Abstract
In this work; Cyclic-Voltammetry (CV) and Electrochemical Impedance Spectroscopy (EIS) were used to study the electrodeposition kinetic of selenium films in potentiostatic mode from aqueous solution containing selenium dioxide and sodium citrate at pH = 4.2. Semiconducting proprieties of obtained deposits were investigated by Mott-Schottky measurements whereas the optical ones were performed by UV–Visible spectrophotometry. The morphological characterization was carried out using the scanning electron microscopy (SEM). The obtained results showed that the electrodeposition process of selenium films in citrate bath occurred under diffusional regime as rate-limiting step. Deposition rate of selenium layers on platinum substrate is much superior than in the case of ITO substrate and up to a value of 0.65 μg/cm2 s. The HSeO3−/Se system becomes more rapid with the increase of the bath temperature. Obtained deposits are photoactive films that belong to p-type semiconductors with number of charge carriers in order of 1021/cm3 and energy band gap about 1.7 eV. The grains of electroplated films have spherical forms, nanometric sizes and strong adhesion on the substrate surface.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Selenium (Se) is a metalloid widely present in nature, generally in association with sulfur minerals in the form of selenide or in rocks of volcanic origin. It is extracted from the byproducts of the processing and refining of certain ores, cements, oils and many processing industries (electronics, production of glass and ceramics, rubber, plastics, pigments, lubricants, cosmetics). It is widely used in animal and human nutrition and pharmacology and it is a very important element in our life [1,2,3]. In addition a recent study has associated the spread of the COVID 19 to a selenium deficiency in the human body due to its deficiency in certain regions in China [4]. In recent years, electrodeposition of selenium attracted considerable attention due to its good semiconducting, optical, thermal, electric and piezoelectric proprieties, it use to make semiconductor compounds such as InSe, In2Se3 and CuInSe2 (CIS), which have many optoelectric applications, including advanced solar cells [5,6,7,8,9]. Selenium appears in different phases. The amorphous phase (red, brown and black) exhibits low electronic conductivity. The crystalline phases, include several solid allotropes, namely, the rhombohedral one, the α, β and γ monoclinic ones and the trigonal (hexagonal) one. Hexagonal Se is gray and it is the densest and the most stable of all phases [10]. Nowadays, various methods are used to synthesis selenium films and its alloys [11,12,13,14,15,16,17,18,19,20,21,22,23,24,25]. Electrodeposition method of thin films is considered among the promising ways, due to its ability to control the thickness, it allows obtaining a large-area deposition. It is a simple process, characterized by good adhesion, reproducibility and a low cost fabrication process [26]. Several studies have used the electrodeposition methods to make electrodeposited selenium and its alloys [23, 27,28,29,30,31,32,33,34,35,36,37]. These methods need a careful study to search the ideal conditions for making pure selenium layer with very good optoelectronic properties in order to meet the desired industrial needs. The biggest problem that we face frequently during the electrodeposition process, is the adherence problem; which depends on several factors, among them, the grain size. The deposits with large germs are not suitable in electroplating because the lack of cohesion between them and the little covering of the substrate surface whereas the deposit with small germs have a good adherence owing to the good cohesion between the germs. For this reason, herein we investigate thoroughly the electrodeposition process of selenium films and its kinetics aspect during the germ growth. We also examine the ideal conditions and kinetic factors (bath composition, temperature, pressure, concentration, deposition rate and pH value) that help to obtain deposits with small granules to meet the requirements of the nanomanufacturing and nanotechnology industry on the one hand and to strengthen the cohesion of the deposited films on the other hand. The electrodeposition kinetic of red selenium on platinum substrate from citrate bath containing selenium dioxide SeO2 was studied using electrochemical (voltammetry, electrochemical impedance spectroscopy) and physicochemical (UV–Visible spectrophotometry, scanning electron microscopy) methods. The deposition rate, the flat band potential Efb, the free carrier concentration Nd, the energy gap Eg and the grain size were also investigated.
2 Experimental
Red selenium dioxide SeO2 (Sigma–Aldrich) and sodium citrates tribasic dihydrate C6H5Na3O7∙2H2O (Fluka) powders are used to prepare the electrodeposition bath. Electrochemical measurements (voltammetry, electrochemical impedance spectroscopy) were carried out using Voltalab PGZ 301 controlled by computer (software VoltaMaster 4) with a cell of three electrodes; the saturated calomel electrode (SCE) as reference electrode, the platinum wire as counter electrode, the work electrode was used as platinum (S = 0.5 cm2) which was cleaned in the nitric acid during a few minutes and rinsed with distilled water before each manipulation. In the case of electrodeposition on ITO (indium tin oxide) substrate we have used a square plate (S = 1 cm2) cleaned in acetone for 5 min, then in the nitric acid (30%) during 2 min and finally well rinsed with distilled water.
In this study, we used for all handling an aqueous solution of 50 mM selenium dioxide SeO2 which dissolves in water and gives HSeO3− according to pourbaix diagram [38] with 0.15 M sodium citrates at ambient temperature, The pH = 4.2 was controlled by digital high precision pH-meter (HANNA instruments brand) and adjusted as needed by adding some drops of concentrated hydrochloric acid solution (HCl). The surface morphology of electrodeposited films was investigated by a JSM-6360 SEM. The UV–VIS transmittance spectrums have been recorded with a Shimadzu UV-1800 UV–Visible Scanning Spectrophotometer. The semiconducting properties of selenium deposited films on platinum were studied using the Mott-Schottky measurements in (0.1 M) KCl aqueous solution with 0.5 kHz employed frequency.
In the case of Mott-Schottky measurements the deposited films were elaborated on platinum substrates to ensure that the analyzed junction is Se/electrolyte and not ITO/electrolyte, but in the optical and micrographic analysis, the films were electrodeposited on the ITO substrates.
3 Results and Discussion
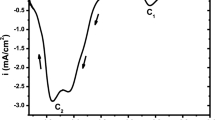
In order to verify the electrochemical behavior of the electrodeposition bath; firstly we have used the cyclic voltammetry to check the electrochemical processes which may occur in the electrolytic solution of the electrodeposition. Figure 1 shows a cyclic voltammogram of an aqueous solution containing SeO2 (50 mM) and sodium citrate medium (0.15 M) plotted on platinum electrode at pH = 4.2 with scanning rate of 5 mV s−1.
In this figure, scanning the potential from more positive values towards the cathodic side, we can observe two cathodic peaks C1 (at − 0.885 V vs. SCE) and C2 (at − 1.076 V vs. SCE) which are related respectively to the formation of (Se) electrodeposited and release of H2Se gas [according to Eqs. (1) and (2)] (chemical formation of elemental selenium can also take place). In the return scan we can see an anodic peak A1 (at 0.72 V vs. SCE) corresponds to the oxidation of electrodeposited selenium. It is obvious that the reduction peak is greater than the oxidation one; this confirms that the deposited quantity was not oxidized completely, but a small part of it only. In addition; the great spacing between the reduction and oxidation peaks which translated by the irreversibility of HSeO3−/Se system [26,27,28, 39,40,41]. In the reverse scan, it is clear that the current density is higher than during the forward scan between − 0.7 and − 0.4 V which shows that the electrodeposited selenium film on the platinum substrate follows the three dimensional nucleation and growth mode. The intersection point during a reverse scan is the beginning of the germination process. In the case of cathodic polarization on ITO substrate in acid medium; the irreversible electroreduction of the tin oxide can also take place [42].
Figure 2 presents the effect of scan rate on the electrochemical behavior of electroplating bath on platinum electrode accompanied by the variation of the critical current density according to the square root of the scan rate. We can observe that the increase of the potential scan rate v increases the current density of peaks corresponding to formation and dissolution of selenium C1 and A1 and shifts potentials of peaks to the cathodic C1 and the anodic A1 sides. When we reach great rates the reduction peak of the dissolved oxygen in the electrolytic medium can be seen in the cyclic voltammogram at 0.07 V vs. SCE. Fig. 2b, c confirmed the linear variation of the current density of cathodic and anodic peaks with the square root of the scanning rate according to Eq. (3), which well showed that the diffusion of the electroactive species is the limitation step of the deposition and oxidation processes of selenium films [43].
Such as: a the transfer coefficient, z the number of electrons transferred, C concentration, D the diffusion coefficient.
The effect of bath temperature on the electrochemical behavior was also studied. The Fig. 3 shows cyclic voltammograms set of SeO2 aqueous solution (50 mM) with sodium citrate (0.15 M) traced on platinum electrode with scan rate of 5 Vm s−1 at pH = 4.2 and different temperatures (20, 40, 60 and 80 °C). In this figure it is clear that the increase of the bath temperature gives different shapes of the voltammograms but the same peaks were observed in all cases. Variation of the shape and increase of peaks in voltammograms with increasing of the bath temperature can be explained by an increase in the activity and movement of electroactive species in the electrolytic solution and the increase in the thermal agitation. We can also see that the increase of bath temperature shifts the potentials of peaks towards small values (oxidation and reduction peaks become closer to each other); i.e. the reverse of the scanning rate influence. In this case we can say that the increase of the bath temperature makes the system more rapid (rapidity of HSeO3−/Se system increases owing to the increase in diffusion rate of electroactive species under the effect of the increase in temperature bath which considered the limiting step rate of the selenium electrodeposition process i.e. the limiting stage of the system speed).
Figure 4 shows the Nyquist impedance measurements for different deposition potentials of selenium on platinum electrode in the frequency range 50 kHz to 100 MHz. We can see the decrease of faradic loops (semi-circle) with the increase in deposition applied potential due to the increase in deposition rate; indicating that the formed layers of selenium have a small charge transfer resistance compared with the platinum surface knowing that the deposited amount increases with increasing applied potential to more negative values (i.e. the coverage of platinum surface by the electrodeposited selenium increases). We can also observe in this figure small inductive loops at low frequencies for certain applied potentials due to the adsorption of the electrolytic solution species on the electrode surface (probably the citrate ion). The electrochemical impedance response of the electrodeposition process of selenium can be modeled by an equivalent electric circuit containing capacitor, two resistances and coil [44,45,46]. Table 1 outlines the average values of electrolyte resistance Rel, charge transfer resistance Rtc and double-layer capacitance Cd for each value of deposition potential.
Table 2 shows the recorded quantity of electricity values Q during the electrodeposition process of selenium films in potentiostatic mode at different applied potentials (PH = 4.2, T = 20 °C) on platinum and ITO substrates for 10 min. In order to ensure that the quantity of electricity recorded by the potentiostat is consumed only by the electrodeposition of selenium films, the working electrode was monitored during the electrodeposition process in order to ensure that no gas bubbles are emitted (the Nyquist diagrams in electrochemical impedance spectroscopy analysis can prove this, when the electrodeposition is accompanied by a noisy release of hydrogen or other gas the shape of the diagram is greatly distorted). Here it should be noted that the value of the quantity of electricity registered by the potentiostat at − 0.9 V vs. SCE applied potential was all consumed in the selenium electrodeposition, but after this potential value E < − 0.9 V, we begin to notice the release of gas bubbles on the work electrode, which means that in this case the process of electrodeposition was accompanied by the release of gases that may be hydrogen or H2Se i.e. there is an interference between the electrodeposition of selenium and other parasitic reactions, for this reason the set of electrodeposition samples was stopped at this potential value (− 0.9 V) and we took the quantity of electricity as an approximation as it was all consumed in the selenium electrodeposition. As we can observe in the table; the consumed amount of electricity increases with the increase of applied potential. The deposition rate r and the thickness d of deposited film d relating to each sample were calculated using Faraday’s law (Eq. 4), it is obvious that the electrodeposition rate of selenium on platinum substrate is very great compared to its electrodeposition rate on the ITO, this is mainly due to the significant difference in conductivity between platinum and tin oxide on the one hand. On the other hand, the lack of active sites on the surface of ITO substrate, with the possibility that not all of the surface was activated during the activation process with nitric acid solution.
Figure 5 presents the Mott-Schottky measurements of selenium thin films electrodeposited in potentiostatic mode on the platinum substrate at different applied potentials for 10 min. the basis of this method is the formation of Schottky barrier between the semiconductor material and electrolytic solution [47,48,49]; it involves measuring the capacitance of the space charge region (Csc) according to electrode tension according the flow equation [50]:
where ε0 is the space permittivity (8.85 × 10–14 F cm−1), ε is the relative dielectric of elemental selenium (6.7), Nd is the concentration of the free carrier in the semiconductor, S is the area in contact with the electrolyte, E is the applied potential, Efb is the flat band potential, K is the Boltzmann constant (1.38 × 10–23 J k−1), T is the absolute temperature (293 K), and e0 is the charge of electron. The plot of 1/\({C}_{SC}^{2}\) as a function of applied potential E for a plane semiconductor can permitted to determine the nature of the semiconductor material n or p as well as the determination of the its electronic properties (the density of the donor and acceptor charge carriers, and the flat band potential).
In this figure, we can observe that all samples exhibited negative slopes, indicating the P-type characteristic of the obtained (Se) semiconductor. The slope of the tangent line in Mott-Schottky plots is proportional to the 1/Nd; and the intersection of this tangent line with the abscissa axis gives us the flat band potential. Table 3 outlines the experimental values of flat band potential Efb and the concentration of free carrier Nd relating to each sample.
Determination of the transition type direct or indirect, as well as the investigation of the energy gap value of a semiconductor can be carried out by UV–Visible spectrophotometry. The semiconductor materials absorb the light at certain wavelength λg which can be related to the gap according the follow relationship [51]:
The extinction of corresponding light follows an exponential law
where l is the penetration length of the light and α is the optical absorption coefficient, this last is a function with the thickness d, the transmittance t and the reflexing coefficient of the semiconductor material R; it can be expressed by the following formula [52,53,54,55].
The reflexing coefficient can be given by the following equation:
\({n}_{R}\) is the refractive index.
On the other hand the absorption coefficient is a function to the energy of incident photons (\(h\nu\)) and the energy gap Eg, according the following formula [56]:
A is a constant depends to the refractive index, the reduced mass and the light speed in the vacuum. The exponent n depends to the transition type:
For a direct gap (direct transition) n = 0.5
For an indirect gap (indirect transition) n = 2.
So, in order to investigate the semiconducting proprieties of each semiconductor materials, it’s possible to plot the curve:
The intersection of the affine part of this curve and the horizontal axis give us the value of energy gap Eg.
The transmittance spectrums UV–Visible of selenium films electrodeposited in potentiostatic mode on ITO substrate at different applied potentials, and the plot (αhυ)2 vs. (hυ) are presented on the Fig. 6. This figure shows that the deposited films of selenium are photoactive materials in the UV–Visible range which translates by the observed transmission bands. The plot (αhυ)2 vs. (hυ) is valid to estimate the energy gap value according to Eq. (11) (n = 0.5 for the red and gray selenium thin layers [57]), from this equation when αhυ tends towards zero hυ tends towards Eg, which leads that extrapolation on the axis (hυ) gives us directly the energy gap value of the analyzed material. The thicknesses of obtained selenium films at these potentials were used to get the absorption coefficient values α from the transmittance values t. Energy gap values Eg according to applied deposition potential are outlined in Table 4; These energy gap values are comparable to those reported in the literature (1.93–2.0 eV by AL-ANI et al. [58], 1.83–1.96 eV by Bhatnagar et al. [59], 1.76 eV by Chaudhuri et al. [60] and 1.86 eV by Fochs [61]).
Morphological characterization of the electrodeposited film was carried out in order to get an idea about microstructure of deposited selenium as well as the grain forms and the grain size. Figure 7 shows scanning electron microscopy (SEM) image of electrodeposited selenium sample obtained at − 0.86 V vs. SCE applied potential on ITO substrate during 10 min in potentiostatic mode. It is clear that this obtained deposit is less dense and composed from spherical grains forms with nanometric grain in different sizes. We can also see the formation of germs with dimensions ranging from 2 to 2.5 μm contain about 15–25 grains. In this case it can be ensure that the cohesion between the grains is greatest and their coverage of the surface is strong even if we increase the time of the electrodeposition process, which can give a deposited film with good adherence. To confirm this good adherence, a simple mechanical test using sandpaper was carried out accompanied with chemical test by dipping the sample in a solution of nitric acid; it was finding that for complete stripping of the deposited film, a strong concentration was required.
4 Conclusion
The electrodeposition kinetic of selenium on platinum and ITO substrates from citrate bath was studied in this paper; from the obtained results it can be recorded that: Electrodeposition of selenium occurs under the diffusional control regime as limitation step. Deposition rate of selenium films on platinum substrate is superior than in the case of ITO substrate and up to a value of 0.65 μg/cm2∙s. The increase of the bath temperature makes the system HSeO3−/Se more rapid. Electrodeposited layers are photoactive films and belongs to p-type semiconductors with number of charge carriers in order of 1021/cm3 and energy gap about 1.7 eV. The grains of electroplated films have spherical forms and nanometric sizes with strong adhesion on the surface substrate.
The recorded results would help us understand the kinetic and the good conditions for electrodeposition of selenium films with nano-grains and good adherence on the substrate surface and open up prospects for research on the same axis.
Abbreviations
- E:
-
Potential
- E0 :
-
Standard potential
- i:
-
Current density
- C:
-
Concentration
- D:
-
Diffusion coefficient
- v:
-
Scan rate
- a:
-
Transfer coefficient
- Cd :
-
Double layer capacitance
- Zreal :
-
Real part of impedance
- Zim :
-
Imaginary part of impedance
- Rtc :
-
Charge transfer resistance
- Rel :
-
Electrolyte resistance
- Eg :
-
Energy gap
- Q:
-
Quantity of electricity
- z:
-
Transferred electrons number,
- α:
-
Absorption coefficient
- d:
-
The thickness
- h:
-
Planck constant
- υ:
-
Frequency
- t:
-
Transmittance
- R:
-
Reflexing coefficient
- n R :
-
Refractive index.
- Csc :
-
Capacitance of the space charge region
- Efb :
-
Flat band potential
- K:
-
Boltzmann constant
- T:
-
Temperature
- Nd :
-
Concentration of free carrier
- S:
-
Electrode surface
- ε0 :
-
The space permittivity
- ε:
-
The relative dielectric
- m:
-
Masse
- F:
-
Faraday’s constant
- r:
-
Electrodeposition rate
References
Khurana A, Tekula S, Saifi MA, Venkatesh P, Godugu C (2019) Biomed Pharmacother 111:802. https://doi.org/10.1016/j.biopha.2018.12.146
Kuria A, Fang X, Li M, Han H, He J, Aaseth JO, Cao Y (2020) Crit Rev Food Sci Nutr 60:684. https://doi.org/10.1080/10408398.2018.1548427
Huo B, He J, Shen X (2020). Biol Trace Elem Res. https://doi.org/10.1007/s12011-020-02070-6
Zhang J, Taylor EW, Bennett K, Saad R, Rayman MP (2020) Am J Clin Nutr 111:1297. https://doi.org/10.1093/ajcn/nqaa095
Gates B, Mayers B, Cattle B, Xia Y (2002) Adv Funct Mater 12:219. https://doi.org/10.1002/1616-3028(200203)12:3%3c219::AID-ADFM219%3e3.0.CO;2-U
Tan SH, Kounaves SP (1998) Electoanalysis 10:364. https://doi.org/10.1002/(SICI)1521-4109(199805)10:6%3c364::AID-ELAN364%3e3.0.CO;2-F
Ferri T, Sangiorgio P (1999) Anal Acta 385:337. https://doi.org/10.1016/S0003-2670(98)00693-X
Badr Y, Mahmoud MA (2005) Phys B 369:278. https://doi.org/10.1016/j.physb.2005.08.027
Kowalik R, Fitzner K (2009) J Electroanal Chem 633:78. https://doi.org/10.1016/j.jelechem.2009.04.029
Kargar Razi M, Maamoury RS, Banihashemi S (2011) Int J Nano Dim 1:261. https://doi.org/10.7508/IJND.2010.04.003
Gurin VS, Prokopenko VB, Alexeenko AA, Wang Sh, Prokoshin PV (2001) Mater Sci Eng C 15:93. https://doi.org/10.1016/S0928-4931(01)00267-3
Jiang H, Yao X, Che J, Wang M, Kong F (2004) Ceram Int 30:1685. https://doi.org/10.1016/j.ceramint.2004.03.030
Zhang W, Chai Y, Cao N, Wang Y (2014) Mater Lett 134:123. https://doi.org/10.1016/j.matlet.2014.07.072
Chen Y, Zhang W, Fan Y, Xu X, Zhang Z (2006) Mater Chem Phys 98:191. https://doi.org/10.1016/j.matchemphys.2005.05.051
Wang Z, Chen X, Liu J, Yang X, Qian Y (2003) Inorg Chem Commun 6:1329. https://doi.org/10.1016/j.inoche.2003.08.009
Martínez-Escobar D, Ramachandran M, Sánchez-Juárez A, Narro Rios JS (2013) Thin Solid Films 535:390. https://doi.org/10.1016/j.tsf.2012.12.081
Ubale AU, Sakhare YS (2014) Vacuum 99:124. https://doi.org/10.1016/j.vacuum.2013.05.004
Jiang X, Mayers B, Wang Y, Cattle B, Xia Y (2004) Chem Phys Lett 385:472. https://doi.org/10.1016/j.cplett.2004.01.033
Zhang S-Y, Zhang J, Liu Y, Ma X, Chen H-Y (2005) Electrochim Acta 50:4365. https://doi.org/10.1016/j.electacta.2005.01.044
Pola J, Bastl Z, Subrt J, Ouchi A (2001) Appl Surf Sci 172:220. https://doi.org/10.1016/S0169-4332(00)00851-5
Mendoza D, Lbpez S, Granandos S, Morales F, Escudero R (1997) Synth Met 89:71. https://doi.org/10.1016/S0379-6779(97)81195-2
Abdel Aal A, Voigts F, Chakarov D, Endres F (2012) Electrochim Acta 59:228. https://doi.org/10.1016/j.electacta.2011.10.064
Maranowski B, Strawski M, Osowiecki W, Szklarczyk M (2015) J Electroanal Chem 752:54. https://doi.org/10.1016/j.jelechem.2015.05.037
Cabral MF, Suffredini HB, Pedrosa VA, Tanimoto ST, Machado SAS (2008) Appl Surf Sci 254:5612. https://doi.org/10.1016/j.apsusc.2008.03.038
Steichen M, Dale P (2011) Electrochem Commun 13:865. https://doi.org/10.1016/j.elecom.2011.05.023
Lai Y, Liu F, Li J, Zhang Z, Liu Y (2010) J Electroanal Chem 639:187. https://doi.org/10.1016/j.jelechem.2009.11.026
Cattarin S, Furlanetto F, Musiani MM (1996) J Electroanal Chem 415:123. https://doi.org/10.1016/S0022-0728(96)04707-9
Zein El Abedin S, Saad AY, Farag HK, Borisenko N, Liu QX, Endres F (2007) Electrochim Acta 52:2746. https://doi.org/10.1016/j.electacta.2006.08.064
Ivanov DK, Poznyak CK, Osipovich NP, Strel’tsov EA (2004) Russ J Electrochem 40:1044. https://doi.org/10.1023/B:RUEL.0000046489.81407.ff
Kowalik R, Kazimierczak H, Żabiński P (2016) Mater Sci Semicond Process 50:43. https://doi.org/10.1016/j.mssp.2016.04.009
Kwiecińska AM, Kutyła D, Kołczyk-Siedlecka K, Skibińska K, Żabiński P, Kowalik R (2019) J Electroanal Chem 848:113278. https://doi.org/10.1016/j.jelechem.2019.113278
Coelho D, Luiz GM, Machado SAS (2018) J Electroanal Chem 820:89. https://doi.org/10.1016/j.jelechem.2018.04.050
Rasche B, Amin HMA, Clarke SJ, Compton RG (2019) J Electroanal Chem 835:239. https://doi.org/10.1016/j.jelechem.2019.01.015
Xu J, Wang W, Zhang X, Chang X, Shi Z, Haarberg GM (2015) J Alloys Compd 632:778. https://doi.org/10.1016/j.jallcom.2015.01.013
Whyte GM, Awada C, Offor PO, Otung FU, Alshoaibi A, Aljaafari A, Ekwealor ABC, Maaza M, Ezema FI (2020) Opt Mater 99:109556. https://doi.org/10.1016/j.optmat.2019.109556
Majidzade VA, Aliyev AS, Guliyev PH, Babanly DM (2020) J Electrochem Sci Eng 10:1. https://doi.org/10.5599/jese.676
Ye X, Chen L, Liu L, Bai Y (2017) Mater Lett 196:381. https://doi.org/10.1016/j.matlet.2017.03.072
Pourbaix M (1963) Atlas d’équilibre électrochimique à 25°c, p:557
Santos MC, Machado SAS (2004) J Electroanal Chem 567:203. https://doi.org/10.1016/j.jelechem.2003.12.026
Cavallini M, Aloisi G, Guidelli R (1999) Langmuir 15:2993. https://doi.org/10.1021/la981231a
Pourbaix M (1963) Atlas d’équilibre électrochimique à 25°c, pp:555–556
Senthilkumar M, Mathiyarasu J, Joseph J, Phani KLN, Yegnaraman V (2008) Mater Chem Phys 108:403. https://doi.org/10.1016/j.matchemphys.2007.10.030
Bard AJ, Faulkner LR (2001) Electrochemical methods: fundamentals and applications. Wiley, New York
Wiart R (1990) Electrochim Acta 35(10):1587. https://doi.org/10.1016/0013-4686(90)80014-F
Gassa LM, Mishima HT, de Mishima BAL, Vilche JR (1997) Electrochim Acta 42(11):1717. https://doi.org/10.1016/S0013-4686(96)00371-4
Fan Y, Yang J, Jiang L, Wang Y, Ng BK, Sun H, Lai Y, Li J, Liu F (2017) J Electrochem Soc 164(4):H225. https://doi.org/10.1149/2.1481704jes
Rousset J, Saucedo E, Lincot D (2009) Chem Mater 21:534. https://doi.org/10.1021/cm802765c
Windisch CF, Exarhos GJ (2000) J Vac Sci Technol 18:1677. https://doi.org/10.1116/1.582406
Mora-Seró I, Fabregat-Santiago F, Denier B, Bisquert J, Tena-Zaera R, Elias J, Lévy-Clément C (2006) Appl Phys Lett 89:203117. https://doi.org/10.1063/1.2390667
Morrison SR (1980) Electrochemistry at semiconductor and oxidized metal electrodes. Plenum Press, New York
Hagfeldt A, Graetzel M (1995) Chem Rev 95:49. https://doi.org/10.1021/cr00033a003
González-Hernández J, Gorley PM, Horley PP, Vartsabyuk OM, Vorobiev YV (2002) Thin Solid Films 403–404:471. https://doi.org/10.1016/S0040-6090(01)01543-7
Yamaguchi T, Yamamoto Y, Tanaka T, Tanahashi N, Yoshida A (1998) Sol Energy Mater Sol Cells 50:1. https://doi.org/10.1016/S0927-0248(97)00091-3
Huang CJ, Meen TH, Lai MY, Chen WR (2004) Sol Energy Mater Sol Cells 82:553. https://doi.org/10.1016/j.solmat.2003.12.008
Sadigov MS, Özkan M, Bacaksiz E, Altunbaş M, Kopya AI (1999) J Mater Sci 34:4579. https://doi.org/10.1023/A:1004670112975
Singh RP, Singh SL, Chandra S (1986) J Phys D: Appl Phys 19:1299. https://doi.org/10.1088/0022-3727/19/7/019
Pejova B, Grozdanov I (2001) Appl Surf Sci 177:152. https://doi.org/10.1016/S0169-4332(01)00191-X
Al-ani SKJ, Al-delaimi MN, Abdul Munaim AH, Jawher HM (1990) Int J Electron 69:87. https://doi.org/10.1080/00207219008920294
Bhatnagar AK, Reddy KV, Srivastava V (1985) J Phys D: Appl Phys 18:L149. https://doi.org/10.1088/0022-3727/18/9/001
Chaudhuri S, Biswas SK, Choudhury A, Goswami K (1983) J Non-Cryst Solids 54:179. https://doi.org/10.1016/0022-3093(70)90026-8
Fochs PD (1956) Proc Phys Soc London Sect B 69:70. https://doi.org/10.1088/0370-1301/69/1/309
Acknowledgements
This work was supported by the Energetic and Solid State Electrochemistry Laboratory (Ferhat Abbas-Setif1 University-Algeria); the authors would like to thank ZAIM Keltoum and DERBAL Sabrine for their assistances.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Dilmi, O., Benaicha, M. & Asseli, R. Electroplating Kinetic of Nanostructured Selenium Films from Citrate Bath. Chemistry Africa 4, 85–93 (2021). https://doi.org/10.1007/s42250-020-00209-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42250-020-00209-5