Abstract
Purpose
The aim of this work is to provide an overview of studies that explore methods of assessment for muscle tone in patients with neurological disorders.
Methods
A scoping review was conducted on the PubMed, IEEE/IET Electronic Library, Web of Science, Scopus, Cochrane, Bireme, Google Scholar, and Science Direct databases. The characteristics analyzed were types of tonus; neurological disorder diagnosis; the method used for assessing muscle tone and the muscle under evaluation. The search found a total of 1593 studies, and after the filtering process, 84 were selected.
Results
The most reported method for assessing muscle tonus was the Modified Ashworth Scale, applied to post-stroke patients. Only two studies evaluated patients with hypotonia. Some studies presented quantitative devices for the evaluation as an alternative to the most commonly used methods.The most evaluated muscles were the elbow and the knee flexors.
Conclusion
The results show the importance of properly assessing the different types of muscle tone, and show a lack in evaluating hypotonia. The results also demonstrate the need to create new devices that provide more objective and reliable methods that can evaluate more than one type of tonus.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Background
The muscles of the human body have a state of permanent tension: even when the muscle is in a resting state, there is a base degree of contraction known as muscle tone or tonus. The presence of adequate muscle tone is essential, as this allows one to perform functions such as activities of daily living (ADL) [1]. Individuals with neurological injuries that may compromise the central nervous system (CNS) or the peripheral nervous system (PNS) may lose the natural ability to control the level of the tonus and display very low or very high resting tone, known as hypotonia and hypertonia, respectively [2].
Hypotonia, hypertonia and rigidity are the most common disorders found in the muscle tone. Hypotonia can be characterized by lower tonus, where the muscle is not able to support the joint well; therefore, it has little to no resistance to external movements, making it easier for another person to manipulate the joint. This type of disorder can be caused by conditions such as cerebral palsy (CP) [3] and peripheral neuropathies. Hypertonia is defined by an abnormally high tonus, and it is otherwise known as spasticity. This type of disorder causes an increase in resistance to movement and results in limitations to the joint [4]. High values of tonus may also prevent movement, coordination and learning. Hypertonia may be present in numerous clinical conditions, such as traumatic brain injury (TBI), stroke or certain neurodegenerative processes [5]. Rigidity can be defined by the presence of very high muscle tone, which severely restricts any kind of movement, and is often present in disorders such as Parkinson’s disease (PD) [6].
Muscle tone assessments using validated methods are essential for clinical decisions. The results of such assessments can be used to find the best course of treatment for each individual patient, identify concerns about the degree of impairment of the evaluated limb, and provide assistance in monitoring the progress of patients undergoing a particular treatment. [7].
Currently, there are several reports in the literature from studies that performed evaluation of muscle tone in patients with neurological disorders using both subjective and objective methods. Subjective methods may include ordinal measurements, such as the Modified Ashworth Scale (MAS), which is an instrument capable of assessing limb resistance to movement [8, 9]. Among the objective methods, some of the quantitative variables measured are kinematic components [10], torque [11, 12], and electromyography [6, 13].
The purpose of this scoping review is to provide an overview of studies involving methods of assessment of muscle tone in patients with neurological disorders, using both objective or subjective methods. Our main goal is to highlight potential trends, as there is currently no gold standard in tonus evaluation either in clinical practice or in the literature.
2 Methods
2.1 Protocol and Registration
This review was registered in OSF (https://doi.org/10.17605/OSF.IO/BCRHT). A scoping review was performed on studies that evaluated the muscle tone, following the methodological recommendations of the Joanna Briggs Institute for systematic scoping reviews [14]. The target population was composed of individuals with neurological disorders, and eligibility factors were defined and adopted to find potential studies in literature. Studies were considered eligible if they met the following inclusion criteria:
-
(1)
Participants of the study presented neurological and/or muscular disorders (with no limitations based on age or sex).
-
(2)
The study was composed of randomized clinical trials, randomized controlled trials, clinical trials, case reports, or cohort study.
-
(3)
The study evaluated tonus using either subjective or objective methods.
-
(4)
Full text written in English.
Exclusion criteria were:
-
(1)
The research was conducted with healthy participants in an experimental group.
-
(2)
Text written in another language (other than English).
-
(3)
The study was a meta-analysis or systematic review.
-
(4)
Ongoing research (poster, patent, chapter of book).
2.2 Search Strategy
The search was conducted without any time restriction, ranging from the creation of each database to March 2021. We systematically searched electronic databases or potentially relevant papers, including PubMed, IEEE/IET Electronic Library, Web of Science, Scopus, Cochrane, Bireme, Google Scholar, and Science Direct.
The following search query was used in all databases: [(muscle OR muscular) AND (tonus OR tension OR Tightness OR Stiffness)] AND (evaluation OR assessment OR estimation) AND [(neurological AND (disorder OR disturb)] OR (central AND nervous AND system AND injury) OR (peripheral AND nervous AND system AND injury).
2.3 Study Selection
After executing the search strategy in each of the databases, the list of eligible works was extracted and organized in Rayyan – Intelligent Systematic Review [15]. At first, studies were filtered based on the reading of title and abstract. The studies that passed the first filtering process were read in full; those that met the criteria were included for analysis in this review.
Subsequent data extraction and quality assessment of the selected studies were performed, in which title, abstract, and full text of each paper were reviewed by 2 reviewers (AR and CA). Disagreements between reviewers were resolved by consensus. If no consensus could be reached, a third reviewer (EN) would decide. All the reviews checked if the studies included were consistent and followed the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) flow diagram [16].
2.4 Data Analyses
Several characteristics were extracted from each of the selected studies, to be subsequently organized and formatted into a table for better visualization of the results. The characteristics extracted were the following: type of study, year of publication, sample size, type of neurological disorder, type of assessment used, method to assess muscle tone and assessed limb.
3 Results
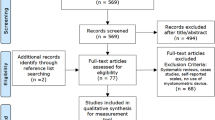
The flow diagram of the filtering process applied to all databases is presented in Fig. 1. After eliminating duplicate works, we found 1369 studies from 8 different databases. Those 1369 underwent a screening process by title and abstract, and 1208 studies were excluded for not meeting the eligibility criteria. In the third step, the 161 remaining studies were fully read, resulting in a total of 84 studies included in this scope review. The other 77 papers were excluded for different reasons: 19 were studies that evaluated muscle tone of healthy individuals rather than individuals with disability; 48 studies did not assess tonus; and 10 papers were reviews, and therefore should not be included.
As stated previously, the characteristics of each study (such as type of tonus and assessment tool) are summarized on Table 1. All three types of muscle tone (hypotonia, spasticity, and rigidity) were discussed among the studies selected. Clinical trials were executed to evaluate the proposed methods in the aforementioned studies with individuals with impairment in the muscle tone, with several methods of tonus evaluation explored in those studies. The Modified Ashworth Scale (MAS), an ordinal measure created to evaluate the resistance to passive movement, was the most commonly applied method among the selected studies, being used in 42 out of 84 works. Another method of assessment often used incorporated quantitative devices (7 out of 84 studies) to evaluate the muscle tonus. Lastly, electromyography is an alternative method (used in a total of 7 studies), where the tonus is measured by muscle activity.
The least type of tonus impairment found in the literature was hypotonia, discussed in only 2 of the 84 studies [17, 18]. Both studies assessed upper limb condition of individuals. In [17], the tonus was measured using an electro-mechanical machine, and [18] used the method of resistance to passive movement to assess participants. On the other hand, spasticity was the most common tonus impairment assessed, mainly due to the existence of the MAS, which is specifically designed for assessment of this impairment, and is widely known and applied in clinical settings.
Table 2 displays the main muscles and the type of tonus impairment focused in the assessment. Regarding the muscles evaluated, the most presented were flexors and extensors of the knee joint (29 studies) and elbow (26 studies), as they are most often affected by disorders while also having the greatest impact on the quality of life.
Regarding the distribution of studies across databases, Fig. 2 shows that the majority of studies (approximately 63.9%) were taken from the PubMed database. The remainder of the studies were selected from the other databases, with Scopus, Web of Science and Bireme being those presenting the most expressive percentages.
Figure 3 presents the number of studies included in this scoping review per decade. Most of the studies recovered were published between 2011 and 2020 (approximately 55.3%). This suggests an increase of over 100% of papers researching the subject in the last decade in comparison with the previous, indicating the growing interest in assessment of muscle tone for individuals with neurological disorders.
The papers were also divided between subjective and objective methods. Subjective methods depend on the experience of the therapist responsible for conducting the evaluation, such as movement resistance scales, including MAS [8] and Tardieu scale [70]. Objective methods use quantitative metrics for analysis of the data obtained during assessment, resulting in a numerical value that indicates the degree of muscle tone, without requiring the interpretation of subjective factors such as therapist expertise. One example of an objective method is using electromyography sensors [61] to assess muscle tone based on the muscle activity. Categorizing the papers in subjective and objective methods revealed that the percentage of types of methods used were very similar, with 48.2% of studies using subjective methods and 51.8% using objective methods.
4 Discussion
This scoping review included 84 studies investigating different methods for tonus evaluation. The studies selected addressed all types of muscle tone conditions (hypotonia, normal, spasticity, and hypertonia), along with several types of tonus assessment methods. A majority of the studies used clinical tools or subjective methods to provide assessment to muscle tone. However, most recent studies have shifted interest to using new tools and quantitative measurements. Only two of the studies eligible in the review investigated hypotonia [17, 18]. Consequently, the vast majority of studies found evaluated either spasticity or rigidity. As established in the eligibility screening, all papers selected applied their respective tonus assessment methods in individuals with tonus impairment through clinical trials.
Hypotonia is a condition originating from different health problems, many of which are inherited. It can occur sometimes in individuals with cerebral palsy (CP). For the two studies evaluating hypotonia, the causes of the impairment were cerebellar ataxia [17] and Williams syndrome [18]. However, none of the papers evaluated hypotonia due to peripheral neuropathies and polyneuropathies. This demonstrates the inexistence of methods to assess this type of muscle tone condition.
Hypertonia encompasses both spasticity and rigidity, the first being dependent on the stretching speed [72]. The assessment of hypertonia is critical in clinical diagnosis, as this tonus disorder affects millions of people around the world [94]. The majority of assessment methods for muscle tone are based on scales such as MAS and Tardieu scale. However, some of the papers also discussed devices using quantitative metrics, showing an increasing search for more reliable means of measurement.
Muscle tone may change when individuals are affected by disorders that impact the CNS. Stroke, for example, is one of the leading causes of death [95], second only to cardiac diseases. In the papers selected for this review, 53 evaluated the muscle tone of post-stroke patients. Stroke is the leading cause of acquired physical disability in adults worldwide [96], including changes in muscle tone, which explains the number of researches involving post-stroke individuals.
The most reported method for assessing muscle tonus in post-stroke patients was MAS. This scale was originally developed for the evaluation of spasticity in post-stroke, although it can be used in other disorders. For example, the MAS can be used alongside Tardieu scale [24] to assess muscle tone condition in patients with cerebral palsy. Most of the studies found concerning Parkinson’s disease used quantitative devices or the UPDRS (Unified Parkinson's Disease Rating Scale) to assess the muscle tone of the patients. The UPDRS is a scale developed to be used in combination with other assessment tools to identify disabilities and impairment related to PD, including abnormal muscle tone [97].
Some studies also used the subjective scales (such as MAS and the UPDRS) in combination with quantitative metrics of assessment. Robotic devices have been developed [11, 43], and other authors have used electronic devices [80, 83]. The combination of the two methods demonstrates the importance of validating these new devices in clinical practice, as well as the lack of quantitative devices able to perform this specific function.
The goal of the quantitative devices used to assess muscle stiffness in patients with PD is to obtain higher reliability [79, 80, 82,83,84]. Sensors such as force transducers [84] and gyroscopes [80] can increase the accuracy of those devices. The studies reported positive outcomes using sensor technology, which reinforces the importance of investing in the development of these technologies using objective metrics of the muscle tone; this can only result in more reliable and efficient evaluations [98, 99]. Despite the increased efforts, the vast majority of researchers are exploring solutions to be applied in spasticity and rigidity measurement, and few for hypotonia.
Subjective assessment methods are still used in most studies selected for the scoping review. The main reasons behind the use of such tools can be attributed to their simplicity, making them useful and accessible in a clinical practice. However, it is also a cause for criticism, since the accuracy of such methods depends entirely on precise clinical observations [100]. As a result, a big challenge is developing devices that are easily accessible in clinical practice, while being able to demonstrate accurate results [94].
The findings of this scoping review support the idea of developing a method to assess more than one type of muscle tone. As seen previously in the study of [101], the muscle tone of an individual can be altered under certain conditions. For example, a patient suffering a stroke may present more than one tonus impairment during the recovery and rehabilitation period, displaying characteristics attributed to hypotonic individuals during the acute phase of stroke and becoming a spastic individual during the chronic phase of the stroke. Using a single method, it would be possible to analyze such changes more reliably, differently from what is currently possible using ordinal measurements.
While the studies applied assessment methods in different parts of the body, they focused on the upper and lower limbs, which are usually the most affected by any change in tonus [94]. For instance, the muscles of the wrist are some of the most affected by the rigidity caused by Parkinson’s disease [79]. However, assessment of the condition of those muscles was conducted in very few of the works found in the literature. In fact, research and development of methods that can be simultaneously applied in different muscles is of great importance, since more information regarding a patient's condition can be obtained from analyzing tonus in more than one muscle group.
4.1 Main Limitations and Clinical Implications
Although this scoping review was conducted following a well-defined protocol, ensuring that the research guidelines were carefully followed, there are limitations inherent to the search parameters and eligibility criteria established. The scoping review provides an overview of the existing evidence regardless of quality. This particular review was limited to studies written in English; as such, potential relevant research reported in other languages were not included in its results.
The assessment of muscle tone in clinical practice bears significance in the creation of new therapies and monitoring of patients with any neurological disorder. The studies included in this scoping review revealed the existing gap generated by the lack of methods capable of evaluating hypotonia, as well as the growth of studies focused in spasticity and rigidity evaluation methods in the last decade.
5 Conclusion
Findings of this study indicated a growing interest in research in the field of assessment of tonus. However, the main focus of such research remains in spasticity and rigidity, resulting in an absence of studies involving hypotonia. The scoping review also highlighted the lack of quantitative methods capable of assessing tonus from several muscles simultaneously and in real-time, since none of the selected papers discussed methods able to perform the assessment of more than one type of tonus, which reinforces the necessity of alternative methods of evaluation.
References
Khonsary, S. (2017). Guyton and Hall: Textbook of medical physiology. Surgical Neurology International. https://doi.org/10.4103/sni.sni_327_17
Maher, A. B. (2016). Neurological assessment. International Journal of Orthopaedic and Trauma Nursing. https://doi.org/10.1016/j.ijotn.2016.01.002
Harris, S. R. (2008). Congenital hypotonia: Clinical and developmental assessment. Developmental Medicine & Child Neurology. https://doi.org/10.1111/j.1469-8749.2008.03097.x
Mayer, N. H., Esquenazi, A., & Childers, M. K. (1997). Common patterns of clinical motor dysfunction. Muscle and Nerve. https://doi.org/10.1002/(SICI)1097-4598(1997)6+%3c21::AID-MUS4%3e3.0.CO;2-L
Martino, D., Espay, A. J., Fasano, A., Morgante, F., Martino, D., Espay, A. J., Fasano, A., & Morgante, F. (2016). Abnormalities of muscle tone. Disorders of Movement (pp. 49–95). Springer.
Marusiak, J., Kisiel-Sajewicz, K., Jaskólska, A., & Jaskólski, A. (2010). Higher muscle passive stiffness in Parkinson’s disease patients than in controls measured by myotonometry. Archives of Physical Medicine and Rehabilitation. https://doi.org/10.1016/j.apmr.2010.01.012
Ward, A. B. (2000). Assessment of muscle tone. Age and Ageing. https://doi.org/10.1093/ageing/29.5.385
Hameau, S., Bensmail, D., Robertson, J., Boudarham, J., Roche, N., & Zory, R. (2014). Isokinetic assessment of the effects of botulinum toxin injection on spasticity and voluntary strength in patients with spastic hemiparesis. European Journal of Physical and Rehabilitation Medicine, 50(5), 515–523.
Woolacott, A. J., & Burne, J. A. (2006). The tonic stretch reflex and spastic hypertonia after spinal cord injury. Experimental Brain Research, 174, 386–396. https://doi.org/10.1007/s00221-006-0478-7
Bar-On, L., Molenaers, G., Aertbeliën, E., Monari, D., Feys, H., & Desloovere, K. (2014). The relation between spasticity and muscle behavior during the swing phase of gait in children with cerebral palsy. Research in Developmental Disabilities. https://doi.org/10.1016/j.ridd.2014.07.053
Posteraro, F., Crea, S., Mazzoleni, S., Bert Eanu, M., Ciobanu, I., Vitiello, N., Cempini, M., Gervasio, S., & Mrachacz-Kersting, N. (2018). Technologically-advanced assessment of upper-limb spasticity: A pilot study. European Journal of Physical and Rehabilitation Medicine, 54, 536–544. https://doi.org/10.23736/S1973-9087.17.04815-8
Peng, Q., Park, H. S., Shah, P., Wilson, N., Ren, Y., Wu, Y. N., Liu, J., Gaebler-Spira, D. J., & Zhang, L. Q. (2011). Quantitative evaluations of ankle spasticity and stiffness in neurological disorders using manual spasticity evaluator. Journal of Rehabilitation Research and Development. https://doi.org/10.1682/JRRD.2010.04.0053
Pilla, A., Trigili, E., McKinney, Z., Fanciullacci, C., Malasoma, C., Posteraro, F., Crea, S., & Vitiello, N. (2020). Robotic rehabilitation and multimodal instrumented assessment of post-stroke elbow motor functions—A randomized controlled trial protocol. Frontiers in Neurology. https://doi.org/10.3389/fneur.2020.587293
Peters, M. D. J., Godfrey, C. M., Khalil, H., McInerney, P., Parker, D., & Soares, C. B. (2015). Guidance for conducting systematic scoping reviews. International Journal of Evidence-Based Healthcare. https://doi.org/10.1097/XEB.0000000000000050
Ouzzani, M., Hammady, H., Fedorowicz, Z., & Elmagarmid, A. (2016). Rayyan-a web and mobile app for systematic reviews. Systematic Reviews. https://doi.org/10.1186/s13643-016-0384-4
Page, M. J., McKenzie, J. E., Bossuyt, P. M., Boutron, I., Hoffmann, T. C., Mulrow, C. D., Shamseer, L., Tetzlaff, J. M., Akl, E. A., Brennan, S. E., Chou, R., Glanville, J., Grimshaw, J. M., Hróbjartsson, A., Lalu, M. M., Li, T., Loder, E. W., Mayo-Wilson, E., McDonald, S., … Moher, D. (2021). The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ. https://doi.org/10.1136/bmj.n71
Iloeje, S. O. (1994). Measurement of muscle tone in children with cerebellar ataxia. East African Medical Journal, 71, 256–260.
Chapman, C. A., Du Plessis, A., & Pober, B. R. (1996). Neurologic findings in children and adults with Williams syndrome. Journal of Child Neurology, 11, 63–65. https://doi.org/10.1177/088307389601100116
Alcantara, C. C., Blanco, J., De Oliveira, L. M., Ribeiro, P. F. S., Herrera, E., Nakagawa, T. H., Reisman, D. S., Michaelsen, S. M., Garcia, L. C., & Russo, T. L. (2019). Cryotherapy reduces muscle hypertonia, but does not affect lower limb strength or gait kinematics post-stroke: A randomized controlled crossover study. Topics in Stroke Rehabilitation, 26, 267–280. https://doi.org/10.1080/10749357.2019.1593613
Bian, R., Luo, Z., Huang, X., Wang, R., Song, R., Wong, S., Li, L. (2019). Relationship between Passive Stretch Resistance in spastic wrist Flexors and Clinical Scales of Stroke Survivors: A Cross-sectional Study. In ICARM 2018–2018 3rd International Conference on Advanced Robotics and Mechatronics. Institute of Electrical and Electronics Engineers Inc., pp 136–139
Bleyenheuft, C., Cockx, S., Caty, G., Stoquart, G., Lejeune, T., & Detrembleur, C. (2009). The effect of botulinum toxin injections on gait control in spastic stroke patients presenting with a stiff-knee gait. Gait & Posture, 30, 168–172. https://doi.org/10.1016/j.gaitpost.2009.04.003
Booth, C. M., Cortina-Borja, M. J. F., & Theologis, T. N. (2001). Collagen accumulation in muscles of children with cerebral palsy and correlation with severity of spasticity. Developmental Medicine and Child Neurology, 43, 314. https://doi.org/10.1017/s0012162201000597
Boudarham, J., Hameau, S., Pradon, D., Bensmail, D., Roche, N., & Zory, R. (2013). Changes in electromyographic activity after botulinum toxin injection of the rectus femoris in patients with hemiparesis walking with a stiff-knee gait. Journal of Electromyography and Kinesiology, 23, 1036–1043. https://doi.org/10.1016/j.jelekin.2013.07.002
Dreher, T., Brunner, R., Vegvari, D., Heitzmann, D., Gantz, S., Maier, M. W., Braatz, F., & Wolf, S. I. (2013). The effects of muscle-tendon surgery on dynamic electromyographic patterns and muscle tone in children with cerebral palsy. Gait & Posture, 38, 215–220. https://doi.org/10.1016/j.gaitpost.2012.11.013
Flansbjer, U. B., Lexell, J., & Brogårdh, C. (2012). Long-term benefits of progressive resistance training in chronic stroke: A 4-year follow-up. Journal of Rehabilitation Medicine, 44, 218–221.
Gao, J., He, W., Du, L. J., Chen, J., Park, D., Wells, M., Fowlkes, B., & O’Dell, M. (2018). Quantitative ultrasound imaging to assess the biceps Brachii muscle in chronic post-stroke spasticity: Preliminary observation. Ultrasound in Medicine and Biology, 44, 1931–1940. https://doi.org/10.1016/j.ultrasmedbio.2017.12.012
Gekht, A. B., Burd, G. S., Selikhova, M. V., Iaish, F., & Beliakov, V. V. (1998). Disorders of muscle tonus and their treatment with sirdalud in patients in the early recovery period of ischemic stroke. Zhurnal Nevrol i psikhiatrii Im SS Korsakova, 98, 22–229.
Given, J. D., Dewald, J. P. A., & Rymer, W. Z. (1995). Joint dependent passive stiffness in paretic and contralateral limbs of spastic patients with hemiparetic stroke. Journal of Neurology, Neurosurgery and Psychiatry, 59, 271–279.
Gracies, J. M., O’Dell, M., Vecchio, M., Hedera, P., Kocer, S., Rudzinska-Bar, M., Rubin, B., Timerbaeva, S. L., Lusakowska, A., Boyer, F. C., Grandoulier, A. S., Vilain, C., & Picaut, P. (2018). Effects of repeated abobotulinumtoxinA injections in upper limb spasticity. Muscle and Nerve, 57, 245–254. https://doi.org/10.1002/mus.25721
Gregson, J. M., Leathley, M., Moore, A. P., Sharma, A. K., Smith, T. L., & Watkins, C. L. (1999). Reliability of the tone assessment scale and the modified Ashworth scale as clinical tools for assessing poststroke spasticity. Archives of Physical Medicine and Rehabilitation, 80, 1013–1016. https://doi.org/10.1016/S0003-9993(99)90053-9
Gross, R., Delporte, L., Arsenault, L., Revol, P., Lefevre, M., Clevenot, D., Boisson, D., Mertens, P., Rossetti, Y., & Luauté, J. (2014). Does the rectus femoris nerve block improve knee recurvatum in adult stroke patients? A kinematic and electromyographic study. Gait & Posture, 39, 761–766. https://doi.org/10.1016/j.gaitpost.2013.10.008
Gupta, M., Lal Rajak, B., Bhatia, D., & Mukherjee, A. (2016). Effect of r-TMS over standard therapy in decreasing muscle tone of spastic cerebral palsy patients. Journal of Medical Engineering & Technology, 40, 210–216. https://doi.org/10.3109/03091902.2016.1161854
Hesse, S., Bertelt, C., Jahnke, M. T., Schaffrin, A. P. T., Baake, P. P. T., Malezic, M., & Mauritz, K. H. (1995). Treadmill training with partial body weight support compared with physiotherapy in nonambulatory hemiparetic patients. Stroke, 26, 976–981. https://doi.org/10.1161/01.STR.26.6.976
Hesse, S., Reiter, F., Konrad, M., & Jahnke, M. T. (1998). Botulinum toxin type A and short-term electrical stimulation in the treatment of upper limb flexor spasticity after stroke: A randomized, double-blind, placebo-controlled trial. Clinical Rehabilitation, 12, 381–388. https://doi.org/10.1191/026921598668275996
Hösl, M., Böhm, H., Eck, J., Döderlein, L., & Arampatzis, A. (2018). Effects of backward-downhill treadmill training versus manual static plantarflexor stretching on muscle-joint pathology and function in children with spastic Cerebral Palsy. Gait & Posture, 65, 121–128. https://doi.org/10.1016/j.gaitpost.2018.07.171
Huang, H. W., Ju, M. S., & Lin, C. C. K. (2016). Flexor and extensor muscle tone evaluated using the quantitative pendulum test in stroke and parkinsonian patients. Journal of Clinical Neuroscience, 27, 48–52. https://doi.org/10.1016/j.jocn.2015.07.031
Kofler, M., Quirbach, E., Schauer, R., Singer, M., & Saltuari, L. (2009). Limitations of intrathecal baclofen for spastic hemiparesis following stroke. Neurorehabilitation and Neural Repair, 23, 26–31. https://doi.org/10.1177/1545968308317700
Krukowska, J., Dalewski, M., & Czernicki, J. (2014). Evaluation of effectiveness of local cryotherapy in patients with post-stroke spasticity. Wiadomosci Lekarskie, 67, 71–75.
Kumru, H., Stetkarova, I., Schindler, C., Vidal, J., & Kofler, M. (2011). Neurophysiological evidence for muscle tone reduction by intrathecal baclofen at the brainstem level. Clinical Neurophysiology, 122, 1229–1237. https://doi.org/10.1016/j.clinph.2010.09.010
Leonard, C. T., Gardipee, K. A., Koontz, J. R., Anderson, J. H., & Wilkins, S. A. (2006). Correlation between impairment and motor performance during reaching tasks in subjects with spastic hemiparesis. Journal of Rehabilitation Medicine, 38, 243–249. https://doi.org/10.1080/16501970600609808
Leonard, C. T., Stephens, J. U., & Stroppel, S. L. (2001). Assessing the spastic condition of individuals with upper motoneuron involvement: Validity of the myotonometer. Archives of Physical Medicine and Rehabilitation, 82, 1416–1420. https://doi.org/10.1053/apmr.2001.26070
Lindberg, P. G., Gäverth, J., Islam, M., Fagergren, A., Borg, J., & Forssberg, H. (2011). Validation of a new biomechanical model to measure muscle tone in spastic muscles. Neurorehabilitation and Neural Repair, 25, 617–625. https://doi.org/10.1177/1545968311403494
Lünenburger, L., Colombo, G., Riener, R., Dietz, V. (2005). Clinical assessments performed during robotic rehabilitation by the gait training robot Lokomat. In Proceedings of the 2005 IEEE 9th International Conference on Rehabilitation Robotics. pp 345–348
Mancini, F., Sandrini, G., Moglia, A., Nappi, G., & Pacchetti, C. (2005). A randomised, double-blind, dose-ranging study to evaluate efficacy and safety of three doses of botulinum toxin type A (Botox) for the treatment of spastic foot. Neurological Sciences, 26, 26–31. https://doi.org/10.1007/s10072-005-0378-9
McGibbon, C. A., Sexton, A., Hughes, G., Wilson, A., Jones, M., O’Connell, C., Parker, K., Adans-Dester, C., O’Brien, A., & Bonato, P. (2018). Evaluation of a toolkit for standardizing clinical measures of muscle tone. Physiological Measurement. https://doi.org/10.1088/1361-6579/aad424
Mirek, E., Opoka, K., Kozioł, K., Filip, M., Pasiut, S., Szymura, J., Legwant, A., Wasielewska, A., Michalski, M., & Tomaszewski, T. (2017). Analysis of the effects of the upper limb improvement programme in patients after ischemic stroke treated with botulinum toxin. Rehabilitation Medicine, 21, 58–63.
Najafi, Z., Rezaeitalab, F., Yaghubi, M., & Manzari, Z. S. (2018). The effect of biofeedback on the motor—Muscular situation in rehabilitation of stroke patients. Journal of Caring Sciences, 7, 89–93. https://doi.org/10.15171/jcs.2018.014
Park, G. Y., & Kwon, D. R. (2012). Sonoelastographic evaluation of medial gastrocnemius muscles intrinsic stiffness after rehabilitation therapy with botulinum toxin a injection in spastic cerebral palsy. Archives of Physical Medicine and Rehabilitation. https://doi.org/10.1016/j.apmr.2012.06.024
Sajedi, F., Alizad, V., Alaeddini, F., Fatemi, R., & Mazaherinezhad, A. (2008). The effect of adding homeopathic treatment to rehabilitation on muscle tone of children with spastic cerebral palsy. Complementary Therapies in Clinical Practice, 14, 33–37. https://doi.org/10.1016/j.ctcp.2007.11.001
Slawek, J., Bogucki, A., & Reclawowicz, D. (2005). Botulinum toxin type A for upper limb spasticity following stroke: An open-label study with individualised, flexible injection regimens. Neurological Sciences, 26, 32–39. https://doi.org/10.1007/s10072-005-0379-8
Sommerfeld, D. K., Eek, E. U. B., Svensson, A. K., Holmqvist, L. W., & Von Arbin, M. H. (2004). Spasticity after stroke: its occurrence and association with motor impairments and activity limitations. Stroke, 35, 134–139. https://doi.org/10.1161/01.STR.0000105386.05173.5E
Supiot, A., Geiger, M., Bensmail, D., Aegerter, P., Pradon, D., & Roche, N. (2018). Effect of botulinum toxin injection on length and force of the rectus femoris and triceps surae muscles during locomotion in patients with chronic hemiparesis (FOLOTOX). BMC Neurology. https://doi.org/10.1186/s12883-018-1110-8
Tok, F., Balaban, B., Yaşar, E., Alaca, R., & Tan, A. K. (2012). The effects of onabotulinum toxin A injection into rectus femoris muscle in hemiplegic stroke patients with stiff-knee gait: A placebo-controlled, nonrandomized trial. American Journal of Physical Medicine and Rehabilitation, 91, 321–326. https://doi.org/10.1097/PHM.0b013e3182465feb
Vola, E. A., Albano, M., Di Luise, C., Servodidio, V., Sansone, M., Russo, S., Corrado, B., Servodio Iammarrone, C., Caprio, M. G., & Vallone, G. (2018). Use of ultrasound shear wave to measure muscle stiffness in children with cerebral palsy. Journal of Ultrasound, 21, 241–247. https://doi.org/10.1007/s40477-018-0313-6
Wang, Y. J., & Gao, B. Q. (2013). Efficacy and safety of serial injections of botulinum toxin A in children with spastic cerebral palsy. World Journal of Pediatrics, 9, 342–345. https://doi.org/10.1007/s12519-013-0442-0
Waninge, A., Rook, R. A., Dijkhuizen, A., Gielen, E., & van der Schans, C. P. (2011). Feasibility, test-retest reliability, and interrater reliability of the Modified Ashworth Scale and Modified Tardieu Scale in persons with profound intellectual and multiple disabilities. Research in Developmental Disabilities, 32, 613–620. https://doi.org/10.1016/j.ridd.2010.12.013
Wu, C. H., Ho, Y. C., Hsiao, M. Y., Chen, W. S., & Wang, T. G. (2017). Evaluation of post-stroke spastic muscle stiffness using shear wave ultrasound elastography. Ultrasound in Medicine and Biology, 43, 1105–1111. https://doi.org/10.1016/j.ultrasmedbio.2016.12.008
Andrews, C. J., Neilson, P., & Knowles, L. (1973). Electromyographic study of the rigidospasticity of athetosis. Journal of Neurology, Neurosurgery and Psychiatry, 36, 94–103. https://doi.org/10.1136/jnnp.36.1.94
Hesse, S., Krajnik, J., Luecke, D., Jahnke, M. T., Gregoric, M., & Mauritz, K. H. (1996). Ankle muscle activity before and after botulinum toxin therapy for lower limb extensor spasticity in chronic hemiparetic patients. Stroke, 27, 455–460. https://doi.org/10.1161/01.STR.27.3.455
Lamontagne, A., Malouin, F., Richards, C. L., & Dumas, F. (2002). Mechanisms of disturbed motor control in ankle weakness during gait after stroke. Gait & Posture, 15, 244–255. https://doi.org/10.1016/S0966-6362(01)00190-4
Lin, P. Y., Yang, Y. R., Cheng, S. J., & Wang, R. Y. (2006). The relation between ankle impairments and gait velocity and symmetry in people with stroke. Archives of Physical Medicine and Rehabilitation, 87, 562–568. https://doi.org/10.1016/j.apmr.2005.12.042
Qi, Y., & Shen, J. (2016). Acupuncture combined with rehabilitation training for foot drop after stroke. Zhongguo Zhen Jiu, 36, 679–682. https://doi.org/10.13703/j.0255-2930.2016.07.002
Yamaguchi, T., Hvass Petersen, T., Kirk, H., Forman, C., Svane, C., Kofoed-Hansen, M., Boesen, F., & Lorentzen, J. (2018). Spasticity in adults with cerebral palsy and multiple sclerosis measured by objective clinically applicable technique. Clinical Neurophysiology, 129, 2010–2021. https://doi.org/10.1016/j.clinph.2018.07.004
Caty, G. D., Detrembleur, C., Bleyenheuft, C., Deltombe, T., & Lejeune, T. M. (2008). Effect of simultaneous botulinum toxin injections into several muscles on impairment, activity, participation, and quality of life among stroke patients presenting with a stiff knee gait. Stroke, 39, 2803–2808. https://doi.org/10.1161/STROKEAHA.108.516153
Gross, R., Robertson, J., Leboeuf, F., Hamel, O., Brochard, S., & Perrouin-Verbe, B. (2017). Neurotomy of the rectus femoris nerve: Short-term effectiveness for spastic stiff knee gait: Clinical assessment and quantitative gait analysis. Gait & Posture, 52, 251–257. https://doi.org/10.1016/j.gaitpost.2016.11.032
Ochs, G., Struppler, A., Meyerson, B. A., Linderoth, B., Gybels, J., Gardner, B. P., Teddy, P., Jamous, A., & Weinmann, P. (1989). Intrathecal baclofen for long-term treatment of spasticity: A multi-centre study. Journal of Neurology, Neurosurgery and Psychiatry, 52, 933–939. https://doi.org/10.1136/jnnp.52.8.933
Więch, P., Ćwirlej-Sozańska, A., Wiśniowska-Szurlej, A., Kilian, J., Lenart-Domka, E., Bejer, A., Domka-Jopek, E., Sozański, B., & Korczowski, B. (2020). The relationship between body composition and muscle tone in children with cerebral palsy: A case-control study. Nutrients. https://doi.org/10.3390/nu12030864
Brown, R. A., Lawson, D. A., Leslie, G. C., & Part, N. J. (1988). Observations on the applicability of the Wartenberg pendulum test to healthy, elderly subjects. Journal of Neurology, Neurosurgery and Psychiatry, 51, 1171–1177. https://doi.org/10.1136/jnnp.51.9.1171
Lin, D. C., & Rymer, W. Z. (1991). A quantitative analysis of pendular motion of the lower leg in spastic human subjects. IEEE Transactions on Biomedical Engineering, 38, 906–918. https://doi.org/10.1109/10.83611
Bernuz, B., Genet, F., Terrat, P., Pradon, D., Barbot, F., Bussel, B., & Bensmail, D. (2012). Botulinum toxin effect on voluntary and stretch reflex-related torque produced by the quadriceps: An isokinetic pilot study. Neurorehabilitation and Neural Repair, 26, 542–547. https://doi.org/10.1177/1545968311423668
Glass, A., & Hannah, A. (1974). A comparison of dantrolene sodium and diazepam in the treatment of spasticity. Paraplegia, 12, 170–174. https://doi.org/10.1038/sc.1974.27
Powers, R. K., Marder-Meyer, J., & Rymer, W. Z. (1988). Quantitative relations between hypertonia and stretch reflex threshold in spastic hemiparesis. Annals of Neurology, 23, 115–124. https://doi.org/10.1002/ana.410230203
Kamper, D. G., Fischer, H. C., Cruz, E. G., & Rymer, W. Z. (2006). Weakness Is the primary contributor to finger impairment in chronic stroke. Archives of Physical Medicine and Rehabilitation, 87, 1262–1269. https://doi.org/10.1016/j.apmr.2006.05.013
Marsden, J., Stevenson, V., McFadden, C., Swain, I., & Taylor, P. (2013). The effects of functional electrical stimulation on walking in hereditary and spontaneous spastic paraparesis. Neuromodulation, 16, 256–260. https://doi.org/10.1111/j.1525-1403.2012.00494.x
Du, L.-J., He, W., Cheng, L.-G., Li, S., Pan, Y.-S., & Gao, J. (2016). Ultrasound shear wave elastography in assessment of muscle stiffness in patients with Parkinson’s disease: A primary observation. Clinical Imaging, 40, 1075–1080. https://doi.org/10.1016/j.clinimag.2016.05.008
Gao, J., Du, L. J., He, W., Li, S., & Cheng, L. G. (2016). Ultrasound strain elastography in assessment of muscle stiffness in acute levodopa challenge test: A feasibility study. Ultrasound in Medicine and Biology, 42, 1084–1089. https://doi.org/10.1016/j.ultrasmedbio.2015.12.014
Gao, J., He, W., Du, L. J., Li, S., Cheng, L. G., Shih, G., & Rubin, J. (2016). Ultrasound strain elastography in assessment of resting biceps brachii muscle stiffness in patients with Parkinson’s disease: A primary observation. Clinical Imaging, 40, 440–444. https://doi.org/10.1016/j.clinimag.2015.12.008
Martínez-Payá, J. J., del Baño-Aledo, M. E., Ríos-Díaz, J., Fornés-Ferrer, V., & Vázquez-Costa, J. F. (2018). Sonoelastography for the assessment of muscle changes in amyotrophic lateral sclerosis: Results of a pilot study. Ultrasound in Medicine and Biology, 44, 2540–2547. https://doi.org/10.1016/j.ultrasmedbio.2018.08.009
Caligiuri, M. P. (1994). Portable device for quantifying parkinsonian wrist rigidity. Movement Disorders, 9, 57–63. https://doi.org/10.1002/mds.870090109
Caligiuri, M. P., Bracha, H. S., & Lohr, J. B. (1989). Asymmetry of neuroleptic-induced rigidity: Development of quantitative methods and clinical correlates. Psychiatry Research, 30, 275–284. https://doi.org/10.1016/0165-1781(89)90019-X
Hömberg, V., & Huttunen, J. (1994). Muscle tone in Huntington’s disease. Journal of the Neurological Sciences, 121, 147–154. https://doi.org/10.1016/0022-510X(94)90343-3
Marusiak, J., Jaskólska, A., Budrewicz, S., Koszewicz, M., & Jaskólski, A. (2011). Increased muscle belly and tendon stiffness in patients with Parkinson’s disease, as measured by myotonometry. Movement Disorders, 26, 2119–2122. https://doi.org/10.1002/mds.23841
Patrick, S. K., Denington, A. A., Gauthier, M. J. A., Gillard, D. M., & Prochazka, A. (2001). Quantification of the UPDRS rigidity scale. IEEE Transactions on Neural Systems and Rehabilitation Engineering, 9, 31–41. https://doi.org/10.1109/7333.918274
Perera, T., Lee, W. L., Jones, M., Tan, J. L., Proud, E. L., Begg, A., Sinclair, N. C., Peppard, R., & McDermott, H. J. (2019). A palm-worn device to quantify rigidity in Parkinson’s disease. Journal of Neuroscience Methods, 317, 113–120. https://doi.org/10.1016/j.jneumeth.2019.02.006
Marusiak, J., Jaskólska, A., Koszewicz, M., Budrewicz, S., & Jaskólski, A. (2012). Myometry revealed medication-induced decrease in resting skeletal muscle stiffness in Parkinson’s disease patients. Clinical Biomechanics. https://doi.org/10.1016/j.clinbiomech.2012.02.001
Rätsep, T., & Asser, T. (2017). The effect of subthalamic stimulation on viscoelastic stiffness of skeletal muscles in patients with Parkinson’s disease. Clinical Biomechanics, 44, 94–98. https://doi.org/10.1016/j.clinbiomech.2017.03.012
Burleigh, A., Horak, F., Nutt, J., & Frank, J. (1995). Levodopa Reduces muscle tone and lower extremity tremor in Parkinson’s disease. Canadian Journal of Neurological Sciences/Journal Canadien des Sciences Neurologiques, 22, 280–285. https://doi.org/10.1017/S0317167100039470
Tanigawa, A., Komiyama, A., & Hasegawa, O. (1998). Truncal muscle tonus in progressive supranuclear palsy. Journal of Neurology, Neurosurgery and Psychiatry, 64, 190–196. https://doi.org/10.1136/jnnp.64.2.190
Kirk, H., Geertsen, S. S., Lorentzen, J., Krarup, K. B., Bandholm, T., & Nielsen, J. B. (2016). Explosive resistance training increases rate of force development in ankle dorsiflexors and gait function in adults with cerebral palsy. Journal of Strength and Conditioning Research, 30, 2749–2760. https://doi.org/10.1519/JSC.0000000000001376
Lee, Y. Y., Lin, K. C., Wu, C. Y., Liao, C. H., Lin, J. C., & Chen, C. L. (2015). Combining afferent stimulation and mirror therapy for improving muscular, sensorimotor, and daily functions after chronic stroke: A randomized, placebo-controlled study. American Journal of Physical Medicine and Rehabilitation, 94, 859–868. https://doi.org/10.1097/PHM.0000000000000271
Brown, R. A., Lawson, D. A., Leslie, G. C., Macarthur, A., Maclennan, W. J., Mcmurdo, M. E. T., Mutch, W. J., & Part, N. J. (1988). Does the Wartenberg pendulum test differentiate quantitatively between spasticity and rigidity? A study in elderly stroke and Parkinsonian patients. Journal of Neurology, Neurosurgery and Psychiatry, 51, 1178–1186. https://doi.org/10.1136/jnnp.51.9.1178
Dalakas, M. C., Rakocevic, G., Dambrosia, J. M., Alexopoulos, H., & McElroy, B. (2017). A double-blind, placebo-controlled study of rituximab in patients with stiff person syndrome. Annals of Neurology, 82, 271–277. https://doi.org/10.1002/ana.25002
Watts, R. L., Wiegner, A. W., & Young, R. R. (1986). Elastic properties of muscles measured at the elbow in man: II. Patients with Parkinsonian rigidity. Journal of Neurology, Neurosurgery and Psychiatry, 49, 1177–1181. https://doi.org/10.1136/jnnp.49.10.1177
Alves, C. M., Rezende, A. R., Marques, I. A., & Martins Naves, E. L. (2021). SpES: A new portable device for objective assessment of hypertonia in clinical practice. Computers in Biology and Medicine. https://doi.org/10.1016/j.compbiomed.2021.104486
Di Carlo, A. (2009). Human and economic burden of stroke. Age and Ageing. https://doi.org/10.1093/ageing/afn282
Broussy, S., Saillour-Glenisson, F., García-Lorenzo, B., Rouanet, F., Lesaine, E., Maugeais, M., Aly, F., Glize, B., Salamon, R., & Sibon, I. (2019). Sequelae and quality of life in patients living at home 1 year after a stroke managed in stroke units. Frontiers in Neurology. https://doi.org/10.3389/fneur.2019.00907
Goetz, C. C. (2003). The Unified Parkinson’s Disease Rating Scale (UPDRS): Status and recommendations. Movement Disorders. https://doi.org/10.1002/mds.10473
Marques, I. A., Alves, C. M., Rezende, A. R., Silva, M. A. M., Cruz, R. G. D., Lucena, V. F., & Naves, E. L. M. (2019). Evaluating the spasticity level during the rehabilitation process: A survey of traditional methods and a new trend. Procedia Computer Science. https://doi.org/10.1016/j.procs.2019.11.033
Marques, I. A., Silva, M. B., Silva, A. N., Luiz, L. M. D., Soares, A. B., & Naves, E. L. M. (2017). Measurement of post-stroke spasticity based on tonic stretch reflex threshold: implications of stretch velocity for clinical practice. Disability and Rehabilitation. https://doi.org/10.1080/09638288.2017.1381183
Fleuren, J. F. M., Voerman, G. E., Erren-Wolters, C. V., Snoek, G. J., Rietman, J. S., Hermens, H. J., & Nene, A. V. (2010). Stop using the Ashworth Scale for the assessment of spasticity. Journal of Neurology, Neurosurgery and Psychiatry, 81, 46–52. https://doi.org/10.1136/jnnp.2009.177071
Marques, I. A., Alves, C. M., Rezende, A. R., Mendes, L. C., de Paiva, T. S., Cyrino, G. F., de Souza, J. T., Silva, M. A. M., de Souza, L. A. P. S., & Naves, E. L. M. (2022). Virtual reality and serious game therapy for post-stroke individuals: A preliminary study with humanized rehabilitation approach protocol. Complementary Therapies in Clinical Practice, 49, 101681. https://doi.org/10.1016/j.ctcp.2022.101681
Acknowledgements
The authors thank the Assistive Technology Lab for the selfless help. IA Marques and CM Alves are fellows of the Program CAPES/COFECUB (Grant Nos. 88887.628121/2021-01, 88887.662002/2022-00). The authors would also like to express their gratitude to Kathryn L Selzer for the English proofreading of the manuscript.
Funding
This work was supported in part by the Coordination for the Improvement of Higher Education Personnel (CAPES) Finance Code 001, the Brazilian National Council for Scientific and Technological Development (CNPq) grant number 307754/2020-0 and the program CAPES/COFECUB-88881.370894/2019-01). I. A. Marques and C. M. Alves are fellow of the Program CAPES/COFECUB (88887.628121/2021-00, 88887.662002/2022-00 respectively).
Author information
Authors and Affiliations
Contributions
ARR and CMA were responsible for searching the databases. IAM contributed in data extraction and compiling the studies selected from the search. ELMN and LAPSS contributed in the development of research ideas, coordinating the team and reviewing the results of the search. All authors contributed to the development of the manuscript and approved the final version.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no conflict of interest.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rezende, A.R., Alves, C.M., Marques, I.A. et al. Muscle Tonus Evaluation in Patients with Neurological Disorders: A Scoping Review. J. Med. Biol. Eng. 43, 1–10 (2023). https://doi.org/10.1007/s40846-023-00773-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40846-023-00773-4