frontoparietal cortical veins, and bilateral proximal jugular veins. The 30-day MRI follow-up after EVT showed the improvement of neuroradiological findings, with the regular evolution of venous infarctions on axial FLAIR images (d, e). PCA sequences (f) demonstrated a better representation of the cerebral venous system, with the almost complete recanalization of left transverse and superior sagittal sinus
The patient was treated with low-weight molecular heparin (LWMH), anti-epileptic, and anti-edema therapy. The transcranial echo-color-doppler sonography performed in ICU revealed abnormal and asymmetric flow in most intracranial arteries, especially in the left posterior cerebral artery (PCA), showing thrombolysis in brain ischemia (TIBI) grade 2 [3]. Cerebral MRA on second day highlighted neuroradiological findings worsening, and therefore the patient underwent venous MT (eFigure 1; eVideo).
Through a 4F right femoral arterial sheath, preliminary cerebral angiography demonstrated a massive CVST, with patency of the extracranial left internal jugular vein, and partial opacification of the left transverse sinus via superficial cerebral venous system (eFigure 1a; eVideo a). Multiple maneuvers of venous MT were performed in transverse sinus bilaterally and in the superior sagittal sinus, using thromboaspiration catheters (AXS Vecta 74 - Stryker) and stent retrievers (AXS Vecta 74 – Stryker and Aperio Hybrid 6×50mm - Candis, Jet 7 –Penumbra Inc. and CatchView 6×50mm - Balt) (eFigure 1b). As the last attempt, MT was performed by double stent retrievers (Embotrap III 5×37mm - Cerenovus, placed in superior sagittal sinus, and CatchView 6×50mm - Balt, in right trasverse sinus), in combined technique with intermediate aspiration catheter (Jet 7 - Penumbra Inc.) (eFigure 1c). During the endovascular procedure, unfractioned-heparin was administered intravenously. Partial recanalization of both transverse-sigmoid sinus and posterior portion of superior sagittal sinus was achieved at the end of the endovascular procedure, allowing a dramatic improvement of cerebral venous drainage (eFigure 1d; eVideo b). Premises and contraindication for endovascular approach are summarized in eTable.
The patient’s consciousness gradually improved after extubation, without new seizures. Therefore, he started a rehabilitation program and was discharged 30 days after admission (NIHSS-0). Oral anticoagulation with acenocumarol had been started. At 30-day follow-up, no neurological deficits and gradual resolution of CVST (Fig. 1 d–f; eFigures-2-3) were present.
Discussion
Neurological manifestations during SARS-CoV-2 infection may be present in at least one-third of all cases, causing both peripheral and central diseases, including CVST [1].
The European guidelines for the management of CVST suggests LWMH as the main therapy. Nevertheless, around 9%–13% of the patients with non-COVID CVST may require MT, because of worsening despite anticoagulation [4]. Recent studies suggested that CVST has a higher incidence in COVID-19 patients than in the general population, as well as different demographic and clinical characteristics [1, 5, 6]. Wherein this group, two patients underwent MT and both died respectively 1 and 6 days after surgery [5, 7]. More recently, one case of CVST associated with COVID-19 has been described reporting successful thrombectomy results [6]. Table 1 summarizes a comprehensive comparison of our case with the aforementioned three cases. Headache was uniformly seen in all patients. Superior sagittal sinus and other sinuses and superficial and deep veins were involved in all four. The pulmonary circulation was not involved in our patient, as well as in case A [6]. In comparison with the present case, case A presented inflammatory markers slightly more increased and similarly mild severity of SARS-CoV-2 symptoms [6], according to the WHO Clinical Progression Scale [8]. The remaining two patients had remarkably raised D-dimers and neutrophil-to-lymphocyte ratio along with moderate severity of COVID-19 disease [5, 7]. No antiviral therapy was given to our patient for COVID-19, likewise case A. Cases B and C received on-site chemical thrombolysis with alteplase along with MT, differently from ours and case A. MT led to recanalization of superior sagittal sinus in all of the cases. In our case, residual thrombi persisted in both transverse-sigmoid sinus and posterior portion of superior sagittal sinus. We achieved recanalization of cortical veins (eFigures 2-3), in contrast to cases A and C, whereas there was no mention of cortical veins in case B. Neurological deterioration in cases B and C played a critical role.
With our collected data, we excluded platelet dysfunction, coagulation alteration, and the presence of anti-phospholipid antibodies as the possible cause of the CVST. Our patient had a heterozygous mutation in FVL rs6025 variant known as G1691A, causing inherited poor response to the anticoagulant effect of activated Protein C. There is currently lack of studies concerning the prevalence of thrombophilia or the association between thrombophilia’s panel and COVID-19 mortality. Hence, we did not identify any clear pathogenic factor, apart from a breakthrough COVID-19 infection.
In contrast to other cases, our patient received a full vaccination against SARS-CoV-2, including a third dose with a different mRNA vaccine platform. Cerebral vascular events have been reported in association with mRNA vaccine platforms, considering multiple causal factors. However, the risks of most of these events were substantially higher and more prolonged after SARS-CoV-2 infection than after vaccination in the same population [9]. In this specific case, the long interval of time between the administration of the third dose and the onset of symptoms (45-days), in addition to the concurrent SARS-CoV-2 detection, made eventual correlation between CVST and previous vaccination implausible (see also eFigure-4).
In this regard, real-world vaccine efficacy has shown differences across the two approved mRNA platforms, suggesting that subtle variation in immune responses may induce differential protection [10]. Mixing vaccines might both enhance the immune response and increase flexibility for booster dose administration [11]. Nevertheless, our patient acquired SARS-CoV-2 despite three doses of vaccination, using two different mRNA platforms. Recently, Accorsi et al. [12] found that a third dose vaccination was associated with protection against both the Omicron and Delta variants, although less protection for Omicron than for Delta was suggested by higher odds ratios for the association with Omicron infection. Specifically, the appearance of the omicron variant has meant a breakthrough in the SARS-CoV-2 pandemic, because of its features, as greater transmissibility, less severity, and peculiar decrease in anosmia, in patients reinfected or who had received vaccination before infection [13]. According to the above-mentioned considerations, we think that this might be the case in our patient.
Finally, a hyperinflammatory syndrome with breakthrough SARS-CoV-2 infection in a fully vaccinated patient is not a common finding. Multi-inflammatory syndrome in adults (MIS-A) with COVID-19 is a rare entity especially after receiving COVID-19 vaccination. An overwhelming antibody-mediated immune activation could result from enhanced serologic response in patients who are vaccinated and have breakthrough COVID-19 infection [14]. Concerning our case, the CDC clinical and laboratory criteria for MIS-A were not fulfilled, because of the absence of severe cardiac illness [15].
In conclusion, CVST associated with COVID-19 may result from a prothrombotic inflammatory state caused by anti-SARS-CoV-2 immune response targeting vessel endothelium. Evaluation of acquired and hereditary thrombophilia in COVID-19 patients, also without a previous history of thrombosis, may be beneficial in an acute phase. CVST associated with either vaccines or COVID-19 seems to evolve much more rapidly and with a higher mortality rate than CVST with different etiology. Even a breakthrough infection with mild severity of SARS-CoV-2 symptoms may present severe vascular complications. High index of suspicion and prompt diagnosis are extremely important to ensure immediate hospitalization and therapy. Clinicians should start early treatment with anticoagulation and additionally consider the endovascular approach as an optimistic choice in refractory CVST.
Data Availability
Anonymized data not published within this article will be made available by request from any qualified investigator.
Code Availability
Not applicable.
References
Baldini T, Asioli GM, Romoli M, Carvalho Dias M, Schulte EC, Hauer L, et al. Cerebral venous thrombosis and severe acute respiratory syndrome coronavirus-2 infection: a systematic review and meta-analysis. Eur J Neurol. 2021;28(10):3478–90.
Accorsi EK, Britton A, Fleming-Dutra KE, Smith ZR, Shang N, Derado G, et al. Association between 3 doses of mRNA COVID-19 vaccine and symptomatic infection caused by the SARS-CoV-2 omicron and delta variants. JAMA. 2022;327(7):639–51. https://pubmed.ncbi.nlm.nih.gov/35060999/
Demchuk AM, Scott Burgin W, Christou I, Felberg RA, Barber PA, Hill MD, et al. Thrombolysis in brain ischemia (TIBI) transcranial Doppler flow grades predict clinical severity, early recovery, and mortality in patients treated with intravenous tissue plasminogen activator. Stroke. 2001;32(1):89–93.
Ferro JM, Bousser MG, Canhão P, Coutinho JM, Crassard I, Dentali F, et al. European Stroke Organization guideline for the diagnosis and treatment of cerebral venous thrombosis – endorsed by the European Academy of Neurology. Eur J Neurol. 2017;24(10):1203–13.
Ostovan VR, Foroughi R, Rostami M, Almasi-Dooghaee M, Esmaili M, Bidaki AA, et al. Cerebral venous sinus thrombosis associated with COVID-19: a case series and literature review. J Neurol. 2021;268(10):3549–60.
Sajjad A, Khan AF, Jafri L, Kamal AK. Successful endovascular mechanical thrombectomy in anticoagulation-resistant COVID-19 associated cerebral venous sinus thrombosis. BMJ Case Rep. 2021;14(12) https://pubmed.ncbi.nlm.nih.gov/34972772/
Cavalcanti DD, Raz E, Shapiro M, Dehkharghani S, Yaghi S, Lillemoe K, et al. Cerebral venous thrombosis associated with COVID-19. Am J Neuroradiol. 2020;41(8):1370–6.
Marshall JC, Murthy S, Diaz J, Adhikari N, Angus DC, Arabi YM, et al. A minimal common outcome measure set for COVID-19 clinical research. Lancet Infect Dis. 2020;20(8):e192–7.
Tobaiqy M, Maclure K, Elkout H, Stewart D. Thrombotic adverse events reported for Moderna, Pfizer and Oxford-AstraZeneca COVID-19 vaccines: comparison of occurrence and clinical outcomes in the EudraVigilance Database. Vaccines. 2021;9(11):1326. https://pubmed.ncbi.nlm.nih.gov/34835256/
Kaplonek P, Cizmeci D, Fischinger S, Collier A, Suscovich T, Linde C, et al. mRNA-1273 and BNT162b2 COVID-19 vaccines elicit antibodies with differences in Fc-mediated effector functions. Sci Transl Med. 2022;14(645):eabm2311.
Atmar RL, Lyke KE, Deming ME, Jackson LA, Branche AR, El Sahly HM, et al. Homologous and heterologous COVID-19 booster vaccinations. N Engl J Med. 2022;386(11):1046–57. https://doi.org/10.1056/NEJMoa2116414.
Accorsi EK, Britton A, Fleming-Dutra KE, Smith ZR, Shang N, Derado G, et al. Association between 3 doses of mRNA COVID-19 vaccine and symptomatic infection caused by the SARS-CoV-2 omicron and delta variants. JAMA. 2022;327(7):639–51. https://jamanetwork.com/journals/jama/fullarticle/2788485
Rodriguez-Sevilla JJ, Güerri-Fernádez R, Bertran RB. Is there less alteration of smell sensation in patients with omicron SARS-CoV-2 variant infection? Front Med. 2022;12(9):1044.
Narvel H, Kaur A, Seo J, Kumar A. Multisystem inflammatory syndrome in adults or hemophagocytic lymphohistiocytosis: a clinical conundrum in fully vaccinated adults with breakthrough COVID-19 infections. Cureus. 2022;14(2):1–12.
Vogel TP, Top KA, Karatzios C, Hilmers DC, Tapia LI, Moceri P, et al. Multisystem inflammatory syndrome in children and adults (MIS-C/A): case definition & guidelines for data collection, analysis, and presentation of immunization safety data. Vaccine. 2021;39(22):3037–49.
Acknowledgements
We thank the patient and the patient’s family for their kind cooperation. We would like to thank all professionals involved in stroke care during the pandemic.
Author information
Authors and Affiliations
Contributions
FGi, FGr, and ATe contributed to the study design. F Gi, AG, and LF performed data collections. ATe and DI performed imaging analysis. AAC, MV, and OB performed endovascular treatment. FGi and AG wrote the article. FGr reviewed and critiqued the manuscript. ATo, PLS, and SLV supervised the research.
Corresponding author
Ethics declarations
Ethics Approval
All procedures performed in the studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The paper does not report on primary research. Our analysis looked retrospectively at outcomes for a large cohort of patients treated. All data analyzed were collected as part of routine diagnosis and treatment.
Consent to Participate
A handwritten signature was obtained from the patient.
Consent for Publication
A handwritten signature was obtained from the patient.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on COVID-19
Supplementary Information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Giammello, F., Grillo, F., Tessitore, A. et al. Management of Severe Cerebral Venous Sinus Thrombosis After Post-vaccination Breakthrough COVID-19 Infection: A Case Report and Review of the Literature. SN Compr. Clin. Med. 5, 167 (2023). https://doi.org/10.1007/s42399-023-01506-z
Accepted:
Published:
DOI: https://doi.org/10.1007/s42399-023-01506-z