Abstract
Heavy metals in the environment have proven to be a major health concern, and there are several health risks associated with it. Seafood consumers are particularly prone to these health risks. This study was designed to investigate heavy metal levels in shellfish from Bodo City and B-Dere, Ogoniland, Rivers State, Nigeria, and to evaluate possible human health risks to consumers of shellfish in these coastal regions. Non-carcinogenic health risk for consumption of shellfish were evaluated using the estimated daily intake (EDI) and target hazard quotients (THQs) for Ni, Cr, As, Cd, Pb, and Fe, while carcinogenic health risk from Ni, Cr, Pb, Cd, As, and Ni was also evaluated. EDI was below the threshold values for Cr but exceeded the threshold for Cd, Pb, As, Ni and Fe. THQs for Cr, Cd, Pb, and Ni were below 1 except for Cd in U. tangeri from Bodo City. THQs values for As and Fe were greater than 1 except for Fe level in C. pallidus from Bodo City and U. tangeri from Bodo City and B-Dere. Also, carcinogenic risk (CR) for Cr in all shellfish exceeded the accepted risk level of 10E−4. Cd CR level for C. pallidus, T. fucastus, U. tangeri, and C. gasar collected from Bodo City and B-Dere also exceeded the accepted risk level of 10E−4 for As and Ni. CR risk for Pb was below the acceptable range of 10E−4. Consumers of shellfish from Bodo City and B-Dere may be exposed to hazardous metals contamination over a period of time.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Water and soil contamination from crude oil in Ogoniland is the primary source of heavy metals in the environment, although there are also varieties of industrial and commercial activities, such as shipping, engineering, power production, and manufacturing activities. Moreover, agricultural and domestic effluents may also contribute significant contaminants along the coastal waters in Ogoniland. There is a growing concern that hazardous metals bio-accumulated in fish muscle tissues may represent a major human health risk, especially for populations like those in Ogoniland with high fish consumption rates (Kumar and Mukherjee 2011; Liao and Ling 2003; Diez et al. 2009; Nkpaa et al. 2016). Fish bio-accumulate hazardous metals from the surrounding environment if the following factors are met: long exposure period, the characteristics of the species examined, the levels of the element, and particularly abiotic factors such as pH, seasonal changes, temperature, salinity (Ginsberg and Toal 2009; Copat et al. 2012; Burger and Gochfeld 2009). Hence, harmful substances such as hazardous heavy metals released by anthropogenic sources will be bio-accumulated in aquatic organisms via the food chain. This is because organisms like fishes are constantly exposed to contaminated water and sediment, thus could act as excellent biological markers of heavy metals pollution and non-essential metals because both are taken in and bio-accumulated in their tissues (Kumar and Mukherjee 2011; Canli and Atli 2003). The result is that human health could be at risk because of consumption of fish contaminated by toxic metals (Copat et al. 2012).
Studies on heavy metals bioaccumulation in fish abound, but in recent times, human health risk assessment for populations have become one of the fastest methods used by environmental scientist and regulatory bodies to investigate the impact of hazardous metals on human health and also to investigate and suggest the level of treatment that can solve the environmental problems (Zhang et al. 2012; Storelli and Marcotrigiano 2008; Imar and Carlos 2011; Michael et al. 2011; USEPA 2000). Sometimes the levels of contaminants exceed the permissible limits set by regulatory bodies; this may particularly represent a potential risk for human health.
Human health risk assessment (HHRA) in this study seeks to investigate the bioaccumulation of heavy metals as a result of their mobility in runoff water and crude oil spill and their absorption by shellfish during wet season which have never been investigated before in these regions. HHRA investigates the implications of human activities and weighs the adverse effects to public health. It is becoming one of the fastest and growing methods used currently by environmental toxicologists to evaluate the impact of the hazardous metals bioaccumulation on human health. HHRA of heavy metals on human health from consumption of seafood in this present study is divided into carcinogenic risks (CR) and non-carcinogenic risks such as target hazard quotients (THQ) and estimated daily intake (EDI) (Han et al. 1998; Yabanli and Alparslan 2015). Heavy metals pollution of seafood and its toxicity has become an inevitable challenge nowadays (Orisakwe et al. 2012). It is very important to estimate non-carcinogenic and carcinogenic risks of heavy metals through the consumption of polluted seafood. HHRA protocols were developed by the US Environmental Protection Agency (USEPA) for the assessment of potential health risks associated with long-term exposure to chemical pollutants (USEPA 1989; Kumar et al. 2010a, b; Harmanescu et al. 2011). HHRA has also been used recently by many researchers (Onuoha et al. 2016; Nkpaa et al. 2016; Adeel and Riffact 2014; Amirah et al. 2013; Copat et al. 2012; Kumar and Mukherjee 2011; Yu-jun et al. 2011; Harmanescu et al. 2011; Declan and Andrea 2008; Wang et al. 2005; Chien et al. 2002) and has been very productive.
Oil exploration and production in Ogoniland began in the early 1990s. The oilfields and installations have since largely remained dormant due to civil unrest by the populace. However, major oil pipelines still pass through Ogoniland, and crude oil spills, especially the oil spills in 2008 and 2009, continue to cause environmental degradation in the region due to such factors as illegal artisanal refining, vandalism to oil facilities, and lack of maintenance. This has resulted in contamination of rivers, creeks, agricultural farm, and forest lands (Kinako and Awiwaadu 2000). Previous reports have shown high levels of heavy metals contamination in fishes and water from Kaa, B-Dere and Bodo City in Ogoniland from crude oil contamination during dry season (Nkpaa et al. 2013, 2016).
Heavy metal mobility in runoff water and absorption by sediments and seafood during wet season may contribute significantly to the levels of contaminants (metals) that an exposed population are at risk to in addition to the direct crude oil spill in coastal water as seen in B-Dere and Bodo City, in Gokana Local Government Area, Ogoniand, Rivers State, Nigeria. This kind of contamination clearly contributes to an increase of heavy metals content in the environment. Thus, in these coastal areas, there is a need for systematic and detailed research on the health risk of heavy metals bioaccumulation as a result of their mobility in runoff water and crude oil spill and their absorption by shellfish during wet season. It is pertinent to treat and protect water contaminants from different sources in different ways (Li et al. 2013) to prevent human health risk. Therefore, the aim of this research is to assess the impact of runoff water from contaminated agricultural land and crude oil spill in coastal water on the levels of heavy metals bioaccumulation in shellfish in wet season and consequently, evaluate the Estimated Daily Intake, Target Hazard Quotient, and Carcinogenic Risk provided in the US-EPA Region III risk-based concentration table (USEPA 2000) in order to investigate possible causes for alerts regarding hazards to human health for those who consume shellfish.
Materials and methods
Study area
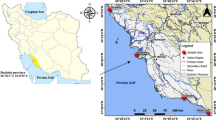
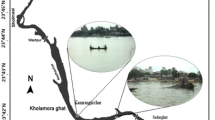
Samples were collected from Bodo city and B-Dere all in Ogoniland (Latitude 4°40′5′′N and 4°43′19.5′′N and Longitude 7°22′53.7′′E and 7°27′9.8′′E), an important and known crude oil producing province in the South-East of Niger Delta, Nigeria (Nkpaa et al. 2016). Ogoniland is known to have a tragic history of pollution from oil spills and oil well fires, although there have been no systematic scientific information available (except the recent UNEP 2011 report) about the ensuing contamination. This region covers some 1000 km2 in the south-east of the Niger Delta basin. Its population is about 832,000, according to the 2006 National Census. The major source of protein for the people living in these coastal regions is seafood.
Oil activities such as exploration began in Ogoniland in the 1950s and extensive oil exploration and production facilities were established during the first 30 years. These facilities were operated by Shell Petroleum Development Company (Nigeria) Ltd (SPDC), a joint venture between the Nigerian National Petroleum Company (NNPC), Shell International, Elf and Agip. Once these facilities began operation, other issues had to be dealt with, such as spills caused during oil production and the disposal of water (often salty and known as ‘produced water’) and flaring of gas (produced gases) generated alongside the oil. All these activities and their effects leave an environmental footprint in Ogoniland (UNEP 2011). It is important to note that the majority of seafood consumed in Ogoniland, especially Gokana Local Government Area (LGA), Rivers State, Nigeria, come from the study sites (B—Dere and Bodo City). These research study sites have witnessed many oil spills as a result of bunkering and artisanal crude oil refining activities and pipeline failures. This presents a special health risk to populace consuming seafood from these sites.
Collection of test samples
Four species of shellfish Callinectes pallidus (crabs), Tympanotonus fucastus (periwinkle), Uca tangeri (fiddler crab), and crassostrea gasar (oysters) mostly consumed by the populace were collected from two sites (February 02nd, 2016) from B-Dere and Bodo City in Gokana Local Government Area, Ogoniand, Rivers State, Nigeria. The sampling period was during the wet season. Wet season was chosen for this study because of the high consumption rate of shellfish and high surface runoff from contaminated agricultural land experienced around this coastal water in the wet season. At each site, twenty (20) individual shellfish were collected, and dirt and particles were removed by washing with double distilled water. The fishes were subsequently blotted with tissue paper and kept frozen in an ice chest before transportation to the laboratory for analysis (Nkpaa et al. 2016).
Quality assurance and quality control
The selection of species was determined by their feeding habits (sediment feeders) which have significant effect on their metal bioaccumulation and retention except C. gasar. Among the analyzed species, T. fucastus has accumulation of sediments on the shell which contribute significantly to the extent of metals bioaccumulation. Deionized water which was double distilled and free from dirts was used for the study. Glassware and plastic ware (Merck, Germany) used were rinsed thoroughly with 10% HNO3 followed by washing with deionized distilled water. For data assurance, the entire test samples were spectrophotometrically analyzed in triplicate. Determination of metals was performed with a GBC Avanta atomic absorption spectrophotometer (ASS) (model: PM ver 2.02 Avanta). The analytical blanks used in the study were determined as the test samples. The heavy metal levels were analyzed using standard solutions which were prepared within the same acid matrix. Standards for the instrument calibration were prepared on the basis of monoelement certified reference solution ICP Standard (Merck, Germany) (Yesudhason et al. 2013; Nkpaa et al. 2016).
Sample processing
The edible part of the samples was carefully removed from its shell and oven dried thoroughly using Memmert drying oven (U27, Germany) for 3 days at a temperature of 60–70°C, after which it was ground to powder form using Silimic mortar (Pyrex). Five grams of the sample was weighed into a crucible container, and then introduced into a furnace to derive the ash at 450–500°C for 6 h. Six hours later, a crucible thug was used to bring down the crucible from the furnace and placed in a desiccator where it was allowed to cool. After cooling, 5 mL of 10% hydrochloric acid was used to dissolve ash content to near dryness. After that, it was filtered into a funnel and cylinder before it was made up to 20 mL with distilled water before the metals analysis using atomic ASS (Nkpaa et al. 2013, 2016).
Atomic absorption spectrophotometer
ASS (model pm ver 2.02 Avanta) methods were used for each metal and was calibrated using standard of the metals (As, Cr, Ni, Cd, Pb and Fe). Cr, Fe, and As were evaluated using hollow cathode lamp (HCL) in a Flame atomizer AAS. Cd, Pb, and Ni were analyzed using electrode less discharge lamp (EDL) in the Flame atomizer AAS (Nkpaa et al. 2013, 2016). The extract was aspirated directly into the ASS machine. The carrier gas was acetylene and air pressure at of 70 psi.
Health risk assessment for shellfish consumption
Estimated daily intake (EDI)
Hazardous heavy metals exposure pathway to human via ingestion of polluted food has been investigated by many researchers (Copat et al. 2012; Xue et al. 2012; Chary et al. 2008; Nkpaa et al. 2016). EDI of individual hazardous metals in this study was evaluated by the equation below:
where EDI is the estimated daily intake; EF is the exposure frequency (365 days−1 year−1); ED is the exposure duration, equivalent to average lifetime (65 years for Nigeria population); FIR is the fresh food ingestion rate (g−1 person−1 day−1), which was considered to be 102 g−1 person−1 day−1 for fish consumers from Bodo City and B-Dere, Ogoniland, Rivers State, Nigeria (Nkpaa et al. 2016); Cm is the heavy metal concentration in foodstuffs (mg kg−1 Dw.) Conversion factor of 0.208 was used to multiply the Cm from fresh weight (FW) to dry weight (Dw.); Wab is the average body weight (bw) (Wab was considered to be 60 kg); TA is the average exposure time for non-carcinogens (equal to EF × ED) (Saha and Zaman 2012).
Target hazard quotients (THQs)
Estimation of human health risk via consumption of hazardous metals polluted shellfish was done using THQ. This was calculated based on the formula provided on USEPA Region III Risk-Based Concentration Table (USEPA 2011). The THQ is an estimate of the risk level (non-carcinogenic) due to pollutant exposure (Yu-Jun et al. 2011). If the ratio is equal to or greater than 1, an exposed population experiences health risks. The methods used for the estimation of THQ have been provided in USEPA Region III Risk-Based Concentration Table, January–June 1996 (Han et al. 1998; USEPA 2011; Chien et al. 2002; USDOE 2011). Given below is the equation used for THQs:
where THQ is the target hazard quotient; RfD is the oral reference dose (mg−1 kg−1 day−1). RfD for Cr, Cd, Pb, As, Ni, and Fe are 1.5, 1E−3, 3.5E−3, 2E−2, 3E−4 and 1.7 mg−1 kg−1 day−1, respectively (USEPA 2000).
Carcinogenic risk (CR)
USEPA stated that the Carcinogenic Risk between 10–6 (1 in 1,000,000) to 10–4 (1 in 10,000) represent a range of permissible predicted lifetime risks for carcinogens (Valberg et al. 1996; USEPA 2011). Chemicals for which the risk factor falls below 10–6 may be eliminated from further consideration as chemicals of concern (Liu et al. 2006). The human health risk associated with the carcinogenic effect of hazardous heavy metal is expressed as the probability that an individual may contract cancer over a lifetime period of 70 years (Liu et al. 2006; USEPA 2011; Nkpaa et al. 2016). The equation used for estimating CR was as follows:
where CR is the carcinogenic risk; CSF is the ingestion cancer slope factors. The ingestion cancer slope factors (CSF) evaluates the probability of an individual contracting cancer from oral exposure to pollutants over a period of a lifetime (ATSDR 2010; Nkpaa et al. 2016). Ingestion cancer slope factors for Cr, Cd, Pb, As, and Ni are 0.5, 0.35, 8.5E−3, 1.7, and 1.5 and are expressed in units of (mg/kg/day)−1.
Statistical analysis
The data were statistically analyzed by SPSS software version 20. One-way ANOVA were applied for evaluating the significant difference between hazardous metals concentration in seafood, and student t-test was applied to evaluate the statistical differences between the two sites.
Results and discussion
The result for Cr, Cd, Pb, As, Ni, and Fe in shellfish sampled in Bodo City and B-Dere Ogoniland are presented on Table 1 with the following ranges: 1.84–4.49, 0.96–3.82, 1.64–3.67, 2.13–4.89, 4.35–8.34, and 1256.2–2593 mg kg− 1 for Cr, Cd, Pb, As, Ni, and Fe respectively. The level of Cr in T. facustus from Bodo City and B-Dere was significantly (p < 0.05) higher than those of other shellfish. It can be seen that the level Cd, Pb, and As in U. tangeri from Bodo City and B-Dere was significantly (p < 0.05) higher than those of other shellfish. C. gasar from Bodo City and B-Dere had Ni mean level higher than those from the other shellfish. But there was no statistically significant difference in the mean level of Fe in all the shellfish analyzed. The result indicates the ability of U. tangeri to concentrate Cd, Pb, and As (2.54–3.82, 3.42–3.67 and 3.76–4.89 mg kg− 1 for Cd, Pb, and As respectively) is much stronger than that of other shellfish. Therefore, the potential risk of consuming U. tangeri is relatively higher than that of other seafood due to the high Cd, Pb, and As bioaccumulation of U. tangeri. Because of high tolerance of shellfish to high level of Cr, Cd, Pb, As, Ni, and Fe, it may not affect the populace directly, but may subsequently transfer toxicity to humans through the food chain and render people consuming the shellfish more prone to contamination (Okazaki and Panietz 1981; Oliver 1997; Han and Hung 1990). This is because in most cases, these shellfish live in an environment with heavy metal contaminations. The FAO, WHO, EU, and other regulatory bodies of many countries have provided the maximum permissible limit of heavy metals in foods (Xue et al. 2012; Chary et al. 2008). The level of each metals determined in this study was above the maximum permissible limit by USEPA (2011). For example, Pb exceeded the permissible limit of 0.3 mg kg−1, while Cd also exceeded maximum tolerable of 2 mg kg−1 by (EU 2006). However, Cr was above the minimum permissible limit of 1 mg kg−1 set by USEPA (2011). From the results, it can be predicted that consumption of shellfish from the Bodo City and B-Dere by humans can lead to chronic heavy metals poisoning.
Human health risk assessment
The results of EDI for shellfish are shown in Table 2. The EDI of Cr from consumption of these shellfish ranged between 0.65 and 1.59 g person−1 day−1 with the highest intake of Cr coming from the consumption of T. fucastus collected from Bodo City (1.59 g−1 person−1 day). This value is above the tolerable daily intake level (1.5 g−1 person−1 day) established by FAO/WHO (1993) and USEPA (2011). EDI of Cr through the consumption of seafood from these study sites could adversely affect balanced glucose metabolism in the population. Chromium has been reported to facilitate insulin action (Nielsen 1993; Vincent 1999; Nkpaa et al. 2016). The EDI for Cd ranged between 0.34 and 1.35 g−1 person−1 day. U. tangeri from Bodo City showed the highest EDI of 1.35 g−1 person−1 day−1 for Cd and exceeded the tolerable daily intake of 0.001 g−1 person−1 day−1 established by USEPA (2011) by 1350 times. The high EDI recorded for Cd in shellfish is an indication that it might lead to severe ingestion of Cd. Cd has been shown to cause acute and chronic intoxications by Chakraborty et al. (2013). If Cd is ingested in higher amounts, it may result to stomach irritation, vomiting and diarrhea. Over long-term exposure, Cd can become deposited in the renal tissues and finally lead to kidney disease, lung damage, and fragile bones (Bernard 2008; Jaishankar et al. 2014). Ingestion of high levels of Cd has been reported to cause acute renal failure in humans (NANS-NRC 1982). Cadmium is a well-known endocrine disruptor capable of increasing the risk of developing prostate cancer and breast cancer in humans (Saha and Zaman 2012; Akoto et al. 2014).
Results also showed that EDI of Pb ranges between 0.58 and 1.30 g−1 person−1 day, with U. tangeri collected from Bodo City having the highest EDI value of 1.30 g−1 person−1 day−1. This EDI value far exceeds the established EDI of Pb by USEPA (2011). Ingestion of Pb via the consumption of shellfish may cause chronic Pb intoxication in adults. It may also result in anemia, some types of cancer, and reproductive malfunction in males, while in young children, it may lead to hormonal imbalance of metabolite of vitamin D (1, 25- dihydroxy-vitamin D) and a drop in IQ (Tandon et al. 2001; Siddiqui et al. 2002; Lindbohm et al. 1991). Chronic lead exposure can also result in mental retardation, birth defects, psychosis, autism, allergies, dyslexia, weight loss, hyperactivity, paralysis, muscular weakness, brain damage, kidney damage, and may even cause death (Martin and Griswold 2009). The EDI of As and Ni ranged between 0.75 and 1.73 and 1.54–2.95 g−1 person−1 day−1, respectively, with the highest As and Ni intake coming from the consumption of U. tangeri collected from Bodo City and U. tangeri from B-Dere, respectively. The provisional tolerable intake for As is 0.0012 g−1 person−1 day−1 USEPA (2011). U. tangeri from B-Dere showed the highest EDI of 2.95 g−1 person−1 day−1 for Ni which exceeded the tolerable daily intake of 0.0012 g−1 person−1 day−1 established by USEPA (2011) by 2458 times. Arsenic may cause protoplastic poison since it affects primarily the sulphydryl group of cells causing malfunctioning of cell respiration, cell enzymes, and mitosis (Gordon and Quastel 1948; Jaishankar et al. 2014). High levels of Nickel observed in this study could cause some genetic abnormalities like DNA strand break, DNA–protein cross links, nucleotide excision, micronuclei, nucleic acid concentration alteration, single gene mutations, sister chromatid exchanges, and cell transformation (Coogan et al. 1989; Das and Dasgupta 2000). Das and Dasgupta (2002) also reported decreased sperm count, sperm motility, alteration of steroidogenesis, suppresses antioxidant enzymes activities, and elevated testicular lipid peroxidation in nickel exposed rats (Gupta et al. 2007). EDI values of Fe ranged between 444.2 and 916.9 g−1 person−1 day−1. EDI rates of 916.9 g−1 person−1 day−1 was obtained from the consumption of C. pallidus from B-Dere. The high level of Fe through the consumption of shellfish may be good sources of Fe in diets of consumers (Nkpaa et al. 2016). At the same time, it may be highly dangerous since too high levels can lead to intracellular breakdown and promote DNA damage (Jaishankar et al. 2014). Fe can also initiate cancer mainly by the process of oxidation of DNA molecules (Bhasin et al. 2002; Jaishankar et al. 2014).
We also calculated the THQs with RfD set by EFSA (2009, 2010) for Cr, Cd, Pb, As, Ni and Fe. THQs results indicate that there is no carcinogenic risk via consumption of shellfish as shown on Table 3. Values of the THQ index for total exposure above 1.0 indicate that the estimated exposure is potentially of concern. However, the THQ does not define a dose–response relationship, and its numerical value should not be regarded as a direct estimate of risk (USEPA 1996; Nkpaa et al. 2016). The THQs for Cr and Cd ranged from 04.30E−4–1.06E−3 and 0.34–1.35. However, the THQ value for Pb, As, Ni and Fe ranged between 0.17 and 0.37, 2.50–5.77, 0.08–0.15, and 0.63–1.25, respectively, with U. tangeri collected from Bodo City showing the highest THQ of 5.77. The THQs result indicated that Cr, Cd, Pb, and Ni were below the safe standard of 1. This observation is suggestive that consumers of seafood from the study sites would not experience significant health risks from intake of individual metals through seafood consumption, except for As and Fe which was above safe standard of 1 (Nkpaa et al. 2016). However, U. tangeri from Bodo City was above 1 for Cr. THQs values below 1 indicate that the level of exposure metal pollution is smaller than the reference dose (Copat et al. 2012). Therefore we can assume that the daily intake shown by results at this stage is more likely to cause deleterious effects during a lifetime in humans from As and Fe toxicity. In other words, the reference dose is not a sharp division line between “‘safe” and “unsafe” intakes. It is by no means certain that intakes at or below the references are risk-free or that intakes above it pose undue risks (Rodricks and Jackson 1992). Li et al. (2014), and Krishna et al. (2014) reported that high THQ value >1 indicate a potential health risk to human beings, especially consumers who reside in area with serious metal pollution.
The averages carcinogenic risk (CR) of Cr, Cd, Pb, As, and Ni via the consumption of shellfish are shown in Fig. 1. The average carcinogenic risk for Cr, Cd, Pb, As, and Ni ranged between 3.25E−4–7.95E−3, 1.29E−4–5.13E−4, 2.29E−6–1.11E−5, 1.13E−3–2.60E−3, and 2.63E−3–5.02E−3, respectively, with T. facustus collected from Bodo City showing the highest CR value of 7.95E−3. CR is measured and expressed as a probability of contracting cancer over a lifetime of 70 years. CR results indicated that the average CR value of cancer risk from Pb accumulation did not show carcinogenicity, this is in agreement with our earlier work (Nkpaa et al. 2016). However, in comparison with established guideline values, data from this study indicate that these shellfish (C. pallidus, T. fucastus, U. tangeri, and C. gasar) collected from Bodo City and B-Dere may not be safe for human consumption and as such consumers of these species of seafood have the increased probability of contracting cancer from As and Ni exposure over a lifetime period of 70 years or more in future.
Conclusion
Data from this study indicated that there are significant health risks associated with heavy metal mobility in runoff water and absorption by sediments and seafood during wet season and its consumption especially for consumers of shellfish. This may contribute significantly to the levels of contaminants (metals) that an exposed population are at risk to in addition to the direct crude oil spill in coastal waters as seen in B-Dere and Bodo City, in Gokana Local Government Area, Ogoniand, Rivers State, Nigeria. The shellfish analyzed reveal potentially toxic level of heavy metals if they enter the food chain. Data obtained on EDI, THQs, and CR indicate that the levels of heavy metals found in the sampled shellfish represent a very high risk to human health. In the light of the above findings, policy makers in government and regulatory bodies should strongly encourage crude oil producing companies to maintain and repair their oil production and transportation facilities on a regular basis; provide strict security to oil installation facilities; and educate the populace, especially the youths, in these coastal on the danger of bunkering and illegal artisanal refining activities. These actions will help prevent human health risks associated with hazardous heavy metals contamination.
References
Adeel M, Riffact NM (2014) Human health risk assessment of heavy metals via consumption of contaminated vegetables collected from different irrigation sources in Lahore, Pakistan. Arabian J Chem 7:91–99
Akoto OF, Bismark ED, Adei E (2014) Concentrations and health risk assessments of heavy metals in from the Fosu Lagoon. Int J Environ Resour 8(2):403–410
Amirah MN, Afiza AS, Faizal WI, Nurliyana MH (2013) Human health risk assessment of metal contamination through consumption of fish. J Environ Pollut Hum Health (Lond) 1(1):1–5
ATSDR (2010) Agency for Toxic Substance and Disease Registry, public health assessment and health consultation. CENEX supply and marketing, Incorporated, Quicy, Grant County, Washington
Ayolagha GA, Peter KD (2013) Effect of remediation on growth parameters, grain and dry matter yield of soybean (Glycine max) in crude oil polluted ultisols in Ogoni Land, South Eastern Nigeria. Afr J Environ Sci Technol 7(2):61–67
Bernard A (2008) Cadmium & its adverse eff ects on human health. Indian J Med Res 128(4):557–564
Bhasin G, Kauser H, Athar M (2002) Iron augments stage-I and stage-II tumor promotion in murine skin. Cancer Lett 183(2):113–122 (Bielicka)
Burger J, Gochfeld M (2009) Perceptions of the risks and benefits of fish consumption: individual choices to reduce risk and increase health benefit. Environ Resour 109: 343–349
Canli M, Atli G (2003) The relationships between heavy metal (Cd, Cr, Cu, Fe, Pb, Zn) levels and the size of six Mediterranean fish species. Environ Pollut 121:129–136
Chakraborty S, Dutta AR, Sural S, Gupta D, Sen S (2013) Ailing bones and failing kidneys: a case of chronic cadmium toxicity. Ann Clin Biochem 50(5):492–495
Chary NS, Kamala CT, Raj DS (2008) Assessing risk of heavy metals from consuming food grown on sewage irrigated soils and food chain transfer. Ecotoxicol Environ Saf 69(3):513–524
Chien LC, Hung Choang KY, Yeh CY, Meng PJ, Shieh MJ, Ha BC (2002) Daily intake of TBT, Cu, Zn, Cd and As for fisherman in Taiwan. Sci Total Environ 285:177–185
Coogan TP, Latta DM, Snow ET, Costa M (1989) Toxicity and carcinogenicity of nickel compounds. In: McClellan RO (ed) Critical reviews in toxicology, vol 19. CRC Press, Boca Raton, pp 341–384
Copat C, Bella F, Castaing M, Fallico R, Sciacca S, Ferrante M (2012) Heavy metals concentrations in fish from Sicily (Mediterranean Sea) and evaluation of possible health risks to consumers. Bull Environ Contam Toxicol 88:78–83
Costa M (1991) Molecular mechanism of nickel carcinogenesis. Annu Rev Pharmacol Toxicol 3:321–337
CXS (2013) Codex general standard for contaminants and toxins in food and feed. Codex Standard 193–1995. Revision 1997, 2006, 2008, 2009. Amendment 2010, 2012, 2013
Das KK, Dasgupta S (2000) Effect of nickel on testicular nucleic acid concentrations of rats on protein restriction. Biol Trace Elem Res 73:175–180
Das KK, Dasgupta S (2002) Effect of nickel sulphate on testicular steroidogenesis in rats during protein restriction. Environ Health Perspect 110:923–926
Declan PN, Andrea P (2008) Heavy metal ions in wines: meta-analysis of target hazard quotients reveal health risks. Chem Cent J 2:22
Diez S, Delgado S, Aguilera I et al (2009) Prenatal and early childhood exposure to mercury and methylmercury in Spain, a high-fish-consumer country. Achieve Environ Contam Toxicol 56: 615–622
EFSA (2009) Cadmium in food. (Request N EFSA-Q-2007-138) (adopted on 30 January 2009) scientific opinion of the panel on contaminants in the food chain. EFSA J 980:1–139
EFSA (2010) Scientific opinion on lead in food (request N. EFSA-Q2007-137) (adopted on 18 March 2010) EFSA panel on contaminants in the food chain (CONTAM). EFSA J 8:1570
European Union (2006) Setting maximum levels for certain contaminants in foodstuffs, Commission Regulation (EC) No 1881/2006; OJ L 364, 20.12, p 5
FAO (1983) Food and Agriculture Organization. Compilation of legal limits for hazardous substances in fish and fishery products. Food and Agriculture Organization of the United Nations, Rome, Italy, pp 5–100
FAO/WHO (1989) National Research Council Recommended Dietary Allowances, 10th edn. National Academy Press, Washington, DC
FAO/WHO (1993) Evaluation of certain food addictives and contaminants. WHO technical report series no. 837
Ginsberg GL, Toal BF (2009) Quantitative approach for incorporating methylmercury risks and omega-3 fatty acid benefits in developing species-specific fish consumption advice. Environ Health Perspect 117:267–275
Gordon J, Quaste J (1948) Effects of organic arsenicals on enzyme systems. Biochem J 42:337
Gupta AD, Dhundasi SA, Ambekar JG, Das KK (2007) Effect of l-ascorbic acid on antioxidant defense system in testes of albinorats exposed to nickel sulphate. J Basic Clin Physiol Pharmacol 18:87–95
Han BC, Hung TC (1990) Green oyster caused by copper pollution on the Taiwan coast. Environ Pollut 65:347–362
Han B, Jeng WL, Chen RY, Fang GT, Hung TC, Tseng RJ (1998) Estimation of target hazard quotients and potential health risks for metals by consumption of seafood in Taiwan. Arch Environ Contam Toxicol 35(4):711–720
Harmanescu M, Alda LM, Bordean DM, Gogoasa I et al (2011) Heavy metals health risk assessment for population via consumption of vegetables grown in old mining area; a case study: Banat County, Romania. Chem Cent J 5:64
Imar MR, Carlos JRS (2011) Metal levels in fish captured in Puerto Rico and estimation of risk from fish consumption. Arch Environ Contam Toxicol 60:132–144
Jaishankar M, Tseten T, Anbalagan N, Matthew BB, Beeregowda KN (2014) Toxicity, mechanism and health effects of some heavy metals. Interdiscip Toxicol 7(2):60–72
Kinako PD, Awi-Waadu GDB (2000) General ecology: a- state-of-the-art compendium of ecology. Belk Publishers limited, Port Harcourt
Krishna PV, Jyothirmayi V, Rao KM (2014) Human health risk assessment of heavy metal accumulation through fish consumption, from Machilipatnam Coast, Andhra Pradesh, India. Int Res J Public. Environ Health 1(5):121–125
Kumar B, Mukherjee DP (2011) Assessment of human health risk for arsenic, copper, nickel, mercury and zinc in fish collected from tropical wetlands in India. Adv Life Sci Technol 2:13–24
Kumar A, Chang B, Xagoraraki I (2010a) Human health risk assessment of pharmaceuticals in water: issues and challenges ahead. Int J Environ Resour Public Health 7(11):3929–3953
Kumar K, Priya M, Mukhopadhyay DP (2010b) Distribution, partitioning, bioaccumulation of trace elements in water, sediment and fish from sewage fed fish ponds in eastern Kolkata, India. Toxicol Environ Chem 92(2):243–260
Li P, Wu J, Qian H, Lyu X, Liu H (2013) Origin and assessment of groundwater pollution and associated health risk: a case study in an industrial park, northwest China. Environ Geochem Health. doi:10.1007/s10653-013-9590-3
Li Z, Zhang D, Wei Y (2014) Risk assessment of trace elements is cultured from freshwater fishes from Jiangxi Provence, China. Environ Monit Assess 186:2185–2194
Liao CM, Ling MP (2003) Assessment of human health risks for arsenic bioaccumulation in tilapia (Oreochromis mossambicus) and large-scale mullet (Liza macrolepis) from blackfoot disease area in Taiwan. Arch Environ Toxicol 45:264–272
Lindbohm ML, Sallmen M, Anttila A, Taskinen H, Hemminki K (1991) Paternal occupational lead exposure and spontaneous abortion. Scand J Work Environ Health 17:95–103
Liu C-W, Liang C-P, Huang FM (2006) Assessing the human health risks from exposure of inorganic arsenic through oyster (Crassostrea gigas) consumption in Taiwan. Sci Total Environ 361:57–66
Martin S, Griswold W (2009) Human health effects of heavy metals. Environ Sci Technol Briefs Citiz 15:1–6
Michael AM, Matthew RM, Michael FEW (2011) Elevated levels of metals and organic pollutants in fish and clams in the Cape Fear River watershed. Arch Environ Contam Toxicol 61:461–471
Mokhtar M (2009). Assessment level of heavy metals in Penaeusmonodon and Oreochromis spp. in selected aquaculture ponds of high densities development area. Eur J Sci Res 30(3): 348e60
NANS-NRC (1982) National, Drinking Water and Health. Academy of Sciences-National Research Council National Academic Press, Washington, DC
Nielsen FH (1993) Chromium. In: Shils ME, Olson JA, Shike M (eds) Modern nutrition in health and disease, 8th edn. Lea & Febiger, Philadelphia, pp 264–268
Nkpaa KW, Wegwu MO, Essien EB (2013) Heavy metals concentrations in four selected seafood from crude oil polluted waters of Ogoniland, Rivers State, Nigeria. Arch Appl Sci Res 5(4):97–104
Nkpaa KW, Patrick-Iwuanyanwu KC, Wegwu MO, Essien EB (2016) Health risk assessment of hazardous metals for population via consumption of seafood from Ogoniland, Rivers State, Nigeria; a case study of Kaa, B-Dere, and Bodo City. Environ Monit Assess 188(1):9
Okazaki RK, Panietz MH (1981) Depuration of twelve trace metals in tissues of the oysters Crassostrea gigas and C. virginica. Mar Biol 13:113–120
Oliver MA (1997) Soil and human health: a review. Eur. J Soil Sci 48:573–592
Onuoha SC, Anelo PC, Nkpaa KW (2016) Nkpaa1 Human Health Risk Assessment of heavy metals in snail (Archachatina marginata) from four contaminated regions in Rivers State, Nigeria. Am Chem Sci J 11(2):1–8
Orisakwe OE, Nduka JK, Amadi CN (2012) Heavy metals health risk assessment for population via consumption of food crops and fruits in Owerri, South Eastern, Nigeria. Chem Cent J 6(77):1–7
Rodricks JV, Jackson BA (1992) Food constituents and contaminants. In: Lippmann M (ed) Environmental toxicants—human exposure and their health effect. Van Nosstrand, New York, pp. 266–298
Saha N, Zaman MR (2012) Evaluation of possible health risks of heavy metals by consumption of food stuffs available in the central market of Rajshahi City, Bangladesh. Environ Monit Assess 185:3867–3878
Siddiqui MK, Srivastava S, Mehrotra PK (2002) Environmental exposure to lead as a risk for prostate cancer. Biomed Environ Sci 15:298–305
Storelli MM, Marcotrigiano GO (2005) Bioindicator organisms: heavy metal pollution evaluation in the Ionian Sea (Mediterranean Sea—Italy). Environ Monit Assess 102:159–166
Tandon SK, Chatterjee M, Bhargava A, Shukla V, Bihari V (2001) Lead poisoning in Indian silver refiners. Sci Total Environ 281:177–182
United Nation Environment Programme (UNEP) (2011) Environmental Assessment of Ogoniland
USDOE (2011) The risk assessment information system (RAIS). U.S. Department of energyork ridge operations office (ORO)
USEPA (1989) Guidance manual for assessing human health risks from chemically contaminated, fish and shellfish. U.S. Environmental Protection Agency, Washington, DC, EPA503/8-89-002
USEPA (1996) Region III risk-based concentration table, January–June 1996. Memorandum from RL Smith, Office of RCRA, Technical and program support branch (3HW70), Washington DC
USEPA (2000) Risk-based concentration table. United States Environmental Protection Agency, Philadelphia
USEPA (2011) USEPA Regional Screening Level (RSL) Summary Table: November 2011. Available at: http://www.epa.gov/regshwmd/risk/human/Index.htm. Last update: 6th December
USFDA (1993) Food and Drug Administration, Guidance document for nickel in shell fish. DHHS/PHS/FDA/CFSAN/office of seafood, Washington DC
Valberg PA, Drivas PJ, McCarthy S (1996) Evaluating the health impacts of incinerator emissions. J Hazard Mater 47:205–227
Vincent JB (1999) Mechanisms of chromium action: low-molecular weight chromium-binding substance. J Am Coll Nutr 18:6–12
Wang X, Sato T, Baoshan X (2005) Health risk of heavy metals to the general public of Tianjin, China via consumption of vegetables and fish. Sci Total Environ 350:28–37
Xue ZJ, Liu SQ, Liu YL (2012) Health risk assessment of heavy metals for edible parts of vegetables grown in sewage-irrigated soils in suburbs of Baoding City, China. Environ Monit Assess 184:3503–3513
Yabanli M, Alparslan Y (2015) Potential health hazard assessment in terms of some heavy metals determined in demersal fishes caught in Eastern Aegean Sea. Bull Environ Contam Toxicol 95(4):494–498
Yesudhason P, Al-Busaidi M, Al-Rahbi WAK, Al-Waili AS (2013) Distribution patterns of toxic metals in the marine oyster Saccostrea cucullata from the Arabian Sea in Oman: spatial, temporal, and size variations. SpringerPlus 2:282
Yu-jun Y, Zhifeng Y, Shanghong Z (2011) Ecological risk assessment of heavy metals in sediment and human health risk assessment of heavy metals in fishes in the middle and lower reaches of the Yangtze River Basin. J Environ Pollut 159:2575–2585
Zhang Y, Zhang B, Li L, Wang Z, Xie S, Shen Y, Li J (2012) Radiation induced reduction: an effective and clean route to synthesize functionalized graphene. J Mater Chem 22(16):7775
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nkpaa, K.W., Onyeso, G.I. & Achugasim, O. Heavy metals levels in shellfish from Bodo City and B-Dere, Ogoniland, Rivers State, Nigeria, and evaluation of possible health risks to consumers. Sustain. Water Resour. Manag. 3, 83–91 (2017). https://doi.org/10.1007/s40899-017-0096-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40899-017-0096-5