Abstract
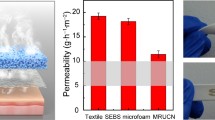
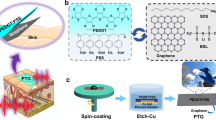
The development of durable and reliable bioelectrodes for high-quality bioelectrical signals acquisition has become a crucial technology in the field of human physiological condition monitoring and human-machine interfaces. However, most existing bioelectrodes are limited to conventional elastomeric substrates that suffer from mechanical mismatch and low permeability, and lack multifaceted attributes and essential synergistic properties akin to those found in biological skins. In this work, we report advanced substrate-free ultra-thin epidermal bioelectrodes (ASU-EBEs) based on free-standing conductive all-polymer (FCAP) films, which integrate the advantages of ultra-conformality, excellent stretchability and breathability into a single device. The resulting ASU-EBEs exhibit excellent conductivity of ∼475 S cm−1, outstanding stretchability of ∼48%, ultra-conformality to the interface of biological tissues and superior breathability. The enhanced electronic and mechanical performance is attributed to the introduction of water-soluble polyethylene oxide into poly(3,4-ethylenedioxythiophene): poly(4-styrenesulfonate) (PEDOT:PSS) to control the molecular π-π stacking distance and promote the formation of nanofiber structures. Hence, ASU-EBEs show much lower skin-contact impedance than the standard gel electrodes, enabling the use for long-term healthcare monitoring in complex daily conditions.
摘要
开发耐用且可靠的生物电极, 用以采集高质量的生物电信号, 已成为人体生理状态监测和人机交互领域的关键技术. 然而, 现有的生物电极多基于传统弹性基底, 这导致了机械性能不匹配和低渗透性等问题, 并且缺乏与生物皮肤类似的多方面属性和必要的协同特性. 本研究中, 我们报道了一种新型的基于自支撑导电全聚合物薄膜的超薄表皮生物电极(ASU-EBE). 该电极将超一致性、优异的拉伸性和透气性集成于一体, 展现了约475 S cm−1的高导电性, 约48%的出色拉伸性, 与生物组织界面的超一致性以及优异的透气性. 该电极的电子和机械性能得到提升, 这归功于在PEDOT:PSS中引入水溶性聚氧化乙烯, 以调节分子间π-π堆积距离, 并促进纳米纤维结构的形成. 因此, ASU-EBE在与皮肤接触时的阻抗远低于标准凝胶电极, 使其成为复杂日常环境下长期医疗监测的理想选择.

Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Zhang M, Tang Z, Liu X, et al. Electronic neural interfaces. Nat Electron, 2020, 3: 191–200
Wang Y, Haick H, Guo S, et al. Skin bioelectronics towards long-term, continuous health monitoring. Chem Soc Rev, 2022, 51: 3759–3793
Wang C, Wang H, Wang B, et al. On-skin paintable biogel for long-term high-fidelity electroencephalogram recording. Sci Adv, 2022, 8: eabo1396
Liu S, Rao Y, Jang H, et al. Strategies for body-conformable electronics. Matter, 2022, 5: 1104–1136
Zhang L, Kumar KS, He H, et al. Fully organic compliant dry electrodes self-adhesive to skin for long-term motion-robust epidermal biopotential monitoring. Nat Commun, 2020, 11: 4683
Miyamoto A, Lee S, Cooray NF, et al. Inflammation-free, gas-permeable, lightweight, stretchable on-skin electronics with nanomeshes. Nat Nanotech, 2017, 12: 907–913
Zhu Y, Haghniaz R, Hartel MC, et al. A breathable, passive-cooling, non-inflammatory, and biodegradable aerogel electronic skin for wearable physical-electrophysiological-chemical analysis. Adv Mater, 2023, 35: 2209300
Someya T, Amagai M. Toward a new generation of smart skins. Nat Biotechnol, 2019, 37: 382–388
Zhao Y, Zhang S, Yu T, et al. Ultra-conformal skin electrodes with synergistically enhanced conductivity for long-time and low-motion artifact epidermal electrophysiology. Nat Commun, 2021, 12: 4880
Fernandez M, Pallas-Areny R. Ag-AgCl electrode noise in high-resolution ECG measurements. Biomed Instrum Technol, 2000, 34: 125–130
Deng J, Yuk H, Wu J, et al. Electrical bioadhesive interface for bioelectronics. Nat Mater, 2021, 20: 229–236
Gogurla N, Kim Y, Cho S, et al. Multifunctional and ultrathin electronic tattoo for on-skin diagnostic and therapeutic applications. Adv Mater, 2021, 33: 2008308
Kabiri Ameri S, Ho R, Jang H, et al. Graphene electronic tattoo sensors. ACS Nano, 2017, 11: 7634–7641
Tang L, Shang J, Jiang X. Multilayered electronic transfer tattoo that can enable the crease amplification effect. Sci Adv, 2021, 7: eabe3778
Jiang Z, Chen N, Yi Z, et al. A 1.3-micrometre-thick elastic conductor for seamless on-skin and implantable sensors. Nat Electron, 2022, 5: 784–793
Kireev D, Sel K, Ibrahim B, et al. Continuous cuffless monitoring of arterial blood pressure via graphene bioimpedance tattoos. Nat Nanotechnol, 2022, 17: 864–870
Jang H, Sel K, Kim E, et al. Graphene e-tattoos for unobstructive ambulatory electrodermal activity sensing on the palm enabled by heterogeneous serpentine ribbons. Nat Commun, 2022, 13: 6604
Song D, Ye G, Zhao Y, et al. An all-in-one, bioderived, air-permeable, and sweat-stable MXene epidermal electrode for muscle theranostics. ACS Nano, 2022, 16: 17168–17178
Yan Z, Xu D, Lin Z, et al. Highly stretchable van der Waals thin films for adaptable and breathable electronic membranes. Science, 2022, 375: 852–859
Wang Y, Zhu C, Pfattner R, et al. A highly stretchable, transparent, and conductive polymer. Sci Adv, 2017, 3: e1602076
Fan X, Nie W, Tsai H, et al. PEDOT:PSS for flexible and stretchable electronics: Modifications, strategies, and applications. Adv Sci, 2019, 6: 1900813
Ju D, Kim D, Yook H, et al. Controlling electrostatic interaction in PEDOT:PSS to overcome thermoelectric tradeoff relation. Adv Funct Mater, 2019, 29: 1905590
Jiang Y, Zhang Z, Wang YX, et al. Topological supramolecular network enabled high-conductivity, stretchable organic bioelectronics. Science, 2022, 375: 1411–1417
Kottner J, Lichterfeld A, Blume-Peytavi U. Transepidermal water loss in young and aged healthy humans: A systematic review and meta-analysis. Arch Dermatol Res, 2013, 305: 315–323
Hong YJ, Jeong H, Cho KW, et al. Wearable and implantable devices for cardiovascular healthcare: From monitoring to therapy based on flexible and stretchable electronics. Adv Funct Mater, 2019, 29: 1808247
Kireev D, Okogbue E, Jayanth RT, et al. Multipurpose and reusable ultrathin electronic tattoos based on PtSe2 and PtTe2. ACS Nano, 2021, 15: 2800–2811
Salatino JW, Ludwig KA, Kozai TDY, et al. Glial responses to implanted electrodes in the brain. Nat Biomed Eng, 2017, 1: 862–877
Ouyang J, Xu Q, Chu CW, et al. On the mechanism of conductivity enhancement in poly(3,4-ethylenedioxythiophene):poly(styrene sulfonate) film through solvent treatment. Polymer, 2004, 45: 8443–8450
Kee S, Kim N, Kim BS, et al. Controlling molecular ordering in aqueous conducting polymers using ionic liquids. Adv Mater, 2016, 28: 8625–8631
Kayser LV, Lipomi DJ. Stretchable conductive polymers and composites based on PEDOT and PEDOT:PSS. Adv Mater, 2019, 31: 1806133
Li P, Sun K, Ouyang J. Stretchable and conductive polymer films prepared by solution blending. ACS Appl Mater Interfaces, 2015, 7: 18415–18423
Roberts T, De Graaf JB, Nicol C, et al. Flexible inkjet-printed multi-electrode arrays for neuromuscular cartography. Adv Healthcare Mater, 2016, 5: 1462–1470
Acknowledgements
This work was supported by the National Key Research and Development Program of China (2023YFB3608904), the National Natural Science Foundation of China (21835003 and 61704077), the Natural Science Foundation of Jiangsu Province (BE2019120 and BK20191374), the Foundation of Key Laboratory of Flexible Electronics of Zhejiang Province (2023FE002), the Natural Science Foundation of Jiangsu Higher Education Institutions of China (18KJB150025), the Program for Jiangsu Specially-Appointed Professor (RK030STP15001), the Postgraduate Research & Practice Innovation Program of Jiangsu Province (KYCX21_0778, SJCX21_0298), the NUPT Scientific Foundation (NY219021 and NY219109), the Leading Talent of Technological Innovation of National Ten-Thousands Talents Program of China, and the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).
Author information
Authors and Affiliations
Contributions
Author contributions Niu J and Lai WY conceived the idea; Li G carried out the material synthesis and performed the material characterization; Fang S, Gong Y, Shao R, and You T contributed to the electrophysiological measurement and design of human-machine interaction programs; Li G, Gong Y, Niu J, and Lai WY co-wrote the paper. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest The authors declare that they have no conflict of interest.
Additional information
Guanjun Li is a PhD candidate at Nanjing Post and Communications University. His research focuses on the development of high-performance electronic skins and epidermal electronics, including adhesive bioelectronic materials and devices, new conductive hydrogels, and biological tissue-electrode interface sensing technology.
Jian Niu is an associate professor at Nanjing University of Posts and Telecommunications. He received his PhD in physics from Lanzhou University in 2011. His research primarily focuses on the development and application of flexible and stretchable electronic devices.
Wen-Yong Lai is a full professor at Nanjing University of Posts and Telecommunications. He received his PhD degree from Fudan University in 2007. He then joined the Key Laboratory for Organic Electronics & Information Displays, Institute of Advanced Materials (IAM), Nanjing University of Posts and Telecommunications. His research mainly focuses on the design, synthesis, and application of organic & polymer optoelectronic materials for organic/flexible electronics. He is also interested in the exploration of novel materials and processes for printed electronics.
Supplementary information Experimental details and supporting data are available in the online version of the paper.
Supporting Information
40843_2024_2823_MOESM1_ESM.pdf
Substrate-Free Ultra-Thin Epidermal Bioelectrodes with Enhanced Conformality and Breathability for Long-Term Physiological Monitoring
Supplementary material, approximately 6.41 MB.
Supplementary material, approximately 15.3 MB.
Supplementary material, approximately 15.7 MB.
Rights and permissions
About this article
Cite this article
Li, G., Gong, Y., Fang, S. et al. Substrate-free ultra-thin epidermal bioelectrodes with enhanced conformality and breathability for long-term physiological monitoring. Sci. China Mater. 67, 1481–1490 (2024). https://doi.org/10.1007/s40843-024-2823-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40843-024-2823-9