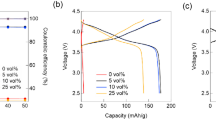
Abstract
High-capacity anode materials, such as SiO and Si/C, are considered promising candidates for high-energy-density lithium-ion batteries. However, the low initial Coulombic efficiency of these anode materials induced by side reactions (forming Li2O and lithium silicate) and the formation of solid electrolyte interface film reduces the active Liions and causes low-discharge capacity. Adding a Li-compensation material in the cathode or anode is an effective strategy to overcome this problem. The most used Li-compensation material is the stabilized lithium metal powder. However, this strategy has high safety risks, high costs, and is challenging to quantify. Herein, the Li-compensation material of Li6CoO4 is synthesized and investigated. The preparation conditions, stability in the air, delithiation mechanism, and structural transformation are analyzed and discussed. Electrochemical tests reveal that the discharge capacity and capacity retention of the full pouch cells (3-Ah) with Li6CoO4 additive is significantly improved. Also, the reason for such improvement is investigated. This work provides an effective strategy of Li-compensating technology to enhance the electrochemical performance of lithium-ion batteries.
摘要
SiO和Si/C具有高容量, 被认为是高能量密度锂离子电池有前景的负极材料. 然而, 副反应(形成Li2O和硅酸锂)和固体电解质界面(SEI)膜的形成, 导致这些负极材料初始库仑效率、活性锂离子数量以及放电容量的降低. 在正极或负极中添加补锂材料是解决这一问题的有效方法. 最常用的补锂材料是稳定锂金属粉末(SLMP), 但这种策略存在安全风险高、成本高且难以实现定量补锂的缺点. 本文合成并研究了Li6CoO4锂补偿材料, 分析和讨论了其制备条件、空气中的稳定性、脱锂机制和结构转变机理. 研究结果表明, 添加Li6CoO4能在发挥补锂作用的同时, 显著提高软包全电池(3-Ah)的循环性能. 这项工作提供了一种有效的补锂策略, 是提高锂离子电池电化学性能的有效方法.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Kong L, Liu Y, Huang H, et al. Interconnected CoS2/NC-CNTs network as high-performance anode materials for lithium-ion batteries. Sci China Mater, 2021, 64: 820–829
Liu Z, Yu Q, Zhao Y, et al. Silicon oxides: A promising family of anode materials for lithium-ion batteries. Chem Soc Rev, 2019, 48: 285–309
Wang YY, Gao MY, Liu S, et al. Yttrium surface gradient doping for enhancing structure and thermal stability of high-Ni layered oxide as cathode for Li-ion batteries. ACS Appl Mater Interfaces, 2021, 13: 7343–7354
Cao K, Jiao L, Liu H, et al. 3D hierarchical porous α-Fe2O3 nanosheets for high-performance lithium-ion batteries. Adv Energy Mater, 2015, 5: 1401421
Su Z, Liu J, Li M, et al. Defect engineering in titanium-based oxides for electrochemical energy storage devices. Electrochem Energ Rev, 2020, 3: 286–343
Yen HJ, Tsai H, Zhou M, et al. Structurally defined 3D nanographene assemblies via bottom-up chemical synthesis for highly efficient lithium storage. Adv Mater, 2016, 28: 10250–10256
Feng Y, Shu N, Xie J, et al. Carbon-coated Fe2O3 hollow sea urchin nanostructures as high-performance anode materials for lithium-ion battery. Sci China Mater, 2021, 64: 307–317
Xin F, Whittingham MS. Challenges and development of tin-based anode with high volumetric capacity for Li-ion batteries. Electrochem Energ Rev, 2020, 3: 643–655
Du Z, Zhang S, Liu Y, et al. Facile fabrication of reticular polypyrrole-silicon core-shell nanofibers for high performance lithium storage. J Mater Chem, 2012, 22: 11636–11641
Cui LF, Hu L, Choi JW, et al. Light-weight free-standing carbon nanotube-silicon films for anodes of lithium ion batteries. ACS Nano, 2010, 4: 3671–3678
Cui LF, Ruffo R, Chan CK, et al. Crystalline-amorphous core-shell silicon nanowires for high capacity and high current battery electrodes. Nano Lett, 2009, 9: 491–495
Ren WF, Le JB, Li JT, et al. Improving the electrochemical property of silicon anodes through hydrogen-bonding cross-linked thiourea-based polymeric binders. ACS Appl Mater Interfaces, 2021, 13: 639–649
Tan T, Lee PK, Zettsu N, et al. Highly stable lithium-ion battery anode with polyimide coating anchored onto micron-size silicon monoxide via self-assembled monolayer. J Power Sources, 2020, 453: 227874
Chen M, Li B, Liu X, et al. Boron-doped porous Si anode materials with high initial coulombic efficiency and long cycling stability. J Mater Chem A, 2018, 6: 3022–3027
Wu W, Wang M, Wang R, et al. Magnesio-mechanochemical reduced SiOx for high-performance lithium ion batteries. J Power Sources, 2018, 407: 112–122
Chen T, Wu J, Zhang Q, et al. Recent advancement of SiOx based anodes for lithium-ion batteries. J Power Sources, 2017, 363: 126–144
Xu Q, Sun JK, Yin YX, et al. Facile synthesis of blocky SiOx/C with graphite-like structure for high-performance lithium-ion battery anodes. Adv Funct Mater, 2018, 28: 1705235
Zhang Y, Mu Z, Lai J, et al. MXene/Si@SiOx@C layer-by-layer superstructure with autoadjustable function for superior stable lithium storage. ACS Nano, 2019, 13: 2167–2175
Yang C, Zhang Y, Zhou J, et al. Hollow Si/SiOx nanosphere/nitrogendoped carbon superstructure with a double shell and void for high-rate and long-life lithium-ion storage. J Mater Chem A, 2018, 6: 8039–8046
Zhuang X, Song P, Chen G, et al. Coralloid-like nanostructured c-nSi/SiOx@Cy anodes for high performance lithium ion battery. ACS Appl Mater Interfaces, 2017, 9: 28464–28472
Jiang B, Zeng S, Wang H, et al. Dual core-shell structured Si@SiOx@C nanocomposite synthesized via a one-step pyrolysis method as a highly stable anode material for lithium-ion batteries. ACS Appl Mater Interfaces, 2016, 8: 31611–31616
Li Y, Fitch B. Effective enhancement of lithium-ion battery performance using SLMP. Electrochem Commun, 2011, 13: 664–667
Aravindan V, Lee YS, Madhavi S. Best practices for mitigating irreversible capacity loss of negative electrodes in Li-ion batteries. Adv Energy Mater, 2017, 7: 1602607
Forney MW, Ganter MJ, Staub JW, et al. Prelithiation of silicon-carbon nanotube anodes for lithium ion batteries by stabilized lithium metal powder (SLMP). Nano Lett, 2013, 13: 4158–4163
Wang L, Fu Y, Battaglia VS, et al. SBR-PVDF based binder for the application of SLMP in graphite anodes. RSC Adv, 2013, 3: 15022–15027
Pan Q, Zuo P, Mu T, et al. Improved electrochemical performance of micro-sized SiO-based composite anode by prelithiation of stabilized lithium metal powder. J Power Sources, 2017, 347: 170–177
Shellikeri A, Watson VG, Adams DL, et al. Pre-lithiation of carbon anodes using different lithium sources. ECS Trans, 2017, 77: 293–303
Zhao H, Wang Z, Lu P, et al. Toward practical application of functional conductive polymer binder for a high-energy lithium-ion battery design. Nano Lett, 2015, 14: 6704–6710
Su X, Lin C, Wang X, et al. A new strategy to mitigate the initial capacity loss of lithium ion batteries. J Power Sources, 2016, 324: 150–157
Zhang L, Dose WM, Vu AD, et al. Mitigating the initial capacity loss and improving the cycling stability of silicon monoxide using Li5FeO4. J Power Sources, 2018, 400: 549–555
Jeżowski P, Fic K, Crosnier O, et al. Lithium rhenium(VII) oxide as a novel material for graphite pre-lithiation in high performance lithiumion capacitors. J Mater Chem A, 2016, 4: 12609–12615
Noh M, Cho J. Role of Li6CoO4 cathode additive in Li-ion cells containing low coulombic efficiency anode material. J Electrochem Soc, 2012, 159: A1329–A1334
Guo Y, Li X, Wang Z, et al. Bifunctional Li6CoO4 serving as prelithiation reagent and pseudocapacitive electrode for lithium ion capacitors. J Energy Chem, 2020, 47: 38–45
Liang M, Sun Y, Song D, et al. Superior electrochemical performance of quasi-concentration-gradient LiNi0.8Co0.15Al0.05 O2 cathode material synthesized with multi-shell precursor and new aluminum source. Electrochim Acta, 2019, 300: 426–436
Lu W, Yuan Z, Xu C, et al. Construction of mesoporous Cu-doped Co9S8 rectangular nanotube arrays for high energy density all-solidstate asymmetric supercapacitors J Mater Chem A, 2019, 7: 5333–5343
Ben Yahia M, Vergnet J, Saubanère M, et al. Unified picture of anionic redox in Li/Na-ion batteries Nat Mater, 2019, 18: 496–502
Li B, Jiang N, Huang W, et al. Thermodynamic activation of charge transfer in anionic redox process for Li-ion batteries Adv Funct Mater, 2018, 28: 1704864
Xiao R, Li H, Chen L Density functional investigation on Li2MnO3. Chem Mater, 2012, 24: 4242–4251
Xiang X, Knight JC, Li W, et al. Understanding the effect of Co3+ substitution on the electrochemical properties of lithium-rich layered oxide cathodes for lithium-ion batteries J Phys Chem C, 2014, 118: 21826–21833
Acknowledgements
This work was supported by the National Key R&D Program of China (2016YFB0100500) and the Young Elite Scientists Sponsorship Program by Tianjin (TJSQNTJ-2017-05)
Author information
Authors and Affiliations
Contributions
Zhang H conceived the idea and designed the experiments Na Z designed and carried out the experiments and wrote the paper. Lai C, Song D, and Shi X analyzed the data and refined the manuscript Zhang L and Zhou J supervised the project All authors contributed to the general discussion.
Corresponding authors
Additional information
Conflict of interest
The authors declare that they have no conflict of interest
Ziyu Na received her bachelor’s degree in 2018. She joined the School of Materials Science and Engineering, Tianjin University of Technology, in 2021 Her current research interests are mainly focused on the Li-compensation material for lithium-ion batteries.
Hongzhou Zhang obtained his PhD degree in Materials Science from Nankai University (China) in 2015. Then, he joined Prof. Lianqi Zhang’s group at Tianjin University of Technology, China. His current research focused on functional materials and their application in energy conversion and storage devices.
Lianqi Zhang is a professor at Tianjin University of Technology, China. He obtained his PhD degree in chemistry from Soga University (Japan) in 2003. His current interest is focused on the applied research in lithium-ion batteries, the structure, and the electrochemical properties of new materials for energy storage and conversion.
Rights and permissions
About this article
Cite this article
Na, Z., Lai, C., Zhou, J. et al. Enhancing the reversible capacity and cycle stability of lithium-ion batteries with Li-compensation material Li6CoO4. Sci. China Mater. 65, 620–628 (2022). https://doi.org/10.1007/s40843-021-1784-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40843-021-1784-0