Abstract
Purpose
The role of adipokines in causing inflammation and insulin resistance in normal weight and obese patients is generally well studied. However, there are often conflicting results regarding their levels in type 1 diabetes mellitus (T1DM) patients and their relationship to micro- and macrovascular disease. We therefore investigated which serum adipokine levels are independently associated with markers of early atherosclerosis and microvascular complications in patients with T1DM.
Methods
A cross-sectional study was performed in the Diabetes Outpatient Clinic of Hippokrateion General Hospital, Thessaloniki, Greece. Sixty T1DM patients (30 females, mean age 38.8 ± 10.6 years, mean diabetes duration 17.4 ± 9.9 years) were included. Plasma adiponectin, leptin, and resistin, carotid artery intima media thickness (cIMT), and arterial stiffness (pulse wave velocity, PWV/SpygmoCor CP System and Mobil-O-Graph 24 h PWA) were assessed.
Results
Leptin and resistin levels were significantly higher in overweight and obese patients (p = 0.002 and p = 0.039, respectively). Adiponectin was the only adipokine negatively correlated with BMI (rs = − 0.41, p = 0.001). We report a bivariate association between serum adiponectin levels and retinopathy (p = 0.007). Resistin was the only adipokine that showed significant correlation with systolic (rs = 0.42, p = 0.001) and diastolic (rs = 0.29, p = 0.024) hypertension and PWV (p = 0.035).
Conclusions
Serum adipokine levels demonstrate similar bivariate associations with anthropometric variables in patients with T1DM to those in normal weight subjects. Although microvascular complications are associated with serum adipokine levels by bivariate analysis, only resistin, an inflammatory marker, is independently associated with arterial stiffness in patients with T1DM.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Type 1 diabetes mellitus (T1DM) is mainly characterized by autoimmune destruction of insulin-secreting beta cells [1]. However, the prevalence of overweight and obesity seems to have significantly increased among individuals with T1DM [2]. Furthermore, although obesity and insulin resistance constitute a state that is mainly associated with T2DM, research has shown that insulin resistance is also associated with T1DM and even precedes its onset [3]. Among people at risk for developing T1DM (positive pancreatic autoantibodies), individuals with the highest insulin resistance at baseline are those who are more likely to develop T1DM in the future [4]. Data from 2011 also revealed that adipokines play a crucial role in the relationship between T1DM and obesity as they are known to have tissue insulin sensitization activity and also regulate glucose metabolism through various mechanisms, such as increased insulin secretion and glucose storage, inhibition of glucagon secretion, and hepatic gluconeogenesis [5].
Previous studies have demonstrated that patients with diabetes have extensive atherosclerosis early in life because of a complex atherogenic process. In this process, adipokines are involved; however, their relationship with other markers of atherosclerosis, including carotid intima media thickness (cIMT), pulse wave velocity (PWV), and other parameters such as body weight (BW) is still debated [6].
The aim of this study was to evaluate the concentrations of selected serum adipokines (leptin, adiponectin, and resistin) in patients with T1DM and to investigate their relationship with BW and markers of early atherosclerosis, including cIMT and arterial stiffness.
Materials and methods
Study population
A total of 60 subjects with T1DM were consecutively recruited from our Diabetes Outpatient Clinic. The diagnosis of T1DM was made according to the 2015 criteria of the American Diabetes Association [7]. The study was conducted in accordance with the principles of the Declaration of Helsinki and was approved by the Ethics in Research Committee of the School of Public Health of the Aristotle University of Thessaloniki, Greece. All participants provided written informed consent.
Methods
Study design
Cross-sectional.
Setting
All procedures were performed in the morning and all subjects were advised to abstain from smoking, coffee, and any other food or drink apart from water for 8 h before the study. Blood samples for biochemical measurements were collected at the time of study enrollment.
Anthropometric measurements
BW was measured using a balanced-beam scale with light clothing without shoes and expressed in kilograms (kg). Height was measured using a wall-mounted stadiometer and expressed in cm. BMI was calculated as weight in kilograms divided by the square of height in meters (kg/m2) and the cut-off 25 used to define normal vs. overweight or obese patients (BMI < 25 = normal, 25 to < 30 = overweight, and > 30 = obesity). The Michigan Neuropathy Screening Instrument (MNSI) was used to assess the presentation of peripheral neuropathy. Assessment of diabetic retinopathy was carried out by a trained ophthalmologist. Patients with T1DM were considered as having hypertension if systolic blood pressure (SBP) was over 140 mmHg and/or diastolic blood pressure (DBP) was over 90 mmHg or if they were receiving medication for hypertension.
Biochemical parameters
Blood samples were collected to measure glycated hemoglobin (HbA1c), uric acid (UA), lipids profile (total cholesterol, high-density lipoprotein, low-density lipoprotein, and triglycerides), and serum creatinine. Urine albumin to creatinine ratio (UACR) was calculated to estimate the presence of albuminuria. Estimated glomerular filtration rate (eGFR) was calculated using the CKD Epidemiology Collaboration (CKD-EPI) equation. Patients with T1DM were considered as having nephropathy if the eGFR was less than 60 ml/min/1.73 m2 and/or if the UACR was over 30 mg/g [8], and dyslipidemia if LDL values were over 100 mg/dl or the patient was receiving hypolipidemic treatment. Urine samples were collected for measurement of albumin and creatinine excretion.
Cytokines
Serum leptin levels were measured by a commercial ELISA kit (Human Leptin Quantikine ELISA R&D Systems Catalog No. DLP00). Serum resistin levels were measured by a commercial ELISA kit (Human Resistin Quantikine ELISA R&D Systems Catalog No. DRSN00). Serum adiponectin levels were measured by a commercial ELISA kit (Human Total Adiponectin/Acrp 30 Quantikine ELISA R&D Systems Catalog No. DRP300). All measurements were carried out in the laboratory of the 2nd Propedeutic Department of Internal Medicine.
Carotid sonography
High-resolution B-mode ultrasonography of the right and left common carotid artery (CCA) was performed with the VividTM–S6 General Electric Medical Systems ultrasound scanner equipped with a linear 13–5 MHz transducer. For all images, IMT measurement was automatically obtained using the General Electric EchoPAC version 12. The maximum IMT (cIMTmax) was calculated for each patient of the two carotid measurements (right and left).
Assessment of arterial stiffness and central hemodynamics (instant and 24-h)
After 5 min of rest in the supine position, PWV and central blood pressure (CBP) were measured with a SpygmoCor CP System and two to three separate recordings were taken if needed. All measurements were performed in accordance with the 2012 expert consensus document on the measurement of aortic stiffness in daily practice. Furthermore, during 24-h ambulatory daily-life conditions, brachial systolic and diastolic blood pressure (SBP and DBP) and PWV were measured by the Mobil-O-Graph 24 h PWA Monitor (IEM, Stolberg, Germany). The differences in PWV values between the two methods were as expected, since the first represents office and the other out-of-office measurements [9].
Statistical analysis
In the statistical analysis, the baseline characteristics of the patients who participated in the study were calculated. Continuous variables are reported as means with standard deviation (SD) or as medians with interquartile range (IQR), while frequencies with percentages for categorical variables n (%). The chi-square test (χ2) was applied to investigate the relationship between categorical variables. If there were expected counts less than five, Fisher’s exact test was applied. The independent samples t-test and the Mann–Whitney non-parametric test were used to compare the means and medians of continuous variables in different categories of categorical variables. Spearman’s rank correlation (rs) coefficients were used in order to find the correlation between continuous variables. A normality test was conducted using the Kolmogorov–Smirnov and Shapiro–Wilk tests, as well as histograms, P-P, and Q-Q plots. Relationships with a p value (p) ≤ 0.05 were considered as statistically significant. All reported p values are two-sided. Univariate and multivariable linear regression analyses were performed in standard and forward stepwise selection to identify independent factors affecting IMT and PWV and to estimate the final predictors of their variability. Univariate and multivariable logistic regression analysis was performed to examine which variables influence retinopathy, nephropathy, and neuropathy and to estimate the final predictors of their variability. Any univariate variable with a p value < 0.20 was selected as a candidate for multivariable analysis. We also added univariate variables of great medical significance to the models. All independent variables (predictors) were entered simultaneously into the model. When data are not shown in the multivariable logistic regression tables, this is due to suboptimal “goodness of fit” of the model. R Statistical software (V 4.0.3) and the “pwr” package were used for sample size estimation, our purpose being to compare the change in leptin levels between obese and normal weight individuals. The aim was to identify any difference between groups of 9 (ng/ml) with a between-subject SD (pooled standard deviation) as 12.8 (ng/ml [10]), 80% power, and 5% type I error. Based on these assumptions, it was determined that we would need 32 people in each group. The data were analyzed in the Statistical Package for the Social Sciences 24.0 (SPSS Inc., Chicago, IL, USA).
Results
Baseline characteristics
The study population consisted of 60 patients (30 females, mean age 38.8 ± 10.6 years, mean diabetes duration 17.4 ± 9.9 years). Half of the patients were overweight or obese. General characteristics of the study participants by BMI status (normal BMI vs. high BMI) are presented in Table 1. There were no statistically significant differences in age, diabetes duration, or eGFR and HbA1c levels between the two groups, although those with high BMI were more likely to have hypercholesterolemia. In addition, men were more likely to be overweight than women. Only four patients had confirmed clinical neuropathy (MNSI ≥ 4). Furthermore, 15 patients had nephropathy, while, specifically, 33 patients had eGFR of > 90 in combination with ACR < 30 mg/g, nine had eGFR of > 90 in combination with ACR > 30 mg/g, 12 had eGFR of 60–90 in combination with ACR < 30 mg/g, five had eGFR of 60–90 in combination with ACR > 30 mg/g, and one had eGFR of < 60 in combination with ACR < 30 mg/g. Markers of atherosclerosis and angiopathy, such as cIMTmax and arterial stiffness (PWV), were significantly higher in overweight/obese patients compared to normal. Finally, leptin and resistin levels were significantly higher, while adiponectin was lower in overweight/obese participants.
Unadjusted correlation analysis between adipokine concentration and metabolic and anthropometric quantitative parameters is presented in Table 2 and association analysis between adipokine concentration and metabolic and anthropometric qualitative parameters is presented in Table 3
Unadjusted correlations revealed that leptin levels were positively correlated with BMI (rs = 0.38, p = 0.003) and associated with female sex (median = 13,880; IQR = 4077.25–25,075, p = 0.012). Also, leptin levels were positively correlated with total cholesterol levels (rs = 0.29; p = 0.024). No other significant correlation was observed between leptin and the remaining measured parameters.
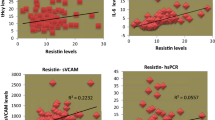
Resistin levels were also positively correlated with BMI (rs = 0.28, p = 0.03) and with presence of both systolic (rs = 0.42, p = 0.001) and diastolic (rs = 0.29, p = 0.024) hypertension. Moreover, resistin levels were positively associated with the presence of albuminuria (AC ≥ 30 mg/g) (median = 11.10; IQR = 8.28–16.38, p = 0.026), but negatively correlated with HDL-C levels (rs = − 0.33, p = 0.011). No other significant correlation was observed between resistin and the remaining measurable parameters.
Adiponectin was the only adipokine negatively correlated with BMI (rs = − 0.41, p = 0.001) and was also negatively correlated to WHR (rs = − 0.42, p = 0.001). Adiponectin levels were found to be lower in patients with systolic (rs = − 0.58, p < 0.001) and diastolic (rs = − 0.56, p < 0.001) hypertension (in contrast to resistin), hyperuricemia (rs = − 0.39, p = 0.002), and increased total daily insulin requirements (rs = − 0.58, p = < 0.001). Women had greater adiponectin levels in comparison to men (median = 11.57; IQR = 7.96–18.18 vs. median = 6.19; IQR = 3.19–8.42, p < 0.001). In addition, patients with low triglyceride levels (rs = − 0.38, p = 0.003) and high HDL-C (rs = 0.67, p < 0.001) had higher adiponectin levels. Lastly, there was an association between serum adiponectin levels and retinopathy (median = 16.69; IQR = 9.29–24.07, p = 0.007).
Finally, cIMTmax (p = 0.025) and arterial stiffness (p = 0.003) were significantly positively associated with BMI, but there was no direct correlation between them and serum adipokine levels. As expected, PWVm and PWVs were positively correlated with traditional cardiovascular risk factors such as age, diabetes duration, WHR, BMI, cIMTmax, SBP, DBP, and CSBP. The same finding applies to cIMTmax, which was found to be also positively correlated with age, diabetes duration, BMI, HbA1c, PWV, SBP, and CSBP.
Univariate and multivariable linear regression analysis of cardiovascular outcomes (IMT, PWV) (see Supplementary Tables S1a, S1b) and univariate and multivariable logistic regression analysis of microvascular complications (retinopathy, nephropathy, and neuropathy) (see Supplementary Tables S2a, S2b, S2c)
As anticipated, unadjusted correlations revealed that older age, higher BMI, longer diabetes duration, and higher HbA1c, SBPm, and LDL-C were associated with a higher IMT (p < 0.05), whereas no association was observed with adipokine levels. After adjustment for covariates (models 1, 2, 3), IMTʼs association with higher HbA1c and LDL-C remained, but the most decisive factor for IMT was found to be older age (p < 0.001) (Table S1a). We also found significant univariate associations of older age, higher BMI, longer diabetes duration, and higher SBPm with PWV progression (p < 0.05), but no direct association was observed with adipokine levels. In multivariable linear regression analysis adjusted for adiponectin or leptin levels and other covariates (models 1, 2, 3), older age was revealed also as the most decisive factor for PWV progression (p < 0.001) (Table S1b). In the final model for resistin when all predictors were added to the model (model 3, Table S1b), the relationship of PWV with resistin levels became significant (p = 0.035), along with age (p < 0.001), BMI (p = 0.03), and LDL-C (p = 0.039).
Unsurprisingly, in the univariate analysis, diabetes duration was a strong independent predictor for retinopathy (p = 0.04) and even after adjustment for adipokine levels, gender, BMI, age, and HbA1c (model 2, Table S2a), their relationship remained significant. Additional adjustment for SBPm and LDL-C attenuated the association between diabetes duration and retinopathy, which became no longer significant. In addition, adiponectin levels were found to be strongly (p = 0.008) associated with retinopathy, even after adjustment for sex and BMI (p = 0.004). Further adjustment for clinically significant covariates such as age, diabetes duration, and HbA1c changed this relationship, and adiponectin was no longer a significant independent predictor factor for retinopathy (model 2, Table S2a). On the other hand, only diabetes duration was found to be a strong independent predictive factor for nephropathy, but this relationship remained no longer significant after adjustment for all the other covariates (models 1, 2, 3) (Table S2b). The univariate and multivariable logistic regression analysis for neuropathy as a dependent variable revealed no further associations (Table S2c).
Discussion
Leptin and resistin levels were found to be significantly higher in overweight and obese patients with T1D (p = 0.002 and p = 0.039, respectively). Adiponectin was the only adipokine negatively correlated with BMI (rs = − 0.41, p = 0.001). Serum adipokine levels in patients with T1DM seem to have a similar dependence on adipose tissue as do healthy subjects without diabetes.
Microvascular complications appear to correlate with serum adipokine levels. Bivariate association between serum adiponectin levels and retinopathy (p = 0.007) was observed. Resistin was the only adipokine that showed significant correlation with systolic (rs = 0.42, p = 0.001) and diastolic (rs = 0.29, p = 0.024) hypertension and PWV (p = 0.035). With the exception of resistin, which seems to play a role in the development of hypertension and arterial stiffness in patients with T1DM, a further direct association between adipokine levels and macrovascular complications was not identified.
Leptin
Leptin is predominantly produced in adipose tissue, and circulating leptin levels correlate well with total body fat, reflecting the individual’s energy status [11]. Leptin’s secretion pattern is similar in obese and lean individuals, but obese subjects have greater leptin concentrations than do lean subjects due to their greater amount of body fat [12]. Furthermore, for the same age and body mass index (BMI), women have greater leptin concentrations than men. Leptin is a target of many trials in T2DM patients, but the number of studies assessing the role of leptin in T1DM is modest. Various authors report divergent results regarding serum leptin levels in T1DM in contrast to those in non-diabetic individuals [13]. In our study, the results are consistent with findings from previous studies which also demonstrated that leptin concentrations are strongly correlated with BMI and gender in T1DM patients [14, 15]. The most likely explanation for this is that leptin levels in patients with T1DM have a similar dependence on adipose tissue as in normal weight patients and increase during diabetes as a result of the effects of insulin on body fat mass [16]. Another explanation involves insulin requirements, as leptin levels were found to decrease in parallel with the decrease of insulin resistance and insulin levels, irrespective of BMI status [17].
In our study, we also observed a positive relationship between leptin and total cholesterol levels, a finding in accordance with previous studies [18]. A possible explanation is that changes in leptin levels and lipid pattern may be associated with obesity and features of metabolic syndrome that coexist, as an altered plasma lipid pattern is a common pathophysiological feature in both metabolic syndrome and obesity. However, a clear etiology for the direct interactions of leptin with lipid metabolism regulation and its associated biochemical alterations continues to be a matter of debate [18].
Adiponectin
Although adiponectin is exclusively secreted by adipose tissue, plasma adiponectin levels are negatively associated with total body fat, with the highest levels observed in lean subjects and the lowest in obese subjects [19]. We also demonstrated that there is an inverse association between adiponectin serum level and BMI. Several studies have shown the same finding in Τ1DM patients [20, 21]. It is not clear why adiponectin secreted from fat cells decreases when fat cells in obesity increase. It has additionally been reported that the aggregation of visceral fat leads to cellular malfunction of adipose tissue, which ultimately results in decrement of serum adiponectin levels in obesity [22]. As with leptin, adiponectin is higher in women than in men (probably due to different concentrations of sex hormones and different distribution of adipose tissue in subcutaneous or intra-abdominal fat) [23].
Though there is a large volume of literature on the role of adiponectin in cellular and systemic physiology, the consensus is that adiponectin generally increases tissue sensitivity to insulin and has anti-inflammatory, anti-atheromatic, and anti-apoptotic effects on different types of cells [24]. In our study, adiponectin levels were found to be lower in patients with systolic and diastolic hypertension in contrast to resistin. Hypoadiponectinemia has been reported to be positively correlated with hypertension in several studies, albeit this association remains controversial [19, 25, 26]. Nevertheless, a 2013 systematic review and meta-analysis including 17,598 adults from 43 non-prospective and five prospective studies found lower adiponectin levels in hypertensive individuals compared with normotensive adults, suggesting that plasma adiponectin levels constitute a biomarker and possible mediator in the development of adiposity-related hypertension [27].
In addition, we found that lower adiponectin levels are associated with dyslipidemia, hyperuricemia, and increased total daily insulin requirements in patients with T1DM. Numerous studies exist that demonstrate the same correlation [19, 28]. These associations of low adiponectin levels with obesity, dyslipidemia, insulin resistance, hypertension, and hyperuricemia indicate that this protein may be an important novel marker of the metabolic syndrome [29].
Lastly, we found that patients with T1DM and retinopathy complications have higher serum adiponectin levels than patients without complications (p = 0.007). Nevertheless, when important clinical covariates such as age, diabetes duration, and HbA1c were added to the model, adiponectin was no longer a significant independent predictor factor for retinopathy (model 2, Table S2a). Our findings are consistent with previous reports, but it remains to be determined whether increased adiponectin levels are pathophysiologically related to the development of microvascular complications or whether they constitute a counter-regulatory response [30, 31].
Resistin
Data on resistin levels in patients with T1DM are limited. In our study, resistin levels were positively correlated with BMI status in T1DM (p = 0.03), findings that are consistent with a 2015 study in adults with T1DM [32]. Reports on resistin levels in T1DM patients are to date conflicting [33,34,35]. Considering the small number of studies on T1DM and the controversial findings concerning resistin levels and adiposity, it is clear that further studies with larger populations are needed to understand this association.
Our results revealed increased resistin concentrations in hypertensive patients with T1DM, while other clinical studies have also reported that circulating resistin levels are associated with hypertension in humans [36, 37], and a recent meta-analysis found that elevated serum resistin levels correlate with development of hypertension [38]. Indeed, plasma levels of resistin are elevated early in life in young healthy offspring of hypertensive subjects compared to young healthy offspring of normotensives [39], while its higher plasma levels have been shown to be associated with an increased risk of developing hypertension in women without diabetes or hypertension [40]. Likewise, our findings that resistin is an independent predictive factor of PWV (p = 0.035) (model 3, Table S1b), even after adjustment for significant clinical regressors (such as gender, BMI, age, diabetes duration, HbA1c, SBPm, and LDL-C), further confirms resistin’s association with hypertension, since hypertension is known to be the most decisive mediator of development of arterial stiffness. Its levels therefore appear to be associated with a predisposition for hypertension, though the pathophysiological mechanism underlying the abovementioned association is not yet established.
In our study, resistin levels were positively correlated with the presence of albuminuria (AC ≥ 30 mg/g). Although limited data exist as to the relationship between serum resistin levels and diabetic nephropathy in T1DM patients, data from T2DM patients have reported increased serum resistin levels in subjects with advanced diabetic nephropathy [13]. A possible explanation includes decreased renal clearance together with subclinical inflammation that is present even in the early stages of CKD [41]. Furthermore, in non-diabetic populations, serum resistin levels are reported to be increased in patients with microalbuminuria [42]. A recent study in 83 T2DM patients found a positive correlation between resistin levels and albuminuria [43]; however, another study in 202 T1DM patients found no correlation between resistin levels and albuminuria [44]. A possible explanation for this discrepancy may have been provided by another study which also reported that serum resistin levels are independently associated with albuminuria in 635 non-diabetic patients, the authors speculating that genetic background and not inflammation might be the missing link between resistin and nephropathy [45].
Adipokines and macrovascular angiopathy (IMT and PWV)
As expected, PWV and IMT were positively correlated with traditional cardiovascular risk factors such as age, diabetes duration, HbA1c, WHR, BMI, SBP, DBP, and CSBP. In our study, resistin was the only adipokine that showed correlation with PWV, but there was no other direct correlation between angiopathy markers and serum adipokine levels. cIMTmax (p = 0.025) and arterial stiffness (p = 0.003) were significantly higher in overweight or obese T1D patients. Obesity, as a major cardiovascular risk factor, is known to be associated with an accelerated atherosclerotic and atherothrombotic process, resulting in increased cardiovascular morbidity and mortality [46]. There is also increasing evidence (largely from experimental studies) that obesity-associated altered adipokine levels are closely involved in the pathogenesis of atherosclerotic vascular diseases [47].
Over the last decade, increasing experimental and clinical data have revealed a direct effect of adipokines on vascular function and atherogenesis, which is independent of their effects on insulin sensitivity and glucose/fat metabolism [25, 48]. There are often conflicting results regarding adipokine levels in T1D patients and their relationship to macrovascular disease markers such as IMT and PWV [49,50,51,52]. The role of adipokines in macrovascular angiopathy remains a matter of debate not only in T1DM patients [53]. The impact of SNPs and mutations on candidate loci encoding different adipokines, the role of the leptin/leptin receptor axis, the genetically driven changes in adiponectin levels, and the emerging concept of “adipokine-resistance,” consisting in the disruption of the cell signaling pathway beyond adipokine receptor activation, are all potential explanations for why changes in adipokine levels might not directly reflect a deteriorated/improved cardiometabolic profile [53].
In conclusion, the association between adipokines and macroangiopathy comes with important limitations including the cross-sectional design of most studies, limiting the ability to infer causality. Few longitudinal studies have been conducted, all with inconsistent results. In addition, findings may be influenced by basal cardiovascular risk and by specific characteristics of the studied populations, making it difficult to generalize from the conclusions. The complexity of the interrelationships in the chain “obesity-cardiovascular risk factors and adipokines” makes it difficult to determine the direct effects of each pair of factors. Prospective studies taking these multiple variables in different populations into account are needed to investigate the role of adipokines in order to understand the mechanisms via which adipokines affect atherosclerotic cardiovascular outcome in the long term [25].
Limitations
The current study is cross-sectional and the sample size is small. Also, the patients were divided into two groups, according to BMI, of normal weight and overweight, and there was no healthy (non-diabetic) control group. Moreover, the male/female ratio in the two groups was significantly different; thus, the comparisons of the studied parameters between the two groups could have been influenced by the gender difference. Therefore, causation cannot be determined for any of the observed relationships. Second, we used a single fasting baseline measurement of adipocytokines; however, considering that hormones might exhibit diurnal and intra-individual fluctuations and the fact that different isoforms of resistin exist that could differ in their biological activities, one sample is not sufficiently accurate to characterize an individual’s hormone levels. Finally, data on macrovascular complications (history of peripheral artery disease, ischemic heart disease, and cerebrovascular disease) were not included in this study. On the other hand, potential strengths of the study are that all patients underwent a 24-h ambulatory BP measurement, which is more trustworthy compared to a single ambulatory BP monitoring. It is important to outline the novelty of the research question given the fact that very few published reports on the relationship of adipokines with insulin resistance, obesity, microangiopathy, and macroangiopathy in T1DM patients currently exist.
Conclusions
Serum adipokine levels in patients with T1DM seem to have a similar dependence on adipose tissue as in healthy subjects without diabetes.
Microvascular complications appear to correlate with serum adipokine levels (retinopathy with adiponectin and albuminuria with resistin). No strong direct association between adipokine levels and markers of early atherosclerosis was identified, but resistin seems to play a crucial role in the development of hypertension and arterial stiffness. Further research is needed on whether the regulation of adipokines could be a promising novel approach for managing cardiovascular outcomes in T1DM and obese patients.
Data availability
The datasets generated during the current study are available from the corresponding author on request.
Code availability
Not applicable.
Abbreviations
- BMI:
-
Body mass index
- BW:
-
Body weight
- CBP:
-
Central blood pressure
- CCA:
-
Common carotid artery
- cIMT:
-
Carotid artery intima media thickness
- cIMTmax:
-
Maximum carotid artery intima media thickness
- MNSI:
-
Michigan Neuropathy Screening Instrument
- CKD:
-
Chronic kidney disease
- DBP:
-
Diastolic blood pressure
- eGFR:
-
Estimated glomerular filtration rate
- HbA1c:
-
Glycated hemoglobin
- PWV:
-
Pulse wave velocity
- SBP:
-
Systolic blood pressure
- T1DM:
-
Type 1 diabetes mellitus
- T2DM:
-
Type 2 diabetes mellitus
- UACR:
-
Urine albumin to creatinine ratio
References
Tuomi T, Santoro N, Caprio S, Cai M, Weng J, Groop L (2014) The many faces of diabetes: a disease with increasing heterogeneity. Lancet 383:1084–1094. https://doi.org/10.1016/S0140-6736(13)62219-9
Minges KE, Whittemore R, Grey M (2013) Overweight and obesity in youth with type 1 diabetes. Annu Rev Nurs Res 31:47–69. https://doi.org/10.1891/0739-6686.31.47
Xu P, Cuthbertson D, Greenbaum C, Palmer JP, Krischer JP, Diabetes Prevention Trial-Type 1 Study Group (2007) Role of insulin resistance in predicting progression to type 1 diabetes. Diabetes Care 30:2314–2320. https://doi.org/10.2337/dc06-2389
Fourlanos S, Narendran P, Byrnes GB, Colman PG, Harrison LC (2004) Insulin resistance is a risk factor for progression to type 1 diabetes. Diabetologia 47:1661–1667. https://doi.org/10.1007/s00125-004-1507-3
Athyros V, Tziomalos K, Karagiannis A, Anagnostis P, Mikhailidis D, Fiorino G, Rovida S, Correale C, Malesci A, Danese S (2009) Should adipokines be considered in the choice of the treatment of obesity-related health problems? Curr Drug Targets 11:122–135. https://doi.org/10.2174/138945010790030992
Yoo HJ (2014) Adipokines as a novel link between obesity and atherosclerosis. World J Diabetes 5:357. https://doi.org/10.4239/wjd.v5.i3.357
(2015) Standards of medical care in diabetes-2015. https://doi.org/10.2337/dc15-S001
Association AD (2021) 11. Microvascular complications and foot care: standards of medical care in diabetes—2021. Diabetes Care 44:S151–S167. https://doi.org/10.2337/DC21-S011
Berukstis A, Jarasunas J, Daskeviciute A, Ryliskyte L, Baranauskas A, Steponeniene R, Laucevicius A (2019) How to interpret 24-h arterial stiffness markers: comparison of 24-h ambulatory Mobil-O-Graph with SphygmoCor office values. Blood Press Monit 24:93–98. https://doi.org/10.1097/MBP.0000000000000369
Kumar R, Mal K, Razaq MK, Magsi M, Memon MK, Memon S, Afroz MN, Siddiqui HF, Rizwan A (2020) Association of leptin with obesity and insulin resistance. Cureus 12.https://doi.org/10.7759/cureus.12178
Park H-KK, Ahima RS (2015) Physiology of leptin: energy homeostasis, neuroendocrine function and metabolism. Metabolism 64:24–34. https://doi.org/10.1016/j.metabol.2014.08.004
Mantzoros CS, Magkos F, Brinkoetter M, Sienkiewicz E, Dardeno TA, Kim S-Y, Hamnvik O-PR, Koniaris A (2011) Leptin in human physiology and pathophysiology. Am J Physiol Endocrinol Metab 301:E567–E584. https://doi.org/10.1152/ajpendo.00315.2011
S. Popovic D, Sekerus V (2016) Levels of different adipocytokines in chronic complications of type 1 diabetes mellitus. Integr Obes Diabetes 2: . https://doi.org/10.15761/IOD.1000156
Verrotti A, Basciani F, De Simone M, Morgese G, Chiarelli F (2000) Leptin concentration in non-obese and obese children with type 1 diabetes mellitus. Biomed Pharmacother 54:69–73. https://doi.org/10.1016/S0753-3322(00)88854-X
Majewska KA, Majewski D, Skowrońska B, Stankiewicz W, Fichna P (2015) Serum leptin and adiponectin levels in children with type 1 diabetes mellitus - relation to body fat mass and disease course. Adv Med Sci 61:117–122. https://doi.org/10.1016/j.advms.2015.10.002
Kiess W, Anil M, Blum WF, Englaro P, Juul A, Attanasio A, Dötsch J, Rascher W (1998) Serum leptin levels in children and adolescents with insulin-dependent diabetes mellitus in relation to metabolic control and body mass index. Eur J Endocrinol 138:501–509
Greco AV, Mingrone G, Giancaterini A, Manco M, Morroni M, Cinti S, Granzotto M, Vettor R, Camastra S, Ferrannini E (2002) Insulin resistance in morbid obesity: reversal with intramyocellular fat depletion. Diabetes 51:144–151. https://doi.org/10.2337/diabetes.51.1.144
Gil-Campos M, Cañete R, Gil A (2004) Hormones regulating lipid metabolism and plasma lipids in childhood obesity. Int J Obes 28:S75–S80. https://doi.org/10.1038/sj.ijo.0802806
Cui J, Panse S, Falkner B (2011) The role of adiponectin in metabolic and vascular disease: a review. Clin Nephrol 75:26–33
Galler A, Gelbrich G, Kratzsch J, Noack N, Kapellen T, Kiess W (2007) Elevated serum levels of adiponectin in children, adolescents and young adults with type 1 diabetes and the impact of age, gender, body mass index and metabolic control: a longitudinal study. Eur J Endocrinol 157:481–489. https://doi.org/10.1530/EJE-07-0250
Blaslov K, Bulum T, Zibar K, Duvnjak L (2013) Relationship between adiponectin level, insulin sensitivity, and metabolic syndrome in type 1 diabetic patients. Int J Endocrinol 2013:535906. https://doi.org/10.1155/2013/535906
Kishida K, Kim KK, Funahashi T, Matsuzawa Y, Kang H-C, Shimomura I (2011) Relationships between circulating adiponectin levels and fat distribution in obese subjects. J Atheroscler Thromb 18:592–595. https://doi.org/10.5551/jat.7625
Cnop M, Havel PJ, Utzschneider KM, Carr DB, Sinha MK, Boyko EJ, Retzlaff BM, Knopp RH, Brunzell JD, Kahn SE (2003) Relationship of adiponectin to body fat distribution, insulin sensitivity and plasma lipoproteins: evidence for independent roles of age and sex. Diabetologia 46:459–469. https://doi.org/10.1007/s00125-003-1074-z
Mantzoros CS, Li T, Manson JE, Meigs JB, Hu FB (2005) Circulating adiponectin levels are associated with better glycemic control, more favorable lipid profile, and reduced inflammation in women with type 2 diabetes. J Clin Endocrinol Metab 90:4542–4548. https://doi.org/10.1210/jc.2005-0372
Sabbatini AR, Fontana V, Laurent S, Moreno H (2015) An update on the role of adipokines in arterial stiffness and hypertension. J Hypertens 33:435–444. https://doi.org/10.1097/HJH.0000000000000444
Van de Voorde J, Pauwels B, Boydens C, Decaluwé K (2013) Adipocytokines in relation to cardiovascular disease. Metabolism 62:1513–1521. https://doi.org/10.1016/j.metabol.2013.06.004
Kim DH, Kim C, Ding EL, Townsend MK, Lipsitz LA (2013) Adiponectin levels and the risk of hypertension. Hypertension 62:27–32. https://doi.org/10.1161/HYPERTENSIONAHA.113.01453
Weyer C, Funahashi T, Tanaka S, Hotta K, Matsuzawa Y, Pratley RE, Tataranni PA (2001) Hypoadiponectinemia in obesity and type 2 diabetes: close association with insulin resistance and hyperinsulinemia. J Clin Endocrinol Metab 86:1930–1935. https://doi.org/10.1210/jcem.86.5.7463
Chandran M, Phillips SA, Ciaraldi T, Henry RR (2003) Adiponectin: more than just another fat cell hormone? Diabetes Care 26:2442–2450
Fu Z, Gong Y, Löfqvist C, Hellström A, Smith LEH (2016) Review: adiponectin in retinopathy. Biochim Biophys Acta - Mol Basis Dis 1862:1392–1400
Hadjadj S, Aubert R, Fumeron F, Pean F, Tichet J, Roussel R, Marre M (2005) Increased plasma adiponectin concentrations are associated with microangiopathy in type 1 diabetic subjects. Diabetologia 48:1088–1092. https://doi.org/10.1007/s00125-005-1747-x
Ferreira-Hermosillo A, Molina-Ayala M, Ramírez-Rentería C, Vargas G, Gonzalez B, Isibasi A, Archundia-Riveros I, Mendoza V (2015) Inflammatory cytokine profile associated with metabolic syndrome in adult patients with type 1 diabetes. J Diabetes Res 2015:972073. https://doi.org/10.1155/2015/972073
Redondo MJ, Rodriguez LM, Haymond MW, Hampe CS, Smith EO, Balasubramanyam A, Devaraj S (2014) Serum adiposity-induced biomarkers in obese and lean children with recently diagnosed autoimmune type 1 diabetes. Pediatr Diabetes 15:543–549. https://doi.org/10.1111/pedi.12159
Schäffler A, Büchler C, Müller-Ladner U, Herfarth H, Ehling A, Paul G, Schölmerich J, Zietz B (2004) Identification of variables influencing resistin serum levels in patients with type 1 and type 2 diabetes mellitus. Horm Metab Res 36:702–707. https://doi.org/10.1055/s-2004-826015
Fehmann H-C, Heyn J (2002) Plasma resistin levels in patients with type 1 and type 2 diabetes mellitus and in healthy controls. Horm Metab Res 34:671–673. https://doi.org/10.1055/s-2002-38241
Takata Y, Osawa H, Kurata M, Kurokawa M, Yamauchi J, Ochi M, Nishida W, Okura T, Higaki J, Makino H (2008) Hyperresistinemia is associated with coexistence of hypertension and type 2 diabetes. Hypertension 51:534–539. https://doi.org/10.1161/HYPERTENSIONAHA.107.103077
Thomopoulos C, Daskalaki M, Papazachou O, Rodolakis N, Bratsas A, Papadopoulos DP, Papavasileiou MV, Perrea D, Makris T (2011) Association of resistin and adiponectin with different clinical blood pressure phenotypes. J Hum Hypertens 25:38–46. https://doi.org/10.1038/jhh.2010.22
Zhang Y, Li Y, Yu L, Zhou L (2017) Association between serum resistin concentration and hypertension: a systematic review and meta-analysis. Oncotarget 8:41529–41537. https://doi.org/10.18632/oncotarget.17561
Papadopoulos DP, Makris TK, Perrea D, Papazachou O, Daskalaki M, Sanidas E, Votteas V (2008) Adiponectin – insulin and resistin plasma levels in young healthy offspring of patients with essential hypertension. Blood Press 17:50–54. https://doi.org/10.1080/08037050701876307
Zhang L, Curhan GC, Forman JP (2010) Plasma resistin levels associate with risk for hypertension among nondiabetic women. J Am Soc Nephrol 21:1185–1191. https://doi.org/10.1681/ASN.2009101053
Kawamura R, Doi Y, Osawa H, Ninomiya T, Hata J, Yonemoto K, Tanizaki Y, Iida M, Makino H, Kiyohara Y (2010) Circulating resistin is increased with decreasing renal function in a general Japanese population: the Hisayama study. Nephrol Dial Transplant 25:3236–3240. https://doi.org/10.1093/ndt/gfq155
Tsioufis C, Dimitriadis K, Selima M, Miliou A, Toutouzas K, Roussos D, Stefanadi E, Tousoulis D, Kallikazaros I, Stefanadis C (2010) Association of resistin with urinary albumin excretion in nondiabetic patients with essential hypertension. Am J Hypertens 23:681–686. https://doi.org/10.1038/ajh.2010.34
Cebeci E, Cakan C, Gursu M, Uzun S, Karadag S, Koldas M, Calhan T, Helvaci S, Ozturk S (2019) The main determinants of serum resistin level in type 2 diabetic patients are renal function and inflammation not presence of microvascular complication, obesity and insulin resistance. Exp Clin Endocrinol Diabetes 127:189–194. https://doi.org/10.1055/s-0043-121262
Bulum T, Vučić Lovrenčić M, Tomić M, Vučković-Rebrina S, Roso V, Kolarić B, Vuksan V, Duvnjak L (2019) Serum adipocytokines are associated with microalbuminuria in patients with type 1 diabetes and incipient chronic complications. Diabetes Metab Syndr Clin Res Rev 13:496–499. https://doi.org/10.1016/j.dsx.2018.11.001
Menzaghi C, Salvemini L, Fini G, Thompson R, Mangiacotti D, Di Paola R, Morini E, Giorelli M, De Bonis C, De Cosmo S, Doria A, Trischitta V (2012) Serum resistin and kidney function: a family-based study in non-diabetic, untreated individuals. PLoS ONE 7:e38414. https://doi.org/10.1371/journal.pone.0038414
Tziomalos K, Athyros V, Karagiannis A (2014) Treating arterial stiffness in young and elderly patients with the metabolic syndrome. Curr Pharm Des 20:6106–6113. https://doi.org/10.2174/1381612820666140417101523
Csongrádi É, Káplár M, Nagy B, Koch CA, Juhász A, Bajnok L, Varga Z, Seres I, Karányi Z, Magyar MT, Oláh L, Facskó A, Kappelmayer J, Paragh G (2017) Adipokines as atherothrombotic risk factors in obese subjects: associations with haemostatic markers and common carotid wall thickness. Nutr Metab Cardiovasc Dis 27:571–580. https://doi.org/10.1016/j.numecd.2017.02.007
Ntaios G, Gatselis NK, Makaritsis K, Dalekos GN (2013) Adipokines as mediators of endothelial function and atherosclerosis. Atherosclerosis 227:216–221. https://doi.org/10.1016/j.atherosclerosis.2012.12.029
Atabek ME, Kurtoglu S, Demir F, Baykara M (2004) Relation of serum leptin and insulin-like growth factor-1 levels to intima-media thickness and functions of common carotid artery in children and adolescents with type 1 diabetes. Acta Paediatr 93:1052–1057
Yazıcı D, Yavuz D, Öğünç AV, Şirikçi Ö, Toprak A, Deyneli O, Akalın S (2012) Serum adipokine levels in type 1 diabetic patients: association with carotid intima media thickness. Metab Syndr Relat Disord 10:26–31. https://doi.org/10.1089/met.2011.0052
Shah AS, Dolan LM, Lauer A, Davis C, Dabelea D, Daniels SR, Hamman RF, Marcovina S, Wadwa RP, Urbina EM (2012) Adiponectin and arterial stiffness in youth with type 1 diabetes: the SEARCH for diabetes in youth study. J Pediatr Endocrinol Metab 25:717–721. https://doi.org/10.1515/jpem-2012-0070
Alman AC, Talton JW, Wadwa RP, Urbina EM, Dolan LM, Hamman RF, D’Agostino RB, Marcovina SM, Dabelea DM (2018) Inflammation, adiposity, and progression of arterial stiffness in adolescents with type 1 diabetes: the SEARCH CVD study. J Diabetes Complications 32:995–999. https://doi.org/10.1016/j.jdiacomp.2018.08.004
Ruscica M, Baragetti A, Catapano AL, Norata GD (2017) Translating the biology of adipokines in atherosclerosis and cardiovascular diseases: gaps and open questions. Nutr Metab Cardiovasc Dis 27:379–395. https://doi.org/10.1016/j.numecd.2016.12.005
Acknowledgements
We wish to thank all volunteers who participated in this trial.
Funding
This work was supported by the Northern Greece Diabetes Association.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
The study was conducted in accordance with the principles of the Declaration of Helsinki and was approved by the Ethics in Research Committee of the School of Public Health of the Aristotle University of Thessaloniki.
Consent to participate
All participants provided written informed consent.
Consent for publication
All authors read and approved the final manuscript before submission.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kollari, E., Zografou, I., Sampanis, C. et al. Serum adipokine levels in patients with type 1 diabetes are associated with degree of obesity but only resistin is independently associated with atherosclerosis markers. Hormones 21, 91–101 (2022). https://doi.org/10.1007/s42000-021-00328-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42000-021-00328-9