Abstract
Seed borne diseases caused by fungal pathogens are one of the most important threats that reduce the quantity and quality of cumin production every year all over the world. Hence, the first step in this research was to identify the seed-borne fungi obtained from seeds of native populations of Iranian cumin (Cuminum cyminum L.), their effect on germination and vigor indices, the location of fungi in seeds and their transmission into seedlings. Based on morphological and molecular characters, the fungi were identified as Alternaria alternata, A. burnsii, Fusarium oxysporum and F. solani. Most of the identified isolates were present in the seed coat, while a few isolates of A. alternata was located in embryos. The results revealed that a negative correlation were found between incidences of naturally fungi infected seed, germination, and vigor indices. The pathogenicity test revealed that 20% of the isolates were non-pathogenic and other isolates were pathogenic or weakly pathogenic. In addition, the percentage seed transmission of fungal phytopathogens to cumin seedlings revealed that A. alternata was the more frequently isolated (67.27%) followed by F. oxysporum (52.80%), A. burnsii (32.74%) and F. solani (20.01%). Then, we investigated correlation among the activities of cell wall degrading enzymes (CWDEs) produced by fungal isolates and variation in pathogenicity and aggressiveness in vivo. Different levels of pathogenicity and aggressiveness were observed for various isolates of Fusarium and Alternaria species. Analysis of CWDEs activities, including cellulase, xylanase, pectinase and lipase showed that cellulase and lipase activities were more important than xylanase and pectinase activities for the pathogenicity of isolates. Therefore, these findings suggested that activity levels of cellulase and lipase are correlated with variation in pathogenicity and aggressiveness of seed-borne fungal isolates on seedling. This is the first report on isolation and identification of seed-borne fungi infection of cumin, together with investigating association between pathogenesis and CWDEs production by isolates isolated from seeds of Iranian cumin native populations. It is also the first time that the location of fungi in seeds and its seed-transmitted into cumin seedlings have been identified and evaluated, respectively.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cumin (Cuminum cyminum L.), is an important seed spice crop with medicinal properties and is believed to be a native of the Mediterranean and Near Eastern regions. In 2019, world production of cumin was about 700,000 thousand tons and the major producing countries include: India, Iran, Syria and Turkey contributing up to 85% of world production. Cumin is cultivated on about 18,000 thousand hectares with a production of 9512 thousand ton in Iran. Khorassan-Razavi province is one of the most important producer of cumin, accounting for about 55% of Iranian cumin production with cultivated area of 10,694 thousand hectares and production of 5281 thousand ton (Ghasemi et al. 2019). Cumin production and growth is seriously affected by various stress factors including diseases like Alternaria blight and vascular wilt which are devastating diseases that occur in major cumin growing areas of the world. Alternaria alternata and A. burnsii are the causal agents of Alternaria blight of cumin, which after flowering stage affects cumin plant and causes complete failure of the cumin crop. Fusarium vascular wilt is caused by several species of Fusarium, but mainly by Fusarium oxysporum f. sp. cumini and F. solani, and results in yield losses of up to 80% (Sharma et al. 2013). Fusarium species are distributed via seeds and soil, while Alternaria sp. is seed-borne and seed-transmitted pathogen.
Seed health plays an important role in increasing the quantity and quality of cumin yield. Seed health refers specifically to the severity and incidence of pathogens in seed and seeds play a vital role in carrying pathogens. Fungal pathogens are one of the most important living factors affecting seed health that may be internally, externally seed-borne and also associated with seeds. Seed-borne fungal pathogens have some deleterious effects on seed such as reducing seed viability, seed necrosis, seed rot, rotting, reduction or elimination of vigor and germination capacity, variation in plant morphology, wilting and blight of seedlings (Singh et al. 2011).
So far, the presence of different species of Alternaria and Fusarium associated with cumin have been identified in world which including Alternaria alternata, A. tenuis, A. infectoria, A. raphani, Fusarium oxysporum, F. solani, F. acuminatum, F. avenaceum, F. equiseti and F. sambucinum (Özer and Bayraktar 2015; Sharma et al. 2013; Beygtash 2013; Ramchandra and Bhatt 2012; Mohammadi and Nooras 2009).
Plant cell wall as the first layer of plant defense is one of the barriers that pathogens need to overcome in order to colonize the host and spread disease. Fungal pathogens directly with short infection-hyphae (for Fusarium spp.) and appressoria-like structures (for Alternaria spp.) penetrate cell walls or can enter through natural openings and wounds (Boedo et al. 2007).
The ability to instead of produce various cell wall degrading enzymes (CWDEs), as the major type of pathogenicity factors, is necessary for fungal pathogens to penetrate into plant cell wall (Khaledi and Hassani 2018). Aggressiveness is an important factor of pathogenic adaptability since it indicates the potential ability of pathogens to cause epidemics and induce the disease in a smaller scale of time to the host plants (Lannou 2012). The production of extracellular enzymes is one of the mechanisms for pathogenicity and aggressiveness of Fusarium and Alternaria species, which enable to penetrate and invade into the host plant seedlings and to cause wilt and blight disease symptoms (Cho et al. 2009).
According to reports by other researchers, Fusarium and Alternaria species are associated with cumin seeds which reduce germination and vigor indices of seedlings (Fatima and Khot 2015; Suthar et al. 2014; Kumar 2005). Crop rotation, planting uninfected and healthy seeds, and chemical and biological control as management strategies have been used to cumin biological control have been used for the management of cumin diseases (Sharma et al. 2013).
Despite the economic importance of diseases in cumin production, little information is available about the seed-borne fungi which cause yield losses in Iranian cumin native populations. On the other hand, our knowledge on CWDEs produced by seed-borne fungi of cumin and their association with pathogenicity and aggressiveness are very limited. Therefore, the main objectives of this study were to identify and determine: (i) the frequency of seed-borne fungal species isolated from cumin seeds, (ii) the effect of seed-borne fungi on germination and vigor indices, (iii) the location of fungi in seeds and its transmission into seedlings, (iv) analyze pathogenicity, aggressiveness and to evaluate production of enzyme activities of fungal isolates and to identify possible correlation.
Materials and methods
Sample collection
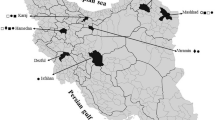
We have randomly sampled the cumin fields with the highest area under cultivation in different regions including Mashhad, Quchan and Fariman (belonging to Khorassan-Razavi province) after harvest in 2019–2020. The sampling was done according to the International Seed Testing Association guidelines (ISTA 2013). The seed samples were packed in a paper envelope and stored in a freezer at the Seed and Plant Certification and Registration Institute (SPCRI) in Iran until used for subsequent studies.
Isolation of fungi
The agar plate method was used to detect seed-borne fungi associated with cumin seeds. The seeds were surface-sterilized in 1% sodium hypochlorite solution for one minute and rinsed with sterile distilled water. Seeds were plated in petri dishes containing potato dextrose agar (PDA) amended with 25 mg L−1 streptomycin sulphate. Petri dishes were incubated for 10 days at 25 ± 1 °C under alternating cycles of 12 h light and darkness.
Morphological identification of fungi
The different fungal colonies on the PDA retrieved from the seeds were identified to the genus level using a stereomicroscope. Emerged fungi were isolated and purified using the single spore technique and/or the hyphal tip method. Fungal cultures identified as Alternaria sp. was cultured on potato carrot agar (PCA) and cultures identified as Fusarium sp. transferred onto carnation leaf agar (CLA). Identification of fungi was carried out by using standard keys as described by Simmons (2007), and Leslie and Summerell (2006).
Molecular identification of isolated fungi
To confirm the morphological identification of the isolated fungi at the species level, conventional polymerase chain reaction (PCR) was performed using specific primers (Table 1). The DNA extraction was performed by DNA extraction kit (Genomic DNA isolation kit IV; DENA Zist Asia, Iran) according to the manufacturer’s instructions. For each reaction, 3 μl (50 ng μL−1) of template DNA was mixed with 12.5 μL of Master Mix (Pishgam, Iran), 1 μL of 2.5 μM of ach species-specific primers each forward and reverse primers and 7.5 μL of sterile water.
The PCR cycle consisted of an initial denaturation at 95 °C for 10 min and then 40 cycles of denaturation (95 °C for 40 s), annealing (at specific annealing temperatures and times listed in Table 1), extension (72 °C for 60 s) and a final extension at 72 °C for 10 min. Amplification products were separated by electrophoresis (80–90 V, 60 mA, 100 W, 30 min) in 1.5% agarose gels in 1 TAE buffer and visualized by SYBR Green staining on a UV gel documentation system.
Data analysis
According to the results of morphological and molecular analysis, the isolation frequency (Fr), relative density (RD) and incidence (In) of fungi were recorded and calculated as follow:
where ns is the number of samples in which a fungus occurred, N is the total number of samples, ni is the number of isolates of a given fungal species, Ni is the total number of fungal isolates obtained, n is the number of infected seeds, Tn is the total number of seeds.
Standard germination test
Seed germination tests were performed in accordance with the International Seed Testing Association (ISTA 2013) rules by following a modified between paper method. After a 14 day incubation period, analysis was done for the germination percentage, normal and abnormal (deformed and diseased) seedlings, shoot and root lengths, fresh and dry weight were determined (by placing them in the oven for 24 h at 75 °C). Also, seedling length vigor index (SLVI) and seedling weight vigor index (SWVI) relations were determined by the following equations;
Plant materials
Iranian cumin native population obtained from the Seed and Plant Improvement Institute of Karaj, Iran, was used for pathogenicity tests. Surface-sterilized seeds were sown in each of the prepared pots and grown in a greenhouse (25 ± 3 °C; 16/8 h light/dark photoperiod). The soil used in this experiment was a combination of peat moss, vermiculite and perlite at the ratio of 2: 1: 1 (v/v/v). For sterilization, the soil was autoclaved at 121 °C for a minimum of 30 min at 100 kPa (15 psi) on 2 successive days.
Inoculum preparation
The fungal inoculum was produced in spezieller nährstoffarmer agar (SNA) medium for Fusarium isolates and PCA medium for Alternaria isolates using the methods described by Khaledi et al. (2017) and Koch et al. (2013), respectively. Conidial suspensions were diluted with sterile distilled water to a final concentration of 1 × 105 conidia mL−1 containing 0.05% (v/v) Tween 20.
Pathogenicity test
To confirm Koch’s postulates, isolates were subjected to pathogenicity test on cumin under controlled conditions. Inoculation was carried out by 10 mL of spore suspension (1 × 105 conidia mL−1) amended with Tween 20 was sprayed onto the leaves of cumin seedlings (Pryor and Gilbertson 2002). Cumin seedlings inoculated with the isolates of Fusarium and Alternaria species were grown for 40 and 15 days, respectively. Control plants were treated only with sterile distilled water. Symptoms of like Alternaria blight and Fusarium wilt were evaluated on seedlings after 10 days of incubation. Diseases severities were rated based on a five-point and six-point scale as described by Gour and Agrawal (1988) and Pryor and Gilbertson (2002), respectively. Finally, the percent disease index was calculated by the following formula:
Assessment of aggressiveness
The aggressiveness of each isolate on seedlings was determined using the method described by Hassani et al. (2019) and Khaledi et al. (2017) based on determining hours post inoculation (hpi) for the appearance of symptoms, such as discoloration, chlorosis and necrosis.
Histopathological and seed-transmitted tests
The location of identified isolates in cumin seed was studied by employing the component plating technique (Maden et al. 1975). Naturally infected cumin seed samples were used for the study.
A seed-transmitted test was carried out using the method described by Mangwende et al. (2018). After surface sterilization, twenty cumin seeds were sown singly in a greenhouse (25 ± 3 °C; 16/8 h light/dark photoperiod, with humidity of 75–85%). Disease severity and index values were calculated as mentioned above. Isolations of the causal organism were made showing symptoms of the disease and plated in petri dishes containing PDA and incubated at conditions mentioned above. Isolated fungi were purified and morphological and molecular identification was done as described above.
Enzymatic activities analysis of pathogenicity factors
Activity of several CWDEs (including cellulase, xylanase, pectinase and lipase) were evaluated in this study within 7 days in vitro. Fungal cultures were prepared for each fungal isolate in basic culture media of cellulase, xylanase, pectinase and lipase activities as described by Khaledi and Hassani (2018), and Ortega et al. (2013). Then, cellulase, xylanase, pectinase and lipase activities were investigated using the method of Wood and Bhat (1988), Khanna and Gauri (1993), Colowich (1995), Ortega et al. (2013), respectively.
The absorbance of cellulase and xylanase activities were spectrophotometrically measured at 550 nm and 540 nm, respectively, and also the amount of reducing sugar released was calculated from the standard curve of glucose. One unit of cellulase and/or xylanase activity was defined as the amount of the enzyme that catalyzed 1.0 µ mol of glucose and/or xylose per minute during the hydrolysis reaction. The absorbance of pectinase activity was measured spectrophotometrically at 540 nm and standard curve was drawn based on the absorbance in different concentrations (µg mL−1) of d-galacturonic acid. Then, unit of pectinase activity was defined as the amount of enzyme that released 1 μ mol of galacturonic acid per minute according to the standard curve. Lipase hydrolytic activity was measured spectrophotometrically at 440 nm with p-nitrophenyl palmitate (p-NPP) and one unit of lipase activity was defined as the amount of enzyme that releases 1 µ mol of p-NPP per minute under the above mentioned reaction conditions.
Statistical analysis
All experiments were set up in a completely randomized design with four replicates and conducted twice. The data were analyzed by one-way analysis of variance (ANOVA) and comparison of means was carried out using the least significant difference (LSD) at the level of P < 0.05. Statistical analysis was performed using SAS software (version 9.2; SAS Institute, Cary, NC, USA).
Results
Morphological and molecular identification of isolated fungi
In this study, a total of 45 fungal isolates were recovered from cumin fields in different regions of Mashhad, Quchan and Fariman. Morphological observations showed that all isolates were classified into two genera, including of Alternaria and Fusarium. Then, based on morphological and molecular criteria using the identification keys and species-specific PCR assay, a total of four fungal species were isolated including Alternaria alternata, A. burnsii, Fusarium oxysporum and F. solani. The morphological identifications were confirmed by PCR amplification of DNA isolates using specific primers (Table 1). F. oxysporum identification was confirmed by using the primer specific to Fusarium oxysporum f. sp. cumini described by Leslie and Summerell (2006), and according to the protocol of Bayraktar and Dolar (2011).
Distribution, frequency and density of isolates
Fungi-infected seeds were observed in all studied seed samples. Among different cumin samples of Iranian cumin native populations in the investigated regions, Mashhad farms showed the highest distribution of fungal isolates. F. oxysporum f. sp. cumini associated with infected seeds was detected in all sampling regions except sample in Fariman region. Alternaria spp. was detected from some fields in Mashhad region. Alternaria spp. was detected form some fields in Quchan and Fariman regions. Among the identified species, A. alternata was the most frequent followed by F. oxysporum, F. solani and A. burnsii (Tables 2, 3).
In our study, the highest and lowest incidences of fungi-infected seeds were observed in samples from Mashhad and Fariman regions, respectively. The results showed that A. alternata was the predominant species with the highest incidence and relative density of 11.7 and 77.8%, respectively (Table 2). Cumin seeds of Mashhad cumin native population had the highest average infected by fungi while the Fariman cumin native population had the lowest average infected by fungi (Tables 3, 5).
Germination and vigor indices of cumin seeds
The results of seed germination tests as affected by natural fungal infection are presented in Table 3. The seed germination percentage ranged from 35.50% to 81.75 in all samples. Standard germination results showed that the percentage of disease seedlings was less than 7.5%; moreover, the percent of deformed seedlings was less than 4% for all samples. The SLVI and SWVI of the seedlings varied from 863.54 to 4657.71 and from 0.71 to 10.16, respectively. Sample ZM110 (belonging to Mashhad region) showed the lowest vigor indices. The shoot and root lengths of the seedlings varied from 8.37 to 24.57 cm and from 15.95 to 32.40 cm, respectively. The fresh and dry weights of the seedlings varied from 0.19 to 0.44 g and from 0.02 to 0.12 g, respectively. The lowest seed germination, shoot and root lengths, fresh and dry weights were observed on sample ZM110 (Table 3).
Location of fungi in seeds and seed-transmitted of fungi pathogens
The location of fungi in the seed are presented in Table 4. All identified isolates were located in seed coats. Only A. alternata was recorded in embryos of seeds (Table 4). Sowing naturally infected seeds showed that not all fungal isolates isolated from seeds are transmission into seedlings. Isolation from tissue infected and morphological and molecular identification showed that transmission of A. alternata (67.27%), A. burnsii (32.74%), F. oxysporum (52.80%) and F. solani (20.01%) were found to be the pathogens and thereby fulfilling Koch’s postulates (Table 4).
Pathogenicity test
A comparison of the data obtained from pathogenicity of fungal isolates isolated in cumin seeds revealed that significant differences occurred the pathogenicity of the different isolates. The pathogenicity test revealed that twenty-seven of the thirty-five isolates of A. alternata and four of the five isolates of F. oxysporum f. sp. cumini were found to be pathogenic or weakly pathogenic and the others were non-pathogenic. All identified isolates of F. solani and A. burnsii were found to be pathogenic or weakly pathogenic. Significant differences in disease index were observed among isolates (any species or genus) tested. The AA14 isolate (belonging to A. alternata) caused the highest pathogenicity on seedlings. The pathogenicity test showed that the disease index of pathogenic and weakly pathogenic isolates for A. alternata ranged from 9.51 ± 0.14 to 85.12 ± 0.86, for A. burnsii from 16.59 ± 0.21 to 23.62 ± 0.64, for F. oxysporum f. sp. cumini from 21.65 ± 0.43 to 53.22 ± 1.03 and for F. solani of 12.74 ± 0.71 (Table 5).
Aggressiveness assay
The results of aggressiveness of Alternaria isolates on seedlings showed that the highest and lowest aggressiveness belonged to the AA14 (120 hpi) and AA18 (240 hpi) isolate, respectively. In overall, the isolate AA14 which showed the highest averages of disease index on seedlings, had also the highest level of aggressiveness (Table 5). Infection assays on seedlings revealed various levels of aggressiveness for fungal isolates. In addition, aggressiveness variation among isolates of Fusarium showed that that the highest and lowest aggressiveness belonged to the FO4 (186 hpi) and FS1 (228 hpi) isolate, respectively (Table 5).
Analysis of CWDEs activity
Analysis of CWDEs showed that not all isolates tested were capable of producing cellulase, xylanase, pectinase and lipase, as the main hydrolytic enzymes involved in pathogenicity of isolates pathogenic or weakly pathogenic. These results suggested that all isolates pathogenic or weakly pathogenic were capable of producing cellulase and lipase (Table 5). Investigating the activities of CWDEs showed that different isolates tested had various CWDEs activity. Level of CWDEs activity among the tested isolates varied from 154.50 to 1068.25 µg mL−1 for cellulase, 158.50 to 1374.25 µg mL−1 for xylanase, 1651 to 2886 µg mL−1 for pectinase, and 21.06 to 30.58 µg mL−1 for lipase (Table 5). The isolate AA14 isolate had maximum of CWDEs activity in vitro compared to other isolates tested. The lowest levels of xylanase and pectinase activities were observed for the AA34 isolate (belonging to A. alternata). The lowest cellulase and lipase activity belonged to the AA18 isolate. Among all CWDEs testes, the highest activity levels were observed for pectinase, followed by xylanase, cellulase and lipase (Table 5).
The AA14 and AA18 isolates, which showed the highest and lowest levels of CWDEs activity had considerably the highest and lowest levels of pathogenicity and aggressiveness in bioassays on seedling, respectively (Table 5). Isolates non-pathogenic only produced low levels of pectinase and xylanase compared to isolates pathogenic or weakly (Table 5). Analysis of CWDEs showed that cellulase and lipase play an important role in pathogenicity by degrading the plant cell wall.
Discussion
In this study, a total of 45 fungal isolates obtained from different regions of Iran cumin fields with the highest area under cultivation were identified using molecular and morphological characters. This is the first report on identification of seed-borne fungal species in native Iranian cumin cultivar. In addition, the relationship among pathogenicity, aggressiveness, and activity of CWDEs produced by seed-borne fungi of cumin seeds were evaluated and compared. Furthermore, the location of fungi in seeds, its seed transmission into seedlings and pathogenicity were determined. We evaluated the effect of seed-borne fungi on germination and vigor indices of cumin seeds.
Based on morphological observations and molecular analyses, all isolates belonging to four species were isolated from cumin seed samples. Thirty-five isolates were identified as isolates of A. alternata, 5 isolates as F. oxysporum f. sp. cumini, 4 isolates as A. burnsii and one isolate as F. solani. Özer and Bayraktar (2015) observed that the main species associated with cumin production of Turkey are A. alternata, A. infectoria, A. burnsii, F. oxysporum f.sp. cumini, F. solani, F. avenaceum, F. equiseti, F. acuminatum, and F. sambucinum, which is supported by our results. Similar results of morphological and molecular identification of fungal species were in accordance with the reports of Khare et al. (2014), Beygtash (2013), and Kumar (2005). So far, F. solani, F. oxysporum f. sp. cumini and A. burnsii from Khorassan-Razavi and Fars provinces have been reported on cumin in Iran.
This diversity of seed-borne fungi may be attributed to cultures of the sensitive Iranian cumin native population, varying environmental conditions, crop rotation with sensitive crops and also not seed disinfection with fungicide cause the high incidences of fungi-infected seeds in the Mashhad region of Iran. This finding is in accordance with the finding of Beygtash (2013), who reported that the frequency and relative density of fungal infection on Iranian cumin native populations highly varied from region to region. Results similar to our findings were previously reported in Turkey (Özer and Bayraktar 2015), and India (Khare et al. 2014).
Overall, all of samples were found to be affected by natural fungal infection in cumin seeds. Our results showed that the highest incidence of natural fungal infection was observed in Mashhad region, followed by Quchan and Fariman regions. Beygtash (2013) reported that Khorassan-Razavi province, where Mashhad region is located had the highest percentage of infected samples which is in accordance with our data.
In this study, 15% incidences of natural fungal infection were recorded in different regions of Iran cumin fields with the highest area under cultivation. The current study revealed that A. alternata and F. oxysporum were the most frequent species (100 and 66.7%, respectively), with high (11.7%) and moderate incidence (1.7%), and with relative densities of 77.8 and 11.1%, respectively, on cumin seeds (Table 2). On the basis of these results, the A. alternata and F. graminearum were the two main species associated with cumin seeds in Iran as previously demonstrated by Beygtash (2013). Results similar to our findings were obtained in India (Singh et al. 2016; Fatima and Khot 2015; Sharma et al. 2013; Kumar 2005), Turkey (Özer and Bayraktar 2015) and Egypt (Hashem et al. 2010). To our knowledge, A. alternata and F. oxysporum f. sp. cumini were found as the main limiting factor in cumin cultivation of Iran, especially in the Khorassan-Razavi province.
The results showed that growth indicators used in this research (shoot and root lengths, fresh and dry weights) decreased with increased natural fungal infection in cumin seeds. Similar results were obtained by Suthar et al. (2014), who observed reduced germination and vigor indices of seedlings of cumin associated with infection by F. equiseti. The number of abnormal (deformed and diseased) seedlings increased significantly in samples that contained more infected seeds. Similar results were reported by Mangwende et al. (2018), who observed that a positive correlation was found between the incidence of A. alternata and the percentage of abnormal seedlings. Hashem et al. (2010) reported that germination and vigor indices were decreased due to increased fungi-infected seeds, which is supported by our results. Similarly, Kumar (2005) reported that the percentage germination of cumin seeds infected by fungi decreased with germination and vigor indices. Also, Fatima and Khot (2015) showed that the fungi associated with cumin seeds significantly affected seed germination and seedling growth, which is in accordance with our observation.
This is the first time the location of fungi pathogens in cumin seeds has been identified. All isolates examined in this study were observed in the seed coat. However, we observed that among the species of fungi detected in cumin seeds, A. alternata in the embryo of seeds. The appearance of disease symptoms on cumin seedlings and its re-isolation from diseased plants showed that the 77.14% of A. alternata isolates, 80% of F. oxysporum isolates; and also all A. burnsii and F. solani isolates to be pathogenic and proved seed-transmitted into seedlings. Previous studies have shown that the location and transmission of fungi pathogens in naturally infected black cumin (Elwakil and Ghoneem 1999), wheat (Hassani et al. 2019), coriander (Mangwende et al. 2018), sorghum and foxtail millet seeds (Yago et al. 2011).
Our results suggest that these pathogens affect the seedlings and cause a decline in growth indicators. The results of our research about the pathogenicity of Alternaria and Fusarium isolates on cumin showed that 79.49% and 83.33% of the isolates were pathogenic and non-pathogenic, respectively, and differences in pathogenicity were found. These findings were in accordance with the results reported by Sharma et al. (2013) and Hashem et al. (2010). Thirty-six (about 80%) of 45 isolates were pathogenic or weakly pathogenic in cumin. The fungal isolates recovered from cumin seeds revealed differences in their pathogenicity. The results showed that isolate AA14 of A. alternata caused the highest disease progress with an average disease index of 85.12 ± 0.86 and also the earliest time of disease symptom after inoculation 120 hpi. In our investigations, the aggressiveness of the fungal isolates varied as much among genus and species and also within a species as among species. This is in accordance with the results obtained by Hassani et al. (2019), and Khaledi et al. (2017). Overall, significant differences in pathogenicity were observed among the seed-borne fungal isolates, which is evidence of differences in aggressiveness.
Alternaria spp. and Fusarium spp. as necrotrophic pathogens utilize a variety of pathogenicity factors especially production of extracellular enzymes throughout the infection process (Khaledi et al. 2017; Cho et al. 2009). The secreted cellulase and lipase produced by Fusarium species is one of important of pathogenicity factors (Khaledi et al. 2017). Comparing CWDEs activities, pathogenicity and aggressiveness of the isolates showed that pectinase and xylanase had no effect on pathogenicity compared to cellulase and lipase. Isolates pathogenic or weakly pathogenic produced considerably higher levels of cellulase, xylanase, pectinase and lipase compared to isolates non-pathogenic. Fungal isolates without the ability of producing cellulase and lipase were unable to cause severe disease in cumin seedlings, but same isolates produced pectinase and xylanase. Similar results were reported by Hubballi et al. (2011), who reported that non-pathogenic isolates produced pectinolytic enzymes (macerating enzymes, pectin methyl esterase and endo polygalacturonase).
According to the results reported by other researchers on different phytopathogens strong association has been found between pathogenicity and amount of extracellular CWDEs activities of isolates (Khaledi et al. 2017; Ortega et al. 2013), as observed in this study. Maximum activities of CWDEs studied were observed for the isolate AA14 of A. alternata, which showed highest levels of pathogenicity and aggressiveness, which is parallel to other reports (Hassani et al. 2019; Khaledi et al. 2017). Therefore, we found a correlation among both pathogenicity and aggressiveness on seedlings and CWDEs produced by fungal isolates in vitro. These findings were in accordance with the results obtained by Hassani et al. (2019) and Khaledi et al. (2017).
In summary, this study indicates that seed-borne fungi caused a reduction in germination, seedling vigor and quality seed. The use of healthy seeds helps to maintain the value traits of Iranian cumin native populations and to protect the cumin from seed-borne fungal infection. Knowledge about seed-borne fungi of cumin and effect it’s on germination and vigor indices could be useful for choosing effective disease management strategies to reduce destructive effects of disease transmitted to seedlings and increase the production and quality of cumin seeds. Moreover, pathogenicity and factors affecting it could be useful for plant breeders in the screening of cumin native populations in the discovery of resistance sources for biotic stresses.
References
Bayraktar H, Dolar FS (2011) Molecular identification and genetic diversity of Fusarium species associated with onion fields in Turkey. J Phytopathol 159:28–34. https://doi.org/10.1111/j.1439-0434.2010.01715.x
Beygtash S (2013) Identification of seedborne fungal pathogenes of cumin and caraway and biological control of dominant pathogens. Masters thesis, University of Zabol
Boedo C, Le Clerc V, Briard M, Simoneau P, Chevalier M, Georgeault S, Poupard P (2007) Impact of carrot resistance on development of the Alternaria leaf blight pathogen (Alternaria dauci). Eur J Plant Pathol 121:55–66. https://doi.org/10.1007/s10658-007-9241-6
Cho Y, Kim KH, Rota ML, Scott D, Santopietro G, Callihan M, Mitchell TK, Lawrenc CB (2009) Identification of novel virulence factors associated with signal transduction pathways in Alternaria brassicicola. J Mol Microbiol 72:1316–1333. https://doi.org/10.1111/j.1365-2958.2009.06689.x
Colowich SP (1995) Methods in enzymology. Academic Press Inc., London
Elwakil MA, Ghoneem K (1999) Detection and location of seed-borne fungi of black cumin and their transmission in seedlings. Pak J Biol Sci 2:559–564
Fatima S, Khot YC (2015) Studies on fungal population of cumin (Nigella sativa L.) from different parts of Marathwada. J Multidiscip Res 2:25–31
Ghasemi G, Fattahi M, Alirezalu A, Ghosta Y (2019) Antioxidant and antifungal activities of a new chemovar of cumin (Cuminum cyminum L.). Food Sci Biotechnol 28:669–677. https://doi.org/10.1007/s10068-018-0506-y
Gour HN, Agrawal S (1988) A wilt toxin from Fusarium oxysporum f. sp. cumini Patel and Prasad. Curr Sci 57:849–851
Hashem M, Moharam AM, Zaied AA (2010) Efficacy of essential oils in the control of cumin root rot disease caused by Fusarium spp. Crop Prot 29:1111–1117. https://doi.org/10.1016/j.cropro.2010.04.020
Hassani F, Zare L, Khaledi N (2019) Evaluation of germination and vigor indices associated with fusarium-infected seeds in pre-basic seeds wheat fields. J Plant Prot Res 59:69–85. https://doi.org/10.24425/jppr.2019.126037
Hubballi M, Sornakili A, Nakkeeran S, Anand T, Raguchander T (2011) Virulence of Alternaria alternate infecting noni associated with production of cell wall degrading enzymes. J Plant Prot Res 51:87–92. https://doi.org/10.2478/v10045-011-0016-x
[ISTA] International Seed Testing Association (2013) The germination test. In: “International Rules for Seed Testing”. ISTA, Bassersdorf, p 56. https://doi.org/10.15258/istarules.2017.05
Khaledi N, Hassani F (2018) Antifungal activity of Bunium persicum essential oil and its constituents on growth and pathogenesis of Colletotrichum lindemuthianum. J Plant Prot Res 58:431–441. https://doi.org/10.24425/jppr.2018.124646
Khaledi N, Taheri P, Falahati-Rastegar M (2017) Evaluation of resistance and the role of some defense responses in wheat cultivars to Fusarium head blight. J Plant Prot Res 57:396–408. https://doi.org/10.1515/jppr-2017-0054
Khanna S, Gauri A (1993) Regulation, purification, and properties of xylanase from Cellulomonas fimi. Enzyme Microb Technol 15:990–995. https://doi.org/10.1016/0141-0229(93)90177-4
Khare MN, Tiwari SP, Sharma YK (2014) Disease problems in the cultivation of Cumin (Cuminum cyminum L.) and Caraway (Carumcarvi L.) and their management leading to the production of high quality pathogen free seed. Int J Seed Spices 4:1–8
Koch A, Kumar N, Weber L, Keller H, Imani J, Kogel KH (2013) Host-induced gene silencing of cytochrome P450 lanosterol C14α-demethylase-encoding genes confers strong resistance to Fusarium species. Proc Natl Acad Sci USA 110:19324–19329. https://doi.org/10.1073/pnas.1306373110
Kumar S (2005) Seed mycoflora of cumin (Cuminum cyminum L.) and their management. Masters thesis, Maharana Pratap University of Agriculture and Technology, Udaipur
Lannou C (2012) Variation and selection of quantitative traits in plant pathogens. Annu Rev Phytopathol 50:319–338. https://doi.org/10.1146/annurev-phyto-081211-173031
Leslie JF, Summerell BA (2006) The Fusarium laboratory manual, 1st edn. Blackwell Publishing Ltd, London
Maden S, Singh D, Mathur SB, Neergard P (1975) Detection and location of seed borne inoculum of Ascochyta rabei and its transmission in chickpea. Seed Sci Technol 3:667–671
Mangwende E, Kritzinger Q, Aveling AST (2018) Control of Alternaria leaf spot of coriander in organic farming. Eur J Plant Pathol 152:409–416. https://doi.org/10.1007/s10658-019-01682-6
Mohammadi A, Nooras NM (2009) Genetic diversity in population of Fusarium solani from cumin in Iran. J Plant Prot Res 49:283–286. https://doi.org/10.2478/v10045-009-0045-x
Ortega LM, Kikot GE, Astoreca AL, Alconada TM (2013) Screening of Fusarium graminearum isolates for enzymes extracellular and deoxynivalenol production. J Mycol 358140:1–7. https://doi.org/10.1155/2013/358140
Özer G, Bayraktar H (2015) Determination of fungal pathogens associated with Cuminum cyminumin Turkey. Plant Protect Sci 51:74–79. https://doi.org/10.17221/51/2014-PPS
Pryor BM, Gilbertson RL (2002) Relationship and taxonomic status of Alternaria radicina, A. carotiinclatae and A. petroselini based on morphological, biochemical and molecular characteristics. Mycologia 94:49–61. https://doi.org/10.1080/15572536.2003.11833248
Ramchandra S, Bhatt PN (2012) First report of Fusarium equiseti causing vascular wilt of cumin in India. Plant Dis 96:1821. https://doi.org/10.1094/PDIS-03-12-0236-PDN
Sharma YK, Choudappa PC, Anwer MM (2013) Efficacy of fungicides for the management of Alternaria blight of cumin. Int J Seed Spices 3:48–49
Simmons EG (2007) Alternaria: an identification manual. CBS, Utrecht, p 775
Singh J, Shikha SS, Sinha A, Bose B (2011) Studies on seed mycoflora of wheat (Triticum aestivum L.) treated with potassium nitrate and its effect on germination during storage. Res J Seed Sci 4:148–156. https://doi.org/10.3923/rjss.2011.148.156
Singh S, Bairwa SK, Trivedi A, Meena RK, Meena SC, Prasad D (2016) Morphological and molecular variability in Alternaria burnsii and Alternaria alternata causing blight disease of Cumin (Cuminum cyminum L.). Int J Bioresour J Stress Manag 7:1336–1345. https://doi.org/10.23910/IJBSM/2016.7.6.1709
Suthar RS, Bhatt DP, Bhatt PN (2014) Effect of culture filtrate of Fusarium equiseti on seed germination and seedling growth of cumin (Cuminum cyminum). Indian Phytopathol 67:193–194
Wood TM, Bhat M (1988) Methods for measuring cellulase activities. Method Enzymol 160:87–112. https://doi.org/10.1016/0076-6879(88)60109-1
Yago JI, Roh JH, Bae SD, Yoon YN, Kim HJ, Nam MH (2011) The effect of seed-borne mycoflora from sorghum and foxtail millet seeds on germination and disease transmission. Mycobiology 39:206–218. https://doi.org/10.5941/MYCO.2011.39.3.206
Acknowledgements
The research was financially supported by Seed and Plant Certification and Registration Institute (SPCRI), Iran (Grant Number: 124-08-08-021-98024-980892).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Khaledi, N., Dehshiri, A. & Hassani, F. Effect of seed-borne fungi on seed health of native populations of Iranian cumin (Cuminum cyminum L.). Indian Phytopathology 74, 659–668 (2021). https://doi.org/10.1007/s42360-021-00350-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42360-021-00350-2