Abstract
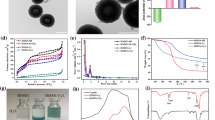
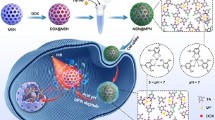
The design and synthesis of multifunctional nanocarriers for efficient synergistic cancer therapy have drawn great research interests in recent years. In this work, a nanoplatform for chemo-photothermal therapy with targeting ligand was developed. Hollow porous structured silica nanotubes (SNTs) with controllable lengths decorated with CuS nanoparticles (NPs) on the surface as photothermal agents were prepared and further conjugated with lactobionic acid groups as a cancer cell target. SNTs with average lengths of 40, 55 and 150 nm were obtained and further functionalized as drug carriers. The smallest bifunctional SNTs with targeting groups show good biocompatibility and highest cellular uptake for HepG2 cells. The release of doxorubicin hydrochloride (DOX) from the SNTs was dependent on the pH of the buffer solution and 808-nm near infrared (NIR) light irradiation. The integration of photothermal therapy (PTT) of CuS NPs and chemotherapy of anticancer drug leads to a better tumor inhibition effect than the individual therapy alone in vitro and in vivo. These results demonstrate potential applications of the nanocomposites as vector for efficient chemo-photothermal therapy.
摘要
多功能药物载体的设计合成并应用于肿瘤的联合治疗得到了研究人员的广泛关注. 本文介绍了一种连接靶向基团的化疗-光热联合治疗纳米平台. 首先制备了尺寸可控的平均长度为40、 55和150 nm的空心多孔氧化硅纳米管, 在表面修饰具有光热功能的硫化铜纳米颗粒, 然后连接乳糖酸基团实现肝癌细胞靶向功能. 平均长度为40 nm、 修饰靶向基团的空心多孔材料显示出良好的生物相容性, 且具有最大的HepG2细胞吞噬量. 负载盐酸阿霉素的纳米复合材料表现出pH和808 nm近红外激光刺激响应的释放效果. 将CuS光热治疗和盐酸阿霉素化疗相结合的方法在体外和体内的抑制肿瘤效果都优于单独治疗. 研究结果表明, 该纳米复合材料在化疗-光热联合治疗方面具有潜在的应用价值.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
He Q, Shi J. MSN anti-cancer nanomedicines: chemotherapy enhancement, overcoming of drug resistance, and metastasis inhibition. Adv Mater, 2014, 26: 391–411
Pan L, He Q, Liu J, et al. Nuclear-targeted drug delivery of TAT peptide-conjugated monodisperse mesoporous silica nanoparticles. J Am Chem Soc, 2012, 134: 5722–5725
Lee SB, Kim HL, Jeong HJ, et al. Mesoporous silica nanoparticle pretargeting for PET imaging based on a rapid bioorthogonal reaction in a living body. Angew Chem Int Ed, 2013, 52: 10549–10552
Cheng L, Yang L, Meng F, Zhong Z. Protein nanotherapeutics as an emerging modality for cancer therapy. Adv Healthcare Mater, 2018, 7: 1800685
Yang B, Chen Y, Shi J. Mesoporous silica/organosilica nano-particles: Synthesis, biological effect and biomedical application. Mater Sci Eng-R-Rep, 2019, 137: 66–105
Zhao W, Gu J, Zhang L, Chen H, Shi J. Fabrication of uniform magnetic nanocomposite spheres with a magnetic core/mesoporous silica shell structure. J Am Chem Soc, 2005, 127: 8916–8917
Chen Y, Chen H, Zeng D, et al. Core/shell structured hollow mesoporous nanocapsules: a potential platform for simultaneous cell imaging and anticancer drug delivery. ACS Nano, 2010, 4: 6001–6013
Hao N, Li L, Zhang Q, et al. The shape effect of PEGylated mesoporous silica nanoparticles on cellular uptake pathway in hela cells. Microporous Mesoporous Mater, 2012, 162: 14–23
Huang X, Teng X, Chen D, Tang F, He J. The effect of the shape of mesoporous silica nanoparticles on cellular uptake and cell function. Biomaterials, 2010, 31: 438–448
Decuzzi P, Godin B, Tanaka T, et al. Size and shape effects in the biodistribution of intravascularly injected particles. J Control Release, 2010, 141: 320–327
Gao C, Zhang Q, Lu Z, Yin Y. Templated synthesis of metal nanorods in silica nanotubes. J Am Chem Soc, 2011, 133: 19706–19709
Gao C, Lu Z, Yin Y. Gram-scale synthesis of silica nanotubes with controlled aspect ratios by templating of nickel-hydrazine complex nanorods. Langmuir, 2011, 27: 12201–12208
Huang L, Ao L, Wang W, et al. Multifunctional magnetic silica nanotubes for MR imaging and targeted drug delivery. Chem Commun, 2015, 51: 3923–3926
Yang X, He D, He X, et al. Synthesis of hollow mesoporous silica nanorods with controllable aspect ratios for intracellular triggered drug release in cancer cells. ACS Appl Mater Interfaces, 2016, 8: 20558–20569
Deng X, Li K, Cai X, et al. A hollow-structured CuS@Cu2S@Au nanohybrid: synergistically enhanced photothermal efficiency and photoswitchable targeting effect for cancer theranostics. Adv Mater, 2017, 29: 1701266
Li Y, Bai G, Zeng S, Hao J. Theranostic carbon dots with innovative NIR-II emission for in vivo renal-excreted optical imaging and photothermal therapy. ACS Appl Mater Interfaces, 2019, 11: 4737–4744
Zhou M, Zhang R, Huang M, et al. A chelator-free multifunctional [64Cu]CuS nanoparticle platform for simultaneous micro-PET/CT imaging and photothermal ablation therapy. J Am Chem Soc, 2010, 132: 15351–15358
Guo L, Panderi I, Yan DD, et al. A comparative study of hollow copper sulfide nanoparticles and hollow gold nanospheres on degradability and toxicity. ACS Nano, 2013, 7: 8780–8793
Xiao Q, Zheng X, Bu W, et al. A core/satellite multifunctional nanotheranostic for in vivo imaging and tumor eradication by radiation/photothermal synergistic therapy. J Am Chem Soc, 2013, 135: 13041–13048
Zha Z, Wang S, Zhang S, et al. Targeted delivery of CuS nano-particles through ultrasound image-guided microbubble destruction for efficient photothermal therapy. Nanoscale, 2013, 5: 3216–3219
Liang S, Deng X, Chang Y, et al. Intelligent hollow Pt-CuS janus architecture for synergistic catalysis-enhanced sonodynamic and photothermal cancer therapy. Nano Lett, 2019, 19: 4134–4145
Zhang Y, Hou Z, Ge Y, et al. DNA-hybrid-gated photothermal mesoporous silica nanoparticles for NIR-responsive and aptamer-targeted drug delivery. ACS Appl Mater Interfaces, 2015, 7: 20696–20706
Chen Y, Hou Z, Liu B, et al. DOX-Cu9S5@mSiO2-PG composite fibers for orthotopic synergistic chemo- and photothermal tumor therapy. Dalton Trans, 2015, 44: 3118–3127
Jiang M, Liu H, Zeng S, Hao J. A general in situ growth strategy of designing theranostic NaLnF4@Cu2−xS nanoplatform for in vivo NIR-II optical imaging beyond 1500 nm and photothermal therapy. Adv Therap, 2019, 2: 1800153
Li Y, Zeng S, Hao J. Non-invasive optical guided tumor metastasis/vessel imaging by using lanthanide nanoprobe with enhanced down-shifting emission beyond 1500 nm. ACS Nano, 2019, 13: 248–259
Wu L, Wu M, Zeng Y, et al. Multifunctional PEG modified DOX loaded mesoporous silica nanoparticle@CuS nanohybrids as photo-thermal agent and thermal-triggered drug release vehicle for hepatocellular carcinoma treatment. Nanotechnology, 2015, 26: 025102
Shi S, Chen F, Cai W. Biomedical applications of functionalized hollow mesoporous silica nanoparticles: focusing on molecular imaging. Nanomedicine, 2013, 8: 2027–2039
Wang X, Li X, Ito A, et al. Stimulation of in vivo antitumor immunity with hollow mesoporous silica nanospheres. Angew Chem Int Ed, 2016, 55: 1899–1903
Liu X, Fu F, Xu K, et al. Folic acid-conjugated hollow mesoporous silica/CuS nanocomposites as a difunctional nanoplatform for targeted chemo-photothermal therapy of cancer cells. J Mater Chem B, 2014, 2: 5358
Xie X, Gao N, Deng R, et al. Mechanistic investigation of photon upconversion in Nd3+-sensitized core-shell nanoparticles. J Am Chem Soc, 2013, 135: 12608–12611
Shen J, Chen G, Vu AM, et al. Engineering the upconversion nanoparticle excitation wavelength: cascade sensitization of tri-doped upconversion colloidal nanoparticles at 800 nm. Adv Opt Mater, 2013, 1: 644–650
Sun LD, Wang YF, Yan CH. Paradigms and challenges for bioapplication of rare earth upconversion luminescent nanoparticles: small size and tunable emission/excitation spectra. Acc Chem Res, 2014, 47: 1001–1009
Jayakumar MKG, Idris NM, Huang K, Zhang Y. A paradigm shift in the excitation wavelength of upconversion nanoparticles. Nanoscale, 2014, 6: 8441–8443
Duan X, Li Y. Physicochemical characteristics of nanoparticles affect circulation, biodistribution, cellular internalization, and trafficking. Small, 2013, 9: 1521–1532
Yang R, Meng F, Ma S, et al. Galactose-decorated cross-linked biodegradable poly(ethylene glycol)-b-poly(ε-caprolactone) block copolymer micelles for enhanced hepatoma-targeting delivery of paclitaxel. Biomacromolecules, 2011, 12: 3047–3055
Sun L, Zhang X, An J, et al. Boronate ester bond-based core-shell nanocarriers with pH response for anticancer drug delivery. RSC Adv, 2014, 4: 20208–20215
Bi H, Dai Y, Lv R, et al. Doxorubicin-conjugated CuS nanoparticles for efficient synergistic therapy triggered by near-infrared light. Dalton Trans, 2016, 45: 5101–5110
Guo Z, Seol ML, Kim MS, et al. Hollow CuO nanospheres uniformly anchored on porous Si nanowires: preparation and their potential use as electrochemical sensors. Nanoscale, 2012, 4: 7525–7531
Liu P, Huang Y, Yan J, Yang Y, Zhao Y. Construction of CuS nanoflakes vertically aligned on magnetically decorated graphene and their enhanced microwave absorption properties. ACS Appl Mater Interfaces, 2016, 8: 5536–5546
Zhao R, Li T, Zheng G, et al. Simultaneous inhibition of growth and metastasis of hepatocellular carcinoma by co-delivery of ursolic acid and sorafenib using lactobionic acid modified and pH-sensitive chitosan-conjugated mesoporous silica nanocomplex. Biomaterials, 2017, 143: 1–16
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (NSFC, 51572257, 51720105015, 51672269, 51929201, 51922097, 51772124 and 51872282), the Science and Technology Cooperation Project between Chinese and Australian Governments (2017YFE0132300), the Key Research Program of Frontier Sciences, CAS (YZDY-SSW-JSC018), the Overseas, Hong Kong & Macao Scholars Collaborated Researching Fund (21728101), the CAS-Croucher Funding Scheme for Joint Laboratories (CAS18204), Jiangmen Innovative Research Team Program (2017), and the Major Program of Basic Research and Applied Research of Guangdong Province (2017KZDXM083)
Author information
Authors and Affiliations
Contributions
Author contributions Huang S performed the experiments and wrote the draft of manuscript. Liu B, Deng X and Xie Z helped design the cell and animal experiments. Wei Y contributed to the CLSM image and analysis. Ma P, Cheng Z and Xing B provided suggestions and comments on the manuscript. Lin J proposed the project and revised the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest The authors declare that they have no conflict of interest.
Additional information
Shanshan Huang received her PhD degree in inorganic chemistry from the University of Chinese Academy of Sciences in 2013. She worked in Changchun Institute of Applied Chemistry (CIAC), Chinese Academy of Sciences (CAS) from 2006 to 2018. Currently, she is an associate professor in the College of Chemistry and Pharmaceutical Engineering, Huanghuai University. Her research interests include multifunctional mesoporous silica materials for drug delivery systems and rare-earth doped luminescent materials.
Ziyong Cheng received his BSc degree in materials engineering from Changchun University of Technology in 1994 and his PhD degree from CIAC, CAS in 2006. Following postdoctoral studies at Max-Planck Institute for Polymer Research (Mainz, Germany), he returned to CIAC (2008) to take up an associate professor position in inorganic chemistry. In 2013, he was promoted to a full professor. His research interests are nanostructured materials including perovskite quantum dots in photoelectric applications and polymer-inorganic nanocomposites for biomaterial related fields.
Jun Lin received his BSc and MSc degrees in inorganic chemistry from Jilin University in 1989 and 1992, respectively, and a PhD degree (inorganic chemistry) from CIAC, CAS in 1995. He worked as a postdoctoral researcher for more than 4 the years in City University of Hong Kong (1996), Institute of New Materials (Germany, 1997), Virginia Commonwealth University (USA, 1998), and University of New Orleans (USA, 1999). He is a professor at the CIAC, CAS. His research interests include luminescent materials and multifunctional composite materials toward their application in display, lighting and biomedical fields.
Rights and permissions
About this article
Cite this article
Huang, S., Ma, P., Wei, Y. et al. Controllable synthesis of hollow porous silica nanotubes/CuS nanoplatform for targeted chemo-photothermal therapy. Sci. China Mater. 63, 864–875 (2020). https://doi.org/10.1007/s40843-019-1235-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40843-019-1235-1