Key summary points
We investigated the relationship between physical activity and functional recovery in post-acute rehabilitation units.
AbstractSection FindingsDecreased sedentary behavior time and increased total physical activity time were associated with high functional recovery.
AbstractSection MessageInterventions for physical activity duration may be effective in improving activities of daily living in post-acute and subacute older patients.
Abstract
Purpose
The effect of increased physical activity duration on functional recovery in older inpatients in subacute settings is not well established. This study aimed to investigate the relationship between physical activity and functional recovery in older patients receiving post-acute and subacute care.
Methods
We analyzed cohort data of hospitalized older patients (age ≥ 65 years) in the post-acute rehabilitation units. The main outcome was functional independence measure (FIM) gain. Physical activity was measured using a triaxial accelerometer. Changes in sedentary behavior and total physical activity time from admission to discharge were measured as changes in each physical activity time. Logistic regression analysis was performed to examine the relationship between changes in physical activity and FIM gain.
Results
A total of 210 patients were eligible for analysis. The mean age of the study patients was 83.6 ± 7.2 years, and 63.8% (n = 134) were female. According to the multivariate regression analysis, changes in sedentary behavior time were significantly associated with high recovery of FIM gain (odds ratio [OR] 0.996, 95% confidence interval [CI]: 0.993–1.000; p = 0.026), and changes in total physical activity time also showed a similar association (OR 1.006, 95% CI 1.000–1.011; p = 0.041).
Conclusion
Decreased sedentary behavior time and increased total physical activity time were significantly associated with high functional recovery in post-acute rehabilitation units. These results suggest that interventions for physical activity duration may be effective in improving activities of daily living in older post-acute and subacute patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Functional decline during hospitalization, which is called “hospital associated disability” is a huge problem especially for older patients. In the acute ward, one-third to one-half of older patients experience functional decline [1,2,3,4,5], and many patients remain restricted in activities of daily living (ADL) a year after discharge [3]. Furthermore, functional decline during hospitalization is associated with institutionalization [6] and mortality [3]. Therefore, post-acute care for functional recovery has become important owing to the increasing aging population [7].
In Japan, community-based integrated care units were established in 2014 to provide post-acute rehabilitation [8]. The primary objective of these units is to facilitate functional recovery in older patients. However, in addition, they provide respite care and other services. Nevertheless, achieving sufficient functional recovery is difficult because many patients experience negative factors for functional improvement such as older age, requiring long-term care, cognitive decline, physical inactivity, and malnutrition [9, 10] in post-acute rehabilitation units. Therefore, it is necessary to improve or support variable factors among these negative factors during early hospitalization.
Reducing sedentary time and promoting physical activity are important for improving functional recovery in older patients. It is well known that hospitalized older patients become inactive from the time of admission [11,12,13], and this lifestyle leads to adverse health outcomes, such as muscle wasting [14, 15] and declining cardiovascular health [16], and even has a negative impact on functional recovery [17]. Among them, the effect of promoting increased activity time/decreased inactivity time on functional recovery is well established for some diseases, such as stroke [18, 19] and postoperative lower extremity [20, 21]. In addition, recent studies have shown that even light-intensity physical activity may effectively improve physical function in patients with poor physical function [22, 23]. However, this has not been established for older inpatients in post-acute settings with a variety of primary conditions and negative factors.
Therefore, this study aimed to investigate the relationship between changes in physical activity and functional improvement in older patients in post-acute rehabilitation units. We believe that the current study provides novel information that will help in the functional improvement of older patients in the post-acute and subacute phases.
Methods
Study design and participants
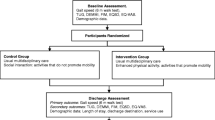
This prospective cohort study included older patients admitted to the community-based integrated care units of Shonan Keiiku Hospital, Fujisawa, Japan, from October 2020 to January 2022. The inclusion criteria were patients aged 65 years and older. The exclusion criteria were patients who did not intend to improve ADL; who were not intended for post-acute rehabilitation, independent ADL at admission, ADL limitations due to rest management, difficulty in communication, hospitalization for less than one week, readmission during the study period, hospital transfer, missing data, and death during hospitalization. The determination of “patients not intending to improve ADL” was made via a pre-admission interview with the patient, family, and medical staff. This study was conducted in accordance with the Declaration of Helsinki, and the study protocol was reviewed and approved by the Ethics Committee of Shonan Keiiku Hospital. Written informed consent was obtained from all the participants or their families.
Rehabilitation program
All patients underwent rehabilitation for 40–60 min daily during hospitalization under the medical insurance system. Rehabilitation programs were individually designed according to health conditions and goals for each patient and included gait training, balance exercises, strength exercise, self-care skills training, and swallowing training by physical, occupational, and speech therapy.
Measurements
Outcome
The outcome of the current study was functional independence measure (FIM) gain. The FIM is an indicator of ADL and consists of a motor domain of 13 sub-items and a cognitive domain of five sub-items [24]. The scores were evaluated on a 7-point scale ranging from 1 (total assistance) to 7 (complete independence). The total FIM scores range from 18 to 126, with lower scores indicating greater dependency. The FIM gain was obtained by subtracting the admission scores from the discharge scores [25]. We operationally defined the third quartile or higher for FIM gain as a high level of recovery for ADL. In this study, well-trained physical therapists assessed the FIM.
Physical activity
We investigated the amount of 3 days’ physical activity at two time points: immediately after admission and immediately before discharge. Physical activity was measured using a triaxial accelerometer (Active Style Pro HJA-750C; Omron Healthcare Co. Ltd. Kyoto, Japan; 52 × 40 × 12 mm, weighing approximately 23 g, and including a battery), which can estimate metabolic equivalents (METs) from a wide range of body movements during ADL [26, 27]. The accelerometer was worn on the waist for 24 h, excluding the dressing and bathing times. We analyzed the acceleration data between 5:00 AM and 0:00 AM each day and estimated the METs every 60 s. From the estimated METs data, we calculated time (min/day) spent in sedentary behavior (≤ 1.5 METs) [28] and in total physical activity (≥ 1.6 METs). We defined total physical activity as an accumulation of light-intensity (1.6 < 3.0 METs), moderate (3.0–6.0 METs), and vigorous (> 6.0 METs) physical activity [29]. We defined the changes in sedentary behavior and total physical activity time from admission to discharge as the change in each physical activity time.
Other variables
Demographic and clinical characteristics, including age, sex, body mass index (BMI), reason for admission to the acute ward, comorbidities, length of hospital stay, requirement for long-term care, cognitive function, depression, physical function and nutritional intake were investigated. Comorbidity was assessed using the Charlson comorbidity index (CCI) [30], and we defined scores 3 or more as disease severity [31]. We defined requiring long-term care as all care need levels (one to five) according to the standards for long-term care requirement certification in Japan [32]. Cognitive function was assessed using the Mini-Mental State Examination (MMSE) [33], which ranges from 0 to 30; we defined scores of 23 or less as cognitive dysfunction [34]. Depression was assessed using the 15-item Geriatric Depression Scale (GDS-15) [35]. Physical function was assessed using the Berg balance scale (BBS), which ranges from 0 to 56; we defined a score of 45 or less as low physical function [36]. Nutritional intake was estimated as the average calorie intake per actual body weight per day based on the intake rate of the main and side dishes recorded by the nurse during the three days (9 meals) immediately after admission [10].
Statistical analysis
We compared all variables between the high recovery of FIM gain group and the low recovery group using the chi-square test, t-test, or Mann–Whitney U test for categorical or continuous variables. Logistic regression analysis was used to estimate the odds ratios (OR) and 95% confidence intervals (CI) of the relationships between changes in physical activity (sedentary behavior and total physical activity) and FIM gain. Demographic details and level of physical activity were used as covariates to adjust for confounding factors in the multivariate logistic regression models (model 1: adjusted for age, sex, CCI, MMSE, and BBS; model 2: model 1 + changes in sedentary behavior or changes in total physical activity). All statistical analyses were performed using the IBM SPSS for Mac (version 27.0; IBM Japan, Tokyo, Japan). Statistical significance was set at p < 0.05.
Results
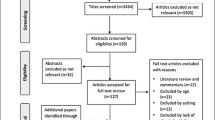
Of the 620 patients enrolled in the study, 410 were excluded based on the aforementioned exclusion criteria, resulting in 210 eligible patients for analysis (Fig. 1). Most of the excluded patients were those not intending to improve ADL, such as those in respite care or for whom rehabilitation was not prescribed because ADL had not declined. The clinical and demographic characteristics of patients in the high recovery (FIM gain) and low recovery groups are shown in Table 1. The mean age of the study patients was 83.6 ± 7.2 years, and 63.8% (n = 134) of them were female. The comparison between the two groups showed significant differences in sex (p = 0.041), requirement for long-term care (p = 0.002), MMSE (p = 0.027), BBS (p < 0.001) and FIM scores at discharge (p < 0.001).
FIM gain, changes in sedentary behavior, and total physical activity time are shown in Table 2. The median FIM gains (interquartile range [IQR]) in the high and low recovery groups were 26 and 9 (3–14), respectively (p < 0.001). The high recovery group showed a significant decrease in sedentary behavior time (p < 0.001) and a significant increase in total physical activity time (p < 0.001) compared with the low recovery group. According to the multivariate logistic regression analysis, changes in sedentary behavior time were significantly associated with high recovery of FIM gain (OR 0.996, 95% CI 0.993–1.000; p = 0.026), and changes in total physical activity time also showed a similar association (OR 1.006, 95% CI 1.000–1.011; p = 0.041) (Table 3).
Discussion
This prospective cohort study investigated the relationship between changes in physical activity and functional recovery in subacute and post-acute older patients. We found that greater changes in sedentary behavior time and amount of physical activity were significantly associated with high functional recovery after adjusting for covariates. This result suggests that reducing sedentary behavior time and increasing amount of physical activity may be valuable factors for functional recovery in older patients in post-acute rehabilitation units.
Two characteristic differences were observed in the group comparisons. First, the high functional recovery group had significantly more female than male patients, similar to previous surveys [37]. In acute rehabilitation units, it has been suggested that women may be more active in physical exercises [38]. Thus, women may experience a better effect of physical activity on functional recovery than men. Second, no group differences were found in comorbidities. Although the CCI indicates the extent of complications, its content does not necessarily contribute to functional recovery. This is because the CCI does not adequately assess geriatric syndrome, which significantly affects functional recovery in older patients.
The amount of physical activity for these study participants was only 1.5 h/day, which is similar to that of patients in acute care wards [39, 40]. It is well known that this inactivity leads to functional decline in acute patients but is not well established in post-acute patients. In the high functional recovery group, part of the sedentary time was replaced with physical activity time during the post-acute hospitalization period. These results suggest the need to promote physical activity for functional recovery in post-acute patients.
The current findings suggest that replacing sedentary time with physical activity may help prevent or improve functional decline caused by disuse. Reducing sedentary behavior in acute care units is important to prevent disuse syndrome [41, 42], and increasing the amount of physical activity leads to the recovery of lower extremity muscle strength and the cardiovascular system after the acute phase [43, 44]. Thus, sedentary and physical activity durations may be more likely to promote ADL recovery. In particular, post-acute patients are more likely to have limited physical activity owing to several impairments, psychological aspects, and disease management [45, 46] and disease management; therefore, replacing sedentary behavior with mild physical activity may have been successful.
This cohort study had two major strengths. First, we used an objective measure of physical activity levels in older post-acute patients. Because many previous studies on physical activity in post-acute older patients used a number of steps and questionnaires [47, 48], it was difficult to apply them clinically based on several findings. Among these, we measured physical activity in detail, as it suggests that encouraging a change from sedentary to active behavior contributes to ADL recovery. Second, this study included patients with ambulatory difficulties and mild to moderate cognitive impairment, which are common in post-acute care units. Previous studies examining physical activity and functional changes have excluded cases of cognitive impairments [4, 17]. The findings of this study can be widely applied to post-acute and subacute older patients.
This cohort study had two limitations. First, patients had a wide range and severity of acute illnesses. The degree of functional recovery varied with the disease type and disease severity and needed to be included as an adjustment variable. Second, physical activity and FIM were measured simultaneously, and this study was cross-sectional; therefore, it was not possible to refer to a causal relationship. In other words, it is possible that the amount of physical activity may have improved as a result of functional recovery. However, several previous studies have indicated that physical activity is related to functional recovery, particularly in individuals with impaired physical functioning [17,18,19,20,21, 23, 47]. Therefore, a similar relationship may be hypothesized in the present study. A larger sample size that considers diseases and interventions is required to clarify these limitations.
Conclusion
In conclusion, decreased sedentary behavior time and increased total physical activity time were significantly associated with high functional recovery in post-acute rehabilitation units. These results suggest that interventions for physical activity duration may be effective in post-acute and subacute older patients with negative factors for functional recovery. Nevertheless, due to the cross-sectional nature of this study and the absence of a definitive causal relationship, further investigation is necessary to elucidate the relationship between physical activity and functional recovery such as multicenter cohort or intervention studies.
Availability of data and materials
The data generated and analyzed during this study are not publicly available but are available from the corresponding author on reasonable request.
References
Zisberg A, Shadmi E, Gur-Yaish N, Tonkikh O, Sinoff G (2015) Hospital-associated functional decline: the role of hospitalization processes beyond individual risk factors. J Am Geriatr Soc 63(1):55–62. https://doi.org/10.1111/jgs.13193
Palleschi L, Fimognari FL, Pierantozzi A et al (2014) Acute functional decline before hospitalization in older patients. Geriatr Gerontol Int 14(4):769–777. https://doi.org/10.1111/ggi.12160
Boyd CM, Landefeld CS, Counsell SR et al (2008) Recovery of activities of daily living in older adults after hospitalization for acute medical illness. J Am Geriatr Soc 56(12):2171–2179. https://doi.org/10.1111/j.1532-5415.2008.02023.x
Tasheva P, Vollenweider P, Kraege V et al (2020) Association between physical activity levels in the hospital setting and hospital-acquired functional decline in elderly patients. JAMA Netw Open 3(1):e1920185. https://doi.org/10.1001/jamanetworkopen.2019.20185. (Published 2020 Jan 3)
Covinsky KE, Palmer RM, Fortinsky RH et al (2003) Loss of independence in activities of daily living in older adults hospitalized with medical illnesses: increased vulnerability with age. J Am Geriatr Soc 51(4):451–458. https://doi.org/10.1046/j.1532-5415.2003.51152.x
Portegijs E, Buurman BM, Essink-Bot ML, Zwinderman AH, de Rooij SE (2012) Failure to regain function at 3 months after acute hospital admission predicts institutionalization within 12 months in older patients. J Am Med Dir Assoc 13(6):569.e1-569.e5697. https://doi.org/10.1016/j.jamda.2012.04.003
Wang YC, Chou MY, Liang CK, Peng LN, Chen LK, Loh CH (2019) Post-acute care as a key component in a healthcare system for older adults. Ann Geriatr Med Res 23(2):54–62. https://doi.org/10.4235/agmr.19.0009
Shimada T, Suzuki Y, Yada Y, Hori S, Ushida K, Momosaki R (2021) Community-based integrated care units: intermediate care units for older adults in Japan. J Am Med Dir Assoc 22(8):1774–1775. https://doi.org/10.1016/j.jamda.2021.03.027
Klenk J, Wekenmann S, Schwickert L, Lindemann U, Becker C, Rapp K (2019) Change of objectively-measured physical activity during geriatric rehabilitation. Sensors (Basel) 19(24):5451. https://doi.org/10.3390/s19245451. (Published 2019 Dec 11)
Maeda K, Koga T, Akagi J (2018) Nutritional variables predict chances of returning home and activities of daily living in post-acute geriatric care. Clin Interv Aging 13:151–157. https://doi.org/10.2147/CIA.S154129. (Published 2018 Jan 26)
Garner J, Smith M (2021) Activity levels of inpatients admitted to two rehabilitation units in regional hospitals: an observational study. Aust J Rural Health 29(3):399–407. https://doi.org/10.1111/ajr.12703
Fini NA, Holland AE, Keating J, Simek J, Bernhardt J (2017) How physically active are people following stroke? Systematic review and quantitative synthesis. Phys Ther 97(7):707–717. https://doi.org/10.1093/ptj/pzx038
Taylor NF, Peiris CL, Kennedy G, Shields N (2016) Walking tolerance of patients recovering from hip fracture: a phase I trial. Disabil Rehabil 38(19):1900–1908. https://doi.org/10.3109/09638288.2015.1107776
Kortebein P, Ferrando A, Lombeida J, Wolfe R, Evans WJ (2007) Effect of 10 days of bed rest on skeletal muscle in healthy older adults. JAMA 297(16):1772–1774. https://doi.org/10.1001/jama.297.16.1772-b
Di Girolamo FG, Fiotti N, Milanović Z et al (2021) The aging muscle in experimental bed rest: a systematic review and meta-analysis. Front Nutr 8:633987. https://doi.org/10.3389/fnut.2021.633987. (Published 2021 Aug 4)
Saunders TJ, McIsaac T, Douillette K et al (2020) Sedentary behaviour and health in adults: an overview of systematic reviews. Appl Physiol Nutr Metab 45(10 (Suppl. 2)):S197–S217. https://doi.org/10.1139/apnm-2020-0272
Pavon JM, Sloane RJ, Pieper CF et al (2020) Accelerometer-measured hospital physical activity and hospital-acquired disability in older adults. J Am Geriatr Soc 68(2):261–265. https://doi.org/10.1111/jgs.16231
Gunnes M, Indredavik B, Langhammer B et al (2019) Associations between adherence to the physical activity and exercise program applied in the LAST study and functional recovery after stroke. Arch Phys Med Rehabil 100(12):2251–2259. https://doi.org/10.1016/j.apmr.2019.04.023
Kimura Y, Ohji S, Nishio N et al (2022) The impact of wheelchair propulsion based physical activity on functional recovery in stroke rehabilitation: a multicenter observational study. Disabil Rehabil 44(10):2027–2032. https://doi.org/10.1080/09638288.2020.1821249
Talkowski JB, Lenze EJ, Munin MC, Harrison C, Brach JS (2009) Patient participation and physical activity during rehabilitation and future functional outcomes in patients after hip fracture. Arch Phys Med Rehabil 90(4):618–622. https://doi.org/10.1016/j.apmr.2008.10.024
Christensen J, Peters C, Gililland J, Stoddard G, Pelt C (2021) Physical activity, pain interference and comorbidities relate to PROMIS physical function in younger adults following total knee arthroplasty. Disabil Rehabil 43(26):3741–3747. https://doi.org/10.1080/09638288.2020.1749944
Terbraak M, Kolk D, Vroomen JLM, Twisk JWR, Buurman BM, van der Schaaf M (2023) Post-discharge light physical activity indicates recovery in acutely hospitalized older adults—the Hospital-ADL study. BMC Geriatr 23(1):311. https://doi.org/10.1186/s12877-023-04031-9. (Published 2023 May 19)
Rojer AGM, Ramsey KA, Trappenburg MC et al (2023) Patterns of objectively measured sedentary behavior and physical activity and their association with changes in physical and functional performance in geriatric rehabilitation inpatients. J Am Med Dir Assoc 24(5):629-637.e11. https://doi.org/10.1016/j.jamda.2023.01.011
Kidd D, Stewart G, Baldry J et al (1995) The functional independence measure: a comparative validity and reliability study. Disabil Rehabil 17(1):10–14. https://doi.org/10.3109/09638289509166622
Koh GC, Chen CH, Petrella R et al (2013) Rehabilitation impact indices and their independent predictors: a systematic review. BMJ Open 3:e003483. https://doi.org/10.1136/bmjopen-2013-003483
Ohkawara K, Oshima Y, Hikihara Y, Ishikawa-Takata K, Tabata I, Tanaka S (2011) Real-time estimation of daily physical activity intensity by a triaxial accelerometer and a gravity-removal classification algorithm. Br J Nutr 105(11):1681–1691. https://doi.org/10.1017/S0007114510005441
Oshima Y, Kawaguchi K, Tanaka S et al (2010) Classifying household and locomotive activities using a triaxial accelerometer. Gait Posture 31(3):370–374. https://doi.org/10.1016/j.gaitpost.2010.01.005
Pate RR, O’Neill JR, Lobelo F (2008) The evolving definition of “sedentary.” Exerc Sport Sci Rev 36(4):173–178. https://doi.org/10.1097/JES.0b013e3181877d1a
Norton K, Norton L, Sadgrove D (2010) Position statement on physical activity and exercise intensity terminology. J Sci Med Sport 13(5):496–502. https://doi.org/10.1016/j.jsams.2009.09.008
Charlson ME, Pompei P, Ales KL, MacKenzie CR (1987) A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 40(5):373–383. https://doi.org/10.1016/0021-9681(87)90171-8
Tuty Kuswardhani RA, Henrina J, Pranata R, Anthonius Lim M, Lawrensia S, Suastika K (2020) Charlson comorbidity index and a composite of poor outcomes in COVID-19 patients: a systematic review and meta-analysis. Diabetes Metab Syndr 14(6):2103–2109. https://doi.org/10.1016/j.dsx.2020.10.022
Iwagami M, Tamiya N (2019) The long-term care insurance system in Japan: past, present, and future. JMA J 2(1):67–69. https://doi.org/10.31662/jmaj.2018-0015
Folstein MF, Folstein SE, McHugh PR (1975) “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 12(3):189–198. https://doi.org/10.1016/0022-3956(75)90026-6
Tsoi KK, Chan JY, Hirai HW, Wong SY, Kwok TC (2015) Cognitive tests to detect dementia: a systematic review and meta-analysis. JAMA Intern Med 175(9):1450–1458. https://doi.org/10.1001/jamainternmed.2015.2152
Cruice M, Worrall L, Hickson L (2011) Reporting on psychological well-being of older adults with chronic aphasia in the context of unaffected peers. Disabil Rehabil 33(3):219–228. https://doi.org/10.3109/09638288.2010.503835
Berg KO, Wood-Dauphinee SL, Williams JI, Maki B (1992) Measuring balance in the elderly: validation of an instrument. Can J Public Health 83(Suppl 2):S7–S11
Seematter-Bagnoud L, Lécureux E, Rochat S, Monod S, Lenoble-Hoskovec C, Büla CJ (2013) Predictors of functional recovery in patients admitted to geriatric postacute rehabilitation. Arch Phys Med Rehabil 94(12):2373–2380. https://doi.org/10.1016/j.apmr.2013.06.024
Verstraeten LMG, Sacchi F, van Wijngaarden JP, Meskers CGM, Maier AB (2023) Sarcopenia, malnutrition and cognition affect physiotherapy frequency during geriatric rehabilitation: RESORT cohort. Ann Phys Rehabil Med 66(6):101735. https://doi.org/10.1016/j.rehab.2023.101735
Jawad BN, Petersen J, Andersen O, Pedersen MM (2022) Variations in physical activity and sedentary behavior during and after hospitalization in acutely admitted older medical patients: a longitudinal study. BMC Geriatr 22(1):209. https://doi.org/10.1186/s12877-022-02917-8. (Published 2022 Mar 15)
Jasper U, Yadav L, Dollard J, Jadczak AD, Yu S, Visvanathan R (2020) Sedentary behaviour in hospitalised older people: a scoping review. Int J Environ Res Public Health 17(24):9359. https://doi.org/10.3390/ijerph17249359. (Published 2020 Dec 14)
Bortz WM 2nd (1984) The disuse syndrome. West J Med 141(5):691–694
Norimoto M, Yamashita M, Yamaoka A et al (2021) Early mobilization reduces the medical care cost and the risk of disuse syndrome in patients with acute osteoporotic vertebral fractures. J Clin Neurosci 93:155–159. https://doi.org/10.1016/j.jocn.2021.09.011
Valenzuela PL, Morales JS, Pareja-Galeano H et al (2018) Physical strategies to prevent disuse-induced functional decline in the elderly. Ageing Res Rev 47:80–88. https://doi.org/10.1016/j.arr.2018.07.003
Kalisch BJ, Lee S, Dabney BW (2014) Outcomes of inpatient mobilization: a literature review. J Clin Nurs 23(11–12):1486–1501. https://doi.org/10.1111/jocn.12315
Wen P, Sun L, Shen T, Wang Z (2024) Factors impeding physical activity in older hospitalised patients: a qualitative meta-synthesis. J Clin Nurs 33(8):3273–3282. https://doi.org/10.1111/jocn.17158
Kuzmik A, Boltz M, Resnick B, Drazich BF, Galvin JE (2023) Gender, pain, and function associated with physical activity after hospitalization in persons living with dementia. Alzheimer Dis Assoc Disord 37(4):357–362. https://doi.org/10.1097/WAD.0000000000000583
Agmon M, Zisberg A, Gil E, Rand D, Gur-Yaish N, Azriel M (2017) Association between 900 steps a day and functional decline in older hospitalized patients. JAMA Intern Med 177(2):272–274. https://doi.org/10.1001/jamainternmed.2016.7266
Douma JAJ, de Beaufort MB, Kampshoff CS et al (2020) Physical activity in patients with cancer: self-report versus accelerometer assessments. Support Care Cancer 28(8):3701–3709. https://doi.org/10.1007/s00520-019-05203-3
Acknowledgements
We thank the nursing staff and rehabilitation therapists of our hospitals for their contributions to data collection. We are grateful to our laboratory members for their helpful advice in drafting this manuscript. We would like to thank Tomoya Ishigaki for providing the activity analysis program.
Funding
This study did not receive any specific grants from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
Conceptualization: [Hiroki Saegusa], [Iwao Kojima], [Minoru Yamada]; Methodology: [Hiroki Saegusa], [Iwao Kojima], [Yusuke Terao], [Shingo Koyama], [Mizue Suzuki], [Yosuke Kimura], [Yuhei Otobe], [Minoru Yamada]; Formal analysis and investigation: [Hiroki Saegusa], [Iwao Kojima], [Yusuke Terao], [Shingo Koyama], [Mizue Suzuki], [Yosuke Kimura], [Yuhei Otobe], [Minoru Yamada]; Writing – original draft preparation: [Hiroki Saegusa]; Writing—review and editing: [Hiroki Saegusa], [Iwao Kojima], [Minoru Yamada]; Methodology: [Hiroki Saegusa], [Iwao Kojima], [Yusuke Terao], [Shingo Koyama], [Mizue Suzuki], [Shu Tanaka], [Yosuke Kimura], [Yuhei Otobe], [Takuya Aoki], [Sho Nishida], [Yasuhiro Kitagawa], [Minoru Yamada]; Resources: [Minoru Yamada]; Supervision: [Minoru Yamada].
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Ethical approval
This study was approved by the Committee of Ethics of Shonan Keiiku Hospital (No. 20-012).
Informed consent
Taken from all patients or their caregivers/relatives.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Saegusa, H., Kojima, I., Terao, Y. et al. The impact of changes in physical activity on functional recovery for older inpatients in post-acute rehabilitation units. Eur Geriatr Med (2024). https://doi.org/10.1007/s41999-024-01051-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s41999-024-01051-5