Abstract
Obstructive sleep apnoea syndrome (OSAS) is a type of breathing disorder with upper airway obstruction, leading to oxy-haemoglobin desaturations and sleep disturbance. However awareness regarding various anthropometric parameters used to analyse OSA syndrome pre-clinically is inadequate. In developing nations, like India, resources are not adequate for analysis of sleep disturbances. That is why the prevalence and validity of various anthropometric parameters including neck circumference, Body mass index and waist circumference to be established and verified regularly. We have also seen the data from oral aspect and its anomalies. Various articles from Pubmed, scopus, google scholar were searched for data. These body measurements may provide pre-clinical aspect of OSA, whether it is present or not. Discussion was done on these anthropometric parameters and which parameter is gold standard for the pre-clinical investigations.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Sleep is defined as a state of human mind and body, characterized by physiological reduction in sensory activity, voluntary muscles and consciousness. Sleep plays a major role in many of the body functions and developments such as learning, memory, neural development, emotional regulation, cardiovascular and metabolic functions [1]. Studies have consolidated in last two decades that a good quality of sleep is essential for the healthier life.
Now a day’s technology and social modernization in our life have led to increase sleep disorder prevalence. A range of pathological changes in respiratory effort during physiological sleep, leading to increased disordered breathing which may be due to nasal or upper airway obstruction referred as sleep disordered breathing (SDB). Obstructive sleep apnoea syndrome (OSAS) is a type of SDB which is characterized by recurrent events of partial or complete upper airway obstruction due to increase in negative intrathoracic pressure during sleep or atonia of tongue during REM sleep, leading to oxy-haemoglobin desaturations [2]. There are enough evidence suggesting OSAS as a risk factor for future development of cardiovascular diseases (CVD) including hypertension, coronary artery disease (CAD) and, metabolic syndrome, neurologic disorders such as stroke, dementia, multiple sclerosis and social effects such traffic trauma [3, 4].
In this review the literature search began with key words such as "sleep apnea," "obesity," "anthropometric parameter," "upper airway anatomy." and "sleep study," using Google scholar, Medline, Scopus and PubMed. For additional sources, reference list of the identified articles were also searched. We tried to report original findings from publications that contain relevant theoretical information on the areas which are addressed in this review. The purpose of this study is to provide a brief review of the present scientific knowledge on the clinical, anthropometric and polysomnographic characteristics of non-obese and obese patients with OSA and finding out literature gaps in these areas.
2 Epidemiology
OSA is a chronic disorder of modern society with estimated prevalence of 2.4–4.9% in men and 1–2.1% in women [5]. According to literature based analysis published in 2019, prevalence of OSA using American academy of sleep medicine (AASM) 2012 diagnostic criteria; it was estimated that 936 million (95% CI 903–970) adults aged 30–69 years (male and female) are having mild to severe OSA and around 425 million (399–450) adults of same age group have moderate to severe OSA globally [12]. Highest numbers of affected individuals are from China, later followed by USA, Brazil and India [12]. In other parts of the world like United States have prevalence of sleep disorders around 56%, Western Europe have 31% and 23% in Japan [7]. In rural population of South India, prevalence of OSAS was estimated using Berlin questionnaire was 8.72% in total, with males estimating around 7.4% and 11.7% in female [6]. Patients who are obese, having type II diabetes mellitus, cardiovascular disorders such as stroke, Heart failure, atherosclerosis and myocardial infarction are more prone to OSA [8]. It was reported in study at South Delhi, that OSA found to be 9% and OSAS is 2.8% [9]. A study did in North Indian hospitals, reported prevalence of OSA and OSAS in males to be 4.4% and 2.5%, 2.5% and 1% in females [10]. Obese population are more at risk of OSA, around 33.5%, reported by Singh et al., in North Indian population [11].
3 Pathophysiology
3.1 Normal Sleep physiology
As per AASM, sleep architecture is majorly divided into two stages:
-
1.
Non-rapid eye movement (NREM)
-
2.
Rapid eye movement (REM)
Sleep begins with NREM sleep which consist of three stages; stage 1 (N1) showing alpha waves in electroencephalogram (EEG) constituting 2–5% of sleep cycle, NREM stage 2 (N2) characterize by sleep spindle and K-complexes constitutes 45 to 55% of sleep cycle, NREM stage 3 is also known as slow wave sleep (SWS), is 10–15% of total sleep cycle [13, 14]. REM stage is associated with dreaming, desynchronized brain activity which constitutes 20–25% of sleep cycle [14,15,16,17]. Total sleep cycle is of about 90 to 120 min that includes three stages of NREM and REM stage [14].
3.2 Upper Airway Anatomy
The upper airway is a tubular structure, which begins with the nasal cavity (choanae) and ends at the vocal folds. This is a multipurpose structure, performing various functions such as swallowing of food, breathing air in or out and speech. The upper airway consists of numerous muscles making it prone to collapse in patient with OSA. The cross sectional area of the upper airway in the patients with OSA is narrower than the healthy subjects [18]. There are many reasons behind the narrowing of upper cross sectional area like larger tonsils, fat deposition at the base of tongue or along the airway in obesity [19]. Another important factor is position of hyoid, which determine the airway length and the collapsibility of upper airway [20]. Upper airway collapse is shown in Fig. 1.
3.3 Mechanism of obstruction in upper airway
The largest dilating muscle of tongue is genioglossus, due to decreased activity in the patients with OSA compared to normal individuals [21]. On contraction of genioglossus, tongue protude, leading to widening of oropharyngeal airway in the anterior–posterior aspect [21]. The hypoglossal nerve (12th cranial nerve) innervates genioglossus and it provides input from chemoreceptor, respiratory neurons and negative pressure receptors of the airways [22]. Inspiration leads to increase activity of genioglossus, to prevent airway collapse from negative airway pressure [23]. In OSA patients, velopharynx is one of the common sites for obstruction [24]. Other muscles which are maintaining the upper airway patency are tensor veli palatini control stiffness, levator veli palatini for elevation, musculus uvulae for uvular position, palatoglossus for positioning of tongue, together composing soft palate [24]. The muscles of hyoid act bi-directionally, anteriorly and upward via mylohyoid and geniohyoid, and caudally via sternohyoid and thyrohyoid, this together helps in airway dilation [26].
3.4 Breathing during awake and sleep
During awake stage, humans are obligate nose breather. While during sleep, they breathe again via nose and it is not affected by any stages of sleep or changes in position of body [27]. SDB is mainly due to nasal obstruction as nose is the foremost anatomical boundary of upper airway. Nasal pathology in SDB is explained by number of pathophysiology mechanisms like Starling resistor model, the nasal ventilator reflex, an unstable upper airway and the role of nitric oxide (NO) [28]. The Starling resistor model explains the basis for treatment of OSA with nasal continuous positive airway pressure (CPAP) [29]. This model consists of two rigid segments as a part of tube, with a collapsible segment interposed in between, within the box. The nose and trachea representing the rigid tube in humans and muscular pharyngeal regions is considered to be collapsible segment. Hence for the normal patency of pharynx during sleep, the pressure inside the pharynx should be greater in comparison to surrounding tissue outside [16]. When the pressure inside the pharynx falls in comparison to outside leading to collapse is known as pharyngeal critical closing pressure [30].
On observing studies it suggested that Pharyngeal critical closing pressure (PCRIT) plays a major role in the pathogenesis of OSA [31]. PCRIT is both sensitive (majority of people have upper airway collapse) and specific (many normal people do not have upper airway collapse) for ruling out OSA [32]. Elevated levels of PCRIT can be attributed to many structural defects in the upper airway and irregular neuromuscular control [33]. It is seen that OSA severity increased during REM in comparison to NREM sleep due to decrease in neuromuscular tone in some patients, specially children and women [34, 35]. Neuromuscular activity in the pharyngeal region is led by many chemical and mechanical reflexes [32]. In conditions like hypercapnia, stimulated neuromuscular activity in upper airway, causing decrease in PCRIT [36] and in Hypocapnia, there is increase in PCRIT [37]. During phasic activation of pharyngeal muscles, upper airway demonstrated decrease in collapse during expiration in comparison to inspiration [38]. Now once there is a collapse in upper airway, there are many factors that response to obstruction and expresses OSA as shown in Fig. 2.
3.5 Role of polysomnography in OSA
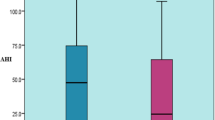
Polysomnography is a sleep study which is performed in the laboratory to analyze the apnea–hypopnea index (AHI), which is required for the diagnosis of OSA. As apnea is complete obstruction of upper airflow anatomy and hypopnea is a partial obstruction of upper airflow anatomy [39, 40]. Hypopnea is also measured by desaturations in the level of oxygen by 3% or more or arousal from the sleep. The AHI is generally calculated by adding all apneas and hypopneas and then they are divided by total sleep time. Together both this should last for 10 s or more to be considered as an event [39, 40]. If both of these occur for 15 or more events per hour, or 5 or more per hour with symptoms of daytime sleepiness, impaired cognition or mood disorder or cardiovascular comorbidities, is considered to be as OSA [39,40,41]. PSG signals are classified into three groups: for the recognition of sleep related events are electroencephalogram (EEG), electrooculogram (EOG), submental electromyogram (EMG), those which are required for the monitoring of cardiac arrhythmic events are electrocardiogram (ECG) and for the airflow or respiratory related events are oximetry, oronasal thermistor, nasal canula and thoracoabdominal effort. A microphone may be used which is attached to the neck for recording of snoring. EEG is used to measure the sleep stages from the central, frontal and occipital regions of the brain. The EEG activity in the reticular nucleus of the thalamus shows hyperpolarization of neurons, generating EEG spindles and afferent input to the cortex are blocked and maintain sleep [42]. During REM sleep, it is generally designated for mild OSA, with AHI range around 5 to 14. Moderate OSA with AHI between 15 to 30, have fragmented sleep with sleep architecture is preserved. Severe OSA is present in both REM and NREM stage with AHI above 30. Althought it more common in NREM sleep related OSA and in older men [43]. REM OSA is having 5 or more events in REM stage and less than 5 events in NREM stage with total REM sleep duration of 30 min [43]. NREM OSA have AHI in NREM stage more than 5 events [43]. REM OSA shows hypotonia of the muscles involving upper airway leading to obstruction [44]. EOG helps in identification of sleep stage (REM or NREM) by noting activity of electropotential difference between the retina and cornea. EMG is used to measure the muscle tension in chin and jawline region in conditions like bruxism and leg movements in condition like REM behavior disorder. ECG is used to analyze any cardiac abnormalities. In this way PSG helps in diagnosing SBD and its variant.
4 Risk factors for OSA
4.1 Genetic disorders
OSA is genetically transmissible and there are much evidence suggesting genetic contribution towards OSA susceptibility [45]. Genetic factors contribute around 40% of variance in AHI [46]. In relation to snoring, twin studies have shown that monozygotic twins are more prone to habitual snoring than in dizygotic twins [47]. Orofacial birth defects such as Pierre-Robin syndrome and Down syndrome are also cause of OSA. Down syndrome is a chromosomal disorder having enlarged tongue, tonsils and adenoids leading to upper airway obstruction causing OSA [48]. Pierre-Robin syndrome is a genetic mutational disorder in which infant has a retrognathia or micrognathia again leading to OSA [49]. In Marfan syndrome, craniofacial dysmorphisms with lax upper airway muscles leads to OSA [50]. OSA is linked to various loci of the major histocompatibility complex (MHC). The genetic relationship between OSA and obesity is implicated with various genetic markers like HLA-A2, HLA-A33, HLA-B7, B65, B63, B73 [51].
4.2 Anthropometric parameters
4.2.1 Body Mass Index
BMI is convenient to measure but does not identify body composition. Studies have shown BMI with body composition relates more closely with AHI [52]. It is also argued that BMI, age, internal laryngeal circumference and NC are important parameters for OSA [53]. BMI with data on body mass and height, indicates nutritional status in many of the epidemiological studies, until they develop body composition for these surveys [54]. And later on papers on body composition is proven to be superior in OSA prediction in comparison to BMI [52].
Higher BMI values were correlated with lower oxygen saturation, higher apnea–hypopnea index (AHI) and decrease in sleep efficiency. BMI greater than 28 kg/m2 increases the risk for OSA in persons around eight to twelve times [57]. Persons with upper body obesity and BMI > 40 kg/m2 are more at risk of OSA [58]. A study by martin et al. reported that in both genders if BMI is greater than 30 kg/m2 it is associated with the development of OSA [59]. In Turkish population, soylu et al. showed that BMI over 27.7 kg/m2 in females and 28.9 kg/m2 in males is risk factor for developing OSA [60]. Caucasian population is showing BMI for obesity at 30 kg/m2 but on the other hand Asian populations have obesity at the cut-off value of 25 kg/m2 [61]. Korean population, shows cut-off values for BMI is around 23.05 kg/m2 for females and 24.95 kg/m2 of males [62]. Mostly Asians are having higher percentage of body fat whether it is upper body or middle than the Caucasian of the same of sex, age and BMI [63]. Due to this reason, a percentage of Asian population who are having BMI below the current WHO BMI cut-off value of 25 kg/m2 are at risk of OSA.
4.2.2 Obesity
Obesity is considered to be one of the major predisposing factors for OSA, so it is relatively important to signify their roles and importance. In around 40% of patients with obesity have severe OSA [64]. A study showed that there is 4–6% more chances of developing OSA with 10% gain in body weight or 6 kg/m2 rise in Body Mass Index (BMI) [65]. It was seen in patients with OSA, if fat is deposited at specific sites it gets worse. Sites including upper airway, results in decrease in the size of lumen and leading to collapse of upper airway, leading to apnea [66]. In addition, if fat is deposited around the thorax (Abdominal obesity), it reduces chest compliance and lung functional residual capacity and may cause in increase oxygen demand [67]. Obesity in visceral region is common in OSA population [68]. There are many studies based on different population that have documented correlation between apnea–hypopnea index (AHI) and BMI. Obesity is graded on the basis of various anthropometric parameters such as BMI, hip circumference (HC), neck circumference (NC), waist cirumference (WC), waist to hip ratio (W:H), Mallampati score (MS) and thyromental distance (TD) [69,70,71]. All these factors contribute to OSA and predict its correlation with OSA [69].
4.2.3 Neck Circumference
NC is measured using non-stretchable plastic tape, subject in standing position, measurement is done in the midway of neck, between mid-cervical spine posterior and mid anterior neck. In males there is laryngeal prominence, so it is measured bellow the prominence. NC is more significantly correlated with OSA than any other anthropometric parameters. NC is higher among male than in females [72]. NC is considered to be marker of central obesity, stated by Onat et al. [19]. NC is considered to be gold standard in 1991 for the prediction of OSA in comparison to other factors [55]. Generally NC is the reflection of upper body obesity and is considerably better marker than BMI for OSA [70]. It is stated that neck circumference greater than 43 cm in male and 38 cm in females are at greater risk of OSA [73]. In a study, 11 women out of 13 had NC greater than 38 cm and 7 man out of 12 had NC more than 43 cm [73]. In 2003, NC was considered better marker for AHI measurement [74].
4.2.4 Waist Circumference
Waist circumference (WC) is measured using non-stretchable tape above hip-bones, over the navel region. WC is a type of abdominal obesity defined at 90 cm or more in male and 80 cm or more in female in Asian population whereas in Middle East and Caucasian population it is 94 cm or more in male and 80 cm or more in female [75]. In a study, cut-off value of WC at 94 cm in males and 80 cm in females is considered to be risk factor for OSA [65]. Soylu et al. studied the cut-off value at 105 cm in males and 101 cm for females in Turkish population [66]. Another study reported WC at 88.5 cm for males and 76.5 cm in females is a risk factor for OSA [75]. Although different races have different criteria for abdominal obesity, with western countries have higher WC than Asian countries.
4.3 Hip Circumference And Waist/Hip Ratio
HC is measured with the help of tape at a level parallel to the floor, placing around the widest circumference of the buttocks. HC is directly related with WC, is inversely associated with the levels of blood glucose and blood pressure [76]. HC provide more specific measure for subcutaneous gluteofemoral adipose tissue than WC. It is of greater possibility that larger HC reflects greater muscle mass. A study reported by Manolopolous et al. that there are evidence from in vitro and in vivo studied of adipose tissue, that gluteofemoral fat is protective for our body by collecting excess fatty acids and exposure from high levels of lipids [77]. HC contribution in OSA is more in relation with WC as waist to hip ratio (WHR). The difference between these adverse effects of increased level of WC and the defensive effects of HC on the other may be due to the observation, that most of the pathological influence of adiposity is due to visceral, instead of the superficial fat [78]. There is increase in superficial fat leading to increase in WC and this misleads the real effect of visceral obesity. In this way WHR improves the assessment of adiposity and help in prediction of obesity and its role in OSA [77].
5 Oral anomalies
In population who are non-obese, an orofacial anomaly such as retrognathia and micrognathia lead to OSA [79]. Additional features that may influence to OSA are enlarged palatine tonsils, large uvula, high-arched palate, deviation of nasal septum, increase in anterior facial height, short anterior cranial base, macroglossia, displaced hyoid bone, long soft palate and decrease in posterior upper airway space [80,81,82,83]. Airway evaluation to be done by checking condition of tongue size and state, grading of tonsils size to be done by universally accepted standard (Grades 1–4) [84]. Classification predicting severity of airway space for intubation is to be graded 1–4 as the Mallampati score [85]. Nasal conchae hypertrophy was also evaluated as one of the cause for airway obstruction and breathing disorder [86]. A study had demonstrated that increase in BMI with tonsillar hypertrophy and higher mallampati score indicates greater risk for OSAS [86]. Mandibular assessment for the effect of its repositioning, both vertical and horizontal, on the upper airway can be done using a device known as George gauge or wax bite [87]. One more technique termed as acoustic reflexion, which evaluates restriction at airway site and the position of mandible which may be restricting the upper airway [88]. Oral appliance therapy is required for the effective treatment craniofacial anomalies. Many authors have suggested for the mandibular advancement or reposition devices (MRD or MAD), which increases efficacy without giving much problem to patients and any temporomandibular jaw (TMJ) problems [89]. One of the appliances such as Kleerway was very effective in reducing AHI to < 15 per hour in 71% of the patients [90]. Surgical procedures such as uvulopalatopharyngoplasty (UPPP) and uvulopalatopharyngoglossoplasty (UPPGP) are performed to open the upper airway.
6 Conclusion
OSA is highly prevalent in modern day society and often unrecognized by healthcare professionals. The long-term pathophysiologic consequences of OSA results in impaired cardiac functioning, sympathetic nervous system overdrive, proinflammatory effects like increase in Interleukin 6, 8 and endothelial dysfunction. Thus early recognition of abnormal anthropometric parameters in asymptomatic patients or patients with comorbidities such as hypertension, stroke, heart failure, CAD etc. is essential to cut down long term complications of OSA. The anthropometric parameters such as NC, WC and BMI are significantly connected with accumulation of adipose tissue in the upper part of the body, correlated well with severity of OSA syndrome. Physiological and pathological analysis of the upper airway helps in providing insight into this disease and how to prevent. Studies have been carried out in recent past, for the improvement of our understanding in physiological mechanism of OSA. Still more research is required to correlate anthropometric parameters and OSA phenotypes. In this way new groups of patients with different phenotypes will be defined based on anthropometric and pathophysiological traits in order to choose precise management approach (Table 1).
References
Punjabi NM. The epidemiology of adult obstructive sleep apnea. Proc Am Thoracic Soc. 2008;5:136–43. https://doi.org/10.1513/pats.200709-155MG.
Medicine AAoS. International classification of sleep disorders 2nd ed: Diagnostic and coding manual. Westchester IL: American Academy of Sleep Medicine. 2005.
Xia Y, Fu Y, Xu H, Guan J, Yi H, Yin S. Changes in cerebral metabolites in obstructive sleep apnea: a systemic review and meta-analysis. Sci Rep. 2016;6:28712. https://doi.org/10.1038/srep28712.
Fornadi K, Ronai KZ, Turanyi CZ, Malavade TS, Shapiro CM, Novak M, Mucsi I, Molnar MZ. Sleep apnea is not associated with worse outcomes in kidney transplant recipients. Sci Rep. 2014;4:6987. https://doi.org/10.1038/srep06987.
Sharma SK, Katoch VM, Mohan A, Kadhiravan T, Elavarasi A, Ragesh R, et al. Consensus and evidence-based Indian initiative on obstructive sleep apnea guidelines 2014 ( first edition) Lung India. 2015;32:422–34.
Ashna M Pinto, Uma Devaraj, Priya Ramachandran, Bobby Joseph, and George A D'Souza. Obstructive Sleep Apnea in a rural population in South India: Feasibility of health care workers to administer level III sleep study. Lung India. 2018; 35(4): 301–306.
Léger D, Poursain B, Neubauer D, Uchiyama M. An international survey of sleeping problems in the general population. Curr Med Res Opin. 2008;24(1):307–17.
Drager LF, Togeiro SM, Polotsky VY, Lorenzi-Filho G. Obstructive sleep apnea: a cardiometabolic risk in obesity and the metabolic syndrome. J Am Coll Cardiol. 2013;62:569–76. https://doi.org/10.1016/j.jacc.2013.05.045.
Reddy EV, Kadhiravan T, Mishra HK, Sreenivas V, Handa KK, Sinha S, et al. Prevalence and risk factors of obstructive sleep apnea among middle-aged urban Indians: a community-based study. Sleep Med. 2009;10:913–8.
Vijayan VK, Patial K. Prevalence of obstructive sleep apnoea syndrome in Delhi. India Chest. 2006;130:149–56.
Singh A, Prasad R, Garg R, Kant S, Hosmane GB, Dubey A, et al. A study to estimate prevalence and risk factors of obstructive sleep apnoea syndrome in a semi-urban Indian population. Monaldi Arch Chest Dis. 2017;87:773.
Benjafield AV, Ayas NT, Eastwood PR, Heinzer R, Ip MSM, Morrell MJ, et al. Estimation of the global prevalence and burden of obstructive sleep apnoea: a literature-based analysis. Lancet Respir Med. 2019;7(8):687–98. https://doi.org/10.1016/S2213-2600(19)30198-5Epub 2019 Jul 9.
Silber MH, Ancoli-Israel S, Bonnet MH, Chokroverty S, Grigg-Damberger MM, et al. The visual scoring of sleep in adults. J Clin Sleep Med. 2007;3(2):121–31.
Carskadon M, Dement W. Normal human sleep: an overview. In: Kryger MH, Roth T, Dement WC, editors. Principles and practice of sleep medicine. 4th ed. Philadelphia: Elsevier Saunders; 2005. p. 13–23.
Gais S, Molle M, Helms K, Born J. Learning-dependent increases in sleep spindle density. J Neurosci. 2002;22(15):6830–4.
Ayappa I, Rapoport DM. The upper airway in sleep: physiology of the pharynx. Sleep Med Rev. 2003;7(1):9–33.
Bader G, Gillberg C, Johnson M, Kadesjö B, Rasmussen P. Activity and sleep in children with ADHD. Sleep. 2003;26:A136.
Mortimore IL, Marshall I, Wraith PK. Neck and total body fat deposition in non-obese and obese patients with sleep apnea compared with that in control subjects. Am J Respir Crit Care Med. 1998;57:280–3.
Onat A, Hergenç G, Yüksel H, Can G, Ayhan E, Kaya Z, Dursunoğlu D. Neck circumference as a measure of central obesity: Associations with metabolic syndrome and obstructive sleep apnea syndrome beyond waist circumference. Clin Nutrition. 2009;28:146–51.
Schwab RJ, Gupta KB, Gefter WB, Metzger LJ, Hoffman EA, Pack AI. Upper airway and soft tissue anatomy in normal subjects and patients with sleep-disordered breathing: significance of the lateral pharyngeal walls. Am J Respir Crit Care Med. 1995;152:1673–89.
Mezzanotte WS, Tangel DJ, White DP. Waking genioglossal electromyogram in sleep apnea patients versus normal controls (a neuromuscular compensatory mechanism). J Clin Invest. 1992;89:1571–9.
Horner RL, Innes JA, Holden HB, Guz A. Afferent pathway(s) for pharyngeal dilator reflex to negative pressure in man: a study using upper airway anaesthesia. J Physiol. 1991;436:31–44.
Mathur R, Mortimore IL, Jan MA, Douglas NJ. Effect of breathing, pressure and posture on palatoglossal and genioglossal tone. Clin Sci (Lond). 1995;89(4):441–5.
Jennifer C, Hsia MD. Anatomy and physiology of the upper airway in obstructive sleep apnea. Oper Tech Otolaryngol. 2015. https://doi.org/10.1016/j.otot.2015.03.005.
McWhorter AJ, Rowley JA, Eisele DW, Smith PL, Schwartz AR. The effect of tensor veli palatini stimulation on upper airway patency. Arch Otolaryngol Head Neck Surg. 1999;125(9):937–40.
Van de Graaff WB, Gottfried SB, Mitra J, van Lunteren E, Cherniack NS, Strohl KP. Respiratory function of hyoid muscles and hyoid arch. J Appl Physiol Respir Environ Exerc Physiol. 1984;57:197–204.
Gleeson K, Zwillich CW, Braier K, White DP. Breathing route during sleep. Am Rev Respir Dis. 1986;134(1):115–20.
Georgalas C. The role of the nose in snoring and obstructive sleep apnoea: an update. Eur Arch Otorhinolaryngol. 2011;268(9):1365–73.
Schwartz AR, Smith PL, Wise RA, Gold AR, Permutt S. Induction of upper airway occlusion in sleeping individuals with subatmospheric nasal pressure. J Appl Physiol. 1985;64(2):535–42.
Ahmed SBH, Tripat S, Smitha G, Karen LA, Kashmira B, Divinagracia EG. Choosing the right interface for positive airway pressure therapy in patients with obstructive sleep apnea. Sleep Breath. 2017. https://doi.org/10.1007/s11325-017-1490-9.
Gleadhill IC, Schwartz AR, Schubert N, et al. Upper airway collapsibility in snorers and in patients with obstructive hypopnea and apnea. Am Rev Respir Dis. 1991;143:1300–3.
Pham LV, Schwartz AR. The pathogenesis of obstructive sleep apnea. J Thorac Dis. 2015;7(8):1358–72. https://doi.org/10.3978/j.issn.2072-1439.2015.07.28.
Brouillette RT, Thach BT. A neuromuscular mechanism maintaining extrathoracic airway patency. J Appl Physiol Respir Environ Exerc Physiol. 1979;46:772–9.
Marcus CL, Moreira GA, Bamford O, et al. Response to inspiratory resistive loading during sleep in normal children and children with obstructive apnea. J Appl Physiol. 1985;1999(87):1448–544.
Resta O, Carpanano GE, Lacedonia D, et al. Gender difference in sleep profile of severely obese patients with obstructive sleep apnea (OSA). Respir Med. 2005;99:91–6.
Schwartz AR, Thut DC, Brower RG, et al. Modulation of maximal inspiratory airflow by neuromuscular activity: effect of CO2. J Appl Physiol. 1985;1993(74):1597–605.
Seelagy MM, Schwartz AR, Russ DB, et al. Reflex modulation of airflow dynamics through the upper airway. J Appl Physiol. 1985;1994(76):2692–700.
Schneider H, Boudewyns A, Smith PL, et al. Modulation of upper airway collapsibility during sleep: influence of respiratory phase and flow regimen. J Appl Physiol. 1985;2002(93):1365–76.
Qaseem A, et al. Diagnosis of obstructive sleep apnea in adults: a clinical practice guideline from the American College of Physicians. Ann Intern Med. 2014;161(3):210–20.
Epstein LJ, Kristo D, Strollo PJ Jr., et al.; Adult Obstructive Sleep Apnea Task Force of the American Academy of Sleep Medicine. Clinical guideline for the evaluation, management and long-term care of obstructive sleep apnea in adults. J Clin Sleep Med. 2009;5(3):263–276
John M. Eisenberg Center for Clinical Decisions and Communications Science. Comparative effectiveness of diagnosis and treatment of obstructive sleep apnea in adults. August 8, 2011. In: Comparative Effectiveness Review Summary Guides for Clinicians. Rockville, Md.: Agency for Healthcare Research and Quality (US); 2011. https://www.ncbi.nlm.nih.gov/books/NBK63965/. Accessed January 15, 2016.
Steriade M, McCormick DA, Sejnowski TJ. Thalamocortical oscillations in the sleeping and aroused brain. Science. 1993;262:679–85.
Al Oweidat K, AlRyalat SA, Al-Essa M, Obeidat N. Comparing REM- and NREM-related obstructive sleep apnea in Jordan: a cross-sectional study. Hindawi. Can Respir J Volume 2018, Article ID 9270329, 6 pages https://doi.org/10.1155/2018/9270329
Grace KP, Hughes SW, Horner RL. Identification of the mechanism mediating genioglossus muscle suppression in REM sleep. Am J Respir Crit Care Med. 2013;187(3):311–9.
Kaparianos A, Sampsonas F, Karkoulias K, Spiropoulos K. Obstructive sleep apnoea syndrome and genes. Neth J Med. 2006;64(8):280–9.
Redline S, Tishler PV. The genetics of sleep apnea. Sleep Med Rev. 2000;4(6):583–602. https://doi.org/10.1053/smrv.2000.0120.
Carmelli D, Bliwise DL, Swan GE, Reed T. Genetic factors in self-reported snoring and excessive daytime sleepiness: a twin study. Am J Respir Crit Care Med. 2001;164:949–52.
Ghosh S, Feingold E, Dey SK. Etiology of Down syndrome: Evidence for consistent association among altered meiotic recombination, nondisjunction, and maternal age across populations. Am J Med Genet A. 2009;149A(7):1415–20.
Tewfik TL, Der Kaloustian VM. Congenital anomalies of the ear, nose, and throat. New York, NY: Oxford University Press; 1997. p. 517.
Hollister DW, Godfrey M, Sakai LY, Pyeritz RE. Immunohistologic abnormalities of the microfibrillar-fiber system in the Marfan syndrome. N Engl J Med. 1990;323:152–9.
Saarelainen S, Lehtimaki T, Kallonene E, Laasonein K, Poussa T, Neiminen MM. No relation between apolipoprotein E alleles and obstructive sleep apnea. Clin Genet. 1998;53:147–8.
Oğretmenoğlu O, Süslü AE, Yücel OT, Onerci TM, Sahin A. Body fat composition: a predictive factor for obstructive sleep apnea. Laryngoscope. 2005;115(8):1493–8.
Katz I, Stradling J, Slutsky AS, Zamel N, Hoffstein V. Do patients with obstructive sleep apnea have thick necks? Am Rev Respir Dis. 1990;141(5 Pt 1):1228–311.
Van Gaal LF. Body fat, body fat distribution and the respiratory system: a “fat neck” syndrome? Eur Respir J. 1992;5(4):375–6.
Obesity: Preventing and managing the global epidemic. Geneva: World health organization; 1997. World health organization. Report of a WHO consultation on obesity; p. 158.
Nuzhat A, Sailaja DK, Praveen Kumar N. Implication of revised consensus body mass indices for Asian Indians on clinical obstetric. Pract J Clin Diagn Res. 2014; 8(5):OC01-OC03
Douglas NJ, Polo O. Pathogenesis of obstructive sleep apnoea/hypopnoea syndrome. The Lancet. 1994;344(8923):653–5.
Kwan SYL, Fleetham JA, Enarson DA, Chan-Yeung M. Snoring, obesity, smoking and systemic hypertension in a working population in British Columbia. Am Rev Respir Dis 1991; 143, article A380.
Martins AB, Tufik S, Moura SM. Physiopathology of obstructive sleep apnea-hypopnea syndrome. J Br Pneumol. 2007;33:93–100.
Soylu AC, Levent E, Sarıman N, Yurtlu S, Alparslan S, et al. Obstructive sleep apnea syndrome and anthropometric obesity indexes. Sleep Breath. 2012;16:1151–8.
No authors listed. Obesity: preventing and managing the global epidemic. Report of a WHO consultation. World Health Organ Tech Resp Ser. 2000;894:1–253.
Hyeon HK, Ji YK, Jick HH, Jongmin L, Sung KK, Hwa SM, Sang HL. The associations between anthropometric indices and obstructive sleep apnea in a Korean population. PLoS ONE. 2014;9(12):e114463. https://doi.org/10.1371/journal.pone.0114463.
WHO Expert Consultation. Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet. 2004;363:157–63.
Van Boxem TJ, de Groot GH. Prevalence and severity of sleep disordered breathing in a group of morbidly obese patients. Neth J Med. 1999;54:202.
Young T, Shahar E, Nieto FJ, et al. Predictors of sleep-disordered breathing in community dwelling adults: the Sleep Heart Health Study. Arch Intern Med. 2002;162:893.
Shelton KE, Woodson H, Gay S, Suratt PM. Pharyngeal fat in obstructive sleep apnea. Am Rev Respir Dis. 1993;148(2):462–6.
Naimark A, Cherniack RM. Compliance of the respiratory system and its components in health and obesity. J Appl Physiol. 1960;15:377–82.
Malhotra A, White DP. Obstructive sleep apnoea. Lancet. 2002;360(9328):237–45.
Davidson TM, Patel MR. Waist circumference and sleep disordered breathing. Laryngoscope. 2008;118(2):339–47.
Davies RJ, Ali NJ, Stradling JR. Neck circumference and other clinical features in the diagnosis of the obstructive sleep apnoea syndrome. Thorax. 1992;47(2):101–5.
Hoffstein V, Mateika S. Differences in abdominal and neck circumferences in patients with and without obstructive sleep apnoea. Eur Respir J. 1992;5(4):377–81.
Tufik S, Silva RS, Taddei JA, et al. Obstructive sleep apnoea syndrome in the sao paulo epidemiolgic sleep study. Sleep Med. 2010;11:441–6.
Schwab RJ, Goldberg AN, Pack AL. Sleep apnea syndromes. In: Fishman AP, editor. Fishman's pulmonary diseases and disorders. McGraw-Hill Book: New York, NY, USA; 1998. p. 1617–1637.
Dixon JB, Schachter LM, OBrien PE. Predicting sleep apnea and excessive day sleepiness in the severely obese: indicators for polysomnography. Chest. 2003;123(4):1134–41.
No authors listed. Executive Summary of The Third Report of The National Cholesterol Education Program (NCEP) expert panel on detection, evaluation, and treatment of high blood cholesterol in adults (Adult TreatmentPanel III). JAMA. 2001;285:2486–97.
Snijder MB, Zimmet PZ, Visser M, Dekker JM, Seidell JC, Shaw JE. Independent and opposite associations of waist and hip circumferences with diabetes, hypertension and dyslipidemia: the AusDiab Study. Int J Obes Relat Metab Disord. 2004;28:402–9.
Manolopoulos KN, Karpe F, Frayn KN. Gluteofemoral body fat as a determinant of metabolic health. Int J Obes. 2010;34:949–59.
Despres JP, Lemieux I, Prud’homme D. Treatment of obesity: need to focus on high risk abdominally obese patients. BMJ 2001;322:716–20.
Miles PG, Vig PS, Weyant RJ, Forrest TD, Rockette HE Jr. Craniofacial structure and obstructive sleep apnea syndrome: a qualitative analysis and meta-analysis of the literature. Am J Orthod Dentofacial Orthop. 1996;109:163–72.
Bacon WH, Kreiger J, Turlot JC, Stierle JL. Craniofacial characteristics in patients with obstructive sleep apnea syndrome. Cleft Palate J. 1988;25:374–8.
Lowe AA, Gionhaku N, Takeuchi K, Fleetham JA. Three dimensional CT reconstructions of tongue and airway in adult subjects with OSA. Am J Orthod Dentofacial Orthop. 1986;90:364–74.
Lowe AA, Fleetham JA, Adachi S, Ryan CF. Cephalometric and computed tomographic predictors of obstructive sleep apnea severity. Am J Orthod Dentofacial Orthop. 1995;107:589–95.
Solow B, Skov S, Ovesen J, Norup PW, Wildschiψdtz G. Airway dimensions and head posture in obstructive sleep apnea. Eur J Orthod. 1996;18:571–9.
Fairbanks DN, Fujita S. Snoring and obstructive sleep apnea. 2nd ed. New York: Raven Press; 1994.
Friedman M, Tanyeri H, LaRosa M, Landsberg R, Vaidyanathan K, Pieri S, et al. Clinical predictors of obstructive sleep apnea. Laryngoscope. 1999;109:1901–7.
Bailey DR, Attanasio R. Dentistry's role in the management of sleep disorders. Recognition and management. Dent Clin North Am. 2001;45:619–30.
George T. A new instrument for functional appliance bite registration. J Clin Orthod. 1992;2:721–3.
George T. Breathing disorders in sleep. Clin Chest Med. 1992;13:383–554.
Schmidt-Nowara W, Lowe A, Wiegand L, Cartwright R, Perez-Guerra F, Menn S. Oral appliances for the treatment of snoring and obstructive sleep apnea: a review. Sleep. 1995;18:501–10.
Liu YH, Lowe A, Fleetham J, Park YC. Cephalometric and physiological predictors of the efficacy of an adjustable oral appliance for the treatment of obstructive sleep apnea. Am J Orthod Dentofac Orthop. 2001;120:639–47.
Funding
There are no funding sources to report for this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflicts of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Thukral, H., Gupta, G., Sinha, S. et al. Anthropometric Correlation with Pathophysiology of Obstructive Sleep Apnea (OSA): A Review. Sleep Vigilance 4, 95–103 (2020). https://doi.org/10.1007/s41782-020-00089-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41782-020-00089-2