Abstract
Eclosion is a rapid process of morphological changes in insects, especially for the wings of butterflies. The orange oakleaf butterfly (Kallima inachus) transits from pupae to adults with a 9.3 fold instant increase in the surface area of their wings. To explore the mechanism for the rapid morphological changes in butterfly wings, we analyzed changes in microstructures in the wings of K. inachus. We found that there were lots of micron-sized foldable units in the wings at the pupal stage. The foldable units could provide as much as 31.35 times of increase in wing surface area. During eclosion, foldable units were flattened sequentially and resulted in a rapid increase in wing surface areas. The unfolding process was regulated by the structures and layouts of wing veins. Based on our observation, foldable units play important roles in both deformation and stretching of wings. The foldable units of microstructures may provide mimics for simulating entities of large-deformational bionic structures with practical application.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
The oakleaf butterfly (Kallima inachus) is a famous example of camouflage due to its nearly perfectly mimic to brown oak leaves. K. inachus undergoes complete metamorphosis with four different stages, including egg, larva, pupa, and adult [1, 2] (Fig. 1, Fig s1). The process of turning into an adult from a pupa is called as eclosion [3]. K. inachus butterflies with large wings emerge from pupae without visible wings in a short period of time. The wing formation appears rapid and wing expansion is drastic.
Organ development is usually a time-consuming process, and organs formed in a short time are often fragile and functionally weak. However, butterfly wings appear instantly after emergence and can withstand long flights that need millions of wing flaps [4, 5]. Wings are vital organs for butterflies, and their integrity also affects the survival ability of the insects [6]. Butterfly wings are complex, deformable, and composed of high-performance structures in large numbers with sizes ranging from macroscopic to nanoscales [4, 7]. These structures have high mechanical properties formed during evolution [8]. They communicate and cooperate appropriately so that the entire organ could perform complex functions. Butterfly wings undergo morphological changes during eclosion, including changes in the form, quantity, and location of structures.
Structural development and morphological changes have been examined to reveal the functions of various structures, including the mechanism involved in eclosion and wing spreading. Understanding the process of wing formation could provide models in designing entities for large-deformational bionic structures via simulation [7,8,9]. During larval development, several structures of pre-wings are initially formed, including an imaginal disc [10,11,12,13,14]. Initially, a layer of irregularly arranged transparent skin cells is observed after pupation [15]. After producing new cells via cell division and removing older cells via apoptosis, the cells are aligned for wing formation after about 32 h [15, 16]. Subsequently, a single cellular layer turns over and folds to form a double-layer structure, which is the basis for the back and abdomen sides of wings [17,18,19]. After that, cells in the middle of wings continue to elongate vertically and connect with adjacent cells to form cell clusters [15]. Two to three days after pupation, wing cells go through programmed cell death, and the remaining cell content turns into wing membrane, which is covered with various sensory organs and wall derivatives [20, 21]. The wall derivative containing trachea gradually develops into wing veins filled with various motor nerves, plasma, and hemolymph [22].
Previous studies were mainly focused on gene regulation, cell development, wing shape and wing pattern formation [14, 23]. Few studies have been carried out on physical structures, butterfly eclosion, and wing spreading mechanisms. In this study, we focus on the structural basis for prompt and drastic changes in wings during eclosion. Our findings could provide models and scientific basis for designing functional bionics based on mimics of insect microstructures.
2 Materials and Methods
2.1 Insects
K. inachus larvae were obtained from a colony maintained at the Butterfly Experimental Station at the Research Institute of Resource Insects, Chinese Academy of Forestry, Yuanjiang, Yunnan, China. When larvae turned to the 5th instar, they were transferred to an artificial climate room and fed with Baphicacanthus cusia (Nees) Bremek under 10/14 light/dark cycles at 30 °C with 70% humidity. Other larval instars were maintained at 25 °C with the same humidity.
2.2 Instruments and Software
Main instruments and software used in this study are Hitachi Scanning Electron Microscope (Hitachi TM-3000), Keyence Ultra Depth of Field 3D Microscope (Keyence vhx-1000), Leica Cryostat (Leica CM1100), Leica Manual Rotary Microtome (Leica RM212rt), 2D drawing software CAXA 2007, and 3D drawing software NX 8.0.
2.3 Collection and Storage of Butterflies
A total of 600 5th instars were collected and reared as described previously. After pupation, individuals were transferred onto a vertically hanging towel. For sample fixation, individual pupae were collected at 0, 24, 72, 96, 120 h after pupation, and were immediately fixed in jars with FAA fixative [50% ethanol. 5% (v/v) acetic acid. 3.7% (v/v) formaldehyde]. Samples were collected at 9:30–10:30 a.m. each day, and each sample contained at least 30 individuals.
2.4 Experimental Method
-
(1)
Measurement of morphological parameters
Whole butterfly wings were removed carefully from insect bodies and dried. Images of individual wings were taken under an Ultra Depth of Field 3D Microscope (Keyence vhx-1000). The wing images were processed for measurements using an image stitching function. The processed images were then measured using the CAXA 2007 software. The length, width, area, and other parameters of the wings were recorded.
-
(2)
Fixation of butterfly pupae at dfferent stages of eclosion
Pupae at different stages were fixed using liquid nitrogen.
-
(3)
Electron microscope scanning
For surface scanning, wings were completely dried, and the surface scales were removed with a brush. Images were taken using a scanning electron microscope (Hitachi TM-3000). For vertical scanning, wings were cut directly with a blade after drying. Images were also taken using the scanning electron microscope (Hitachi TM-3000). For images of the underside of wings, moisturized wings were spread with tweezers along wing veins, the wings were then dried, and imaged using the scanning electron microscope (Hitachi TM-3000).
3 Results
3.1 Changes in Butterfly Wings During Development
At 30 °C under 70% humidity, it took 120 h on average for pupation. On the other hand, it took only 15 min to complete eclosion.
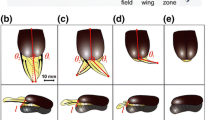
When a K. inachus larva turns into a pupa, wings are already attached to both sides of the abdomen of the pupa [24]. During the pupal stage that lasted for 6 days, no significant changes in terms of the surface area of wings were observed (Fig. 2a 0–120 h). However, during the 15 min of eclosion, a significant increase in sizes of wings was observed (Fig. 2a 120 h to 120 h 15 min). Specifically, there were dramatic increases in the total surface area (b, c), the length (d), and the width (e) of wings during the 15 min of eclosion (Table s1).
Morphological changes of K. inachus right forewings at different pupal stages. a The morphological changes of the right forewing over time during pupation. The length of the scale was 10 mm. b Shapes of front right forewings at different pupal stages. c Changes in sizes of right forewings. The dotted line was the dividing line between the pupal stage and the eclosion stage. d Changes in the length of right forewings. e Changes in the width of right forewings
3.2 Structural Units of Butterfly Wings
The dramatic changes in sizes of butterfly wings should have resulted from changeable tissue structures in the wings. Butterfly wings are mainly composed of wing membrane, wing veins, and scales. Among these three components, scales are the attachments of wing membranes and should have no effect on changes in wing sizes (Fig. 2).
3.2.1 Structure of Wing Membrane
The area of membrane accounted for more than 91% of total wing size (Tables 1, s2). Since morphological changes of wings are largely due to changes in membrane, we examined possible structural changes in membrane to reveal the basis for wing deformation. We found that there were a large number of similarly shaped, foldable structural units in wing membrane (Fig. 3a, b). The foldable units were gradually formed during pupal development. At 0 h, two layers of wing membrane were bonded together without apparent foldable units. At 24 h, irregular foldable units began to appear and were stacked together. During 48 h to 120 h, irregular foldable units became more regular by forming bundles between units (Fig. s4). At 120 h, the space between two layers of wing membrane increased, and bundles remained between the two layers of wing membranes. Eventually, the shapes of foldable units gradually became similar to each other and were arranged orderly (Fig. 3a–b). Therefore, it takes 120 h for K.inachus to form a complete foldable unit (Fig. s3). Sub-structurally, a foldable unit was pyramidal in the membrane and composed of a large number of small double-layer structures (Fig. 3c, d, g). Each microscopic structure was 10–60 μm with a triangular projection (Fig. 3b, g).
Morphology and development of foldable units. a Cross-section mages of foldable units at different time. The length of the scale was 50 μm. At 0 h, wing membranes on both sides were close to each other with a thin layer of gel type substance in between. At 24 h, wing membranes began to form foldable units, and bundles began to be formed. At 48–96 h, foldable units underwent morphological changes and expansion. At 120 h, foldable units became mature, double-layer structures with upright bundles between membranes. At 120 h 15 min, the foldable units and bundles disappeared instantly, and wings became thinner and flattened. b Longitudinal-section images of foldable units. At 0 h, there was no obvious unit structure in wing membrane. At 24 h, foldable units began to emerge, and the foldable units were stacked together. 48–120 h, the stacked foldable units were separated into independent units and arranged in rows. At 120 h 10 min when eclosion began, the foldable units began to unfold. At 120 h 15 min, the foldable units were unfolded completely and bundles between membranes disappeared. Before eclosion (120 h), the vertical view of a foldable unit was triangular (red triangle). After eclosion, triangular foldable units were flattened and bundles disappeared. c Schematic presentation of cross sections of foldable units. d Schematic representation of the vertical view of foldable units. L1 and L2 were curves of foldable units. There are a large number of small folded substructures, with each one substructure taken as a foldable unit. Each foldable unit consisted of a double layered structure. a1 and a2 were the vertical projection of L1 and L2, respectively. e Projected expansion potential of foldable units, which was calculated as below. f Mean transverse lengths of the foldable units
Because of the foldable units, butterfly wings increased in size rapidly by just opening up these units (Fig. 3b). Microscopic observation showed that all foldable units were completely flattened within 15 min. After the foldable units are open up, the two layers of wing membranes were bonded together again. The foldable structures on the wing membrane and the bundles disappeared (Fig. 3a, b).
3.2.2 Projection of Extensibility of Foldable Units
Since membrane is the main component of butterfly wings, the extensibility of a foldable unit during eclosion should be proportional to the rapid increase of wing size. Since each foldable unit can be taken as a triangle (Fig. 3a, b), the hypothetical extensibility of a foldable unit is the sum of the lengths of the two sides that are extended to form the surface of the wing membrane. Therefore, the following equation was used calculated the hypothetical length (L) of an extended foldable unit after eclosion (Fig. 3d, e):
where L is the longitudinal length and the length of a foldable unit after extension, a is the projected length of L before extension, and the number 57.008 is the transverse length of the triangle before it is fully extended. The relationship between L and a is Eq. (1). Based on Eq. (1), the theoretical extensibility of a foldable unit is 6.7264 fold during eclosion (Fig. s5).
The average value (∆l) of the transverse length of a foldable unit was calculated according to Eq. (2).
where ρ is the area ratio, s is the area of the triangle before eclosion, S is the area of the triangle after eclosion, dl is the transverse length of a single foldable unit, CL is the sum of the transverse length of N foldable units, N is the number of foldable units, s = \({\text{dl}}_{120}\times {\it{\text{LL}}}_{120}\text{/2}\), S = \({\text{dl}}_{\text{120h15m}}\times {\it{\text{LL}}}_{{120}\mathrm{h}{15}\mathrm{m}}\text{/}{2}\), TL is the transverse length, and LL is longitudinal length. The value of small letter s was calculated using the equation s = \({\mathrm{dl}}_{120}\times {LL}_{120}/2\), S = \({\mathrm{dl}}_{120\mathrm{h}15\mathrm{m}}\times {LL}_{120\mathrm{h}15\mathrm{m}}/2\). Based on this equation, the sum of the transverse length of the foldable units did not change much, and the longitudinal length was 8.86 μm (Fig. 3f) at 0 to 120 h. After eclosion, the transverse length of the foldable units increased to 41.28 μm, i.e., 4.66 times the original value (Fig. s5). Once a foldable unit is completely flattened, the theoretical size of the flattened area is ρ = 31.35 times. However, the actual change ratio was only 9.30 (Fig. s5).
3.2.3 Composition and Structure of Wing Veins
Wing veins are the second largest structure of butterfly wings, accounting for about 9% of wing size (Tables 1, s2). Wing veins act as a skeleton, supporting the shape of butterfly wings. Wing veins play an important role during eclosion and unfolding of butterfly wings.
The structure of a wing vein consists of four main substructures: membranoid substance, wing vein membrane, trachea, and filaments (Fig. 4a). These substructures were composed of a large number of foldable units as well (Fig. 4b), which could undergo morphological changes during eclosion (Fig. s8). The vein structures (Fig. 4a) directly interact with the foldable units of the wing vein membrane (marked by the white circle). All wing vein structures were connected to the wing membrane via filaments.
Structure and development of wing veins. a The basic structure of wing veins. The length of the scale was 100 μm without number. The wing veins were mainly composed of four structures: membranoid substance, wing vein membrane, trachea, and filaments. Membranoid substance, wing vein membrane, and trachea were all composed of foldable units. Membranoid substance, trachea, and vein membrane were connect via filaments (represented by the white dotted circle). b Morphological changes of wing veins. A green dashed line represent an outline of a developing wing vein, the black arrows point to wing veins, and a red arrow points to hemolymph. At 0 h, the wing vein membrane was not yet formed, but trachea was visible. At 24 h, the diameter of the wing vein increased. At 24 h to 120 h, wing vein membrane developed, forming a large number of foldable units consisting of wing vein membrane and trachea. At 120 h 10 m, the foldable units of the wing vein membrane was flattened, a large amount of air was generated in trachea, resulting in increased diameter of the trachea. A large amount of hemolymph was distributed around trachea. At 120 h 15 m, hemolymph disappeared, foldable units of trachea disappeared, and the diameter of trachea increased. At this time, the diameter of the trachea was close to the diameter of the wing vein. c The morphological change of trachea. The length of the scale was 100 μm, and the black arrow pointing to a wing vein. At 0 h, when a larva just became pupa, wing had already developed trachea. At 48–120 h, foldable units were formed in trachea. At 120 h 15 m, foldable units of trachea were fully unfolded, and the volume of trachea became larger. d Ratio of the wing vein to trachea. The symbol η is the ratio between wing vein area and trachea area. The symbol δ is the ratio between perimeters of wing vein and trachea
As shown in Fig. 4, the perimeter (L) and area (S) of wing veins, and the perimeter (l) and area (s) of trachea were measured to calculate their perimeter ratio (δ) and area ratio (η), which were used as parameters for morphological changes of wing veins. During 0–24 h, trachea was formed, but wing veins were still developing (Fig. 4b green dashed line, Fig s6), with maximal δ and η. During 48–120 h, wing veins and trachea formed a large number of foldable units. The number and diameter of foldable units of trachea increased, with gradually decreased δ and η (Fig. 4b, c). During 120 h–120 h 15 min, eclosion of butterflies began, and the foldable units of wing veins and trachea were completely unfolded, resulting in a sharp decrease of δ until the values of δ and η close to 1. At this time, the size of the trachea and wing veins were the same (Fig. 4b–d). During eclosion, the sharp drop of δ indicated that the morphological change rate of the trachea \({\text{v}}_{\text{t}}\) was greater than the morphological change rate of wing veins \(v_{v} (v_{t} > v_{v} )\), and the relative area \(\Delta s\) of trachea and wing veins decreased (\(\Delta s = S - s\)).
where i is the time period between samples, j is the sample number of the period, n is the number of samples in the period, L is the perimeter of a wing vein, S is the area of a wing vein, l is the perimeter of trachea, and s is the area of trachea.
During eclosion, butterflies take in air to increase air in trachea [25], resulting in enlarged trachea (Fig. 4b). During the enlargement of trachea, the relative area Δs of trachea and wing veins decreased, along with reduced hemolymph [26]. The incoming air might cause higher pressure that served as power for wing unfolding. During 0–120 h, trachea and wing veins formed a large number of foldable units. During 120 h–120 h 15 min, the foldable units of trachea and wing veins were gradually unfolded (Fig. 3b, c), consistent with our hypothesis that incoming air served as the power for butterfly wings to spread [26].
3.2.4 Main Function of Bundles
Foldable units were formed along with bundles at the same time (Fig. 3a). Structurally, bundles were formed with a large number of filaments bonded together. Bundles connect with foldable units of wing membrane (Fig. s7). According to the arrangement of bundles, wing membrane could be divided into three areas, area (Area A) without any bundles (Fig. 5a–c); area (Area B) with bundles connecting to wing membrane in the same layer (Fig. 5a, e, f); and area (Area C) with bundles connecting to the upper and lower wing membranes (Fig. 5d, g, h). Area A was mainly located in the middle of the closed chamber in the hind wing. Because it lacked bundles, Area A should be unfolded easily during the flattening process. Since the bundles in area B were connected to wing membrane on the same layer, the power driving unfolding should be easily transmitted to other areas, resulting in the acceleration of unfolding other parts of a foldable unit. Area C was mainly located in open chambers where bundles were connected to the upper and lower layers of wing membranes. The diameters of wing veins in area C were relatively small and thus the power this area could provide during the unfolding process should be relatively small as well.
Distribution of bundles. a–h Three different areas of bundle distribution. a Area without any bundles (Area A), as indicated by the yellow dashed line. Outside the yellow dashed line was the area (Area B) with bundles connecting to the upper and lower wing membranes in the same layer. The length of the scale was 100 μm without number. b Is enlarged portion of a, showing foldable units without bundles. c Is the partially enlarged portion of b, showing a single foldable unit without any bundles. d The area (Area C) with bundles connecting the upper and lower wing membranes at different layers. e Is another enlarged portion of a, showing the bundles that connected the wing membrane in the same layer. f is a partially enlarged portion of e, showing that the top and bottom of a foldable unit were connected by scattered filaments. g is a partially enlarged portion of d, showing that bundles connecting to the bottom and top of the upper and lower wing membranes. h is an enlarged portion of g, showing bundles connecting to fascicular. i–l Change of bundles at different stages. i Bundles were perpendicular to the upper and lower wing membranes. j Bundles inclined between upper and lower wing membranes. k Bundles flattened between the two wing membranes, ready for eclosion. m–p Change of bundles during eclosion. m Bundles connecting to foldable units of wing membrane in the same layer at the beginning of eclosion (at 0 min). n Flattened bundles and foldable units 2 min later after the beginning of eclosion. o Area with few bundles on foldable units 10 min later after the beginning of exlosion. p Residue filaments forming a thin layer between wing membranes 15 min later after the beginning of exlosion. q Bundle-associated cells (pointed by red arrows). l Branches of bundles before eclosion. The branches were indicated by green arrows
Bundles gradually inclined and flattened between the two wing membranes (Fig. 5 i–l) during the unfolding process of foldable units. After eclosion, flattened bundles were sandwiched between the wing membranes, providing physical support to the wings (Fig. 5p). Cells at both ends of a bundle control the division and proliferation of the bundle (Fig. 5q). When a foldable unit divided, new bundles were formed from filaments produced from the cells (Fig. s4). Therefore, the number of bundles closely related with the number of foldable units.
3.3 Unfolding Patterns of Butterfly Wings
Figure 6 shows morphological changes of butterfly wings during the unfolding process, exhibiting a small and straight wing, a wrinkled wing, a slender wing, and large and straight wings (Fig. 6a–b). Interestingly, the expansion of wings was uneven in terms of the width and length at different time points of eclosion. Specifically, during the first 3 min of eclosion, the width of wings increased from 9.53 to 11.53 mm. However, the length of wings increased much more greatly, from 12.89 to 32.93 mm. Most changes in wing sizes happened at the last minute when butterflies crawled out of their puparia (Fig. 6c). During the period of 3–15 min of eclosion, the width of wings increased greatly, from 11.53 to 31.91 mm. On the other hand, the length of wings only slightly increased, from 32.93 to 31.37 mm (Fig. 6c, d).
Changes in K. inachus wings during eclosion. The length of the scale in figure e was 5.00 mm, and the other figures are 50 μm. a Wings at different time points during eclosion. At 0 min, the butterfly was confined inside its puparium and wings were not visible. At 1 min, the head of the butterfly crawled out of the puparium, and wing shoulders protruded from the puparium. At 1.5 min, half of the wings protruded out from the puparium. At 2 min, the abdomen and the left front wing crawled out of the puparium. At 3 min, the butterfly completely crawled out of the puparium. At this time, the length of its wings increased greatly. At 10 min, the width of its wings increased greatly. As the wings unfolding, wrinkles on the wings reduced greatly, and the remaining wrinkles were mainly distributed on the edge of the wings. At 15 min, wings were fully unfolded. b Morphological changes of the left forewing. At 0 min, the wing was confined in the pupa. The wing dissected out from the pupal case was small without wrinkles. At 1 min, the wing was still in the pupal case, but wrinkles appeared on the dissected wing. At 1.5 min, the wing shoulder protruded out of the pupal case, but remained folded in most part of the wing. At 2 min, the wing was fully protruded out of the pupal case. The length of the wing increased significantly. At 3 min, wrinkles reduced and length of the wing continued to increase. At 10 min, the width of the wing increased significantly and wrinkles reduced greatly. At 15 min, the wing became flattened and wrinkles disappeared. c Dynamic change of wing width. d Dynamic change of wing length. e The wing in emergence. la and lb are incision. f The parameters of foldable unit. The foldable unit of la is closer to the foldable unit of adult, and the foldable unit of lb is close to the foldable unit of pupa. g–i The sections of right forewing at la. g The foldable unit of wing membranes were unfolded. h The surface area of foldable unit increase. i The wing veins have expanded. j–l The sections of right forewing at lb. j The foldable unit of wing membranes is still folded. k The surface area of foldable unit slightly increase. l The wing veins have expanded and wing membranes have not bonded together yet
Butterfly wing was changed section by section in the process of eclosion, the reason is that the foldable units changed at different times (Fig. 6e–l). Initially, the change in wing vein drive the change in the wing membrane and provides power (Fig. 6i, l). On the other hand, the wing shoulders preceded the change of other position (Fig. 6e, f), making wing shoulders have greater morphological variation. The length of folding unit at lb is 129.083 μm which is very close to the length of adult. But, the width was just 16.788 μm, much closer to pupa wing. For this reason, during the period of 0–3 min of eclosion, the process of emergence firstly change the wing in length followed by width. The width is further increased when butterflies are outside the puparium.
4 Discussion
Based on our observation, the development process of butterfly wings during eclosion can be summarized as following. During the pupal stage, large number of foldable units are formed, including foldable units in wing veins, membrane and trachea. These foldable units are connected to each other via filaments. The morphology of foldable units is different among vein, membrane and trachea (Fig. 7a). When eclosion begins, foldable units dislocated results in reduction of distance (Fig. 7b). During eclosion, extra air taken by trachea causes its increase in volume, compressing hemolymph in wing veins to unfold the wing power (Fig. 7c). After eclosion, trachea decreases in volume and becomes flattened within membrane. Along with flattened trachea, wing veins and membrane also become completely flattened, resulting in completion of unfolding and morphological changes of wings during eclosion (Fig. 7d).
3D simulation of wing microstructures during eclosion. a Microstructures before eclosion. Wings were mainly composed of six structures: foldable units of wing vein (represented by the yellow part), foldable units of wing membranes (represented by the brown part), trachea (represented by the silver part), bundles from trachea to wing vein (represented by the blue part), bundles from wing vein to wing membranes (represented by the red part) and bundles between wing membranes (represented by the green part). b Foldable units become dislocated when eclosion begins. c Foldable units become unfolded. The diameters of trachea and wing veins increased. At this time, the area of wing membranes increased and bundles are scattered. d Fully unfolded wings. Foldable units disintegrated completely and bundles are transformed into thin layers
The shape of the wing does n’t change much during different developmental stages (Fig. 1b), indicating that the foldable units are distributed evenly and expansion of the wing during eclosion is relatively in proportion towards different directions. Even though no significant changes in wing size during the pupal stage, structural changes inside wings are accumulating, including formation of foldable units and development of bundles. Foldable units provide the basis for later wing expansion. However, foldable units can expend towards any direction. We found that bundles regulate the direction of expansion of foldable units. Bundles and other filament structures may be crucial to ensure expansion of foldable units in both the upper and lower wing membranes expand proportionally to avoid wing deformation.
K. inachus is a typical insect that undergoes complete metamorphosis. Wings of lepidopterans undergo significant morphological changes during eclosion [2, 17]. The ability of dramatic expansion of wings are due to large number of foldable units developed at the pupal stage. Structurally, foldable units in wing membrane and veins provide the foundation for wing expansion, and the air taken via wing veins provides power for wing unfolding. The structure and layout of the wing veins are crucial for wing unfolding.
Although foldable units can be unfolded in any direction, the actual spreading dimension is determined by bundles and wing spreading power. When eclosion begins, incoming air in wing veins forces foldable units gradually unfold in a certain way. Similar to K. inachus, many other insect species that undergo complete metamorphosis share the same mechanism for wing spread during eclosion [27]. The large number of foldable units in wing veins, trachea, and membrane provide not only the structural basis for wing expansion, but also protect wings from damage by providing wing stability and dispersing stress during deformation.
Wing unfolding during eclosion is apparently regulated orderly. It is interesting to note that the length of wings increase first during wing expansion, followed by wing width. The exact mechanism for the directionally regulated expansion remains delineated. We speculate that the arrangements of the wing veins and bundles are responsible for this phenomenon. The directionally regulated expansion of wings is also more power efficient. Once a wing becomes fully expanded longitudinally, power can be concentrated on expansion vertically (the width direction) (Fig. 5i–l). Once foldable units are unfolded, the shape of wings are completely fixed [18, 19]. The eclosion process directly determines the shape and function of wings, that also affects the flight ability and subsequent survivability of the butterfly.
5 Conclusions
Eclosion is a rapid and significant process of morphological changes in insects, especially for butterfly wings. The orange oakleaf butterfly transits from pupae to adults with a 9.3 fold instant increase in the surface area of their wings. The ability of dramatic expansion of wings is due to presence of a large number of foldable units developed at the pupal stage. The foldable structural units was a double foldable structure in flattened conditions, the surface area of lepidotic wings will obtain 31.35 times change. Therefore, the foldable structural units can provide greater deformability than the conditions requirements of the wing. Structurally, foldable units in wing membrane and veins provide the foundation for wing expansion, and the air taken via wing veins provides power for wing unfolding. The structure and layout of the wing veins are crucial for wing unfolding. Multiple structures work together to ensure the expansion of the wings.
References
Mcmahon, D. P., & Hayward, A. (2016). Why grow up? A perspective on insect strategies to avoid metamorphosis. Ecological Entomology, 41, 1–11.
Truman, J. W., & Riddiford, L. M. (2019). The evolution of insect metamorphosis. Current Biology, 29, 1252–1268.
Piszter, G., Kertesz, K., Horvath, Z. E., Balint, Z., & Biro, L. P. (2019). Reproducible phenotype alteration due to prolonged cooling of the pupae of Polyommatus icarus butterflies. PLoS ONE, 14, 1–24.
Combes, S. A. (2010). Materials, structure, and dynamics of insect wings as bioinspiration for MAVs (Vol. 7, pp. 1–8). Wiley.
Tsai, C. C., Richard, A. C., Shi, N. N., Ren, C., Pelaez, J. N., Bernard, G. D., Pierce, N. E., & Yu, N. F. (2020). Physical and behavioral adaptations to prevent overheating of the living wings of butterflies. Nature Communications, 551, 1–14.
Mountcastle, A. M., & Combes, S. A. (2014). Biomechanical strategies for mitigating collision damage in insect wings: Structural design versus embedded elastic materials. Journal of Experimental Biology, 217, 1108–1115.
Zhang, D., Zhang, W., Gu, J., Fan, T. X., Liu, Q. L., Su, H., & Zhu, S. (2015). Inspiration from butterfly and moth wing scales: Characterization, modeling, and fabrication. Progress in Materials Science, 68, 67–96.
Niu, S. C., Li, B., Mu, Z. Z., Yang, M., Zhang, J. Q., Han, Z. W., & Ren, L. Q. (2015). Excellent structure-based multifunction of morpho butterfly wings: A review. Journal of Bionic Engineering, 12, 170–189.
Wu, L., He, J., Shang, W., Deng, T., Gu, J. J., Su, H. L., Liu, Q. L., Zhang, W., & Zhang, D. (2016). Optical functional materials inspired by biology. Advanced Optical Materials, 4, 195–224.
Nijhout, H. F., & Emlen, D. J. (1998). Competition among body parts in the development and evolution of insect morphology. Proceedings of the National Academy of Ences, 95, 3685–3689.
Marcus, J. M. (2001). The development and evolution of crossveins in insect wings. Journal of Anatomy, 1, 211–216.
Fujiwara, H., & Ogai, S. (2001). Ecdysteroid-induced programmed cell death and cell proliferation during pupal wing development of the silkworm, bombyx mori. Development Genes & Evolution, 211, 118–123.
Shimmi, O., Matsuda, S., & Hatakeyama, M. (2014). Insights into the molecular mechanisms underlying diversified wing venation among insects. Proceedings of the Royal Society B: Biological Sciences, 281, 20140264.
Nienhaus, U., Tinri, A. W., & Aegerter, C. M. (2012). In vivo imaging of the drosophila wing imaginal discover time: Novel insights on growth and boundary formation. PLoS ONE, 7, 103–110.
Iwasaki, M., Ohno, Y., & Otaki, J. M. (2017). Butterfly eyespot organiser: In vivo imaging of the prospective focal cells in pupal wing tissues. Scientific Reports, 7, 40705.
Masaki, I., Yoshikazu, O., & Otaki, J. M. (2014). Real-time in vivo imaging of butterfly wing development: Revealing the cellular dynamics of the pupal wing tissue. PLoS ONE, 9, 233–239.
Macdonald, W. P., Martin, A., & Reed, R. D. (2010). Butterfly wings shaped by a molecular cookie cutter: Evolutionary radiation of lepidopteran wing shapes associated with a derived Cut/wingless wing margin boundary system. Evolution & Development, 12, 296–304.
Gordon, N., Olinger, M. D., Janice, A., Schweiger, R. N., Timothy, A., & Galbraith, M. D. (1987). Primary repair of acute ascending aortic dissection. The Annals of Thoracic Surgery, 44, 389–393.
Polic, D., Fiedler, K., Nell, C., & Grill, A. (2014). Mobility of ringlet butterflies in high-elevation alpine grassland: Effects of habitat barriers, resources and age. Journal of Insect Conservation, 18, 1153–1161.
Kodama, R., Yohida, A., & Mitsui, T. (1995). Programmed cell death at the periphery of the pupal wing of the butterfly, Pieris rapae. Roux’s Archives of Developmental Biology, 204, 418–126.
Gunther, P. (2018). Beyond aerodynamics: The critical roles of the circulatory and tracheal systems in maintaining insect wing functionality. Arthropod Structure & Development, 47, 391–407.
Chintapalli, R. T., & Hillyer, J. F. (2016). Hemolymph circulation in insect flight appendages: Physiology of the wing heart and circulatory flow in the wings of the mosquito Anopheles gambiae. The Journal of Experimental Biology, 219, 3945–3951.
Vincent, J. F. V., & Wegst, U. G. K. (2004). Design and mechanical properties of insect cuticle. Arthropod Structure & Development, 33, 187–199.
Dinwiddie, A., Null, R., Pizzano, M., Chuong, P., Krup, A. L., Tan, H. E., & Patel, N. H. (2014). Dynamics of F-actin prefigure the structure of butterfly wing scales. Developmental Biology, 392, 404–418.
Elliott, C. J. H. (1981). The expansion of Schistocerca gregaria at the imaginal ecdysis: The mechanical properties of the cuticle and the internal pressure. Journal of Insect Physiology, 27, 695–704.
Wasserthal, L. T. (1976). Heartbeat reversal and its coordination with accessory pulsatile organs and abdominal movements in lepidoptera. Experientia, 32, 577–579.
Natori, M. C., Sakamoto, H., Katsumata, N., Yamakawa, H., & Kishimoto, N. (2015). Conceptual model study using origami for membrane space structures a perspective of origami-based engineering. Mechanical Engineering Reviews, 2, 00368.
Acknowledgements
We gratefully acknowledge Jun Yao provided with the pictures of oakleaf butterfly. This research was funded by the Innovation Team Cultivation Project of Yunnan (202005AE160011), the National Special Support Program for High-level Personnel Recruitment (W02070188) and the Fundamental Research Funds of CAF (CAFYBB2017QA013).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, J., Chen, X., Lu, Q. et al. Foldable Units and Wing Expansion of the Oakleaf Butterfly During Eclosion. J Bionic Eng 19, 724–736 (2022). https://doi.org/10.1007/s42235-022-00178-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42235-022-00178-0