Abstract
Introduction
IARC released on 14th December the updated Globocan 2020 with new estimates on the global cancer burden, indicating that it has risen to 19.3 million cases and 10 million cancer deaths in 2020. Ovarian cancer is the 8th most commonly occurring cancer in women and the 18th most common cancer overall. There were more than 313,000 new cases of ovarian cancer in 2020 as per the Globocan 2020. Despite advances in the diagnosis and treatment, there has been little change in the mortality rate of ovarian cancer as most of patients were diagnosed at advanced stage. Tumour markers are the biochemical substances which are detected in the presence of tumours.Usually they are either the products of tumour tissues or secreted from the normal cells which are in the close with tumour tissue (2). Serum CA125 assay has low sensitivity in the early stages and also can be increased in certain conditions such as menstruation or endometriosis. Many studies reported that HE4 is absent in normal ovarian surface epithelium but is expressed specifically in 100% of endometroid and serous epithelial ovarian cancers (2). Furthermore, this combined measure can correct the variations in HE4 and CA125 which are due to other pathology.
Aims and Objective
1. To evaluate HE4 and CA125 in ovarian tumour patients preoperatively. 2. To compare the utility of HE4 and CA 125 for diagnosing and distinguishing malignant ovarian tumour.
Materials and Methods
In this prospective study, 149 patients of ovarian tumour were evaluated for serum CA125 and HE4. Out of which, 18 patients were lost to follow-up. Their levels were determined by the electrochemiluminescence (ECLIA) technique. Positive cut-off of CA 125 was taken > 35 U/mL. The HE4 positive cut-off values for premenopausal and postmenopausal women were > 70 pmol/l and > 140 pmol/l, respectively. Sensitivity, specificity and predictive value were calculated for each tumour marker.
Result
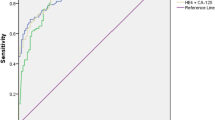
In patients with benign diseases, abnormal serum levels of HE4 and CA125 were found in 0.86% and 9.4% of patients, respectively. Specificity of HE4 and CA125 of diagnosing benign tumour were 99.03% and 81.55%, respectively. Tumour marker sensitivity in ovarian cancer was 100% for HE4 and 85.71% for CA 125. Among patients with ovarian epithelial cancer, HE4 (in contrast to CA 125) had significantly higher concentrations than in other malignancies (p < 0.001). In differentiating benign gynaecological diseases from malignant ovarian cancer, HE4 was found to be more positive predictive than CA125 (96.55% and 55.81%, respectively).
Conclusion
In this study, HE4 was found to be more sensitivity and specificity (p < 0.001) for ovarian malignancyas compare to CA 125. HE4 is better in diagnosing and distinguishing ovarian cancer from benign ovarian tumour as compare to CA125 preoperatively. It may be considered as novel ovarian tumour marker.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
It is estimated that ovarian cancer to be the third most common cancer in Indian women and eighth overall as per the Globocan 2020 Fact sheet, constituting 1.6% (313,959) of all cancer cases and 207,252 death (2.1%) [1].
It is also a leading cause of death from cancer, with 3.34% (24,015) of all cancer deaths in Indian women in the same year. five-year survival from ovarian cancer (when diagnosed in stage I) is 94%, but only 15% of cases are diagnosed at this stage. Most (62%) of cases are diagnosed in Stages III and IV and 5-year survival rate of this patient is only 28% [2].
The common symptoms of ovarian cancer are vague and sometime similar to those observed in other benign gynaecological conditions [3], so most patients are diagnosed at late stage. The diagnostic approach based on the use of CA 125 in association with ultrasonography has been suggested for the early diagnosis of ovarian cancer [4, 5]. However, this has several drawbacks including low sensitivity and specificity [4]. Abnormal CA 125 serum levels can be found in malignancies of different origin including epithelial (endometrial, endocervix and lung cancer) and non-epithelial malignancies (lymphomas) [6, 7]. Abnormal CA 125 serum levels may be also found in several benign diseases, mainly those with effusions, liver or renal failure and benign gynaecological conditions (ovarian cysts, myomas and endometriosis) [8]. Sensitivity of CA 125 in ovarian cancer is mainly related to tumour stage, with abnormal CA 125 serum levels in found inapproximately 50% of stage I patients and 80–90% in patients of stages III–IV [5,6,7,8,9].
Recently, another tumour marker for ovarian cancer has been proposed, the HE4 protein. It frequently overexpressed in ovarian cancers, especially in serous and endometrioid histology [10]. However, HE4 is not specific of ovarian cancer and some expression has also been found in other malignancies mainly pulmonary and endometrial adenocarcinomas, but it is not raised in any benign condition [11, 12]. Our study also reported that HE4 was more sensitive and specific than CA 125 in benign and malignant conditions [13]. Various studies suggest that HE4 has a similar sensitivity to CA 125, but an increased specificity in patients with gynaecological malignancies as compared with those with benign gynaecological disease [14, 15].
The aims of this study were:
-
1
To evaluate the HE4 and CA 125 serum levels in ovarian tumour patients preoperatively.
-
2
To compare the utility of HE4 and CA 125 for diagnosing and distinguishingmalignant ovarian tumour preoperatively.
Materials and Methods
Study Design
This prospective observational study was conducted from December 2020 to November 2022 at Indira Gandhi Institute of medical science, Patna. After proper history and clinical examination, CA125 and HE4 levels were measured prior to surgery in patients with radiologically proven pelvic mass. Serum HE4 levels vary in smokers and in hormonal contraceptive users, thus it always be included in the patient’s clinical history. The study was conducted in accordance with the principles of the Declaration of Helsinki and Good Clinical Practice and was approved by ethical committee. Consent from all the patients were taken.
Sample Collection
Patients with ovarian tumour were investigated preoperatively. Blood samples for HE4 and CA125 were obtained by venous puncture and then collected in 10-ml clothing activating tubes (BD Vacutainer Serum Tube, ref. 369,033; Becton–Dickinson, Erembodegem, Belgium). Serum tubes were centrifuged at 800 g for 10 min. Serum was collected, dispensed into multiple cryotubes and frozen at −70 °C. The time between blood sampling and freezing of the serum and presence of haemolysis was noted. The targeted time limit between sampling and freezing was 4 h. Tumour markers were determined by use of a chemiluminescent enzyme immunoassay on an Architect® (Abbott Laboratories); we have considered 35 U/mLas the upper limits of normality for CA 125. Positive cut-off values of HE4 for premenopausal and postmenopausal women were > 70 pmol/l and > 140 pmol/l respectively. This protocol was approved by the ethical committee of the institute.
Statistical Analysis
In this study, sensitivity, specificity and predictive value of HE4 and CA125 were calculated. The level of statistical significance was set at p < 0.05. Sensitivity was considered as the ratio between the numbers of patients with malignancy whose marker levels were elevated over the total number of patients with malignancy. Specificity was calculated as the ratio between the number of patients without malignancy and normal tumour marker values by the total number of patients without malignancy. Positive predictive values were calculated as the ratio among the cases with elevated tumour markers and malignancy and the sum of all the cases with elevated tumour markers. The negative predictive value was calculated by the ratio among the patients with negative results and without malignancy and the total number of patients with negative results.
Result
In this study, 131 patients of ovarian tumour were undergone surgery out of which 103 were found benign and 28 were found malignant histologically (Tables 1 and 2).
In this study most of the patients were of postmenopausal age group. Malignant tumours were found mainly in more older women (71.43%) with mean age 54.6 years. Patients having family history of breast and ovarian cancer were found to be significantly associated with ovarian cancer. As patients were mostly menopausal and postmenopausal, so we had taken HE4 > 140 pmol/L as significantly elevated (Tables 3, 4 and 5).
In this study, we found that CA125 was not significantly raised in early stage (stageI) as compare to HE4. CA125 was raised in 57.14% of patient with stage I ovarian cancer, whereas HE4 was raised in 100% patients of early malignant ovarian tumour with p value < 0.005. Elevated HE4 level corresponds to tumour burden. Higher level was found in late stage of ovarian tumour.
Discussion
Carbohydrate antigen 125 (CA 125), sometimes named as cancer antigen 125 or tumour antigen 125, is a mucin-type glycoprotein, produced by the MUC16 gene and associated with the cellular membrane [16]. This biomarker is most often used for ovarian lesions. Its upper limit is 35 U/mL in pre- and post-menopausal patients [17,18,19,20,21]. However, this measurement is not very sensitive in the early phases of ovarian cancer (only reported to be elevated in 23 to 50% of stage I cases) [16]. Our study also reported to be elevated in 57.14% in early stagetumour (Table 5). In addition, elevated serum CA125 levels may be observed in other physiological or pathological conditions (menstruation, pregnancy, endometriosis, inflammatory diseases of the peritoneum) [20,21,22,23]. In a meta-analysis by Dikmen et al. [22], the specificity of CA125 for detecting ovarian cancer was 78% (95% CI 76–80). Suggesting that it was probably not the ideal marker for diagnosing ovarian cancer. This study reported that sensitivity and specificity of CA125 were 85.71% and 81.55%, respectively (Table 3).
In practice, CA125 is often measured in cases of ovarian cysts, but according to its low specificity and the observed increased levels in different physiological situations, it is not considered as a very good differentiating biomarker for ovarian tumours. For this reason, new biomarkers have been evaluated in an attempt to improve early diagnosis of ovarian cancer [17].
Human epididymis protein 4 (HE4) is a new biomarker which has been currently evaluated for diagnosing ovarian malignant tumours [16]. It is a glycoprotein belonging to the family of whey acidic four-disulfide core proteins, accounting for its alternative name of WFDC2 and the larger protein family called “WAP” for whey acidic proteins. The main genes coding for the WAP proteins are mainly located on chromosome 20q12–13.1 [17]. This biomarker is weakly expressed in the epithelium tissues of respiratory and reproductive organs, but it is overexpressed in ovarian tumours, especially in endometrioid ovarian cancer [18,19,20]. In addition, it appears that HE4 is not as strongly expressed in clear cell ovarian carcinomas as in other epithelial ovarian cancers. HE4 level varies in smokers and in contraceptive combining oestrogen plus progestin users; simultaneous CA125 evaluation which is not affected by these variables should allow better interpretation of abnormal HE4 levels.
The cut-off level of 70 pmol/L is often used for pre-menopause patients and 140 pmol/L for menopause patients, but sometimes the threshold level of 140 pmol/L is employed.
The combined use of CA125 and HE4 is only sometimes studied [24]. Chen et al. [25] reported a specificity of 65.7% using the ECLIA immunological method with a cut-off value for HE4 of 140 pmol/L. In a different study, using another technique to assess serum HE4, the specificity of the association CA125 and HE4 was much better (80%) [24].
Our study suggested that HE4 had higher sensitivity and specificity more towards 100% and hence considered as more effective ovarian tumour marker. But along with CA125, it becomes more novel tumour marker for diagnosing malignant ovarian tumour and hence can be used for the proper management and follow-up of ovarian tumour.
Conclusion
To predict the risk of ovarian cancer in patients with suspected ovarian tumour, combination of CA125 and HE4 is found to be the best biological diagnostic tool. If the level of CA125 and HE4 is increased or only HE4 is raised, it is necessary to rule out a malignant lesion and therefore surgical treatment for histopathological confirmation becomes important. Our study report HE4 is very important in distinguishing malignant ovarian tumour from benign ovarian tumour. Though it is costlier than CA125, it is a novel serum marker in ovarian tumour.
References
Sung H, Ferlay J, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71:209–49. https://doi.org/10.3322/caac.21660. (American Cancer Society).
Henderson JT, Webber EM, et al. Screening for ovarian cancer updated evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2018;319(6):595–606. https://doi.org/10.1001/jama.2017.21421.
Heintz AP, Odicino F, Maisonneuve P, Quinn MA, Benedet JL, Creasman WT, Ngan HY, Pecorelli S, Beller U. Carcinoma of the fallopian tube. FIGO 26th annual report on the results of treatment in gynecological Cancer. Int J Gynaecol Obstet. 2006;95(Suppl 1):S145–60. https://doi.org/10.1016/S0020-7292(6)60033-7.
Enakpene CA, Omigbodun AO, Goecke TW, Odukogbe AT, Beckmann MW. Preoperative evaluation and triage of women with suspicious adnexal masses using risk of malignancy index. J Obstet Gynaecol Res. 2009;35:131–8. https://doi.org/10.1111/j.1447-0756.2008.00869.x.
Karlsen MA, Sandhu N, Høgdall C, Christensen IJ, Nedergaard L, Lundvall L, Engelholm SA, Pedersen AT, Hartwell D, Lydolph M, et al. Evaluation of HE4, CA125, risk of ovarian malignancy algorithm (ROMA) and risk of malignancy index (RMI) as diagnostic tools of epithelial ovarian cancer in patients with a pelvic mass. Gynecol Oncol. 2012;127:379–83. https://doi.org/10.1016/jgyno.2012.07.106.
Sun ML, Yang ZY, et al. The role of human epididymis protein 4 in the diagnosis and prognosis of diseases: an umbrella review of systematic reviews and meta-analyses of observational studies. Front Med (Lausanne). 2022;24(9): 842002. https://doi.org/10.3389/fmed.2022.842002. (PMID: 35402435).
Stiekema A, Lok C, Korse CM, et al. Serum HE4 is correlated to prognostic factors and survival in patients with endometrial cancer. Virchows Arch. 2017;470(6):655–64. https://doi.org/10.1007/s00428-017-2115-1. (Epub 2017 Apr 11).
El-Nadeim MZ, Ahmed YKB, Mowad HH, et al. Does CA 125 have a Role in Early Diagnosis of Ovarian Malignancy in non-menopausal women? J Biomed Res Environ Sci. 2022;3(4):393–6. https://doi.org/10.37871/jbres1454. (Article ID: jbres 1454).
Su Wei, Hui Li, Bei Zhang. The diagnostic value of serum HE4 and CA125 and ROMA index in ovarian cancer. Biomed Rep. Spandidos publication 2016;5:41–6. https://doi.org/10.3892/br.2016.682
Levanon K, Crum C, Drapkin R. New Insight into the pathogenesis of serous Ovarian Cancer and its clinical impact. J Clin Oncol. 2008;26(32):5284–93. https://doi.org/10.1200/JCO.2008.18.1107.
Anton C, Carvalho FM. Comparison of CA125, HE4, risk ovarian malignancy algorithm (ROMA), and risk malignancy index (RMI) for the classification of ovarian masses. Clinical Sciences. Clinics. 2012;67(5). https://doi.org/10.6061/clinics/2012(05)06
Zeng Q, Liu M, Zhou N, Liu L, Song X. Serum human epididymis protein 4 (HE4) may be a better tumor marker in early lung cancer. Clin Chim Acta. 2016;455:102–6. https://doi.org/10.1016/j.cca.2016.02.002.
Bingle L, Cross SS, High AS, Wallace WA, Rassl D, Yuan G, et al. WFDC2 (HE4): a potential role in the innate immunity of the oral cavity and respiratory tract and the development of adenocarcinomas of the lung. Respir Res. 2006;7:61. https://doi.org/10.1186/1465-9921-7-61.
Bouchard D, Morisset D, Bourbonnais Y, Tremblay GM. Proteins with whey-acidic-protein motifs and cancer. Lancet Oncol. 2006;7:167–74. https://doi.org/10.1016/S1470-2045(06)70579-4.
Granato T, Porpora MG, Longo F, Angeloni A, Manganaro L, Anastasi E. HE4 in the differential diagnosis of ovarian cancer. Clin Chim Acta. 2015;15(446):147–55. https://doi.org/10.1016/j.cca.2015.03.047. (Epub 2015 Apr 16).
Richards A, Herbst U, Manalang J, Pather S, Saidi S, Tejada-Berges T, Tan K, Williams P, Carter J. HE4, CA125, the Risk of Malignancy Algorithm and the Risk of Malignancy Index and complex pelvic masses – a peospectivecomparision in the pre- operative evaluation of pelvic masses in an Australian population. Aust N Z J ObstetGynaecol. 2015;55(5):493–7. https://doi.org/10.1111/ajo.12363. (Epub 2015 Jul 14).
Ferraro S, Borille S, Caruso S, et al. Body mass index does not influence human epididymis protein 4 concentrations in serum. Clin Chim Acta. 2015;446:163–4. https://doi.org/10.1016/j.cca.2015.04.028.
Ferraro S, Schiumarini D, Panteghini M. Human epididymis protein 4: factors of variation. Clin Chim Acta. 2015;438:171–7. https://doi.org/10.1016/j.cca.2014.08.020.
LeBleu VS, Teng Y, O’Connell JT, Charytan D, Muller GA, Muller CA, et al. Identification of human epididymis protein-4 as a fibroblast-derived mediator of fibrosis. Nat Med. 2013;19:227–31. https://doi.org/10.1038/nm.2989.
Piek A, Meijers WC, Schroten NF, Gansevoort RT, de Boer RA, Sillje HH. HE4 serum levels are associated with heart failure severity in patients with chronic heart failure. J Card Fail. 2017;23:12–9. https://doi.org/10.1016/j.cardfail.2016.05.002.
Kurman RJ, Shih I-M. Molecular pathogenesis and extraovarian origin of epithelial ovarian cancer-shifting the paradigm. Hum Pathol. 2011;42(7):918–31. https://doi.org/10.1016/j.humpath.2011.03.003.
Dikmen ZG, Colak A, Dogan P, Tuncer S, Akbiyik F. Diagnostic performances of CA125, HE4, and ROMA index in ovarian cancer. Eur J Gynaecol Oncol. 2015; XXXVI(4). https://doi.org/10.12892/ejgo2714.2015. ISSN: 0392-2936.
Suri A, Perumal V, Ammalli P, Suryan V, Bansal SK. Diagnostic measures comparison for ovarian malignancy risk in Epithelial ovarian cancer patients: a meta-analysis. Sci Rep. 2021;11(1):17308. https://doi.org/10.1038/s41598-021-96552-9.
Ferraro S, Braga F, Lanzoni M, et al. Serum human epididymis protein 4 vs carbohydrate antigen 125 for ovarian cancer diagnosis: a systematic review. J Clin Pathol. 2013;66:273–81.
Chen X, Zhou H, Chen R, et al. Development of a multimarker assay for differential diagnosis of benign and malignant pelvic masses. Clin Chim Acta. 2014;440C:57–63.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
We declare that this manuscript is original; it has not been published anywhere before and is not currently being considered for publication elsewhere. We also confirm that the authors do not have any conflict of interest associated with publication of this work and no significant financial support/funding for this work has been received to influenced the outcome. The manuscript is read and approved, and consent is given by all the authors. We give our permission to reproduce any material of the article.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed Consent
Informed consent was obtained from all individual participants included in the study. This article does not contain any studies with animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kumari, J., Amrita, Pankaj, S. et al. HE-4 A Novel Tumour Marker for Ovarian Mass and Its Comparison with CA 125. Indian J Gynecol Oncolog 22, 72 (2024). https://doi.org/10.1007/s40944-024-00838-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40944-024-00838-5