Abstract
Purpose
Chronic neck pain may affect sensorimotor integration, which is critical for postural control. Accordingly, this study investigated whether individuals with chronic neck pain display altered muscle activation and postural control compared with those of healthy adults while performing functional reach tasks.
Methods
Three-dimensional kinematic analyses and electromyographic analyses were performed. 20 individuals with chronic neck pain and twenty healthy adults were recruited. The reach distance, anterior displacements of the centers of mass and pressure, and activity of the cervical muscles were recorded during functional reach tasks.
Results
Compared with the healthy adults, the individuals with chronic neck pain showed significantly smaller anterior displacements of the centers of mass and pressure and reduced muscle activity of the upper trapezius muscle. However, no difference in the reach distance was observed between the two groups.
Conclusions
It is speculated that a functional reach task may not be sensitive enough to detect changes in the postural control of individuals with chronic neck pain. For individuals with chronic neck pain, a combination of clinical measures and quantitative assessments of the centers of mass and pressure is suggested to facilitate the early detection of changes in postural control.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Neck pain is the second most common form of musculoskeletal disorder, with 1-year prevalence rates varying from 30 to 50% [1,2,3]. Neck pain symptoms are commonly reported to be persistent and recurrent [4], and are one of the most common causes of long-term physical and psychological disabilities [5, 6]. The consequences of neck pain include considerable healthcare costs, productivity loss, and fear of movement, which may lead to reduced physical activity [7,8,9,10,11].
The cervical spine, with abundant muscle spindles and mechanoreceptors, plays an important role in integrating various sensory inputs, including those related to proprioceptive, vestibular, visual, and somatosensory information [12,13,14,15]. Studies based on the use of neck muscle vibration to impair proprioceptive inputs have observed increased body sway and an inclination of the body toward the side opposite the vibrated muscle during stance [16, 17]. Unilateral long-lasting vibration has been found to result in body rotation toward the contralateral side during stepping in place [18]. These findings may be attributed to the involvement of the cervical input to the vestibulospinal nuclei in postural control, which suggests that afferent input from the neck muscle plays an important role in the control of stance as well as locomotion [19, 20].
There is growing evidence for the role of the cervical spine in postural control. For example, patients who have suffered whiplash injuries often complain of an impaired regulation of balance [21,22,23,24]. Similarly, an alteration of postural control has consistently been reported in patients with neck pain [25,26,27]. Several studies have reported that abnormal information from the cervical spine may influence the integration of inputs within the sensorimotor control system [13, 28].
Functional forward reach, which is commonly performed in activities of daily living, requires the coordination of the arm and trunk to move the body center of mass (COM) toward the front edge of the base of support in a well-controlled manner [29]. The reach distance, defined as the maximum distance that the COM can be safely moved without changing the base of support, is a widely used measure of balance ability and is strongly related to frailty, the risk of falling, and the ability to perform functional tasks in elderly people [30,31,32]. Patients with chronic neck pain (CNP) often display atypical postures and movement patterns, and the performance of upper limb movement is frequently compromised due to an alteration of sensorimotor control associated with pain [33]. However, despite the significance of functional forward reach and the prevalence of CNP, the impact of CNP on functional forward reach is still unclear.
A better understanding of the impact of CNP on functional forward reach is essential for minimizing the risk of falls in individuals with CNP. Accordingly, this study compares the difference in performance and muscle activation during functional reach tasks among individuals with CNP and healthy controls. It is hypothesized that individuals with CNP will demonstrate a shorter reach distance and lower activation of the neck muscles during functional forward reach than will healthy controls.
2 Methods
2.1 Subjects
Twenty individuals with CNP and 20 age- and gender-matched healthy controls were recruited. The inclusion criteria for the CNP group were (1) mechanical neck pain duration of at least 3 months in the past half year and (2) pain frequency of at least once a month in the past 3 months. Healthy participants with no previous or current neck pain or history of neck trauma were assigned to the healthy control group. Individuals with a history of cervical spine surgery, neck trauma, neurological signs, vestibular deficits, lower extremity injuries, diabetes mellitus, or pregnancy were excluded. The site of neck pain (left or right side) was documented for all subjects and used as the with-subject factor in the statistical analyses. The study was approved by the Institutional Review Board of National Cheng Kung University Hospital (No. A-ER-102-224). All subjects provided informed consent prior to participation.
2.2 Data Collection and Analysis
The pressure pain threshold of the targeted muscles was measured using a hand-held digital algometer (model OE-220, ITO-Physiotherapy & Rehabilitation, Japan). The measurements were performed over the suboccipital muscles, levator scapulae, and upper trapezius [34]. For each muscle, the pressure pain threshold was measured three times, with a 30-s rest interval between measurements.
A total of 40 reflective markers were placed on the bony landmarks of the whole body, including the top of the head, the bilateral tragus of the ear, the lateral orbit of the eyes, the inferior orbit of the right eye, the 7th cervical spinal process, the 4th thoracic spinal process, and the bilateral acromion processes, olecranons, ulnar styloid processes, radial styloid processes, hand, sacrum, anterior superior iliac spines, greater trochanters, lateral and medial epicondyle of the knee, lateral and medial malleoli of the ankle, midpoints of the lateral thigh and shank, 3rd metatarsal heads, and heels. The three-dimensional motion of the placed markers was detected using a Qualisys motion capture system (ProReflex MCU, 170 240, Gothenburg, Sweden) at a sampling rate of 100 Hz. Two triaxial force plates (Bertec Corp., Columbus, OH) were synchronized with the motion analysis system to collect the ground reaction force at a sampling rate of 1000 Hz. The muscle activity of the cervical muscles was recorded at a sampling rate of 1000 Hz using a surface electromyography (EMG) system (Myomonitor® Trigno EMG Systems, Delsys Incorporated, USA) with bipolar electrodes placed on the muscle bellies of the splenius capitis, upper trapezius, and sternocleidomastoid [35,36,37].
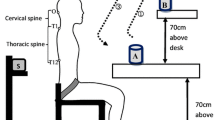
The demographic data, visual analogue pain scale, Neck Disability Index (NDI), and Neck Pain and Disability Scale (NPDS) were recorded for both groups. Pressure pain threshold measurements were performed on all participants over the neck extensors [38, 39]. The neck pain group was asked to watch a video on a tablet (ASUS Memo Pad FHD 10, Taiwan) placed on the lap to reproduce symptoms with a sustained neck flexion posture during sitting. This protocol was designed to simulate the condition of using a smartphone or tablet for a period of time to induce a moderate level of pain that required an immediate break for relief (lasting often 30–45 min). Functional reach tests were performed by the control group and by the neck pain group before and right after pain was induced (Fig. 1). To maintain consistent neck pain intensity throughout the tests, the pain induction protocol was reapplied whenever the level of pain subsided. The intensity of pain, headache, and dizziness as well as the pain pressure threshold were measured both before and after the pain induction protocol for the subjects with CNP.
In the reach tests, the participants first stood with their feet shoulder-distance apart and one arm raised to 90° for 3 s. The participants were then instructed to reach forward as far as possible toward the target at shoulder level without moving their feet, bending their knees, or losing balance, and to hold this position for a further 3 s. Reaching forward beyond the target was allowed. An arm height lower than the target was considered a failed trial.
The head tilt angle was defined as the angle between the global Z-axis unit vector and the line connecting the intersection of the right/left tragus and right/left orbits [37]. The shoulder protraction angle was defined as the angle between the global Z-axis unit vector and the line connecting the acromion to C7 [40]. The trunk tilt angle was defined as the angle between the global Z-axis unit vector and the line connecting the sacrum and C7.
The reach distance was defined as the difference between the starting position and the farthest position of the hand marker in the anterior–posterior direction. The hip posterior displacement was defined as the displacement of the midpoint of the right and left hip markers in the anterior–posterior direction. The whole body COM was calculated by summing up the products of each segment mass distribution and the corresponding COM. The 14-segment model consisted of the head and neck, upper arms, forearms and hands, trunk, pelvis, thighs, shanks, and feet. The COM displacement was defined as the distance between the farthest position and the starting position of the COM in the X-axis (anterior–posterior) direction. The displacement of the center of pressure (COP) between the starting position and the farthest position (normalized by foot length) in the anterior–posterior direction was also calculated.
The EMG raw data were band-pass filtered at 40–400 Hz. The calculated root-mean-square (RMS) values were normalized by the corresponding maximum voluntary isometric contraction and used to quantify muscle activity.
2.3 Statistical Analysis
One-way analysis of variance (ANOVA) tests were performed to examine the difference in the visual analogue scale (neck pain) and pressure pain threshold of the neck pain group before and after pain was induced and the control group. Independent t-tests were additionally performed if a significant main group effect was observed. Furthermore, 3 × 2 mixed ANOVA tests were performed to detect the effects of group (neck pain group before and after pain induction, and control group) and site of pain (painful side vs. non-painful side) on the kinematics, COP, and EMG data. Finally, post hoc independent t-tests and Tukey tests were performed if significant main effects or interaction effects were obtained in the 3 × 2 mixed ANOVA tests, respectively. All of the analyses were conducted using SPSS Statistics 17.0 software (SPSS, Chicago, IL, USA) with the significance level set to P < 0.05.
3 Results
The demographic data of the two groups can be found in our previous study [41]. There were no significant differences in age, standing height, or body weight between the two groups. Before the pain protocol, each neck pain patient performed functional reach tasks. The neck pain group showed significantly higher scores compared with those of the control group in the mean NDI and NPDS as well as the three subscales of the NPDS.
A significant group effect was obtained for the intensity of neck pain, (F = 22.919, P < 0.01). The intensity of neck pain was 2.57 ± 1.84, 5.77 ± 1.69, and 5.87 ± 1.72 before and after pain was induced and in daily life, respectively. The post hoc tests showed that the intensity of neck pain experienced in daily life and after the pain induction protocol was significantly larger than that before the pain induction protocol. The intensity of headache was significantly different among the three conditions; the highest intensity was in daily life and the lowest intensity was prior to the pain induction protocol (F = 10.04, P < 0.01). No significant difference was observed in the intensity of dizziness among the three conditions (F = 2.60, P = 0.078).
A significant group effect was found for the pressure pain threshold of all three cervical muscles (F = 14.70 and 10.57, P < 0.01 for right and left suboccipital, respectively; F = 9.02 and 5.88, P < 0.01 for right and left upper trapezius, respectively; F = 5.31 and 6.41, P < 0.01 for right and left levator scapulae, respectively). It was hence inferred that the pressure pain threshold was significantly lower for the neck pain group both before and after the pain induction protocol than for the control group.
Significant main group effects were observed for both the COM and the COP forward displacements during the functional reach tests (F = 5.89 and 4.98, P < 0.01). The post hoc tests showed that the COM and COP forward displacements in the control group were significantly longer than those in the neck pain group both before and after pain was induced (Fig. 2). However, the reach distance did not differ between the two groups (F = 2.53, P = 0.09). Similarly, no significant group effect was found for the hip posterior displacement, acromioclavicular joint displacement, head tilt angle, shoulder protraction angle, or trunk tilt angle of the two groups during functional reach (Table 1).
A significant group effect was observed only for the muscle activation level of the right upper trapezius at the farthest position during functional reach (F = 3.40, P = 0.04). The post hoc tests showed that the EMG activity of the right upper trapezius was significantly higher in the control group than in the neck pain group before and after pain was induced (Fig. 3).
4 Discussion
The present study aimed to understand the possible impact of CNP on postural control and muscle activation during functional reach tasks. Overall, the results show that compared with healthy controls, individuals with CNP have a lower pressure pain threshold for the neck muscles, a higher disability score, reduced activation of the right upper trapezius muscle, and smaller COP and COM displacements during functional reach.
Tissue injury and inflammation may lead to sensitization of the peripheral nociceptors, which may result in an exaggerated pain response to a normally noxious stimulus applied to the injured tissue in individuals with chronic pain [42, 43]. Moreover, hypersensitivity to pressure may extend beyond the local zone of injury due to an altered processing of the nociceptive information at the level of the spinal cord or higher centers [44]. Studies of patients with whiplash injury have shown widespread sensory hypersensitivity in the cervical region, which may result in poor short- and long-term outcomes [45, 46]. The pressure pain thresholds of the neck muscles in the neck pain group in the present study are similar to those reported by Lluch et al. [47]. These results suggest that individuals with CNP have lower pain thresholds for mechanical stimuli and higher sensory hypersensitivity. The pressure pain thresholds obtained for individuals with CNP in the present study are also similar to those reported by Lluch et al. [47], confirming the validity of the present pain induction protocol and the generalizability of our findings. Although previous studies have reported that CNP is commonly associated with significant disability in the general population [48,49,50], the NDI scores obtained in the present study suggest that individuals with CNP may suffer only mild disability. Additionally, the significant differences in the NPDS scores of the two groups suggest that in addition to causing neck-related disability, CNP results in discomfort in the emotion and affective domains [51].
The altered muscle activity level during functional reach in individuals with CNP observed here has been previously reported by studies on the muscle activation pattern of the upper limb in individuals with CNP [52,53,54]. According to the pain adaptation model, the pain experienced by individuals with CNP may result in reduced contractile capability of the agonistic muscle during task performance [55]. The pain intensity in the present study was significantly higher following the pain induction protocol, and may therefore account for the lower EMG amplitude in the right upper trapezius muscle in individuals with CNP compared with that in the control group. However, no increase in the activity of the antagonistic or synergistic muscles was observed, which may be explained by the different tasks performed by the subjects in the present study and those in prior studies [56].
To maintain balance, the body must generate a quick movement of the COP to exceed the current position of the COM and reverse it in the opposite direction [57]. The ability to modulate the relationship between the COP and the COM during motion for individuals with CNP could be influenced by abundant cervical mechanoreceptors for the integration of proprioceptive, vestibular, visual, and somatosensory information [58,59,60,61]. Altered postural control in individuals with CNP has been reported in previous studies, where the abnormal cervical afferent input may be proprioceptive and/or nociceptive [13, 25, 61, 62]. The deterioration of proprioceptive information from the neck may result in poorer estimation of the COM position, leading to the adoption of an increased safety margin of adaptive COP shifts in response to predicted COM oscillations [63]. Moreover, pain may also cause increased pre-synaptic inhibition and affect the central modulation of the proprioceptive muscle spindles, leading to decreased muscle control and increased postural sway [64, 65]. Previous studies have shown that individuals with CNP display larger body sway during standing or perturbation as the result of compromised postural control [66, 67]. However, the significantly reduced anterior displacements of the COM and COP in individuals with CNP during functional reach observed here suggests that the ability of individuals with CNP to perceive the limit of stability is altered and impairs their ability to coordinate the movement of the COM and COP during forward motion. Hence, a change of the COP and COM displacements during functional reach may provide an important indicator of impaired CNP postural control in individuals with CNP and may thus serve as a useful trigger for potential intervention aimed at reducing the risk of falls.
Several important limitations should be considered when interpreting the present findings. First, the kinematic parameters collected during the functional reach tests were limited to the sagittal plane, even though rotational movements inevitably occur in the transverse and coronal planes. Thus, a more thorough understanding of the impact of CNP on balance control can be achieved by incorporating movements in all three planes during the performance of functional reach tasks. Second, the present NDI scores indicate that the subjects with CNP suffer only mild disability. Hence, care is required when generalizing the present findings and assertions to individuals with CNP who suffer greater disability. Third, the pain induction protocol employed in the present study is targeted mainly at the neck extensors, which may not necessarily represent the source of neck pain for all subjects. Nevertheless, the protocol is capable of reproducing a similar pain intensity to that experienced by CNP subjects in daily life, and hence accurately reflects the possible impact of CNP on their postural control.
5 Conclusion
The present study provides evidence of altered muscle activation and postural control with a smaller activation level of the trapezius muscle of the dominant arm, together with reduced displacements of the COM and COP, during functional reach tasks by individuals with CNP. The lack of difference in the reach distance between the CNP subjects and healthy controls highlights the need to incorporate quantitative measurements of the COP and COM displacements into clinical tests to facilitate early rehabilitation intervention to decrease the risk of falls.
References
Hogg-Johnson, S., van der Velde, G., Carroll, L. J., et al. (2008). The burden and determinants of neck pain in the general population: Results of the bone and joint decade 2000–2010 task force on neck pain and its associated disorders. Spine, 33(4 Suppl.), S39–S51.
Fejer, R., Kyvik, K. O., & Hartvigsen, J. (2006). The prevalence of neck pain in the world population: A systematic critical review of the literature. European Spine Journal, 15(6), 834–848.
Carroll, L. J., Hogg-Johnson, S., van der Velde, G., et al. (2008). Course and prognostic factors for neck pain in the general population: Results of the bone and joint decade 2000–2010 task force on neck pain and its associated disorders. Spine, 33(4 Suppl), S75-82.
Martin, B. I., Deyo, R. A., Mirza, S. K., et al. (2008). Expenditures and health status among adults with back and neck problems. Journal of the American Medical Association, 299(6), 656–664.
Leclerc, A., Niedhammer, I., Landre, M. F., Ozguler, A., Etore, P., & Pietri-Taleb, F. (1999). One year predictive factors for various aspects of neck disorders. Spine, 24(14), 1455–1462.
Sterling, M., Kenardy, J., Jull, G., & Vicenzino, B. (2003). The development of psychological changes following whiplash injury. Pain, 106(3), 481–489.
Loeppke, R., Taitel, M., Haufle, V., et al. (2009). Health and productivity as a business strategy: A multiemployer study. Journal of Occupational and Environmental Medicine, 51(4), 411–428.
Cheung, J., Kajaks, T., & Macdermid, J. C. (2013). The relationship between neck pain and physical activity. The Open Orthopaedics Journal, 20(7), 521–529.
Cote, P., Cassidy, J. D., Carroll, L. J., & Kristman, V. (2004). The annual incidence and course of neck pain in the general population: A population-based cohort study. Pain, 112(3), 267–273.
Côté, P., Hogg-Johnson, S., Cassidy, J. D., Carroll, L., & Frank, J. W. (2001). The association between neck pain intensity, physical functioning, depressive symptomatology and time-to-claim-closure after whiplash. Journal of Clinical Epidemiology, 54(3), 275–286.
Hoving, J. L., de Vet, H. C. W., Twisk, J. W., et al. (2004). Prognostic factors for neck pain in general practice. Pain, 110(3), 639–645.
Gandevia, S. C., McClosekey, D. I., & Burke, D. (1992). Kinesthetic signals and muscle contraction. Trends in Neurosciences, 15(2), 62–65.
Treleaven, J. (2008). Sensorimotor disturbances in neck disorders affecting postural stability, head and eye movement control. Manual Therapy, 13(1), 2–11.
Morningstar, M. W., Pettibon, B. R., Schlappi, H., Schlappi, M., & Ireland, T. V. (2005). Reflex control of the spine and posture: A review of the literature from a chiropractic perspective. Chiropractic and Osteopathy, 13, 16.
Bolton, P. (1998). The somatosensory system of the neck and its effects on the central nervous system. Journal of Manipulative and Physiological Therapeutics, 21(8), 553–563.
Koskimies, K., Sutinen, P., & Aalto, H. (1997). Postural stability, neck proprioception and tension neck. Acta Oto-Laryngologica. Supplementum, 529, 95–97.
Kavounoudias, A., Gilhodes, J. C., Roll, R., & Roll, J. P. (1999). From balance regulation to body orientation: Two goals for muscle proprioceptive information processing? Experimental Brain Research, 124(1), 80–88.
Bove, M., Courtine, G., & Schieppat, M. (2002). Neck muscle vibration and spatial orientation during stepping in place in humans. Journal of Neurophysiology, 88(5), 2232–2241.
Mazzini, L., & Schieppati, M. (1994). Short-latency neck muscle responses to vertical body tilt in normal subjects and in patients with spasmodic torticollis. Electroencephalography and Clinical Neurophysiology, 93(4), 265–275.
Gdowski, G. T., & McCrea, R. A. (2000). Neck proprioceptive inputs to primate vestibular nucleus neurons. Experimental Brain Research, 135(4), 511–526.
Chester, J. B. (1991). Whiplash, postural control and the inner ear. Spine, 16(7), 716–720.
Evans, R. W. (1992). Some observations on whiplash injuries. Neurologic Clinics, 10(4), 975–997.
Spitzer, W. O., Skovron, M. L., Salmi, L. R., et al. (1995). Scientific monograph of the Quebec task force on whiplash-associated disorders: Redefining “whiplash” and its management. Spine, 20(8 Suppl), 1S-73S.
Hinoki, M. (1985). Vertigo due to whiplash injury: A neurotological approach. Acta Oto-Laryngologica. Supplementum, 419, 9–29.
Sjöström, H., Allum, J. H., Carpenter, M. G., et al. (2003). Trunk sway measures of postural stability during clinical balance tests in patients with chronic whiplash injury symptoms. Spine, 28(15), 1725–1734.
Dehner, C., Heym, B., Maier, D., et al. (2008). Postural control deficit in acute QTF grade II whiplash injuries. Gait and Posture, 28(1), 113–119.
Ruhe, A., Fejer, R., & Walker, B. (2011). Altered postural sway in patients suffering from non-specific neck pain and whiplash associated disorder—a systematic review of the literature. Chiropractic and Manual Therapies, 19, 13.
Kristjansson, E., & Treleaven, J. (2009). Sensorimotor function and dizziness in neck pain: Implications for assessment and management. The Journal of Orthopaedic and Sports Physical Therapy, 39(5), 364–377.
Duncan, P. W., Weiner, D. K., Chandler, J., & Studenski, S. (1990). Functional reach: A new clinical measure of balance. Journal of Gerontology, 45(6), M192–M197.
Weiner, D. K., Duncan, P. W., Chandler, J., & Studenski, S. A. (1992). Functional reach: A marker of physical frailty. Journal of the American Geriatrics Society, 40(3), 203–207.
Duncan, P. W., Studenski, S., Chandler, J., & Prescott, B. (1992). Functional reach: Predictive validity in a sample of elderly male veterans. Journal of Gerontology, 47(3), M93–M98.
Wallman, H. W. (2001). Comparison of elderly nonfallers and fallers on performance measures of functional reach, sensory organization, and limits of stability. The Journals of Gerontology. Series A, Biological Sciences and Medical Sciences, 56(9), M580-583.
Sandlund, J., Roijezon, U., Bjorklund, M., & Djupsjobacka, M. (2008). Acuity of goal-directed arm movements to visible targets in chronic neck pain. Journal of Rehabilitation Medicine, 40(5), 366–374.
Ylinen, J., Nykanen, M., Kautiainen, H., & Häkkinen, A. (2007). Evaluation of repeatability of pressure algometry on the neck muscles for clinical use. Manual Therapy, 12(2), 192–197.
Joines, S. M., Sommerich, C. M., Mirka, G. A., Wilson, J. R., & Moon, S. D. (2006). Low-level exertions of the neck musculature: A study of research methods. Journal of Electromyography and Kinesiology, 16(5), 485–497.
Cram, J. R., & Kasman, G. S. (1998). Introduction to surface electromyogrphy. Gaithersburg, Maryland: Aspen Publishers.
Falla, D., Dall’Alba, P., Rainoldi, A., Merletti, R., & Jull, G. (2002). Location of innervation zones of sternocleidomastoid and scalene muscles–a basis for clinical and research electromyography applications. Clinical Neurophysiology, 113(1), 57–63.
Vernon, H., & Mior, S. (1991). The Neck Disability Index: A study of reliability and validity. Journal of Manipulative and Physiological Therapeutics, 14(7), 409–415.
Goolkasian, P., Wheeler, A. H., & Gretz, S. S. (2002). The neck pain and disability scale: Test-retest reliability and construct validity. The Clinical Journal of Pain, 18(4), 245–250.
Edmondston, S. J., Chan, H. Y., Ngai, G. C., et al. (2007). Postural neck pain: An investigation of habitual sitting posture, perception of ‘good’ posture and cervicothoracic kinaesthesia. Manual Therapy, 12(4), 363–371.
Lin, C. C., Hua, S. H., Lin, C. L., Cheng, C. H., Liao, L. J., & Lin, C. F. (2020). Impact of prolonged tablet computer usage with head forward and neck flexion posture on pain intensity, cervical joint position sense and balance control in mechanical neck pain subjects. Journal of Medical and Biological Engineering, 40, 372–382.
Woolf, C. J., & Salter, M. W. (2000). Neuronal plasticity: Increasing the gain of pain. Science, 288(5472), 1765–1768.
Merskey, H., & Bogaduk, N. (1994). Classification of chronic pain: Descriptions of chronic pain syndromes and definitions of pain terms (2nd ed.). Seattle, WA: IASP Press.
Walton, D. M., Macdermid, J. C., Nielson, W., et al. (2011). A descriptive study of pressure pain threshold at 2 standardized sites in people with acute or subacute neck pain. The Journal of Orthopaedic and Sports Physical Therapy, 41(9), 651–657.
Sterling, M. (2010). Differential development of sensory hypersensitivity and a measure of spinal cord hyperexcitability following whiplash injury. Pain, 150(3), 501–506.
Sterling, M., Jull, G., Vicenzino, B., & Kenardy, J. (2003). Sensory hypersensitivity occurs soon after whiplash injury and is associated with poor recovery. Pain, 104(3), 509–517.
Lluch, E., Arguisuelas, M. D., Coloma, P. S., et al. (2013). Effects of deep cervical flexor training on pressure pain thresholds over myofascial trigger points in patients with chronic neck pain. Journal of Manipulative and Physiological Therapeutics, 36(9), 604–611.
Côté, P., Cassidy, D., & Corroll, L. (1998). The Saskatchewan health and back pain survey. The prevalence of neck pain and related disability in Saskatchewan adults. Spine, 23(15), 1689–1698.
Picavet, H., & Schouten, J. (2003). Musculoskeletal pain in the Netherlands: prevalences, consequences and risk groups, the DMC3-study. Pain, 102(1–2), 167–178.
van der Windt, D., Croft, P., & Penninx, B. (2002). Neck and upper limb pain: more pain is associated with psychological distress and consultation rate in primary care. The Journal of Rheumatology, 29(3), 564–569.
Wheeler, A. H., Goolkasian, P., Baird, A. C., et al. (1999). Development of the Neck Pain and Disability Scale. Item analysis, face, and criterion-related validity. Spine, 24(13), 1290–1294.
Falla, D., Bilenkij, G., & Jull, G. (2004). Patients with chronic neck pain demonstrate altered patterns of muscle activation during performance of a functional upper limb task. Spine, 29(13), 1436–1440.
O’Leary, S., Falla, D., & Jull, G. (2003). Recent advances in therapeutic exercise for the neck: Implications for patients with head and neck pain. Australian Endodontic Journal, 29(3), 138–142.
Bansevicius, D., & Sjaastad, O. (1996). Cervicogenic headache: The influence of mental load on pain level and EMG of shoulder-neck and facial muscles. Headache, 36(3), 372–378.
Lund, J. P., Donga, R., Widmer, C. G., & Stohler, C. S. (1991). The pain-adaptation model: A discussion of the relationship between chronic musculoskeletal pain and motor activity. Canadian Journal of Physiology and Pharmacology, 69(5), 683–694.
Falla, D. (2004). Unravelling the complexity of muscle impairment in chronic neck pain. Manual Therapy, 9(3), 125–133.
Baratto, L., Morasso, P. G., Re, C., & Spada, G. (2002). A new look at posturographic analysis in the clinical context: Sway-density versus other parameterization techniques. Motor Control, 6(3), 246–270.
Cavanaugh, J. M., Lu, Y., Chen, C., & Kallakuri, S. (2006). Pain generation in lumbar and cervical facet joints. The Journal of Bone and Joint Surgery, 88(Suppl 2), 63–67.
Winkelstein, B. A., Nightingale, R. W., Richardson, W. J., et al. (2000). The cervical facet capsule and its role in whiplash injury: A biomechanical investigation. Spine, 25(10), 1238–1246.
Yin, W., & Bogduk, N. (2008). The nature of neck pain in a private pain clinic in the United States. Pain Medicne, 9(2), 196–203.
Armstrong, B., McNair, P., & Taylor, D. (2008). Head and neck position sense. Sports Medicine, 38(2), 101–117.
Moseley, G. L., & Hodges, P. W. (2005). Are the changes in postural control associated with low back pain caused by pain interference? The Clinical Journal of Pain, 21(4), 323–329.
Casadio, M., Morasso, P. G., & Sanguineti, V. (2005). Direct measurement of ankle stiffness during quiet standing: Implications for control modelling and clinical application. Gait and Posture, 21(4), 410–424.
Sibley, K. M., Carpenter, M. G., Perry, J. C., & Frank, J. S. (2007). Effects of postural anxiety on the soleus H-reflex. Human Movement Science, 26(1), 103–112.
Capra, N. F., & Ro, J. Y. (2000). Experimental muscle pain produces central modulation of proprioceptive signals arising from jaw muscle spindles. Pain, 86(1–2), 151–162.
Michaelson, P., Michaelson, M., Jaric, S., et al. (2003). Vertical posture and head stability in patients with chronic neck pain. Journal of Rehabilitation Medicine, 35(5), 229–235.
Röijezon, U., Björklund, M., & Djupsjöbacka, M. (2001). The slow and fast components of postural sway in chronic neck pain. Manual Therapy, 16(3), 273–278.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lin, CC., Lin, CF., Hua, SH. et al. Chronic Neck Pain Affects Shoulder Muscle Activity and Postural Control During Functional Reach. J. Med. Biol. Eng. 41, 77–83 (2021). https://doi.org/10.1007/s40846-020-00591-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40846-020-00591-y