Abstract
As part of a marine ecology study aimed to discover the diversity of fish parasites, we described a new species representing Magnanchor n. gen. and provided new and supplementary morphometric information for Mexicana bychowskyi, both monogenoidean species collected from the gill lamellae of the humpback grunt Microlepidotus brevipinnis (Haemulidae) from the Pacific coast of Mexico. The new genus is proposed for its unique species, M. raris n. sp. which is characterized in having overlapping gonads (testis posterodorsal to the germarium), a copulatory complex consisting of counterclockwise coils of the copulatory organ that is unarticulated to the accessory piece, a haptor armed with two pairs of anchors (each with broad base and well differentiated roots), dorsal and ventral bars, a dextral vaginal aperture, and 6 pairs of hooks with uniform shank and upright acute thumb. Data on morphological and biometric variability of the six species of Mexicana (including illustrations of the haptoral structures of the present specimens of M. bychowskyi) previously described and/or reported parasitizing different haemulid hosts from the Pacific and Atlantic oceans in the Tropics are also provided. Due to the morphological similarities among the amphiamerican species of Mexicana, they could represent sister-species pairs in the Tropics. Magnanchor raris n. sp. and M. bychowskyi (type species of the genus) are the first dactylogyrids described and/or reported on M. brevipinnis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Haemulidae (Perciformes) with about 137 species belonging 21 genera are known commonly as grunts and represent a conspicuous group, mainly, of marine shorefishes in the tropical West Atlantic, Eastern Pacific, and Indian Oceans (Tavera and Wainwright 2019; Hernández-Gómez et al. 2021). In Mexico, haemulids are well represented along the Pacific and Gulf of Mexico slope, as well as on coral reefs as those from the Caribbean Sea (Torres-Hernández et al. 2016). Currently, these haemulids have been reported to be parasitized by gill lamellae ectoparasitic monogenoids, i.e., species of Haliotrematoides Kritsky, Yang and Sun 2009, Pseudotetrancistrum Caballero and Bravo-Hollis 1961, Mexicana, Caballero and Bravo-Hollis 1961, Euryhaliotrema (Kritsky and Boeger 2002) Kritsky 2012 (Dactylogyridae), Choricotyle Van Beneden and Hesse, 1863, Pseudoeurysorchis Caballero and Bravo-Hollis 1962 (Diclidophoridae), Paracalceostoma Parona and Perugia, 1890 (Calceostomatidae), Cynoscionicola Price, 1962 (Microcotylidae), Magniexcipula Bravo-Hollis, 1981, and Encotyllabe Diesing, 1850 (Capsalidae) (Lamothe-Argumedo et al. 1998; Mendoza-Garfías and Pérez-Ponce de León 1998; Mendoza-Franco et al. 2018; Montoya-Mendoza et al. 2022). As part of a research project on fish parasite biodiversity as an important component within marine ecosystems (Timi and Poulin 2020) from the Pacific coast of the Guerrero State in Mexico, monogenoidean specimens of Mexicana bychowskyi Caballero and Bravo-Hollis 1959 and of an undescribed species of the Dactylogyridae, Magnanchor raris n. gen, n. sp. were recovered from the humpback grunt Microlepidotus brevipinnis (Steindachner 1869) (Haemulidae). In the present paper, the new species is described and illustrated, and new illustrations and supplemental observations for M. bychowskyi as well as comparable data on biometrical variability of other previously described species of the genus from haemulid hosts are provided. Finally, the Amphiamerican occurrence of known species of Mexicana from the Atlantic and Pacific American coasts of the tropic is briefly discussed.
Materials and methods
A total of 38 fish specimens of M. brevipinnis were caught in the coastal municipality of Acapulco in the Guerrero state (16º51’N; 99º52’W) (Mexican Pacific) from September 2017 to June 2021 by local fishermen and/or purchased at local markets. Gills were excised from fish, immediately placed in Petri dishes with tap water, and then examined for monogenoids by using a dissecting microscope. Then, monogenoids were detached from the gills using fine needles under stereo-microscope and kept within vials containing 4% formaldehyde for fixation of attached helminths. Subsequently, the vials were shipped to the Laboratory of Parasitology of the Instituto EPOMEX (Autonomous University of Campeche, Mexico) for further examination. In the laboratory, parasites were picked from the sediments or gill lamellae using a small probe and dissecting under a stereomicroscope Leica EZ4. Some specimens were mounted unstained in Gray and Wess medium or in a mixture of lactic-acid and glycerin-ammonium picrate, and then remounted in Canada balsam (Mendoza Franco et al. 2013) to obtain measurements and line drawings of haptoral structures and the copulatory complex; other specimens were stained with Gomori’s trichrome and mounted whole in Canada balsam for observing and measuring internal soft organs. Illustrations were made with the aid of a drawing tube, using a Leica microscope DM 2500 with Nomarski interference contrast. Measurements, all in micrometers (µm), represent straight-line distances between extreme points of the structures measured and are expressed as the mean followed by the range and number (n) in parentheses. Direction of the coil (when present, i.e., counterclockwise orientation) of the copulatory organ followed Kritsky et al. (1985). The numbering of hook pairs are those of Mizelle (1936). Reference specimens were deposited in the National Helminthological Collection of Mexico (CNHE), Institute of Biology, National Autonomous University of Mexico, Mexico.
Results
Class Monogenoidea Bychowsky, 1937.
Subclass Polyonchoinea Bychowsky, 1937.
Order Dactylogyridea Bychowsky, 1937.
Dactylogyridae Bychowsky, 1933.
Magnanchor n. gen.
Type-species: Magnanchor raris n. sp.
Etymology: The generic name is an adjective from Latin (magna = prominent, conspicuous + anchor) and refers to the shape of the ventral anchor.
Diagnosis: Body comprising body proper (cephalic region, trunk and peduncle) and haptor. Tegument smooth. Two terminal cephalic lobes, bilateral lobes poorly developed or absent; 3 pairs of bilateral head organs. Eyespots present. Mouth subterminal, midventral, prepharyngeal; pharynx made up of muscular, glandular bulb; esophagus, intestinal ceca 2, confluent posterior to gonads, lacking diverticula. Common genital pore midventral, immediately posterior to intestinal bifurcation. Gonads intercaecal, overlapping; testis dorsal to germarium. Vas deferens, seminal vesicle not observed; prostatic reservoir present. Copulatory complex comprising unarticulated male copulatory organ (MCO), accessory piece. MCO tubular, coiled, with funnel-shaped base; coil with counterclockwise rings. Seminal receptacle not observed; vaginal aperture dextroventral. Vitellaria in trunk, peduncle, absent in regions of other reproductive organs. Haptor bilobed, armed with dorsal, ventral anchor-bar complexes, six pairs of similar hooks, each with upright acute thumb, slender shank comprising of single subunit. Type-and only species: Magnanchor raris n. gen., n. sp. from the gills of Microlepidotus brevipinnis (Haemulidae).
Magnanchor raris n. sp. (Fig. 1)
Type-host: Humpback grunt Microlepidotus brevipinnis (Steindachner 1869) (Haemulidae).
Type locality: Acapulco Bay, Pacific coast of Mexico (16º51’ N; 99º52’ W).
Site in host: Gill lamellae.
Specimens deposited: Holotype and 1 paratype in CNHE (12851 and 12852, respectively).
Etymology: The specific name (an adjective) is from Latin (raris = infrequent) and refers to the fact that this species is relatively rare with only 2 specimens found on 38 examined individuals of M. brevipinnis.
Description: Body 425–437 long, fusiform; greatest width 105 (n = 2) usually near mid-length of trunk. Tegument smooth. Cephalic region broad; cephalic lobes poorly developed. Four eyespots; members of respective pairs equidistant; posterior eyespots slightly larger than those of anterior pair; accessory chromatic granules ovate, variable in size, scattered in cephalic, anterior trunk regions. Pharynx subspherical, 24 (n = 2) in diameter. Peduncle broad; haptor 90–97 wide, not distinctly set off from body, with poorly differentiated bilateral lobes containing hook pairs 2–4, 6 and 7. Ventral anchor 62 (60–63 n = 4) long, prominent, with large base and subequal and well-differentiated roots, elongated straight shaft, point with lightly subterminal bent (shaft and point extending posteriorly from haptor); 28 (23–31; n = 4) wide. Dorsal anchor 40 (39–42; n = 4) long, with well-defined tapered superficial root, subrectangular deep root, curved shaft, elongate point; base 10 (9–12; n = 3) wide. Ventral bar 57–70 long, robust, rod-shaped with 2 posteriorly directed thumb-like processes at each end; dorsal bar 33–36 long, slender with posteriorly directed ends. Hook 13 (n = 6) long, with uniform shank, upright acute thumb and recurved point; FH loop about 60% shank length. Sucker-like structure 16 (13–19; n = 2) wide, anterior to genital pore. Copulatory complex, 25–30 long; MCO with funnel shaped base from which arise a shaft in counterclockwise ring, having about 2 rings. Accessory piece tortuous, covering distal portion of MCO. Margin of testis undefined; vas deferens, seminal vesicle not observed; prostatic reservoir subspherical. Germarium 65 long, 25 wide, ovate; oviduct, oötype uterus not observed. Mehlis’ gland-cells well developed, in body mid-line. Vaginal pore dextral, submarginal, with small distal vestibule; seminal receptacle not observed. Vitellarium in form of dense follicles in lateral fields; intestinal caeca obscured by dense vitellarium. Egg not observed.
Remarks:
Present specimens exhibit diagnostic features (i.e., postovarian testis, vagina dextral, and haptor armed with a dorsal, ventral anchor-bar complexes), some of which are also present in other dactylogyrids. However, the suite of features does not fit with the diagnosis of currently known dactylogyrid genera with species infecting Mexican or tropical marine fishes (see the Introduction section). Therefore, although only two specimens were found, they were fixed in good conditions allowing us their description and the proposal of a new genus to accommodate them as Magnanchor raris n. gen., n. sp. The new species exhibits a similar haptor morphology (i.e., truncated haptor or not distinctly set off from body) with that of species of Pseudempleurosoma (Yamaguti, 1965) Santos, Mourão and Cardenas, 2001 on rectum, gills, esophagus and pharynx of marine fishes of the Hoplichthyidae, Chlorophthalmidae, Carangidae, Holocentridae, Sciaenidae, and Sillaginidae (Mendoza-Franco and Vidal-Martínez 2011). It differs from Pseudempleurosoma spp. by having a coiled MCO (slender and/or twisted in Pseudempleurosoma spp.) and by lacking a single quadrangular dorsal bar and paired irregular ventral bars (present in Pseudempleurosoma spp.). In other features, M. raris n. gen., n. sp. also differs from other known dactylogyrids on haemulids in the absence of hook pairs 1, 2, and 3 on the ventral surface of peduncle (present in Haliotrematoides spp.), gonads in tandem with horse-shoe shaped germarium (present in Pseudotetrancistrum), a sinuous MCO tube (present in Mexicana – see this latter in the remarks sections below) and a bulbous base of the MCO (present in Euryhaliotrema spp.). With the present recognition of a distinct species of the Dactylogyridae on M. brevipinnis from the Mexican Pacific, this latter host has now only two dactylogyrid species: the present one and M. bychowskyi (present study). Only it is noteworthy that in present specimens the hook pair 1 were not observed (possibly absent) (see haptor in Fig. 1).
Mexicana bychowskyi Caballero and Bravo-Hollis 1959 (Fig. 2)
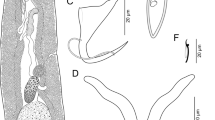
Mexicana bychowskyi Caballero and Bravo-Hollis 1959 on Microlepidotus brevipinnis. A, Copulatory complex; B, ventral anchor; C, dorsal anchor; D, hook pair 5; E, hook pair 4; F, ventral bar; G, dorsal bar. Abbreviation: AC, accessory piece
See measurements in Table 1.
Host: Humpback grunt Microlepidotus brevipinnis (Steindachner, 1869) (Haemulidae).
Locality: Acapulco Bay, Pacific coast of Mexico (16º51’N; 99º52’W).
Site in host: Gill lamellae.
Specimens deposited: 32 reference specimens in CNHE (12853).
Remarks
The specimens found on humpback grunt M. brevipinnis from the Guerrero state correspond in their morphology (bilobed testis, sinuous MCO tube with accessory piece on its midportion, MCO base with bilateral hornlike terminations directed posteriorly, vagina dextral in inverted cone shape, and butterfly-shaped ventral bar [see Fig. 2]) to M. bychowskyi as originally described by Caballero and Bravo-Hollis (1959) (see also the emended generic diagnosis provided by Caballero and Bravo-Hollis 1961) from the Pacific coast of Mexico. All measurements of present specimens ranged within those of type specimens of this monogenoidean species, particularly of the dorsal bar length (see Table 1). There are currently 6 described species of Mexicana in the Tropics (Pacific coast of Mexico and Peru, and Atlantic coast of Brazil) (M. bychowskyi, M. iannaconi Chero, Cruces, Sáez and Alvariño, 2014, M. littoralis, M. anisotremum Cezar, Paschoal and Luque, 2012, M. atlantica Luque et al. 1992, and M. rubra Camargo et al. 2017), parasitizing haemulid species of Microlepidotus, Haemulon, Anisotremus, and Orthopristis (see Table 1). Only, M. bychowskyi has originally been described on an unidentified sciaenid host, since then, it has not been formally reported (i.e., based on a published taxonomic work) on a sciaenid or another haemulid host until present study. Even though M. brevipinnis is distributed from the Gulf of California to Panama (Eastern Central Pacific) (Gomez-Vanega et al. 2021), it only has previously been reported to host species of Choricotyle leonilavazquezae Lamothe-Argumedo et al. 1998 and Pseudoeurysorchis travassosi Caballero and Bravo-Hollis 1962 (both Diclidophoridae) from the Pacific coast of Mexico (Caballero and Bravo-Hollis 1962; Lamothe-Argumedo et al. 1998; Paschoal et al. 2015). The taxonomic identification of species of Mexicana has mainly been based on morphology of their anchors/bar complexes rather than using metrical data which appear to be overlapped among species (see Table 1 in Chero et al. 2014, Table 2 in Camargo et al. 2017, and Table 1 in the present study). In the present study, morphology of the haptoral structures of M. bychowskyi are illustrated in order to clarify them from its original description, i.e., hook pair 5 with expanded shank (see Fig. 2D) (not described and illustrated in the original description) and butterfly-shaped ventral bar (see Fig. 2F) (only described and poorly depicted in the original description, but redrawn as that from photograph of a voucher of this species by Camargo et al. 2017). In fact, measurements of anchors width are not provided in the original description because these structures in the types were in an undefined position (i.e., in longitudinal-lateral view) when mounted onto slides which also precluded well defined line drawings of the anchor/bars (see comments/figures in Caballero and Bravo-Hollis 1959; Table 1 and Fig. 2B,C,F and G). Additionally, the accessory piece, originally described as a sac with very transparent wall covering MCO is herein illustrated (see AC in Fig. 2).
Discussion
The present study on M. brevipinnis constitute the third record (the first and second being that of C. leonilavazquezae and P. travassosi—[Diclidophoridae], respectively) of monogenoidean species on this latter host from the Pacific coast of Mexico. Mexicana bychowskyi on M. brevipinnis, the type species of the genus, had not been recorded since its original description on an unidentified sciaenid host off the Pacific coast of Mexico (see Caballero and Bravo-Hollis 1959). Whereas the host range may not be completely known for all described and/or reported species of Mexicana they appear to be host-specific on haemulid fishes. Mendoza-Garfias et al. (2017) recorded M. bychowskyi on M. brevipinnis based on unpublished bachelor thesis (see Aranda-Cruz (2006) cited by latter authors). However, revising the content of that thesis available from the internet it does not described and illustrated details of the haptoral structures (i.e., hooks, anchors/bars) of M. bychowskyi (sic) as well as any reference specimen deposited in a museum for species confirmation. Thus, we consider the Mendoza-Garfias et al. (2017) an invalid record and the present specimens represent the first valid account of M. bychowskyi on M. brevipinnis. The morphology of the haptoral sclerites, copulatory complex and internal structures (these latter not herein illustrated) of the newly collected specimens of M. bychowskyi from M. brevipinnis revealed that it is consistent with that of the original description of this species (see Figs. 3–7 in Caballero and Bravo-Hollis (1959)). However, while these latter structures are satisfactory for specific identification, others are not clearly identified in the original description, i.e., the morphology of anchors and bars as characters also previously used to identify their congeneric species- see Table 2 in Camargo et al. (2017). Therefore, we provided new morphometrics and illustration of the anchor/bar complex of M. bychowskyi to clarify the morphology of this species by including also that of the hooks and accessory piece (see that membranous, shaped like a delicate vane, encircling MCO on its midportion in Fig. 2). The six known species of Mexicana on haemulids from the Pacific and Atlantic American oceans in the tropics are in general, nearly identical morphologically based on characteristics of its haptoral and copulatory sclerites (see Table 1 in Chero et al. (2014)). Based on comparative morphology of the sinuous MCO tube and its base, and all other features of the reproductive and digestive systems, for example, M. bychowskyi from the Pacific more closely resemble M. atlantica on H. steindachneri from Brazil (Atlantic) (see Luque et al. (1992)) than they do other congeneric species. That resemblance of these monogenoidean species could represent cryptic sister species or amphiamerican “geminate” species pairs occurring on their respective hosts along the Pacific and Atlantic American oceans. Although exist continuing documented evidence of geminate species pairs among monogenoids as well as within other groups of organisms (i.e., fish parasitic crustaceans) from which their isolation/speciation has been associated with formation of the Panamanian Isthmus (3 mya), there are other tropical monogenoids (i.e., species of Euryhaliotrema, Diplectanocotyla and Neohaliotrema) showing to be morphologically related to their pairs parasitizing lutjanid, megalopid and pomacentrid fishes from the Indo-West Pacific Ocean associated with the tectonic of Tethys Sea (12–18 mya) (see Kritsky (2012); May-Tec et al. (2022); Mendoza-Franco et al. (2022)). However, not record of species of Mexicana to occur on haemulids from the Indo Pacific so far. Finally, although only two dactylogyrid specimens representing a new species of Magnanchor n. gen. were collected from M brevipinnis off Mexican Pacific, the features of the haptoral structures and copulatory complex allowed us justify proposal of that new genus to accommodate M. raris n. sp.
Data Availability
Not applicable.
References
Caballero CE, Bravo-Hollis M (1959) Trematodos de peces de aguas mexicanas del Pacífico XVII. Dos nuevos géneros de Monogenoidea Bychowsky, 1937. An Inst Biol Univ Nac Autóm México 30:167–181
Caballero CE, Bravo-Hollis M (1961) Trematodos de peces de aguas mexicanas del Pacífico XX. Tres especies de Monogenoidea Bychowsky, 1937. An Inst Biol Univ Nac Autóm México 32:201–217
Caballero CE, Bravo-Hollis M (1962) Trematodos de peces de aguas mexicanas del Pacífico XXI. Sobre un nuevo género de la familia Diclidophoridae Fuhrmann, 1928. Rev Bras Biol 22:107–114
Camargo ACA, Luque JL, Santos CP (2017) Mexicana rubra sp. nov. and Encotyllabe cf. spari Yamaguti, 1934 (Monogenea) of Orthopristis ruber (Cuvier, 1830) from the Brazilian coast off Rio de Janeiro. Helminthol 54:336–347. https://doi.org/10.1515/helm-2017-0046
Chero J, Cruces C, Sáez G, Alvariño L (2014) Mexicana iannaconi n. sp. (Monogenea: Ancyrocephalidae) parasite of chere-chere grunt Haemulon steindachneri (Perciformes: Haemulidae) (Jordan & Gilbert, 1882) from the Peruvian coast. Neotrop Helminthol 8:429–438. https://www.researchgate.net/publication/270823883
Gomez-Vanega HD, Espino-Barr E, López-Uriarte E (2021) Ichthyofauna composition (Actinopterygii: Teleostei) caught by Jalisco’s small-scale fisheries in the Mexican Central Pacific coast. Lat Am J Aquat Res 49:788–803. https://doi.org/10.3856/vol49-issue5-fulltext-2712
Hernández-Gómez RE, Rodiles-Hernández R, Mendoza-Carranza M, Valenzuela-Córdova I, Perera-García MA (2021) Record of Rhonciscus crocro (Cuvier, 1830) (Haemulidae: Haemulinae) in the Usumacinta River, Mexico. FishTaxa 22:16–22
Kritsky DC (2012) Dactylogyrids (Monogenoidea: Polyonchoinea) parasitizing the gills of snappers (Perciformes: Lutjanidae): Revision of Euryhaliotrema with new and previously described species from the Red Sea, Persian Gulf, the eastern and Indo-west Pacific Ocean, and the Gulf of Mexico. Zoologia 3:227–276. https://doi.org/10.1590/S1984-46702012000300006
Kritsky DC, Boeger WA, Thatcher VE (1985) Neotropical Monogenea. 7. Parasites of the pirarucu Arapaima gigas (Cuvier), with descriptions of two new species and redescription of Dawestrema cycloancistrium Price and Nowlin, 1967 (Dactylogyridae: Ancyrocephalinae). Proc Biol Soc Wash 98:321–331
Lamothe-Argumedo R, Aranda-Cruz C, Pérez-Ponce de León G (1998) Choricotyle leonilvazquezae sp. n. (Monogenea: Diclidophoridae) parasitic on Microlepidotus brevipinnis (Osteichthyes: Haemulidae) from Chamela Bay, Jalisco. Mexico J Helminthol Soc Wash 65:24–30
Luque JL, Amato JFR, Takemoto RM (1992) A new species of Mexicana (Monogenea: Dactylogyridae) parasitic on Haemulon steindachneri (Jordan & Gilbert) (Osteichthyes: Haemulidae) from the Brazilian coast. Rev Bras Parasitol Vet 1(2):85–88
May-Tec AL, Baños-Ojeda C, Mendoza-Franco EF (2022) Parasitic crustaceans (Branchiura and Copepoda) parasitizing the gills of puffer fish species (Tetraodontidae) from the coast of Campeche, Gulf of Mexico. Zookeys 1089:73–92. https://doi.org/10.3897/zookeys.1089.79999
Mendoza Franco EF, Caspeta-Mandujano JM, Salgado-Maldonado G (2013) New species of Cacatuocotyle (Monogenoidea, Dactylogyridae) parasitizing the anus and the gill lamellae of Astyanax aeneus (Pisces, Ostariophysi: Characidae) from the Rio Lacantún basin in the Biosphere Reserve of Montes Azules, Chiapas. Mexico Parasitol Res 112(1):199–205. https://doi.org/10.1007/s00436-012-3126-0
Mendoza-Franco EF, Vidal-Martínez VM (2011) First records of known endoparasitic species of Pseudempleurosoma Yamaguti, 1965 (Monogenoidea: Dactylogyridae) from tetraodontid and rachycentrid fish off the Northern coast of the Yucatan Peninsula. Mexico J Parasitol 97:1020–1025. https://doi.org/10.1645/GE-2727.1
Mendoza-Franco EF, Simöes N, Vidal-Martínez VM, Aguirre-Macedo ML (2022) Rediscovering monogenoids (Platyhelminthes) parasitizing pomacentrid and chaetodontid fishes from Cayo Arcas reef, Gulf of Mexico. Diversity 985:2–24. https://doi.org/10.3390/d14110985
Mendoza-Franco EF, Rosado TMC, Duarte AAD, Rodríguez RER (2018) Morphological and molecular (28S rRNA) data of monogeneans (Platyhelminthes) infecting the gill lamellae of marine fishes in the Campeche Bank, southwest Gulf of Mexico. Zookeys 125–161. https://doi.org/10.3897/zookeys.783.26218.
Mendoza-Garfias B, García-Prieto L, Pérez-Ponce de León GP (2017) Checklist of the Monogenea (Platyhelminthes) parasitic in Mexican aquatic vertebrates. Zoosystema 39:501–598. https://doi.org/10.5252/z2017n4a5
Mendoza-Garfías B, Pérez-Ponce de León G (1998) Microcotílidos (Monogenea: Microcotylidae) parásitos de peces marinos de la Bahía de Chamela, Jalisco, México. An Inst Biol Univ Nac Autóm México, Zool 69:139–153
Mizelle JD (1936) New species of trematodes from the gills of Illinois fishes. Amer Midl Naturalist 17:785–806
Montoya-Mendoza J, Salgado-Maldonado G, Blanco-Segovia CR, Mendoza-Franco EF, Lango-Reynoso F (2022) Helminth Community of the Black Margate Anisotremus surinamensis (Teleostei: Haemulidae), from Coral Reefs off the Veracruz Coast, Mexico, Southern Gulf of Mexico. Diversity 14:368. https://doi.org/10.3390/d14050368
Paschoal F, Cezar AD, Luque JL (2015) Checklist of metazoan associated with grunts (Perciformes, Haemulidae) from the Neartic and Neotropical regions. Checklist 11:1501. https://doi.org/10.15560/11.1.1501
Tavera JJ, Wainwright PC (2019) Geography of speciation affects rate of trait divergence in haemulid fishes. Proc R Soc B 286:20182852. https://doi.org/10.1098/rspb.2018.2852
Timi JT, Poulin R (2020) Why ignoring parasites on fish ecology is a mistake. Int J Parasitol 50:755–761. https://doi.org/10.1016/j.ijpara.2020.04.007
Torres-Hernández E, Palacios-Morales G, Romero-Gallardo S, Salazar-Araujo P, García-Meraz A, Madrigal-Guridi X, Del Moral-Flores LF, Domínguez-Domínguez O (2016) Annotated checklist of the coastal ichthyofauna from Michoacán State, Mexico. ZooKeys 606:99–126. https://doi.org/10.3897/zookeys.606.9004
Acknowledgements
The authors are indebted to fishermen who caught the fish in the Acapulco Bay, as well as to the students of the Marine Ecology Faculty (UAGro) for their assistance during field and laboratory work. FVA was supported by a Master student fellowship (scholarship number 1079874) from the Consejo Nacional de Humanidades, Ciencias y Tecnologías (CONAHCYT), Mexico.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
E.F.M.-F., F.V.-A. and J.V-G conceptualized manuscript; E.F.M.-F., F.V.-A. and J.V-G. structured methodology; E.F.M.-F., F.V.-A. and J.V-G. validated manuscript; E.F.M.-F. and J.V-G. made investigation; F.V.-A. and J.V-G. provided resources; E.F.M.-F. wrote the main manuscript text and prepared figures; E.F.M.-F., F.V.-A. and J.V-G. reviewed and edited manuscript; E.F.M.-F. and J.V-G supervised manuscript. All authors reviewed manuscript.
Corresponding author
Ethics declarations
Ethical Approval
All applicable institutional, national and international guidelines for the care and use of animals were followed.
Competing Interests
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mendoza-Franco, E.F., Valente-Alarcon, F. & Violante-Gonzalez, J. Dactylogyrids (Monogenoidea) Parasitizing Microlepidotus brevipinnis (Haemulidae) from the Pacific Coast of Mexico: Magnanchor raris n. gen., n. sp. and Supplementary Taxonomic Information for Mexicana bychowskyi Caballero and Bravo-Hollis 1959. Thalassas 40, 1223–1229 (2024). https://doi.org/10.1007/s41208-024-00675-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41208-024-00675-5