Abstract
Drug residues are now ubiquitous in the environment due to the extensive and widespread use for animal and human health care. Current technologies do not allow to completely remove drug residues during waste water treatment process. Consequently, drug residues are widespread in the aquatic environment and present thus potential impacts on biodiversity, ecosystem functioning, and public health. As a result, their residues have attracted particular attention from government and research community. High concentrations of antibiotic residues detected in the environment have prompted scientists to advocate for the implementation of various laws around the world. This work aims to contribute to the lack of information on this emerging and essential subject. Twenty-eight antibiotics including five tetracyclines, two macrolides, seven fluoroquinolones, and seven sulfonamides were analyzed in the input and output of two wastewater treatment plans (WWTP) in Tunis, in Northern Tunisia, in a tap and well water. The quantification was performed using online SPE-LC-MS/MS after the sample’s extraction/purification with offline SPE. At least 19 drugs were present at the quantifiable level. In the input and output of WWTP, drug residues were determined at high level with the Σ28drugs can be up to 239.8 ± 47.1 ng/L and 139.4 ± 31.9 ng/L, respectively. One WWTP was not operational during the sampling. The concentration level in the output was similar to which found in the input (no elimination process). Another WWTP allowed eliminating only 48% of the Σ28drugs. As a result, the Σ28drugs were detected at 109.5 ± 25.5 and 36.8 ± 8.7 ng/L respectively in well and tap water.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
For several decades, organic micro-pollutants such as hydrocarbons, pesticides, and PCBs have been studied. However, there are lack data in the literature on drug residues in the natural environment on a worldwide scale. Drug residues used in human and animal health care can be quickly introduced into the aquatic environment via wastewater treatment plants (WWTPs) and other processes, including leaching from agricultural soil where there is a practice of spreading with sewage sludge and pig manure or poultry. Indeed, WWPT or lagoons cannot eliminate these compounds from wastewater or agricultural effluent1- 8,
Antibiotic use is increasing in both developed and developing countries worldwide. It is estimated that 100,000 to 200,000 tons of antibiotics are consumed globally, which has resulted in their frequent detection in wastewater treatment plants’ (WWTPs) effluent, surface waters, and sediments around the world, resulting in the development of antibiotic resistance in the environment2-3. In Africa, 1800 active molecules are marketed. Tunisia is one of the big consumers of drugs. Drug residues are found ubiquitously today in hospital, urban and industrial effluents, as well as livestock manure, WWPT outputs, and surface water7, 9–12. These releases have a non-negligible environmental risk because drugs were initially produced to be biologically active. Indeed, although the effects of pharmaceutical products are studied through safety and toxicology studies, their potential effects on the environment are still unknown and have thus become subjects of interest to the scientific community12. A recent study showed that drug residues could cause endocrine disruption, changes in behavior, and genetic responses13. It was also shown that surface water, groundwater, and drinking water contain hormones from contraceptive treatments, anticancer drugs, anti-inflammatory and antibiotics7, 14–18. The pollution caused in aquatic environments by using human and veterinary pharmaceutical residues has become the source of severe concerns and requires studies to provide appropriate solutions. This work aim to study the occurrence of 28 antibiotics in the input and output of WWTP, in tap and well water collected from Northern Tunisia.
2 Materials and Methods
2.1 Chemicals and Materials
All reagents used were of analytical grade with > 95% of purity. The twenty-eight human and veterinary products were purchased from Sigma Aldrich except for gemfibrozil (GEM), carbamazepine (CBZ) and sulfamethoxazole (SMA) from Dr Ehrenstorfer (Augsburg, Germany). Carbamazepine-d10, sulfamethoxazol-d4 and trimethoprim-d3 were used as internal standards and were purchased from Dr Ehrenstorfer (Augsburg, Germany). All chemicals used in the analysis were purchased from VWR Prolabo and analytical and HPLC grades as ultra-pure water. The cartridges used for SPE were Oasis HLB (200 mg/6 mL) obtained from Waters (Milford MA, USA). Standard mixtures were prepared daily by appropriate dilution of the stock standard solutions using the same solvents as for stock solutions. The standard stock solutions can be preserved for one month7. This study prepared standard solutions weekly from commercial standards to avoid false positives or overestimated results. Glassware was first washed with diluted nitric acid, rinsed with distilled water, and then dried in an oven at 180°C for 2 h prior to use.
2.2 Study Sites and Sampling
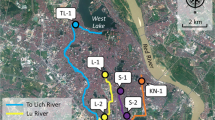
The samples were collected from the Tunis capital, northern of Tunisia (Fig. 1). Even these areas present industrial and agricultural activities; only few data is available for drug residues. (i) The first WWTP is Chotrana (S1) located in Route Sidi Salah km 5, Chotrana 1 Ariana. Chotrana WWTP was operational in 1986 and was dimensioned with the biological process with the treatment capacity of 40000 kg/BOD5/day18 or 78000 m3/day. Aerated pond combined with digestion techniques were used for the treatment. The volume of treated water is 20million m3/year and it is the biggest WWTP of the Tunis capital. It receives mainly domestic effluents which represent 87%; industrial and touristic activities represented 12% and 1% respectively. (ii) The second WWTP is located in Médina Jadida-Ben Arous (S2). It was operational for the first time in 1982 with the capacity to treat 37500 m3/day (9million m3/year) of wastewater. However, it was non-operational during the sampling campaign. Ben Arous is the largest industrial city in Tunis and particularly pharmaceutical industry. Ben Arous WWTP received the industrial effluent including effluent from pharmaceutical industry.
These WWTPs receive various types of wastewater and apply different treatment techniques. Ben Arous WWTP receives exclusively industrial wastewater (100%). Activated sludge combined with chemical treatment was applied for Ben Arous WWTP. Sub-samples were first collected, mixed thoroughly then divided into three samples and finally stored at -5°C in the WWTP. (iii) Bab Saadoun health center (S3) regroups six centers and hospitals; it is the most important health care center in Tunisia. The sampling campaign was conducted in early spring 2016 from 12/03/2016 to 25/03/2016. All the samples were collected with intelligent sampling (10 days) using 2.5 L bottles and immediately capped with Teflon-lined lid.
2.3 Sample Preparation and Extraction
Water samples were directly filtered back to the laboratory using previously calcinated 0.7 µm Whatman glass microfiber filters. Filtered waters were firstly spiked with internal standards (Carbamazepine-d10, sulfamethoxazol-d4 and trimethoprim-d3). Spiked samples were extracted using solid phase extraction (SPE) technique according to the method developed previously by Gros19. The procedural blank was performed in triplicate for each set of samples. Briefly, the cartridge was firstly conditioned with 6 mL of methanol followed by 6 mL of water HPLC grade (previously acidified at pH 2.5) with a flow rate of 1 mL/min. The samples were then passed through the cartridges at a flow rate of 1 mL/min using a vacuum SPE manifold. After sample loading, the cartridge was washed with 6 mL of HPLC grade water. The cartridges were then dried under a nitrogen flow for 30 min using Supelco vacuum manifold (Sigma-Aldrich). Finally, the targeted drug residues were eluted with 6 ml of methanol at a flow rate of 1 mL/min.
2.4 On-line SPE-LC-MS/MS Analysis
The analysis was performed using an off-line SPE combined with an on-line SPE-LC-MS/MS according to the method developed by Tlili7. 28 drugs were analyzed and their therapeutic groups, abbreviation, molecular weight (MW), retention time (RT), parent ion, fragment ions, method yield and limit of detection (LOD) of each selected drug residues were presented in Table 1.
3 Results and Discussions
3.1 Occurrence of the 28 Drug Residues in the Different Water Matrices
The concentration of the 28 selected compounds was investigated in each sample and in triplicate. The individual concentration varied depending on the sample’s origin. The highest level of Σ28Drugs was quantified at Chotrana WWTP input and followed by Ben Arous effluent at respectively 239.8 ± 47.1 ng/L and 199.7 ± 52.7 ng/L. These concentrations were two times lower than those detected in Arras WWTP (470.9 ± 149.2 ng/L) and Fresnoy aerated lagoon (470.9 ± 136.9 ng/L) in Northern France. Tlili4, Oke1 and Vieno20 have reported high levels for fluoroquinolones in Spain, France, Germany and Belgium, where the concentration of ciprofloxacin and ofloxacin were in the range of 1-100 µg/L and 0.5 ng/L -10 µg/L respectively. Among the 28 drugs, CTC, MIN, DAN, CAR and GEM were quantified dominants (Table 2). However, OFL was < LOQ for all samples and NOR was detected only in Ben Arous WWTP effluent at 6.6 ± 2.8 ng/L. These results are the same order to which reported by Tahrani et al (2018)43. DIC was detected only in the untreated wastewater (Fig. 2). Among the 28 drug residues, 15 drugs were detected in the tap water of Tunis Hospital Center, 18 in the well water of Tunis Health Center44, 24 in Ben Arous WWTP, 25 in the output of Chotrana WWTP and 22 in the input of Chotrana WWTP.
High levels of drug residues were found in the output of WWTPs which might indicate that the wastewater treatment processes used in the selection WWTPs were not appropriate for removing satisfactory the antibiotics. Indeed, these WWTPs consists of pretreatment, primary settling and activated sludge 40,41,42. In view of our results, others treatment process should be added to improve the efficiency of the treatment. A similar observation was also reported in the literature with the references herein 10,11, 18,39,40. Moreover, Ben Arous WWTP was not operational during the sampling. Therefore, the contamination levels found in the input were found in the output and then in the receiving aquatic environment. While WWTP Chotrana could eliminate 61%, 58% and 43% respectively for CT, FQ and SN. The elimination yield of Chotrana WWTP were determined in average of 48% was higher than which found for WWTP Arras in northern France with only 20%4, 5, 18. Higher removal efficiency detected for the Chotrana WWTP was possibly due to the aerated pond combined with digestion at higher temperature 2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36, 42.
Among the 28 selected drugs, 15 have been detected in the tap water collected near the Tunis health center. However, their concentrations were detected at lower level than which detected in the input/output of WWTP and the well water selected in this work. Individual concentration varied from < LOQ to 6.8 ± 1.9 ng/L. Twenty of the 28 selected were detected in the well water, usually used for irrigation and domestic activities. Individual concentration varied from < LOQ to 16.7 ± 2.1 ng/L for the ∑28Drugs of 109.5 ± 25.5 ng/L (Fig. 2). Similar results have been reported int the literature 3,10, 22, 23,37,38,41.
Figure 2 shows the sum of TQs, FQs, SNs, and the other nine compounds in input and output of WWTPs, in well and tap water. The highest concentration of TQs was detected at Ben Arous WWTP where other drug residues were also present at a significant level. Additionally, this WWTP was not operational which indicates that the totality of these pollutants could be rejected in the surface water of the receiving natural environment. TQs and FQs were found at high level in well water and were similar to which found in the output of Chotrana WWTP.
3.2 Individual Concentration of Tetracyclines
Among the 5 TCs, the CTC and MIN were dominants in the Chotrana and Ben Arous WWTP (Fig. 3). CTC was detected around 40 ng/L in both the input of Chotrana and Ben Arous WWTPs. However, MIN was detected at the higher level (> 40 ng/L) in Ben Arous WWTP and it was 3 times higher than Chotrana. The DOX, OTC and TC were determined with the low average which did not exceed 10 ng/L. The OTC and DOX were detected at < LOQ value in output Chotrana WWTP which indicated the good elimination by the existing process. These results are similar to which reported by Tarhana et al41. For TC, the concentration was detected in each sample except for the Ben Arous WWTP (Fig. 3). This may be due to the fact that TC could be transformed to epitetracycline, epioxytetracycline and other products3,4, 21, 39,40,49.
3.3 Individual Concentration of Sulfonilamides
SNs concentration (STZ, SDMX, SDZ, SBZ, STZ, SMX and SN) were lower than TCs concentrations. SDMX and SDZ were dominants in Chotrana input and output. SNs were detected in the input and output of WWTP and tap and well water (Fig. 4). Their present could be harmful to human health and need to reinforce the treatment for drinking water22. SDMX is detected in all the samples with high concentrations ranging from 1 ng/L in the tap of Rabta hospital to 14.2 ng/L in Chotrana WWTP input. This ubiquitous may be due to its wide use in human medicine for the treatment of bacterial infections5,23,42. The Chotrana WWTP processing system removed the majority of SNs with a removal rate of 100% for SBZ, 70% for SMZ and 50% for SDZ. High elimination rates were observed for SNs, up to 100%. A similar observation was reported in the literature27,28,29, 40,49.
3.4 Individual Concentration of Fluoroquinolones
FQs (CIP, DAN, DIF, ENR, NOR, OFL and ORB) commonly
used as veterinary medicine in the past were found from < LOD to few dozen ng/L depending on compounds and the matrix. FQs were used as veterinary drugs therapeutic purposes. However, their high concentration can cause problem for human and environment due to their potential carcinogenic and possible development of antibiotic resistance in animals30,40. All the FQs except OFL were detected in Ben Arous WWTP effluent ranging from 3.1 ng/L for DIF until 11.2 ng/L for DAN.
The DIF and ENR were not detected in the effluent (Fig. 5), probably due to their presences in low concentrations or completely removed in the sewer system before arriving at the WWTP31,40. It was found that they can be partly removed by sorption and photo degradation32,48. The highest concentration was detected for DAN with the concentration ranging from 4.3 ng/L in the tap of Rabta hospital to 27.7 ng/L in Chotrana WWTP input. 63% of DAN has been eliminated by Chotrana WWTP. A similar finding was reported in the literature39,40,41,42.
3.5 Nine Other Drugs
Table 2 shows that among the nine other antibiotics, CAR was detected dominant with concentration can be up to 40.8 ng/L in Ben Arous WWTP. It was detected in all the samples with high levels. Indeed, CAR was used as an anti-convulsion drug mainly for the treatment of epilepsy. It is one of the most frequently detected compounds in surface waters12, 16, 33,34,35 ,37,38,39, 45. The Chotrana WWTP could eliminated DIC, TYL and AMP with good efficiency. Their concentration detected in the output was < LOD. Their non-quantifiable level in the WWTP may be due to its presence in low concentrations respectively 5.3, 1.8, and 1.5 ng/L, or they were completely removed by the treatment system 8, 28, 37. For TRI, 45% has been eliminated by Chotrana WWTP; 26.4 ng/L was detected in the input versus 14.5 ng/L in the output. While Florfenicol is an aminoglycoside and high heterogeneity of its concentration was observed between samples. The highest concentration of Florfenicol was detected at 7.6 ng/L in the input of Chotrana WWTP. The elimination yield was calculated at < 50% for Chotrana WWTP. These findings are consistent with those reported by Tahrani et al40.
3.6 Ecological Risk for Antibiotics in Water Samples
In order to evaluate the potential risk related to the presence of micropollutants in the aquatic environment, the European Union has defined the environmental risk quotient (RQ). The ecological risk quotient can be calculated by dividing the maximum measured environmental concentration (MECmax) by the predicted no-effectconcentration (PNEC) as follow:
3.7 RQ = MECmax/PNEC
The ecological risk can be classified into four levels: insignificant risk (RQ < 0.01), low risk (0.01 < RQ < 0.1), medium risk (0.1 < RQ < 1) and high risk (RQ > 1). These classification levels were used to evaluate the ecological risk of the antibiotics in our sample and results were presented in the Table 3.
4 Conclusion
This work presents the contamination level of 28 drug residues in two WWTPs, one tape water and one well water. The samples were extracted using off-line SPE then analyzed by on-line SPE-LC- MS/MS. The results showed that the treatment processes used in the two selected WWTPs were non-appropriates to eliminate the selected antibiotics. Low elimination was observed for the most drugs selected in this research. The results showed also high contamination level of drug residues in tape and well water. CTC, MIN, SDMX, SDZ, DAN, TRI, and CAR were detected dominant in the effluents. More intensive research is needed to better understand how these drugs disperse into the environment and reach groundwater. Their fate and transport in the aquatic environment should also be study to evaluate the global risk of drugs residues. Moreover, to reduce the contamination level in the aquatic environment, it is necessary to develop cost-effective technologies to remove drug residues from wastewaters and contaminated sites to ensure the need of the future generation.
References
Oke-Omuferen, Maseko B et al (2022) Occurrence of antibiotics in wastewater from hospital and convectional wastewater treatment plants and their impact on the effluent receiving rivers: current knowledge between 2010 and 2019. Environ Monit Assess (2022) 194: 306
Hassen W, Mehri I et al (2022) Chemical and Microbiological Assessment of Wastewater Discharged along the Mediterranean Sea. Sustainability 14:2746
Joseas-Waleng N, Nosizo-Nomngongo P (2022) Occurrence of pharmaceuticals in the environmental waters: Africanand Asian perspectives. Environ Chem Ecotoxicol 4:50–66
Baber F, Curini R, Corcia AD, Nazzari M, Samperi R (2001) Solid-phase extraction followed by liquid chromatography-mass-spectrometry for trace determination of lactam antibiotics in bovine milk. J Agric Food Chem 49:3463–3470
Li X, Zheng W, Kelly WR (2013) Occurrence and removal of pharmaceutical and hormone contaminants in rural wastewater treatment lagoons. Sci Tot Environ 445–446:22–28
6. Tlili I, Caria G, Ouddane B, Ghorbel-Abid I, Ternane R, Trabelsi-Ayadi M, S. Net S. (2016) Simultaneous detection of antibiotics and other residues in the dissolved and particulate phases of water by an off-line SPE combined with on-line SPE-LC-MS/MS: Method development and application. Sci of Total Environ, 53-564:424-433
Heberer T (2002) Occurrence, fate, and removal of pharmaceutical residues in the aquatic environment: a review of recent research data. Toxicol Lett 131:5–17
Mansour D, Fourcade F et al (2015) Mineralization of synthetic and industrial pharmaceutical effluent containing trimethoprim by combining electro-fenten and activated sludge treatment. J. of the Taiwan institute of Chem Eng, 2015, 53:58–67
Net S, Rabodonirina S et al (2015) Distribution of phthalates, pesticides and drug residues in the dissolved, particulate and sedimentary phases from tranboudary rivers (France- Belgium),521–522:152–159
Boxall A, Fogg L et al (2004) Veterinary medicines in the environment. Rev Environ Contam Toxicol 180:1–91
Amiard-Triquet C, Mouneyrac C et al (2008) Biological indices, energy reserves, steroid hormones and sexual maturity in the infaunal bivalve Scrobicularia plana from three sites differing by their level of contamination. Gen Comp Endocrin 157:133–141
Tauxe-Wuersch A, De Alencastro LF et al (2005) Occurrence of several acidic drugs in sewage treatment plants in Switzerland and risk assessment. Wat Res 39:1761–1772
Togola A, Budzinski H (2008) Multi-residue analysis of pharmaceutical compounds in aqueous samples. J Chrom A 1177:150–158
Cary L, Casanova J (2013) el al Combining born isotopes and carbamazepine to trace sewage in salinized groundwater: a case of study in Cap Bon, Tunisia. Applied Geochemistry, 34:126–139
Belhaj D, Baccar R et al (2015) Fate of selected estrogenic hormones in an urban sewage treatment plant in Tunisia (North Africa). Sci Total Environ 505:154–160
Tahrani L, Van-Loco J et al (2016) Occurrence of Antibiotics in Pharmaceutical Industrial Wastewater, Wastewater Treatment Plant and Sea Waters in Tunisia. J of Water and Health 14:208–213
Gros M, Petrovic M, Barcelo D (2006) Development of a multi-residue analytical methodology based on liquid chromatography-tandem mass spectrometry (LC-MS/MS) for screening and trace level determination of pharmaceuticals in surface and wastewaters. Talanta 70:678–690
Vieno N, Tuhkanen T, Kronberg L (2006) Analysis of neutral and basic pharmaceuticals in sewage treatment plants and in recipient rivers using solid phase extraction and liquid chromatography-tandem mass spectrometry detection. J Chromatogr A 1134:101–111
Adachi -F, Yamamoto FA et al (2013) Occurrence of fluoroquinolones and fluoroquinolone-resistance genes in the aquatic environment. Sci Total Environ 444:508–514
Kolpin D, Furlong E el al (2002) Pharmaceuticals, hormones, and other organic wastewater contaminants in US streams. 1990–2000: A national reconnaissance. Environ Sci Technol 36:1202–1211
Carrara C, Ptacek C et al (2008) Fate of pharmaceutical and trace organic compounds in three septic system plumes, Ontario, Canada. Environ Sci Technol 42:2805–2811
Skraskova K, Santos L et al (2013) Fast and sensitive UHPLC methods with fluorescence and tandem mass spectrometry detection for the determination of tetracycline antibiotics in surface waters. J of chrom B 927:201–207
Sukul P, Spiteller M (2006) Sulfonilamides in the environment as veterinary drugs. Rev of environ cont and toxico 187:67
Seifrtová M, Nováková L et al (2009) An overview of analytical methodologies for the determination of antibiotics in environmental waters. Anal Chim Act 649:158–179
Xu W, Zhang G et al (2007) Occurrence and elimination of antibiotics at four sewage treatment plants in the Pearl River Delta (PRD), South of China. Water Res 41:4526–4534
Ben Mansour H, Mosrati R et al (2012) Bioremediation of industrial pharmaceutical drugs. Drug Chem Toxicol 35:145–158
Gros M, Ginebreda A, Barceló D (2010) Removal of pharmaceuticals during wastewater treatment and environmental risk assessment using hazard indexes. Environ Inter 36:15–26
Tong -L, Li LP (2009) P el al Analysis of veterinary antibiotic residues in swine wastewater and environmental water samples using optimized SPE-LC/MS/MS. Chemosphere, 74:1090–1097
He K, Soares AD et al (2014) Detection of a wide variety of human and veterinary fluoroquinolone antibiotics in municipal wastewater and wastewater-impacted surface water. J Pharma Biomedical Anal 106:136–143
Halling-sorensen B, Nielson S el al (1998) Occurrence, fate and effects of pharmaceutical substances in the environment: a review. Chemsphere 36:357–393
Garcia-Ac A, Segura P et al (2009) On-line solid phase extraction of large-volume injections coupled to liquid chromatography-tandem mass spectrometry for the quantification and confirmation of 14 selected trace organic contaminants in drinking and surface water. J of chromat A 1216:8518–8527
Radjenovic J, Pettrovic M, Barcelo D (2007) Advanced mass spectrometric methods applied to the study of fate and removal of pharmaceuticals in wastewater treatment. Trends Anal Chem 26(11):1132–1144
Drewers JE (2007) Removal of pharmaceutical residues during wastewater treatment. Anal Chem 50:427–449
Gros M, Petrovic M, Barceló D (2007) Wastewater treatment plants as a pathway for aquatic contamination by pharmaceuticals in the Ebro river basin (NE Spain). Environ Toxicol Chem 26:1553–1562
Tahrani L, Van-Loco J et al (2017) Identification and risk assessment of human and veterinary antibiotics in the wastewater treatment plants and the adjacent sea in Tunisia. Water Sci Technol 76(11):3000–3021
Moslah B, Hepeshi E (2018) Pharmaceuticals and illicit drugs in wastewater samples in north-eastern Tunisia. Environ Sci Pollut Res 25:18226–18241
Deogratias- Ekwanzala M, Florence-Lehutso R et al (2020) Environmental Dissemination of Selected Antibiotics from Hospital Wastewater to the Aquatic Environment. Antibiotics 9(7):431
Afsa S, Hamden K et al (2020) Occurrence of 40 pharmaceutically active compounds in hospital and urban wastewaters and their contribution to Mahdia coastal seawater contamination. Env Sci and Pollut Res 27:1941–1955
Tahrani L, Van Loco J et al (2016) Occurrence of antibiotics in pharmaceutical industrial wastewater, wastewater treatment plant and sea waters in Tunisia. J Water Health 14(2):208–213
Tahrani L, Van Loco J, Anthonissen R, Verschaeve L, Ben Mansour H, Reyns T (2017 Dec) Identification and risk assessment of human and veterinary antibiotics in the wastewater treatment plants and the adjacent sea in Tunisia. Water Sci Technol 76(11–12):3000–3021. doi: https://doi.org/10.2166/wst.2017.465
Tahrani L, Mehri I, Reyns T, Anthonissen R, Verschaeve L, Khalifa ABH, Loco JV, Abdenaceur H, Mansour HB (2018) Mai UPLC-MS/MS analysis of antibiotics in pharmaceutical effluent in Tunisia: ecotoxicological impact and multi-resistant bacteria dissemination Arch Microbiol. ; 200(4):553–565. https://doi.org/10.1007/s00203-017-1467-x
Nasri E, Subirats J, Sànchez-Melsió A, Mansour HB, Borrego CM, Balcázar JL (2017 Oct) Abundance of carbapenemase genes (blaKPC, blaNDM and blaOXA-48) in wastewater effluents from Tunisian hospitals. Environ Pollut 229:371–374. https://doi.org/10.1016/j.envpol.2017.05.095
European Commission. Technical Guidance Document on Risk Assessment, (TGD); Part II, Technical Report, European Commission (EC) (2003) ; ; pp.1917–1942. Available online:https://echa.europa.eu/documents/10162/987906/tgdpart2_2ed_en.pdf/138b7b71-a069-428e-9036-62f4300b752f (06/08/2022)
Jahnel J, Neamtu M, Schudoma D, Frimmel FH(2006) : Bestimmung von Umweltqualitätsnormen für potenziell gewässerrelevante Stoffe. Acta hydrochim. hydrobiol. 34: p.389– 397
Heye K, Wiebusch J et al (2019) Ecotoxicological characterization of the antiepileptic drug carbamazepine using eight aquatic species: baseline study for future higher tier tests. J Environ Sci Health Part A 54(5):441–451. DOI: https://doi.org/10.1080/10934529.2018.1562819
Chen M, Meng M et al (2022) “Rapid Degradation of Chlortetracycline Using Hydrodynamic Cavitation with Hydrogen Peroxide”. Int J Environ Res Public Health 19(7):4167. https://doi.org/10.3390/ijerph19074167
Grung M, Heimstad ES, Moe M, Schlabach M, Svenson A, Thomas K, Woldegiorgis A(2007) Human and Veterinary Pharmaceuticals, Narcotics, and Personal Care Products in the Environment. TA-2325/2007 ISBN 978-82-7655-52-524-0. https://www.miljodirektoratet.no/globalassets/publikasjoner/klif2/publikasjoner/2325/ta2325.pdf (Access on 06/08/2022)
Hassen W, Mehri I et al(2022) Chemical and Microbiological Assessment of WastewaterDischarged along the Mediterranean Sea.Sustainability, 14, 274
Bekir K, Beltifa A, Maatouk F, Nezar H, Barhoumi H, Ben Mansour H (2022) DNA as a Next-Generation Biomonitoring Tool of HospitalEffluent Contamination. Sustainability 14:2440
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tlili, I., Caria, G., Sghaier, R.B. et al. Occurrence of 28 Human and Veterinary Antibiotics Residues in Waters, North-Eastern Tunisia by Liquid Chromatography-Tandem Mass Spectrometry. Chemistry Africa 5, 2163–2172 (2022). https://doi.org/10.1007/s42250-022-00470-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42250-022-00470-w