Abstract
Purpose
This study has compared morphological characteristics of dentin and enamel surfaces after cavity preparation by conventional methods and Er:YAG laser irradiation in different settings by using an Er:YAG laser scanner.
Methods
Thirty-five human teeth were randomly divided into seven groups that received cavity preparations as follows: G1 and G2 with high-speed drill with diamond and carbide burs, respectively, and G3 to G7 with Er:YAG laser scanner (λ = 2940 nm) operating at pulse energies (energy densities) of 420 mJ (840 J/cm2), 490 mJ (980 J/cm2), 560 mJ (1120 J/cm2), 630 mJ (1260 J/cm2) and 700 mJ (1400 J/cm2), respectively. The lased groups were treated with Er:YAG laser scanner in non-contact mode, keeping both frequency (20 Hz) and pulse duration (300 μs) fixed. Samples were sectioned and scanning electron micrographs of the lateral wall of the cavities were taken at different magnifications.
Results
The results showed that both groups of conventional cavity preparations exhibited enamel and dentin surfaces covered by smear layer, with no dentin-enamel boundary being identifiable. All lased groups showed clear enamel-dentin boundaries and clean surfaces of enamel and dentin with different micro-morphologies. The morphology of lased dental surfaces presented homogeneous alterations.
Conclusions
The Er:YAG scanner-assisted cavity preparation produced topographies in the entire dental surfaces and whose morphological characteristics were more favourable to further adhesion of resin restorations than the conventional ones.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The Er:YAG (erbium:yttrium-aluminium-garnet) laser is an effective tool for dental hard tissue removal [1, 2]. This high-power laser removes enamel and dentin with less thermal damage to the surrounding tissue or dental pulp [2]. Additionally, this laser system can produce clean cuts in dental hard tissues with reduced amount of bacteria [3] and other debris [4]. Furthermore, Er:YAG laser has better patient compliance compared to conventional high-speed drills [5].
The Er:YAG emits light with a wavelength of 2.94 μm which is coincident with the absorption peak of the major components of the dental hard tissue, i.e. hydroxyapatite, water and collagen, which have absorption peak in the 2.9-μm region [6, 7]. Dental hard tissue ablation is based on the concept of thermomechanical process [8, 9]. In this process, light energy is absorbed by water molecules and hydroxyl groups in hydroxyapatite. This causes vaporization followed by micro-explosions resulting from the increased steam pressure within the tissue before ejection of microscopic organic and inorganic particles of the substrate.
In many in vitro and in vivo studies in which Er:YAG laser was used for caries removal and/or cavity preparation [10, 11], the laser irradiation has been performed in a manual mode by using a regular handpiece. This technique, although efficient, is not capable of ablating homogeneously the whole substrate surface. To circumvent this problem, especially in vitro studies, it would be better to work with an Er:YAG laser equipment capable of irradiating the dental surfaces in a scanned mode.
An Er:YAG laser scanner equipment meant for dermatological uses is available (Fotona XS Dynamis, Fotona, Slovenia). This laser enables to manually adjust the pilot beam of the Er:YAG scanner with a pre-programmed lasing pattern, which ensures that the whole surface to be treated will receive an equal amount of energy, resulting in a uniformly lased area. Additionally, this makes it possible to prepare a cavity throughout the tooth surface with exact diameter and homogeneous ablation.
These characteristics of the Er:YAG laser scanner would be of importance for more accurate comparisons of dental surface morphologies obtained with different laser irradiation parameters. For that reason, this study aimed to qualitatively compare the morphological characteristics of dentin and enamel surfaces after cavity preparation by conventional methods and Er:YAG laser irradiation in different settings by using an Er:YAG laser scanner. It was expected that the SEM examination of enamel and dentin after cavity preparation showed important topographical differences between the substrates obtained with laser and those obtained with burs (conventional cavity preparation). Our null hypothesis is that both cavity preparation methods would produce similar superficial topographies.
Materials and methods
Human third molars extracted for therapeutic purposes were used after due permission from the patients and then selected for study. Thirty-five freshly extracted teeth, all sound and free from caries, were selected before being cleaned with running water and detergent and stored in normal saline solution at 4 °C for further experimental procedures. The teeth were divided into seven groups, as follows:
-
G1: (n = 5) conventionally prepared with diamond bur
-
G2: (n = 5) conventionally prepared with carbide bur
-
G3: (n = 5) laser prepared with 420-mJ pulse energy
-
G4: (n = 5) laser prepared with 490-mJ pulse energy
-
G5: (n = 5) laser prepared with 560-mJ pulse energy
-
G6: (n = 5) laser prepared with 630-mJ pulse energy
-
G7: (n = 5) laser prepared with 700-mJ pulse energy
The teeth were then mounted on silicon to proceed with laser-assisted cavity preparation. Buccal or lingual surfaces of the molar were selected for class V cavity preparation as these surfaces are relatively broad and flat, which helps visualizing the shape and size of the pilot beam. The cavities presented a 3-mm diameter circular shape, as programmed through the machine, with a depth of approximately 3–4 mm.
Laser and settings
The specimens were irradiated with an Er:YAG laser (λ = 2940 nm) equipped with a scanner handpiece (Fotona XS Dynamis; Fotona, Slovenia). The irradiations were done in both non-contact and focus mode by keeping the frequency (20 Hz) and pulse duration (300 μs) fixed for all experimental groups, with five different pulse energies being used (Table 1). The Fotona XS Dynamis has a scanner pattern of lasing of different shapes. The round pattern was chosen, which was applied with the exact diameter of cavity by manually adjusting the pilot beam of the Er:YAG scanner. After selection of the shape and diameter of the pilot beam, only holding the handpiece in a fixed position produced a well-defined round-shaped cavity. The pilot beam was adjusted to irradiate a 3-mm diameter area (spot size of 0.7 mm). As the dermatological Er:YAG laser scanner is not equipped with a suitable water supply to avoid intense temperature increase, an air-water spray device was used concomitantly (Biolase Waterlase MD Turbo, Biolase, USA) to provide cooling at a ratio of 60% air and 60% water (for 15 ml water/min) during irradiations.
For preparation of conventional cavities, an airotor handpiece (Kavo Super LUX2, Kavo, Germany) with diamond or carbide burs was used. The diameters of the cavities were the same of those of the lased cavities (3 mm).
Scanning electron microscopy
Immediately after cavity preparation, all the teeth were sectioned at the enamel-cement junction to separate the crown from the roots. Next, the crowns were sectioned perpendicular to their long axis at the centre of the prepared cavities in order to expose their lateral walls.
For further scanning electron microscopy examination, the specimens were fixed in 2.5% glutaraldehyde and 0.1 M phosphate-buffered solution for 24 h at room temperature, dehydrated in ascending grades of ethanol, submitted to chemical drying in hexamethyldisilazane (HMDS, Electron Microscopy Sciences, Fort Washington, PA, USA) and finally sputter-coated with 30-nm gold layer (Sputter SCD030, Duchmesser 54mm, Balzers Union FL-9496, Lichtenstein). Scanning electron micrographs of two specimens per group were obtained with an ESEM XL30 FEG microscope (Philips, Frankfurt/Main, Germany) operating at 10.00 kV in different magnifications (×65, ×1000 and ×6500).
Results
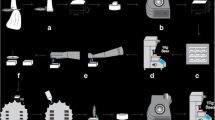
Scanning electron micrographs (Fig. 1) illustrate the overall micro-morphology of the lateral walls of the cavities in all groups at low magnification (×65).
Scanning electron micrographs of the overall walls of the specimens in all groups: conventional cavity preparation with diamond bur (G1) (a) and carbide bur (G2) (b); laser cavity preparation with 420-mJ pulse energy (G3) (c); 490-mJ pulse energy (G4) (d); 560-mJ pulse energy (G5) (e); 630-mJ pulse energy (G6) (f); and 700-mJ pulse energy (G7) (g). Original magnification at ×6500
Both groups of conventional cavity preparations (G1 and G2) exhibited enamel and dentin surfaces covered by smear layer (Fig. 1a, b, respectively), with no clear dentin-enamel boundaries being identifiable. In those cavities prepared with diamond bur, the entire surface presented a striatum morphology resembling a negative image of the diamond bur surface (Fig. 1a). However, cavities prepared with carbide bur exhibited a relatively smooth surface (Fig. 1b).
Figure 1c–g illustrates the lateral walls of cavities prepared with laser irradiation (G3 to G7). Surfaces were free of smear layer and it was possible to observe clear enamel-dentin boundaries. However, at this magnification, it was not possible to observe remarkable differences between the lased groups regarding enamel and dentin topographies.
The scanning electron micrographs (Fig. 2) illustrate the enamel micro-morphology in the lateral walls of the cavities in all experimental groups at higher magnifications (×1000 and ×6500). These images showed a more evident presence of smear layer in the conventionally prepared cavities (G1 and G2) and no enamel prisms could be identified in the electron micrographs (Fig. 2a–d). In addition, each laser group (G3 to G7) exhibited different enamel morphologies (Fig. 2e–n).
Scanning electron micrographs of the lateral walls of the enamel in all groups: conventional cavity preparation with diamond bur (G1) (a, b) and with carbide bur (G2) (c, d); laser cavity preparation with 420-mJ pulse energy (G3) (e, f); 490-mJ pulse energy (G4) (g, h); 560-mJ pulse energy (G5) (i, j); 630-mJ pulse energy (G6) (k, l); and 700-mJ pulse energy (G7) (m, n). Original magnification at ×1000 and inserts at ×6500
Cavities prepared with 420-mJ pulse energy (G3) exhibited a rough enamel surface represented by irregular dissolution of prismatic structure, with open prisms and a glass-like appearance in the inter-prismatic areas (Fig. 2e, f). The typical keyhole pattern of the enamel was not evident (Fig. 2e) and some sharp jagged projections of the prisms were seen (Fig. 2f). Cavities prepared with 490-mJ pulse energy (G4) exhibited enamel surface that was also rough, but the prism exposure seemed to follow a regular pattern through shallow grooves (Fig. 2g, h). There was a parallel sequence of densely packed prisms alternating with lateral and sharp jagged projections of the prisms (Fig. 2h). The prismatic structure of enamel was well visualized in cavities prepared with 560-mJ pulse energy (G5—Fig. 2i, j), including the keyhole pattern with an overall roughness on the surface. The prisms also seemed to follow a regular pattern through shallow and sinuous grooves (Fig. 2i), which at higher magnification showed to be composed of exposed prisms in different directions (Fig. 2j). The 630-mJ pulse energy (G6) also created a rough enamel surface similar to that of 560-mJ, i.e. composed by jagged structures of enamel prisms (Fig. 2k, l) and structures resembling inorganic re-crystallization. The 700-mJ pulse energy (G7) created a rough and porous enamel surface. Occasional cracks and signs of re-crystallization were also seen forming thin scales on the top of the prisms (Fig. 2m), with the prismatic structure being somewhat masked (Fig. 2n).
The scanning electron micrographs (Fig. 3) illustrate dentin micro-morphologies in the lateral walls of the cavities in all experimental groups at higher magnifications (×1000 and ×6500). In cavities conventionally prepared with burs, these surfaces appeared to be covered with a thick and irregular smear layer (Fig. 3a–d) and no open dentin tubule was observed. However, dentine surfaces of cavities prepared with laser were mostly characterized by opened dentinal tubules in an irregularly ablated inter-tubular dentin (Fig. 3e–n). These surfaces were free of smear layer and the dentinal tubules exhibited a protuberant peritubular collar in a scaled inter-tubular dentin. Such characteristics were more evident in those cavities prepared with pulse energies of 560 mJ (G5—Fig. 3i, j) and 630 mJ (G6—Fig. 3k, l). In these groups, the dentin exhibited a honeycomb appearance. In groups where lower pulse energies were used (G3 and G4), a smaller number of open tubules was observed (Fig. 3e–h, respectively). On the other hand, when higher pulse energy was used (700 mJ, G7), only a few open dentin tubules were found and no peritubular collar was observed either (Fig. 3m, n).
Scanning electron micrographs of the lateral walls of the dentin in all groups: conventional cavity preparation with diamond bur (G1) (a, b) and carbide bur (G2) (c, d); laser cavity preparation with 420-mJ pulse energy (G3) (e, f); 490-mJ pulse energy (G4) (g, h); 560-mJ pulse energy (G5) (i, j); 630-mJ pulse energy (G6) (k, l); and 700-mJ pulse energy (G7) (m, n). Original magnification at ×1000 and inserts at ×6500
Discussion
Er:YAG laser is widely used nowadays by dentists worldwide to perform various soft tissue and hard tissue procedures. The delivery systems in which dental equipment are almost the same in every case are represented by a contra-angled handpiece fitted with sapphire tips. Manual movement of the handpiece is necessary to perform the procedures. Even though numerous research works have been performed with this equipment, in such cases, one can never state with certainty that every area of the tissue is receiving the same amount of energy. In addition, the human perception is the only way to judge which is the best procedure. So, it is highly logical to perform a pre-programmed uniform lasing action on a certain measured area by using a scanner handpiece in research works in order to obtain an even more precise result. At the time of this study, such a scanner handpiece was not available, and for this reason, the Fotona XS Dynamis scanner device (which is essentially a dermatological Er:YAG scanner equipment) was used.
In the present study, it was expected that the SEM examination of enamel and dentin after cavity preparation showed important topographical differences between the substrates obtained with laser and those obtained with burs (conventional cavity preparation). The cavity surfaces prepared with conventional method using air-motor handpiece with diamond and carbide burs produced relatively smooth and flat surfaces. These surfaces were completely covered by debris and smear layer, exhibiting the typical morphological pattern observed elsewhere [4, 12, 13]. The dentin-enamel junction as well as the enamel prisms and dentin tubules were not evident due to the abundant smear layer throughout the enamel and dentin surfaces. This surface topography resulting from the conventional cavity preparation is not suitable for adhesion; thus, the search for other cavity preparation methods is justified, such as the use of Er:YAG laser.
In fact, the morphology of the enamel and dentin surfaces obtained after cavity preparation with Er:YAG laser revealed a completely different topography compared to that of conventional cavity preparation. The Er:YAG laser irradiation resulted in a rough topography with a micro-retentive morphological pattern [14], which is characteristic of the thermomechanical ablation process. Additionally, differences in the topography were also observed as a result of different pulse energy applied during laser cavity preparation. Basic structural elements of enamel and dentin were clearly distinguishable.
The topography of both substrates (e.g. dentin and enamel) obtained with laser irradiation was mostly favourable for adhesion. The Er:YAG laser is able to determine these topographies, as widely described in the literature [12, 13, 15, 16]. Lin et al. [17] also proposed that the shear bond strength value is much higher in non-etched laser cavities than in non-etched bur ones. Additionally, other authors have shown higher values of bond strength when cavities were prepared with laser [18, 19] compared to those prepared with conventional instruments.
On the other hand, some studies show a decrease of bond strength values in laser-treated surfaces compared to the surface prepared by conventional drill [20–25]. One of the reasons for the adverse effect on adhesion might be that erbium lasers induce a thermal damage to the collagen fibre network [16], which hampers the diffusion of resin into the surface [20] and forms a hybrid layer [26], thus resulting in lower shear bond strength [20].
The dentin has higher absorption coefficient and lower ablation threshold for Er:YAG laser compared to the enamel tissue, as demonstrated by Stock et al. [27], who used the same parameters, irradiation spot size and optical fibre. This occurs because the dentin has greater organic content [1, 27, 28] and water (12% weight and 25% volume) [29–31], resulting in faster and higher mass removal.
Based on the water content of enamel (3% weight and 12% vol), studies showed that this tissue water is rapidly superheated by the laser irradiation until the water is absorbed [29–31]. The optical to thermal energy exchange will create a rapid phase conversion of water to steam and increase the internal pressure due to a sudden volume expansion, resulting in ejection of the inorganic structure [1, 32, 33]. These specific absorption and rapid expansion properties of water afford the principles of the water-mediated photothermal-mechanical ablation on the enamel surface by the Er:YAG laser [34].
In the present study, the first signs of vitrification were found at an energy output of 630 mJ, with more sites being seen at a higher pulse energy output of 700 mJ. A previous study detected the first signs of enamel vitrification at an energy output of 250 mJ, and more abundant and widespread areas of vitrification at an energy of 400 mJ [35]. Some authors reported that the occurrence of vitrification can be associated to the effect of the water-cooling system [35]. By correlating our study to the previous one, we can infer that the late vitrification found in our work could be explained by the use of an efficient external cooling system during irradiation of the specimens. Water appears to act as a surface coolant by reducing or eliminating the local thermal stress induced by the laser irradiation [36], especially when higher energy settings are used.
Vitrification is a result of the re-crystallization of the dentinal apatite and formation of an additional phase of calcium phosphate. The degree of vitrification is associated with the amount of laser energy applied to the tissue, which inevitably generates heat. The heat generated by laser might induce an intense and rapid temperature increase in the dentin below the irradiated site. Due to the low coefficient of thermal diffusion in the dentinal tissue, the heat is not dispersed and instead concentrated in a limited area below the lased surface. Therefore, rapid and substantial heat output from the laser produces an intense thermal expansion of the dentin below the lased surface. This is followed by a rapid contraction during the subsequent cooling phase, resulting in high internal tension. In addition, light energy can be transmitted to the underlying dentinal layers, causing a collision wave. This mechanical action as well as the internal tensions generated may contribute to the formation of micro-fractures and micro-cracks in the hard dental tissue [37], resulting in negative influence on the bond strength [38].
The morphological analysis of the dental hard tissue irradiated with Er:YAG scanner at 560 mJ showed enamel with irregular surface, better visualization of the prismatic structure with keyhole appearance and no smear layer. The irradiated dentin surface also revealed absence of smear layer, exposing open dentinal tubules with a micro-retentive pattern and honeycomb appearance. These morphological characteristics are in accordance with previous studies [39–41].
Final considerations
Thus, it is conceivable to suggest that irradiating the hard dental tissue with Er:YAG scanner-assisted cavity preparation at 560 mJ and suitable refrigeration produces a surface topography which is more appropriate for further resin restoration and with no alterations compared to the conventional methods. Based on the present study, it is possible to infer that the development of a scanner handpiece for dental laser equipment would make dental hard tissue management (e.g. cavity preparation and surface conditioning) much easier, faster and efficient for the clinician. Even though the microscopic morphology produced by scanner seemed to be much more favourable in terms of future tooth restoration, further studies are necessary to prove this precisely. Moreover, bond strength studies should be conducted to validate the results obtained in the present study.
References
Hibst R, Keller U (1989) Experimental studies of the application of the Er:YAG laser on dental hard substances: I. Measurement of the ablation rate. Lasers Surg Med 9:338–344
Keller U, Hibst R (1989) Experimental studies of the application of the Er:YAG laser on dental hard substances: II. Light microscopic and SEM investigations. Lasers Surg Med 9:345–351
Ando Y, Aoki A, Watanabe H, Ishikawa I (1996) Bactericidal effect of erbium YAG laser on periodontopathic bacteria. Lasers Surg Med 19:190–200
Sharon-Buller A, Block C, Savion I, Mordehai S (2003) Reduced bacteria levels in cavities prepared by Er:YAG laser. J Oral Laser Applic 3:153–155
Matsumoto K, Nakamura Y, Mazeki K, Kimura Y (1996) Clinical dental application of Er:YAG laser for class V cavity preparation. J Clin Laser Med Surg 14:123–127
Sonntag KD, Kutzman B, Burkes J, Hoke J, Moshonov J (1996) Pulpal response to cavity preparation with the Er:YAG and Mark 111 free electron lasers. Oral Surg Oral Med Oral Pathol Oral Radio1 Endod 81:695–702
Cozean C, Arcoria CJ, Pelagalli J, Powell GL (1997) Dentistry for the 21st century? Erbium:YAG laser for teeth. J Am Dent Assoc 128:1080–1087
Bertrand MF, Brulat N, Lazzarini V, Marcato G, Namur S, Roca JP (2008) Er:YAG laser cavity preparation and semi-direct composite resin restoration: a microleakage study. Photom Laser Surg 26:473–477. doi:10.1089/pho.2007.2182
Frentzen M, Koort HJ (1992) Histological investigation of mid infrared laser ablation of dental hard tissue. In: International Congress on Laser in Dentistry, 3. Salt Lake City, Aug., 6–8,. Proceedings. Bologna: ISLD; 243–244.
Mello AM, Mayer MP, Mello FA, Matos AB, Marques MM (2006) Effects of Er:YAG laser on the sealing of glass ionomer cement restorations of bacterial artificial root caries. Photomed Laser Sur 24:467–473
Matsumoto K, Wang X, Zhang C, Kinoshita J (2007) Effect of a novel Er:YAG laser in caries removal and cavity preparation: a clinical observation. Photomed Laser Surg 25:8–13
Manhães L, Oliveira DC, Marques MM, Matos AB (2005) Influence of Er:YAG laser surface treatment and primer application methods on microtensile bond strength self-etching systems. Photomed Laser Surg 23:304–312
Tachibana A, Marques MM, Soler JM, Matos AB (2008) Erbium, chromium:yttrium scandium gallium garnet laser for caries removal: influence on bonding of a self-etching adhesive system. Lasers Med Sci 23:435–441
Trevelin LT, Marques MM, Aranha AC, Aranha-Chavez VE, Matos AB (2015) Effect of super short pulse Er:YAG laser on human dentin—scanning electron microscopy analysis. Microsc. Res Tech 78:472–478. doi:10.1002/jemt.22496
Aoki A, Ishikawa I, Yamada T, Otsuki M, Watanabe H, Tagami J, Ando Y, Yamamoto H (1998) Comparison between Er:YAG laser and conventional technique for root caries treatment in vitro. J Dent Res 77:1404–1414
Aranha ACC, Eduardo CP, Gutknecht N, Marques MM, Ramalho KM, Apel C (2007) Analysis of the interfacial micromorphology of adhesive systems in cavities prepared with Er,Cr:YSGG, Er:YAG laser and bur. Microsc Res Tech 70:745–751
Lin S, Caputo AA, Eversole LR, Rizoiu I (1999) Topographical characteristics and shear bond strength of tooth surfaces cut with a laser-powered hydrokinetic system. J Prosthet Dent 82:451–455
Bertrand MF, Semez G, Leforestier E, Muller-Bolla M, Nammour S, Rocca JP (2006) Er:YAG laser cavity preparation and composite resin bonding with a single-component adhesive system: relationship between shear bond strength and microleakage. Lasers Surg Med 38:615–623
Visuri SR, Gilbert JL, Wright DD, Wigdor HA, Walsh JT Jr (1996) Shear strength of composite bonded to Er:YAG laser prepared dentin. J Dent Res 75:599–605
Ceballos L, Toledano M, Osorio R, Tay FR, Marshall GW (2002) Bonding to Er-YAG-laser-treated dentin. J Dent Res 81:119–122
Barceleiro Mde O, de Mello JB, de Mello GS, Dias KR, de Miranda MS, Sampaio Filho HR (2005) Hybrid layer thickness and morphology: the influence of cavity preparation with Er:YAG laser. Oper Dent 30:304–310
Botta SB, Vieira SN, Cordon R, Marques MM, Matos AB (2009) Can the method of primer application influence adhesion to Er:YAG-laser irradiated dentin? J Contemp Dent Pract 10:49–57
Ishizaka Y, Eguro T, Maeda T, Tanaka H (2002) Effects of Er:YAG laser irradiation on human dentin: polarizing microscopic, light microscopic and microradiographic observations, and FT-IR analysis. Lasers Surg Med 31:171–176
Sassi JF, Chimello DT, Borsatto MC, Corona SAM, Pecora JD, Palma-Dibb RG (2004) Comparative study of the dentin/adhesive systems interface after treatment with Er:YAG laser and acid etching using scanning electron microscope. Lasers Surg Med 34:385–390
Oliveira DC, Manhães LA, Marques MM, Matos AB (2005) Microtensile bond strength analysis of different adhesive systems and dentin prepared with high-speed and Er:YAG laser: a comparative study. Photomed Laser Surg 23:219–224
Bertrand MF, Hessleyer D, Bolla MM, Nammour S, Rocca JP (2004) Scanning electron microscopic evaluation of resin–dentin interface after Er:YAG laser preparation. Lasers Surg Med 35:51–57
Stock K, Hibst R, Keller U (1997) Comparison of Er:YAG and Er:YSGG laser ablation on dental hard tissue. In Altshuler GB, Birngruber R, Dal Fante M, Hibst R, Hoenigsmann H, Krasner N, Laffite F, (eds.) Medical applications of lasers in dermatology, ophthalmology, dentistry and endoscopy (Proceedings SPIE. Vol.3192). SPIE. pp.88–95
Li ZZ, Code JE, Van De Merwe WP (1992) Er:YAG laser ablation of enamel and dentin of human teeth: determination of ablation rates at various fluences and pulse repetition rates. Lasers Surg Med 12:625–630
Arends J, Ruben JL, Inaba D (1997) Major topics in quantitative microradiography of enamel, and dentin: R parameter, mineral distribution visualization, and hyper-remineralization. Adv Dent Res 11:403–414
Duck FA 1990. Tissue composition. In Physical properties of tissue. Academic Press, San Diego. 322p.36
Dibdin GH (1993) The water in human dental enamel and its diffusional exchange measured by clearance of tritiated water from enamel slab of varying thickness. Caries Res 27:81–86
Ramos R, Chimello DT, Chinelatti MA, Nonaka T, Pécora JD, Palma Dibb RG (2002) Effect of Er:YAG laser on bond strength to dentin of a self-etching primer and two single-bottle adhesive systems. Lasers Surg Med 31:164–170
Sasaki KM, Aoki A, Ichinose S, Ishikawa I (2002) Ultrastructural analysis of bone tissue irradiated by Er:YAG laser. Laser Surg Med 31:79–78
Yung FY, Gutknecht N, Franzen R, Fischer H (2013) Shear strength of composite bonded to Er:YAG laser-prepared enamel: an in vitro comparative study. Lasers Med Sci 28:879–889. doi:10.1007/s10103-012-1169-1
Delmé KI, De Moor RJ (2007) Scanning electron microscopic evaluation of enamel and dentin surface after Er:YAG laser preparation and laser conditioning. Photomed Laser Surg 25:393–401
Atrill DC, Farrar SR, King TA, Dickinson MR, Davies R, Blinkhorn AS (2000) Er:YAG (λ=2,94μm) laser etching of dental enamel as an alternative to acid etching. Lasers Med Sci 15:154–161
Giachetti L, Russo DS, Scarpelli F, Vitale M (2004) SEM analysis of dentin treated with the Er:YAG laser: a pilot study of the consequences resulting from laser use on adhesion mechanism. J Clin Laser Med Surg 22:35–41
Staninec M, Gardner AK, Le CQ, Sarma AV, Fried D (2006) Adhesion of composite to enamel and dentin surfaces irradiated by IR laser pulses of 0.5–35 micros duration. J Biomed Mater Res B Appl Biomater 79:193–201
Freitas PM, Navarro RS, Barros JA, Eduardo CP (2007) The use of Er:YAG laser for cavity preparation: an SEM evaluation. Microsc Res Tech 70:803–808
Hossain M, Nakamura Y, Yamada Y, Kimura Y, Nakamura G, Matsumoto K (1999) Ablation depths and morphological changes in human enamel and dentin after Er:YAG laser irradiation with or without water mist. J Clin Laser Med Surg 17:105–109
Kayano T, Ochiai S, Kiyono K, Yamamoto H, Nakajima S, Mochizuki T (2009) Effect of Er:YAG laser irradiation on human extracted teeth. J Clin Laser Med Surg 9:147–150. doi:10.1089/clm.1991.9.147
Acknowledgements
The authors would like to thank the Aachen University and the University of Sao Paulo for their support. Prof. Marques is supported by CNPq.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Chowdhury, S.R., Marques, M.M., Franzen, R. et al. Comparative ultrastructural analysis of Er:YAG laser scanner and conventional method for tooth cavity preparation. Laser Dent Sci 1, 23–31 (2017). https://doi.org/10.1007/s41547-017-0003-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41547-017-0003-2