Abstract
Objective
A goal of behavioral neuroendocrinology is to understand how basal hormone levels relate to behavior. Studies of human participants sometimes measure self-reported personality traits, in addition to or instead of direct behavioral observation. Although personality traits often predict their respective behaviors, whether personality explains hormone-behavior relationships remains unclear.
Methods
We obtained data from eight previous studies (total N = 985) that examined baseline testosterone and cortisol as predictors of status-relevant behavior (competitiveness, dominance, risk-taking, aggression, affiliation, and social status). We tested whether the previously reported hormone-behavior relationships are mediated by self-reported personality traits (e.g., trait dominance, prestige, extraversion). As a secondary research question, we also tested whether trait dominance moderated the testosterone-behavior relationships.
Results
As expected, self-reported personality traits often predicted status-relevant behaviors, but there was little evidence that traits also correlated with basal testosterone or the testosterone × cortisol interaction. Across all eight studies, personality traits did not significantly mediate hormone-behavior relationships. Indeed, the effect sizes of the hormone-behavior relationships were robust to the inclusion of personality traits as covariates. Further, we did not find strong or consistent evidence that trait dominance moderates the testosterone-behavior association.
Conclusion
Results suggest that basal testosterone and cortisol predict status-related behavior independent of self-reported personality. We discuss how these results may have broader implications for the physiological mechanisms by which testosterone and cortisol influence behavior, a process that could be unconscious and automatic. We also discuss alternative explanations, limitations, and future directions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
A major objective of behavioral neuroendocrinology is to understand how basal hormone levels predict behavior. Research with human participants sometimes includes self-reported personality questionnaires, in addition to or instead of direct behavioral observation. Self-reported personality traits represent general self-perceptions about one’s own behavior, and are often positively related to their respective behaviors. However, it remains unclear whether self-reported personality traits explain basal hormone-behavior relationships. In the current project, we re-examined data from eight previously published studies to test whether self-reported personality traits mediate associations between basal hormone levels (testosterone or the testosterone by cortisol interaction) and status-related behaviors. The degree to which individual differences in self-reported personality explain basal hormone-behavior relationships in these studies could provide initial insight into the underlying mechanism by which hormones influence behavior, and highlight the utility of personality as a variable in future research.
Testosterone is a steroid hormone involved in the energetic and behavioral investment in mate pursuit and competition for mates (Gray et al., 2020; Grebe et al., 2019b; Muller, 2017; Roney, 2016; Rosvall et al., 2020; Wingfield et al., 2001) and, more broadly, social contests for status (Archer, 2006). Influential theoretical frameworks postulate that increased testosterone levels are beneficial for mounting the appropriate behavioral response to achieve success in these situations (Archer, 2006; Mazur & Booth, 1998; Roney, 2016; Zilioli & Bird, 2017). In line with this theorizing, research indicates that higher testosterone concentrations are positively related to behaviors implicated in the pursuit and maintenance of higher status, including competitive behavior, social dominance, aggressive behavior, risk taking, and actual status attainment (for review, Carré & Archer, 2018; Casto & Mehta, 2019; Eisenegger et al., 2011; Geniole et al., 2020). However, other studies revealed non-significant associations (e.g., Casto et al., 2020; Dekkers et al., 2019), and some work found negative associations between testosterone concentrations and similar behaviors (e.g., Buades-Rotger et al., 2016). Further, meta-analyses have shown weak aggregate effects and substantial heterogeneity in the associations between individual differences in basal testosterone levels and status-relevant behaviors (aggression: Geniole et al., 2020; risk-taking: Kurath & Mata, 2018; leadership: van der Meij et al., 2016)Footnote 1.
Cortisol is a glucocorticoid produced by the hypothalamic-pituitary-adrenal (HPA) axis in response to psychological and physical stress (Dickerson et al., 2009; Kemeny, 2009; Putman & Roelofs, 2011). Individual differences in levels of basal cortisol are thought to reflect relative differences in stress exposure, with higher levels being linked to greater social avoidance and mood disorder symptomology (Bertsch et al., 2011; Lombardo et al., 2019) as well as diminished social status (for review, Sherman & Mehta, 2020 – although these relationships are somewhat inconsistent: McEwen, 2019; Staufenbiel et al., 2013). The dual-hormone hypothesis, a model that accounts for the moderating role of cortisol, was proposed as one explanation for some of the inconsistent findings regarding basal testosterone’s relationship with status-relevant behavior (Knight et al., 2020b; Mehta & Josephs, 2010). According to the original formulation of the dual-hormone hypothesis, higher levels of basal testosterone are hypothesized to be related to increased status-seeking behaviors when basal cortisol levels are low. However, when basal cortisol levels are high, higher basal testosterone levels are expected to be unrelated or negatively related to status-seeking behaviors.
A number of studies have provided initial support for the dual-hormone hypothesis with a broad set of outcome variables relating to social standing and status-seeking motivation (for review, Knight et al. 2020, b; Mehta & Prasad, 2015; Sarkar et al., 2018). Other studies, however, found non-significant dual-hormone interactions (e.g., Geniole et al., 2013; Mehta et al., 2017) or significant interactions in the opposite direction from what is predicted by the dual-hormone hypothesis (e.g., Denson et al., 2013; Lee et al., 2015). That is, these latter studies showed a “reverse” dual-hormone effect, whereby higher testosterone was positively related to aggressive and cheating behaviors among individuals with relatively high cortisol levels (for review, Knight et al.2020, b). A recent meta-analysis reported that overall support for the dual-hormone hypothesis is weak (Dekkers et al., 2019; Grebe, Del Giudice, et al., 2019), and that there is considerable heterogeneity in the direction and magnitude of the effect across studies (for review, Knight et al. 2020, b). Given the mixed evidence, there is a need for greater theoretical clarity and specificity regarding testosterone and cortisol’s associations with status-relevant behavior.
One potential reason for inconsistencies in both the direct and interactive effects of these hormones is that some studies use self-reported personality traits as the outcome of interest instead of direct behavioral observation (e.g., Grebe, Del Giudice, et al., 2019; Sundin et al., 2021; Torrance et al., 2018). Doing so makes the assumption that general self-awareness about status-motivated behavior is an effective proxy for status-related behavior in attempting to identify a hormone-behavior relationship. Indeed, basal levels of cortisol and testosterone show moderate day-to-day stability when measured around the same time of day (Liening et al., 2010; Sellers et al., 2007). Similarly, personality is defined as a behavioral tendency that is stable over time (Wagner et al., 2019), and is generally positively correlated with a task-based or naturalistic observation of that behavior. However, the mechanism of particular hormone-behavior relationships within an eliciting context could be more tightly coupled than generalized self-awareness about that behavior across contexts (Carré & Archer, 2018; Roney, 2016; Zilioli & Bird, 2017). Thus, it may be the case that hormones regulate status-relevant behavior independent of self-reported personality traits.
Several lines of research support the possibility that hormone-behavior relationships are coordinated through mechanisms that circumvent self-awareness about personality. Whereas self-report inventories represent explicit consciously-accessible perceptions about the self (e.g., “I am a dominant person”), there is increasing evidence that hormones, including testosterone and cortisol, alter perceptual systems and behavior implicitly, outside of conscious awareness (Mossink et al., 2015; Quirin et al., 2009; Schultheiss, 2013; Stanton & Schultheiss, 2009; Wegner et al., 2015). For example, Terburg & van Honk (2013) concluded that “…testosterone’s influence on reactive-reflexive dominance and social aggression is automatic, unconscious, and subcortically driven by phylogenetically ancient brain mechanisms humans share with most other vertebrates.”
Although not a direct test of this assertion, studies have tested whether relationships between hormones and behavior remained after controlling for self-reported personality traits. In one study, baseline testosterone predicted status-relevant behavioral outcomes, with and without controlling for self-reported trait dominance (Study 3: Josephs et al., 2006). In a similar vein, Geniole et al. (2013) reported that increased testosterone was positively related to aggressive behavior with and without controlling for self-reported psychopathic traits. Another study measured basal testosterone and cortisol, self-reported personality traits, and group-level performance among business students (Akinola et al., 2016). Results from this study showed that group-level mean testosterone and cortisol interacted to predict how well the group performed on a computerized group decision-making exercise in models that included and excluded self-reported traits (e.g., dominance). Other research found that testosterone and cortisol predicted cheating behavior, with highly similar results when excluding and including self-reported trait anxiety (Lee et al., 2015)Footnote 2. Overall, there is only initial evidence in a limited set of studies suggesting that testosterone and cortisol predict status-relevant behavior while controlling for status-related personality traits.
However, other studies have indicated that testosterone and cortisol could show modest relationships with some personality traits (e.g., baseline testosterone and dominance or extraversion, Grebe, Del Giudice, et al., 2019; Sellers et al., 2007; Smeets-Janssen et al., 2015). Trait dominance has also been found to interact with testosterone to predict status-relevant behavior (Carré et al., 2017; Geniole et al., 2013; Mehta, van Son et al., 2015; Slatcher et al., 2011). Thus, personality may still play an important part in basal hormone effects on status-relevant behavior (but see Sundin et al., 2021; Knight et al., 2022).
The present research draws on data from eight prior studies that examined basal testosterone or its interaction with basal cortisol in relation to competitive behavior, dominant behavior, risk-taking behavior, aggressive behavior, affiliative behavior, and social status. In these prior studies, self-reported traits that correspond to each behavior of interest were also collected (e.g., trait dominance corresponding to observed dominant behavior). Here, we report new analyses that examine whether personality mediates the observed hormone-behavior relationships (Baron & Kenny, 1986; Hayes, 2020). As a secondary set of analyses, we also test whether trait dominance moderates the testosterone-behavior relationships for six of the eight studies that have relevant data. We examine these questions across multiple studies where a variety of status-related behavioral outcomes were assessed under various contexts. This approach allows for a broad understanding of basal testosterone, cortisol, and behavior when accounting for personality.
Although the present research does not provide a direct test of implicit or other alternative mechanisms, this new research does represent a critical initial step in investigating the extent to which basal testosterone and cortisol associate with status-relevant behavior independently from explicit self-reported personality traits. Examining our research questions across these eight studies can provide initial insight into the mechanism by which hormones influence behavior, and can inform future work that differentiates between the effects of implicit versus explicit mental processes on behavior.
Methods
For the present research, we culled datasets from prior published studies and conducted new analyses with self-reported personality traits. Our criteria for inclusion were that the prior dataset was available for additional analyses or was shared upon request by the corresponding authors. Additionally, datasets were selected if they included all three of the following variable types: (1) a measure of basal testosterone (which could also include basal cortisol); (2) a measure of behavior broadly related to status-seeking, leadership, or dominance; and (3) a self-reported personality trait broadly related to the behavioral outcome measure or status-seeking. These traits were selected because we expected an association between the trait and the behavioral outcome measure included in the respective study. An overview of the methods for each study is summarized in Table 1.
Participants
A total of 985 participants from eight separate prior studies were included in the present report. Two of the studies recruited community participants (Study 1: Mean Age: 44.29, SD: 10.96; Study 5: Mean Age: 27.9, SD = 4.9). The other six studies recruited student samples (Study 3: Mean Age = 20.7, SD = 6.1; Study 4: Mean Age = 20.6, SD = 3.0; Study 6: Mean Age = 21.9, SD = 2.9; Study 7: Mean Age = 19.0, SD = 1.1; Study 8: Mean Age = 20.3, SD = 2.1; age data were not available for Study 2). Race/ethnicity data are only reported for the sample in Study 4 (38.2% Caucasian, 19.4% Black, 18.1% Asian, 4.8% Latino, 0.6% Native American, and 18.8% Other), Study 5 (85.4% Caucasian; 4.9% Asian, 7.3%; Mixed Race, 2.4% Hispanic), and Study 8 (41.5% Caucasians, 20.3% African American, 9.3% Asian, 0.8% Native American, 10.2% Middle Eastern, 5.1% Multiracial and 10.2% others). Study 3 excluded oral contraceptive users from participating.
Behavioral Tasks
There were a wide range of tasks employed to measure status-relevant behavior across the studies (Table 1). More detailed descriptions of the procedures are included in the prior publications, but for the purpose of understanding the various contexts and operationalizations of status-seeking for the present study, we detail key aspects of the behavioral tasks and study designs below.
Study 1- Affiliative behaviors
This was a naturalistic study of individuals who took part in a dog agility competition. Dog-handler teams compete by moving through an obstacle course wherein the dog must respond appropriately to the guiding cues of the handler without leash or physical contact from the handler. The speed and accuracy of course completion is judged and ranked among competitors. The context is status-relevant in that dog-handler teams prepare and train for weeks or longer and their performance, in the presence of large numbers of spectators, often qualifies the teams for higher-value future events. The behavioral outcome of interest was time spent interacting with the dog in an affiliative manner in the two minutes immediately following their performance. Three trained researchers coded video clips of the handlers’ behaviors towards their dog, recording how many seconds each handler performed each behavior (inter-rater reliability = 90.2%). Affiliative behaviors included play (e.g., tug-of-war, chase) and petting of the areas around the ears, neck, and chin. Affiliative behavior in the present study is viewed as the converse to the behavioral expression to social dominance. This theorizing is based on the challenge hypothesis, which postulates that elevated testosterone levels during periods of social challenge (e.g., competition) are related to increased dominant behaviors and reduced affiliative behaviors (e.g., Wingfield et al., 1990).
Study 2- Dominant leadership behaviors
Participants were randomly assigned to the position of “leader” or “follower” (Newman et al., 2005) and then completed a leadership block design task while being videotaped (the WAIS-III Block Design Task, Wechsler, 1997). The follower was seated at a table with blocks while the leader stood behind the follower. The leader directed the follower to move blocks in order to make the block design, which was repeated for nine block design puzzles. Participants alternated leadership positions so that leadership behaviors could be observed in both participants within each dyad. Seven independent observers (4 women, 3 men) watched the videotaped interactions and rated all leaders using a measure that was specially developed to assess dominance in leaders in the block design task (Anderson & Kilduff, 2009; Buss & Craik, 1980). The leadership measure included 19 items that tapped into dominance (e.g., leader-like, confident, shy/timid (reverse-scored), gave clear instructions, assertive, indecisive (reverse-scored), dominant, comfortable giving instructions) using a 7-point Likert scale. The 19 items were rated by all observers and were averaged to create an overall index of dominance in leaders (for more details, see Mehta and Josephs, 2010). Dominant behavior has been positively related to higher social status in prior research (Anderson & Kilduff, 2009). Dominance in this study was conceptualized as a behavioral style that is assertive and self-assured, and the 19 items were selected accordingly (Anderson & Kilduff, 2009). This conceptualization of dominance is different from another conceptualization of dominance that involves threatening and intimidating behaviors that induce fear in others (Cheng et al., 2010). This study did not examine the latter, more anti-social form of dominance.
Study 3- Reactive aggression
This study used a two-part behavioral task that included an initial provocation procedure in which a fictitious female participant, who was actually a confederate, insulted the quality and delivery of a two-minute speech given by the female participant. This was followed by a competitive reaction-time task (speed of on-screen button pressing) against the same fictitious female participant. Participants also decided the loudness and duration of a white noise blast to be delivered to their opponent. The status-relevant outcome of interest, reactive aggression, was operationalized as the composite of intensity and duration scores (0—10) selected by participants on the first of the 25 trials of the reaction time competition. This score has been previously validated by Denson et al., (2010; 2011) as a measure of reactive aggression. Reactive aggression is theorized to be motivated by a desire to protect or enhance one’s social status (Daly & Wilson, 1988). Further, aggression in a competitive context is considered one viable method, albeit antisocial, of achieving social status and power over others (Cheng et al., 2010).
Study 4- Risk-taking
This study employed the Balloon Analog Risk Task (BART) (Lejuez et al., 2003), a validated laboratory task of risk-taking behavior. For this task, participants earned “money points” for each click of an on-screen button (collected points contributed towards a raffle to win a $150 gift card). Each press of the button gradually inflated an on-screen image of a balloon. If the balloon burst, no money points were awarded. Because the balloon was randomly programed to burst anywhere between 1 and 30 presses, each decision to press incured more risk of bursting. Thus, risk-taking was operationalized as the average number of presses, or “pumps”, of all non-burst balloons. Risk-taking is considered a status-relevant behavior based on previous theorizing and research because engaging in competitions risks loss or personal injury, but is also necessary for status to be gained (e.g. Ellis et al., 2012).
Study 5- Social network centrality
To assess the social network centrality of each member of a rugby team, the Cognitive Social Structures (CSS, Casciaro et al., 1999; Krackhardt, 1987) procedure was followed. Each member of the rugby team was asked to answer the questions, “Among your teammates, who likes to hang out with i?”, and, “Among your teammates, who does i like to hang out with?”, where i was every player on the team, including the respondent. From these responses, relationship matrices were constructed for each individual and three measures of centrality were determined: (1) betweenness (the number of times a person lies within the shortest pathway between two others in the network), the level of social influence this subject has over the flow of information within the network; (2) popularity (in-degree centrality), the number of teammates that reported liking to hang out with that participant; (3) gregariousness (out-degree centrality), the number of teammates each participant reported to like hanging out with. Social network centrality is related to higher status attainment. Of the three measures of centrality examined in this study, betweenness centrality is especially indicative of higher social status, such as leadership emergence (Ponzi, Ziloli et al., 2016). For more information on how network centrality scores were calculated, see Ponzi, Zilioli et al. (2016).
Study 6- Risk-taking
This study also employed the Balloon Analog Risk Task (BART) described above. The procedure was largely the same as in Study 4, the only difference being that the probability of balloon bursting (no money awarded for that trial) was increased incrementally with each pump (1/128 for the first pump, 1/127 for the second pump, etc. – average breakpoint was 64 pumps). Further, participants were paid 10% of their winnings at the conclusion of the experiment (Meuro = 0.76, SD = 0.21) and informed that the participant who earned the most across all sessions would receive a cash prize of 50 euros. As with Study 4, risk-taking was operationalized as the average number of presses, or “pumps”, of all non-burst balloons.
Study 7- Competitive persistence
This study was designed to explore the relationship between testosterone and persistence in a test of “competitive will.” Participants held a small amount of weight (a common dumbbell used for resistance training; 1 lb. for women, 2 lbs. for men) at arm’s length and shoulder height, with arm extended from the body at a 90° angle, for as long as they felt they could in competition against other participants, according to the procedure in Casto et al. (2020). To incentivize the competition, participants were informed that a $20 cash prize would be offered to the one participant who held their arm up the longest of all the participants. Competitive performance or persistence in this task was operationalized as the time in seconds that a participant held up their arm before quitting the contest. Performance in the competitive will task is positively correlated with self-reported status-seeking (e.g. competitiveness, dominance motivation, and task-specific desire to win; Casto et al., 2020).
Study 8- Task persistence
Participants completed a persistence task in which they were told that performance indicated differences in “problem solving abilities.” The task consisted of tracing geometric shapes completely without interrupting the line or retracing, and participants were told to complete as many puzzles as they would like and to alert the experimenter when they no longer wished to continue. After a small set of solvable puzzles, participants were given a set of impossible (unsolvable) puzzles. Task persistence was operationalized as the amount of time spent attempting to solve the unsolvable puzzles. Persistence behavior has been previously linked to status attainment (Welker & Carré, 2015).
Other Aspects of the Study Designs
As reported in the original published papers, the behavioral tasks were conducted in status-relevant contexts (e.g., competition) and the studies sometimes included additional context factors as potential moderators of hormone-behavior associations. In the present research, we examined associations between basal hormones, behavior, and personality across the whole sample for each study independent of context factors because our primary research question focused on personality. Personality, by definition, captures a person’s behavioral tendency across situations. In accordance with this view, previous research examined basal hormones and personality associations independent of context. The current research builds on this previous work in order to examine the extent to which basal hormone-behavior associations, independent of context factors, are explained by self-reported personality traits.
Personality Measures
A variety of personality inventories were used across the prior studies to assess individual differences in self-reported traits related to status-striving (Table 1) such as extraversion, assertiveness, dominance, prestige, competitiveness, self-control, self-esteem, and confidence. In some cases, these inventories were included as a part of the design, but not included in the analyses used in prior publications. In other cases, the inventory was reported as a part of the study methods and analyses but was not included as a covariate or mediating variable in the original analyses. Thus, in all cases, what we present here are, to the best of our knowledge, new analyses of the data for the purpose of testing the explanatory role of self-reported personality in the associations between basal hormones and status-relevant behavioral outcomes. All of the personality assessments have been used in prior published studies, and (some more than others) have demonstrated reliability and validity evidence (references to sources for each measure are listed in the notes of Table 1).
Saliva Sampling and Hormonal Assays
For all studies, basal testosterone, and in most cases cortisol, were assessed via saliva or hair (Study 6) samples, using standard collection and storage procedures. For studies assaying hormones from saliva, a small volume (e.g., 1–3 mL) of saliva was provided into a plastic (e.g., polystyrene) tube via unstimulated (Studies 3, 4, 5, 7, 8) or stimulated (Study 1, using sugar free gum) passive drool (Study 2 did not specify). A summary of the saliva collection timings and assay procedure for each study are briefly listed below.
Study 1
Saliva samples were collected 90 min prior to the competition, between 12:00 and 1:00 PM. Hormone assays were completed using enzyme immunoassay kits from Salimetrics (State College, PA). The inter-assay coefficient of variance (CV) for testosterone was 3.7% and for cortisol was 2.5%. If the intra-assay CV for a given sample was greater than 7.5%, the sample was assayed again; this occurred for seven samples.
Study 2
Saliva samples were collected after arrival to the laboratory between the hours of 11:30 AM and 4:30 PM. Hormone assays were completed at the Yerkes Biomarkers Laboratory (Emory University, Atlanta, GA) using radioimmunoassays kits from Diagnostic Systems Laboratories (Webster, TX). The inter-assay CVs for testosterone was 10.67% and for cortisol was 3.65%. The intra-assay CV for both testosterone and cortisol was less than 20%.
Study 3
Saliva samples were provided following a 30-min nature video (to best capture resting baseline) between the hours of 11:30 AM and 7:30 PM. Salivettes were used to obtain samples for cortisol and a separate passive drool sample was taken for testosterone. Hormone assays were completed using chemiluminescence-immunoassay kits from IBL International (Hamburg, Germany). Intra- and inter-assay CVs were below 10%.
Study 4
Saliva samples were provided following the completion of questionnaires for approximately 25–30 min between the hours of 11:00 AM and 5:00 PM. Hormone assays were completed using enzyme immunoassay kits from DRG international (Springfield, NJ). The inter-assay CV for testosterone was 9% and for cortisol was 6%. The intra-assay CV for testosterone was 6%, and for cortisol was 6%.
Study 5
Saliva samples were provided between 6:00 and 7:00 PM. Hormone assays were completed using enzyme immunoassays from Salimetrics (State College, PA). The inter-assay CV for testosterone was 5.85% and for cortisol was 6.39%. The intra-assay CV for testosterone was 3.84% and for cortisol 8.89%.
Study 6
Testosterone and cortisol concentrations were determined from hair samples with liquid chromatography tandem mass spectrometry (LC-MS/MS; Gao et al., 2013). Three hair strands were cut with scissors as close as possible from the scalp from a posterior vertex position and tied with a thread, placed in aluminum foils and sent to the Dresden Lab (Germany) for analyses. Inter- and intra-assay CVs for testosterone and cortisol were between 3.1% and 8.8%.
Study 7
Saliva samples were provided following approximately 15 min of completing questionnaires between 2:00 and 4:00 PM. Hormone assays were completed by Emory Clinical Translational Research Laboratory (Atlanta, GA) using enzyme immunoassay kits from Salimetrics (State College, PA). Inter-assay CV for testosterone for low and high controls were 18.0 and 6.2% (Intra-assay values could not be calculated).
Study 8
Saliva samples were collected between 11:00 AM and 5:00 PM. Hormone assays were completed using enzyme immunoassay kits from DRG International (Springfield, NJ). The inter-assay CV for testosterone was 16.59% and intra-assay CV was 9.19%.
Statistical Analysis Strategy
Data transformations and analyses
Testosterone concentrations were standardized within sex and winsorized to values at three SD of the meanFootnote 3. Cortisol concentrations were natural log-transformed, and subsequently standardized and winsorized to concentrations at 3 SD of the mean. These data transformations were largely consistent with the approach adopted by prior publications for each dataset. We winsorized outliers instead of excluding them because the former approach allowed us to retain all datapoints while accounting for their extreme place within the distribution of values.
Primary Analyses
Across eight studies, we tested the direct effects of testosterone (Studies 1, 7, & 8), and the interactive effects of testosterone and cortisol (Studies 2–6), predicting status-relevant behaviors using a mediation model. Because the purpose of the present study was to explore the explanatory role of personality traits in prior hormone-behavior findings, the decisions to test testosterone’s main effects or dual-hormone interactions were based on whether the original studies focused on direct effects of testosterone or dual-hormone interaction effects (see R code: https://osf.io/phgky/ for additional analyses with these datasets)Footnote 4.
To conduct the mediation analysis, we used the Baron and Kenny (1986) method (see also, Frazier et al., 2004; Judd & Kenny, 1981) in which multiple regressions were applied via a step-wise approach to explore each pathway in the mediation model. First, we tested the effects of hormones on status-relevant behaviors (path c of the mediation model; this step replicates prior analyses with these datasets). In the regression models examining the direct effects of testosterone, status-relevant behaviors were regressed on basal testosterone concentrations (standardized within sex). In the models testing the interactive effects of testosterone and cortisol, status-relevant behaviors were regressed on basal testosterone concentrations, basal cortisol, and their interaction. Second, we tested whether hormones predicted personality traits: personality traits (the outcome variable) were regressed on testosterone or testosterone × cortisol interactions (path a of the mediation model). Third, to examine the extent to which personality traits predicted status-relevant behaviors, controlling for hormone levels (path b of the mediation model), we regressed status-relevant behaviors on both personality traits and hormones (testosterone or the testosterone × cortisol interactions). Fourth, using a similar regression model as the previous step, we assessed whether hormones predicted status-relevant behaviors when controlling for personality traits (individually and all together in the same model; path c’ of the mediation model). This fourth step allowed us to determine the magnitude of mediation by comparing the hormonal effects on behavior when controlling for personality traits (standardized betas of path c’) to the original hormonal-behavioral effects when not controlling for personality traits (standardized betas of path c). The models with testosterone × cortisol interactions also controlled for the main effects of testosterone and cortisol.
We used the PROCESS macro in R to compute the effect size for mediation, i.e., the indirect effect (path a × path b), and to compute the bias-corrected bootstrapped confidence intervals of the indirect effect (Hayes, 2020; Hayes & Scharkow, 2013; Tibbe & Montoya, 2022). In the PROCESS models that tested mediation of the direct effect of testosterone, testosterone was treated as the independent variable, status-relevant behaviors as the outcomes, and self-reported personality traits as mediators (Model 4 in PROCESS). In the PROCESS models that tested mediation of the dual-hormone interaction, cortisol was included as a moderator for the paths of testosterone predicting self-reported personality, and of testosterone predicting behavior (Model 8 in PROCESS).
Secondary Analyses
Some prior work suggests that testosterone’s effects on status-relevant behaviors depend on levels of trait dominance (e.g., Carré et al., 2009; Geniole et al., 2019; Knight et al., 2017; Mehta, Son, et al.,2015; Slatcher et al., 2011), though other studies did not find clear evidence for trait dominance × testosterone interactions (e.g., Knight et al., 2022). For most of the studies included in this report (all but Study 1 and 6), trait dominance was one of the status-relevant personality traits that was assessed (Table 1). To contribute to the prior literature on the moderating role of trait dominance, we used linear regressions to test testosterone × trait dominance effects on status-relevant behaviors.
All statistical analyses were conducted in R (version 4.2.1). Linear regressions were conducted using the lm command in base R. Simple slope estimates and graphical visualization of the interactions were obtained using the interactions package (Long, 2021). Standardized coefficients for the linear regressions were also estimated using the sjplot package, which refits models on standardized data (Gelman, 2008; Lüdecke et al., 2021).
Results
Descriptive Statistics
Table S1 shows the descriptive statistics of status-relevant behaviors, self-reported personality traits, and raw testosterone and cortisol concentrations, across all studies.
Primary Analyses: Do Personality Traits Mediate hormone-behavior Associations?
Hormones predicting status-relevant behaviors (path c)
Across the eight datasets, testosterone independently (Studies 1, 7, and 8), and the testosterone × cortisol interaction (Studies 2–6), predicted a suite of status-relevant behaviors consistent with findings reported in the original studies (see Table 2 Column 1 and Table S2 Column 1 for a summary of hormone-behavior results across all studies). Further, these findings remained robust to the inclusion of relevant, non-personality covariates (see Tables S2 Column 2).
Hormones predicting self-reported personality traits (path a)
Generally, we did not find strong evidence that testosterone and testosterone × cortisol interactions predicted self-reported personality traits (see Table 2 Column 2 and Table S3). There were three exceptions to this general pattern. In Study 1, testosterone was negatively related to extraversion. The direction of this association is consistent with Sundin et al., (2021) but inconsistent with Smeets-Janssen et al. (2015). In Study 3, the testosterone × cortisol interaction predicted trait aggression with a positive slope, which is in the same direction as the behavioral effect found in that study. This positive interaction slope is also somewhat consistent with the interaction effect result for women in one previous study (Armstrong et al., 2021) but in the opposite direction from the interaction effects found in some other studies (Grotzinger et al., 2018; Popma et al., 2007). In Study 6, the testosterone × cortisol interaction in men predicted the dark triad of personality: at low levels of cortisol, testosterone was positively associated with the dark triad, but at high levels of cortisol, the testosterone-personality association was suppressed. This interaction effect was directionally inconsistent with the effect reported for one of the dark triad traits (trait psychopathy) in Welker et al., (2014); (cf. Pfattheicher, 2016 for null interaction effects).
Personality traits predicting status-relevant behaviors (path b)
Personality traits often predicted behavior in the expected direction both with the inclusion of hormones as covariates (path b; see Table 2 Column 3 and Table S4 Column 2) and without the inclusion of hormones as covariates (see Table S4 Column 1). In particular, trait agreeableness was positively related to affiliative behavior (Study 1); trait dominance, extraversion, and assertiveness were positively related to dominant leadership behavior (Study 2); trait prestige was positively related to social network centrality (betweenness, popularity, and gregariousness, Study 5); and trait competitiveness as well as trait dominance were positively related to competitive performance (Study 7). In some cases, the evidence for personality-behavior associations was more ambiguous. For example, in Study 3, the point estimate indicated a small positive relationship between trait aggression and aggressive behavior, which was in the expected direction, but the 95% CI overlapped with zero. We did not find any instance in which the personality-behavior association was robust yet in the opposite direction than expected.
Hormones predicting status-relevant behaviors controlling for self-reported personality traits (path c’)
Testosterone and testosterone × cortisol interactions predicted status-relevant behaviors over and above the inclusion of self-reported personality traits (entered individually and all together) as covariates (see Table 2 Column 4 and Table S5 Columns 1 and 2). To understand the directional pattern of the testosterone × cortisol effects, the interactions were further broken down into their simple slopes (see Table S5 Column 2) and are graphically represented (Figures S1 to S5) in the Supplement. Consistent with the original studies, there was some nuance in the direction of the dual-hormone effect. Studies 2, 4, 5, and male (but not female) participants in Study 6 showed a testosterone × cortisol interaction effect with a negative slope in line with the original formulation of the dual hormone hypothesis: relatively high testosterone was positively related to status-related behavior, but only among those with relatively low cortisol. Study 3 results showed a dual-hormone interaction effect with a positive slope (a “reverse” dual-hormone effect), where higher testosterone positively predicted reactive aggression only for those relatively high in cortisol (consistent with the original findings in Denson et al., 2013).
The effect sizes (standardized betas) of the testosterone and testosterone × cortisol interaction effects controlling for personality traits were consistent with the size for the original hormonal effects that did not include personality covariates (see Table 2 Column 4 for standardized betas). The 95% CIs of the standardized betas in the models that included personality variables generally included the standardized beta of the original hormonal effects (see Table 2 Columns 1 and 4). We also found that these models were generally robust to the inclusion of non-personality covariates (e.g., time of day), in addition to the personality covariates (see Table S5 Column 3).
Simple slopes analyses for the dual-hormone models that included personality traits also provided support for testosterone-behavior associations at high vs. low levels of cortisol in the same direction as the original findings (see Table S5 Column 2 and Figures S1 to S5). We also computed the median percentage change in the standardized beta point estimates for the main effects of testosterone and the testosterone × cortisol interaction effects reported in Table 2 Column 1 (models with no personality covariates) versus Column 4 (models with personality covariates included). There was a median shrinkage of 4.35% in the standardized betas when personality covariates were added to the models. These analyses indicate that the hormone-behavior associations were of similar magnitude in models with and without personality covariates.
Indirect effects of basal hormones on status-relevant behaviors via self-reported personality traits
Finally, we explored the indirect effect of self-reported personality traits mediating the relationship of testosterone and testosterone × cortisol interactions on status-relevant behaviors. We did not find support for an indirect personality-mediated pathway between the hormones and behaviors (see Table 2 Column 5). That is, personality traits did not significantly mediate the pathway between the direct effects of testosterone, and the interactive effects of testosterone and cortisol, on status-relevant behaviors.
In summary, self-reported personality traits often modestly predicted behavioral outcome measures in the expected directions (path b), even when controlling for hormone concentrations. However, there was little evidence for hormones predicting self-reported personality traits (path a). More importantly, hormones continued to predict status-relevant behaviors even after controlling for personality traits, and these hormone-behavior relationships had the same directional pattern and similar effect sizes as the original effects. The clear lack of evidence for a personality-mediated pathway between hormones and behavior may highlight the presence of alternative pathways for hormone-behavior relationships. We return to this point in the discussion.
Secondary Analyses: Moderation by Trait Dominance
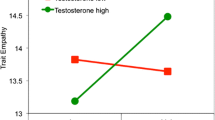
In 5 of the 6 datasets, self-reported trait dominance did not significantly moderate the effects of testosterone on status-relevant behaviors (see Table S6 for key statistics from individual models and Figures S6 to S11 for the directional pattern). However, in Study 4, there was a significant testosterone × trait dominance interaction in the opposite of the expected direction: for those relatively high in dominance, basal testosterone was negatively related to risk-taking (Fig S8). Collectively, there is not strong and consistent evidence that trait dominance moderates the association between basal testosterone and status-oriented behavior, although this effect may be specific to the particular task and behavior.
Discussion
The broad purpose of the present research was to examine the role of personality in explaining previously-identified relationships between basal hormones and status-related behaviors. Data from eight prior studies on basal testosterone and basal cortisol as predictors of status-relevant behavior were re-examined with new analyses that included self-reported status-related personality traits (total N = 985). Self-reported traits were often correlated with the behavioral outcomes in the expected direction. For example, trait dominance was positively related to dominant leadership behavior in a videotaped interaction (Study 2), and trait competitiveness was positively related to competitive performance in a weight-holding task (Study 7). However, the collective results show no compelling evidence that personality mediates the hormone-behavior relationships that were tested. Indeed, the magnitude of these relationships was largely unaffected by the inclusion of personality in the models. That is, the direct effect of basal testosterone and the interactive effect of basal testosterone and basal cortisol on status-relevant behavior emerged above and beyond the variance explained by personality.
These consistent findings across eight studies, along with related evidence in previous research, provide insight into the mechanisms for hormone-behavior relationships (Akinola et al., 2016; Geniole et al., 2013; Grebe et al., 2019a; Josephs et al., 2006; Lee et al., 2015; Sundin et al., 2021). One potential explanation for these results is that hormone regulation of behavior may be mechanistically independent from self-reported personality. That is, it is possible that the physiological effects of testosterone and cortisol on neural pathways that regulate behavior do not simultaneously act directly on perceptual systems involved in conscious awareness. Self-reported personality questionnaires are explicit measures that involve reporting one’s consciously available, stable self-perceptions. Thus, while behaviors play an important role in forming these explicit self-perceptions over time, hormone-behavior relationships could be unconscious, automatic, and independent of self-reported personality. An implicit pathway for testosterone-behavior associations is supported by evidence from neuroscientific research (Bos et al., 2012; Dedovic et al., 2009; Duell et al., 2021; Losecaat Vermeer et al., 2016; Putman et al., 2007; Radke et al., 2015; Terburg & van Honk, 2013; Volman et al., 2011).
An implicit mechanism for testosterone and cortisol’s effects on behavior is also in line with prior research and theorizing about hormonal modulation of implicit dominance motives, threat perception, and stress response (Köllner et al., 2018; Mossink et al., 2015; Quirin et al., 2009; Schultheiss, 2013; Stanton & Schultheiss, 2009; van Honk et al., 2004; Wegner et al., 2015). For example, Terburg et al. (2012) found that participants given testosterone treatment compared to placebo showed significantly greater social dominance in a non-conscious (rapidly shown) face gaze-aversion task but showed no differences in self-reported affective state. Complementary research by Wirth and Schultheiss (2007) found that endogenous testosterone was positively related to instrumental learning in responses to angry faces when the faces were presented outside of conscious awareness, but testosterone was unrelated to learning when faces were presented supraliminally (enabling conscious processing). Another study found that exogenous testosterone affected an automatic early threat bias (within 200 milliseconds of face presentation), but not later neural processes (greater than 200 milliseconds; van Peer et al., 2017).
Cortisol also appears to influence behavior implicitly. Across a variety of validated self-report measures including real-time sampling, cortisol is either unrelated or only weakly related to subjective experiences of stress (Gidlow et al., 2016; Joseph et al., 2021; Schmidt et al., 2020; Wells et al., 2014). Studies that have employed both explicit and implicit measures of stress-related mood and affect showed that the implicit tests predicted various metrics of basal and reactive cortisol, but the explicit tests did not (Mossink et al., 2015; Quirin et al., 2009). One such study even found that a measure of implicit power motivation, but not explicit dominance, predicted cortisol reactivity to psychosocial stress (Wegner et al., 2015). Thus, testosterone and cortisol and their interaction may implicitly relate to behavior largely independent of explicitly measured, i.e., self-reported, personality traits.
The endocrine system is understood to operate by coordinating adaptive and context-relevant behavioral responses to specific environmental stimuli and conditions. Whereas self-reflection and social learning can provide us with a generally accurate understanding of how we typically behave in most contexts (as reflected in self-reported personality traits), hormone levels may reflect more automatic and reflexive needs and motives that are activated “in the heat of the moment” within specific contexts. Indeed, context tends to play an essential role in moderating hormone-behavior effects (e.g., Carré & Archer, 2018; Duell et al., 2021; Josephs et al., 2006; Losecaat Vermeer et al., 2020; Prasad et al., 2017). In contrast, personality is, by definition, relatively context-independent. Thus, it is possible that hormonal effects on behavior bypass the general self-knowledge indexed by self-reported personality traits and instead, influence behavior through context-dependent mechanisms that occur outside of conscious awareness. Although the present research cannot speak to context-dependence directly, future research should be conducted to compare variability in hormone-behavior relationships and personality-behavior relationships across different social contexts.
Some previous studies have found that self-reported trait dominance moderates the association between testosterone and status-relevant behavior (Carré et al., 2009, 2017; Geniole et al., 2019; Knight et al., 2017; Mehta et al., 2015c; Slatcher et al., 2011). Specifically, testosterone has shown stronger effects on dominance, aggression, and competitive behavior among individuals high in trait dominance. Other studies, however, have not found robust evidence for trait dominance × testosterone interactions in predicting status-relevant behavior (e.g., Knight et al., 2022; Casto et al., 2020). In the present research, in secondary analyses, we examined whether an effect of basal testosterone on status-relevant behavior depends on individual differences in trait dominance. We found mixed support. In one study, higher basal testosterone predicted more risk-taking behavior, but only among those low in trait dominance; for those high in trait dominance, higher basal testosterone predicted less risk-taking (see Figure S8). In another study, higher basal testosterone was modestly related to higher persistence behavior especially among those high in trait dominance, but the confidence interval of the interaction term slightly overlapped with zero (see Figure S11). In the other studies, the trait dominance × testosterone effect sizes were small with confidence intervals that overlapped substantially with zero and with no consistent pattern in the direction of the interaction effects (see Figures S6, S7, S9, and S10). Collectively, this mixed pattern of evidence suggests that trait dominance × testosterone interaction effects may not be robust. We recommend additional research that further examines this hormone by trait interaction effect to identify important boundary conditions.
Limitations and Future Directions
In all but one of the eight studies, basal hormone levels were determined by a single sample of saliva at baseline. Although basal hormone levels are relatively stable day-to-day within an individual, multiple samples at rest would produce a more valid estimate of individual differences in testosterone and cortisol (e.g., Crewther et al., 2020). Study 6 examined hormone levels from hair. Hair hormone levels could be a more useful test than saliva for estimating long-term testosterone and cortisol exposure levels in future research (Greff et al., 2019; Grotzinger et al., 2018; Shields et al., 2021). Further, salivary hormone concentrations were assessed using immunoassays in all but one of the eight studies. Recent work suggests that immunoassays may be limited in their capacity to accurately measure salivary testosterone, especially at low levels (e.g., in females; Chafkin et al., 2022; Prasad et al., 2019; Taieb et al., 2003). Future studies may consider using mass spectrometry to assess testosterone and cortisol concentrations (Schultheiss et al., 2019), as was done in Study 6.
Our theorizing about implicit mechanisms for hormonal influences on behavior versus the explicit nature of self-reported traits could be bolstered by directly testing how testosterone and cortisol predict self-reported personality in comparison to implicit motive tests (as in Mossink et al., 2015; Wegner et al., 2015). This theorizing could also be bolstered by neuroscience studies that directly examine hormone-brain-behavior pathways in human participants (e.g., Duell et al., 2021; Mehta & Beer, 2010).
Self-reported traits capture people’s understanding of how they generally behave across various situations. However, knowledge about general behavior and awareness of one’s behavior in the moment are different cognitive experiences and the relevant self-knowledge is formed across different timescales. As argued earlier, hormone levels may influence behavior through automatic and reflexive mechanisms that are activated “in the heat of the moment” within specific contexts. It is possible that self-reported states, rather than traits, capture part of this contextually-activated mechanism for hormone-behavior associations. This possibility can be addressed in future work by measuring relevant states (e.g., personality states, Kordsmeyer & Penke, 2019; reward-related states, Geniole et al., 2019; Mehta et al., 2017; Mehta, Snyder, Mehta et al. 2015, b).
In the current research, there was no compelling evidence that self-reported personality traits mediated the observed hormone-behavior associations. This collective pattern across eight studies is consistent with some initial research (Akinola et al., 2016; Geniole et al., 2013; Josephs et al., 2006; Lee et al., 2015). Mediation effects could be detected for other hormones, traits, or behaviors in future studies. Further, if mediation effects are non-zero but very small, they may be detected in future studies with larger sample sizes. These larger studies would also enable adequate tests of more complex three-way interaction models that include hormones, personality variables, and contextual factors (e.g., win/loss).
The current project focused on basal hormone levels. We did not find compelling evidence that personality traits mediated basal hormone-behavior associations, but personality may still be involved in explaining acute hormone reactivity (for discussion, see Carré & Archer, 2018; Casto et al., 2020; Kordsmeyer & Penke, 2019). For example, self-reported personality traits have been shown to predict acute testosterone and cortisol reactivity to competition (e.g., Geniole & Carré, 2018; Kordsmeyer & Penke, 2019; Mehta, Mor, et al.,2015a). Furthermore, acute testosterone reactivity alone or in interaction with acute cortisol reactivity have been shown to mediate associations between self-reported personality traits and competitive behaviors (Casto et al., 2020; Mehta, Mor, et al.,2015a). These findings suggest that acute hormone changes may serve as biological mechanisms for personality-behavior relationships. Thus, personality might determine who finds a particular context status-relevant or stressful, indicating for whom acute testosterone and cortisol response is activated. These personality-driven biological changes, in turn, are expected to influence subsequent status-relevant behavior (Casto & Edwards, 2021; Geniole et al., 2019; Mehta, Mor, et al., 2015a). Future projects can be designed to provide a more in-depth understanding of personality traits as predictors of acute hormone reactivity in status-relevant contexts.
Across all studies included in this report, personality traits were measured with self-report inventories. Although self-report is the most common approach to personality measurement and self-reported traits do indeed predict behavior, there are other approaches to personality measurement, including informant reports (Vazire, 2010) and the analysis of online social media profiles (Facebook: Back et al., 2010). Some of these other approaches to personality assessment capture variance in behavioral outcomes above and beyond self-report questionnaires (Back & Vazire, 2015; Vazire, 2010). Thus, it is possible that personality assessment beyond self-report may partially explain hormone-behavior associations, which can be tested in future studies.
This project used cross-sectional correlational designs, and thus, the causal pathways linking hormones, personality, and behavior remain unclear. Future longitudinal and experimental studies would be helpful to enable causal inference. However, it is challenging to conduct relevant studies that allow for strong causal conclusions. For example, existing pharmacological challenge paradigms involve acute changes in hormone levels, whereas experimental manipulation of basal hormone levels in humans likely requires different approaches such as long-term hormone administration or chemical castration in clinical samples. Even if a researcher could overcome the feasibility challenges of doing such work, it is unclear whether these experimental approaches are directly comparable to naturally occurring endogenous hormone levels in healthy volunteers. Similarly, personality traits are difficult to manipulate experimentally on short timescales, though longer interventions such as clinical therapy have been shown to alter personality traits (Roberts et al., 2017). Thus, although we recommend experimental studies to examine causality, we acknowledge that this type of work has its own limitations and challenges.
Hormone-behavior relationships varied across the eight studies we re-examinedFootnote 6. Basal testosterone significantly predicted behavior in some of the studies, whereas the interaction between basal testosterone and cortisol was a significant predictor of behavior in other studies. Further, the direction of the effects were inconsistent; dual-hormone interactions went in opposite directions across some of the studies. One plausible explanation for these variable effects is context-dependence. As discussed earlier, personality is, by definition, relatively context independent, whereas context plays an important role in moderating hormone-behavior relationships (e.g., Carré & Archer, 2018; Duell et al., 2021; Josephs et al., 2006; Losecaat Vermeer et al., 2020; Knight et al., 2022; Prasad et al., 2017). The lack of compelling evidence for personality mediation of hormone-behavior pathways in the eight studies presented here is consistent with the notion that hormones may influence behavior through context-dependent neural pathways, rather than context-independent, personality-mediated pathways. Future research is required to identify the specific contextual factors that explain apparent inconsistencies in hormone-behavior effects.
Conclusion
Behavioral neuroendocrinology aims to understand how hormones influence behavior. The present research investigated the role of self-reported personality traits in explaining hormone-behavior relationships. We examined eight prior studies of basal testosterone and basal cortisol as predictors of status-relevant behavior and tested whether these effects were mediated by status-relevant self-reported personality traits. We did not find compelling evidence that self-reported personality traits mediated the hormone-behavior relationships. Basal testosterone and its interaction with basal cortisol predicted status-relevant behavior, but in most cases, did not significantly predict self-reported personality traits. Although personality traits were often associated with the related behaviors in the predicted direction, the magnitude of the hormone-behavior relationships were robust to the inclusion of these explicit trait factors. Further, there was no overall compelling and consistent evidence that the testosterone-behavior relationships depended on individual differences in trait dominance.
In line with prior theorizing, we discuss these results in light of two proposed explanations: (1) that hormonal regulation of behavior occurs implicitly, primarily outside of conscious awareness, and (2) that hormone-behavior relationships are context-dependent, whereas personality-behavior relationships are consistent across contexts. In advancing theorizing about testosterone’s direct and interactive effect with cortisol on status-relevant behavior, this research and discussion contributes to a broader understanding of hormone-behavior relationships.
Materials and Code availability
The datasets for this study and code used analyze the data are available at the Open Science Framework repository: https://osf.io/phgky/.
Change history
21 March 2023
A Correction to this paper has been published: https://doi.org/10.1007/s40750-023-00214-1
Notes
Although the current study is focused on basal hormone levels, some research suggests that effect sizes for associations between context-specific acute testosterone changes and behavior may be larger compared to effect sizes for associations between basal testosterone levels and behavior (e.g., Geniole et al., 2020; Wingfield et al., 1990).
This conclusion comes from the published paper as well as personal communication with the first author. As reported earlier in the introduction, the basal testosterone x basal cortisol interaction slope was positive in this study, consistent with a “reverse” dual-hormone effect.
In Study 3, testosterone concentrations were log-transformed prior to standardization because skew in the distribution contributed to violations of regression model assumptions; this approach was consistent that adopted in the original paper (Denson et al., 2013). In Study 6, testosterone and cortisol concentrations were log-transformed, not standardized, and analyzed separately across men and women, consistent with the approach adopted by the original paper (Ronay et al., 2018).
In Study 1, while cortisol was assayed and is reported in two other publications (Mehta et al., 2008; Sherman et al., 2017), cortisol change rather than basal cortisol was the focus in these prior papers, and neither of these papers examined interactions between basal cortisol and basal testosterone to test the dual-hormone hypothesis. In Study 7, cortisol levels were assayed but not reported in any published manuscript. We include additional R code (https://osf.io/phgky/) for tests of the dual-hormone interaction hypothesis in these datasets.
Results are reported separately across males (M) and females (F) (consistent with that reported in the original paper; Ronay et al., 2018).
This conclusion is based on a qualitative assessment of the results. It is beyond the scope of the present research to quantify the degree of variability in the hormone-behavior associations, but future research can do so by estimating variance components across a given set of studies using a meta-analytic framework (Knight, McShane, et al., 2020a).
References
Akinola, M., Page-Gould, E., Mehta, P. H., & Lu, J. G. (2016). Collective hormonal profiles predict group performance. Proceedings of the National Academy of Sciences, 113(35), 9774–9779. https://doi.org/10.1073/pnas.1603443113
Anderson, C., & Kilduff, G. J. (2009). Why do dominant personalities attain influence in face-to-face groups? The competence-signaling effects of trait dominance. Journal of Personality and Social Psychology, 96, 491–503. https://doi.org/10.1037/a0014201.
Archer, J. (2006). Testosterone and human aggression: an evaluation of the challenge hypothesis. Neuroscience & Biobehavioral Reviews, 30(3), 319–345. https://doi.org/10.1016/j.neubiorev.2004.12.007.
Armstrong, T., Wells, J., Boisvert, D. L., Lewis, R. H., Cooke, E. M., Woeckener, M., & Kavish, N. (2021). An exploratory analysis of testosterone, cortisol, and aggressive behavior type in men and women. Biological Psychology, 161, 108073. https://doi.org/10.1016/j.biopsycho.2021.108073.
Back, M. D., Stopfer, J. M., Vazire, S., Gaddis, S., Schmukle, S. C., Egloff, B., & Gosling, S. D. (2010). Facebook Profiles reflect actual personality, not self-idealization. Psychological Science, 21(3), 372–374. https://doi.org/10.1177/0956797609360756.
Back, M. D., & Vazire, S. (2015). The Social Consequences of personality: six suggestions for Future Research. European Journal of Personality, 29(2), 296–307. https://doi.org/10.1002/per.1998.
Baron, R. M., & Kenny, D. A. (1986). The moderator–mediator variable distinction in social psychological research: conceptual, strategic, and statistical considerations. Journal of Personality and Social Psychology, 51, 1173–1182. https://doi.org/10.1037/0022-3514.51.6.1173.
Bertsch, K., Böhnke, R., Kruk, M. R., Richter, S., & Naumann, E. (2011). Exogenous cortisol facilitates responses to social threat under high provocation. Hormones and Behavior, 59(4), 428–434. https://doi.org/10.1016/j.yhbeh.2010.12.010.
Bos, P. A., Panksepp, J., Bluthé, R. M., & van Honk, J. (2012). Acute effects of steroid hormones and neuropeptides on human social–emotional behavior: a review of single administration studies. Frontiers in Neuroendocrinology, 33(1), 17–35. https://doi.org/10.1016/j.yfrne.2011.01.002.
Buades-Rotger, M., Engelke, C., Beyer, F., Keevil, B. G., Brabant, G., & Krämer, U. M. (2016). Endogenous testosterone is associated with lower amygdala reactivity to angry faces and reduced aggressive behavior in healthy young women. Scientific Reports, 6(1), https://doi.org/10.1038/srep38538.
Buss, A. H., & Perry, M. (1992). The Aggression Questionnaire. Journal of Personality and Social Psychology, 63, 452–459. https://doi.org/10.1037/0022-3514.63.3.452.
Buss, D. M., & Craik, K. H. (1980). The frequency concept of disposition: dominance and prototypically dominant acts1. Journal of Personality, 48(3), 379–392. https://doi.org/10.1111/j.1467-6494.1980.tb00840.x.
Carré, J. M., & Archer, J. (2018). Testosterone and human behavior: the role of individual and contextual variables. Current Opinion in Psychology, 19, 149–153. https://doi.org/10.1016/j.copsyc.2017.03.021.
Carré, J. M., Geniole, S. N., Ortiz, T. L., Bird, B. M., Videto, A., & Bonin, P. L. (2017). Exogenous testosterone rapidly increases aggressive Behavior in Dominant and Impulsive Men. Biological Psychiatry, 82(4), 249–256. https://doi.org/10.1016/j.biopsych.2016.06.009.
Carré, J. M., Putnam, S. K., & McCormick, C. M. (2009). Testosterone responses to competition predict future aggressive behaviour at a cost to reward in men. Psychoneuroendocrinology, 34(4), 561–570. https://doi.org/10.1016/j.psyneuen.2008.10.018.
Casciaro, T., Carley, K. M., & Krackhardt, D. (1999). Positive affectivity and accuracy in Social Network Perception. Motivation and Emotion, 23(4), 285–306. https://doi.org/10.1023/A:1021390826308.
Casto, K. V., Arthur, L. C., Hamilton, D. K., & Edwards, D. A. (2021). Testosterone, athletic context, oral contraceptive use, and competitive persistence in women. Adaptive Human Behavior and Physiology. https://doi.org/10.1007/s40750-021-00180-6.
Casto, K. V., & Edwards, D. A. (2021). Individual differences in hormonal responsiveness to social encounters: Commentary on Félix et al., 2020 and review of pertinent issues. Hormones and Behavior, 129, 104921. https://doi.org/10.1016/j.yhbeh.2020.104921
Casto, K. V., Edwards, D. A., Akinola, M., Davis, C., & Mehta, P. H. (2020). Testosterone reactivity to competition and competitive endurance in men and women. Hormones and Behavior, 123, 104665. https://doi.org/10.1016/j.yhbeh.2019.104665.
Casto, K. V., & Mehta, P. H. (2019). Competition, Dominance, and Social Hierarchy. In The Oxford Handbook of Evolutionary Psychology and Behavioral Endocrinology, Lisa L. M. Welling and Todd K. Shackelford (Eds). https://doi.org/10.1093/oxfordhb/9780190649739.013.16
Chafkin, J. E., O’Brien, J. M., Medrano, F. N., Lee, H. Y., Yeager, D. S., & Josephs, R. A. (2022). Chemiluminescent immunoassay overestimates hormone concentrations and obscures testosterone sex differences relative to LC-MS/MS in a field study of diverse adolescents. Comprehensive Psychoneuroendocrinology, 10, 100132. https://doi.org/10.1016/j.cpnec.2022.100132.
Cheng, J. T., Tracy, J. L., & Henrich, J. (2010). Pride, personality, and the evolutionary foundations of human social status. Evolution and Human Behavior, 31(5), 334–347. https://doi.org/10.1016/j.evolhumbehav.2010.02.004.
Costa, P. T., & McCrae, R. R. (2008). The revised NEO personality inventory (NEO-PI-R). The SAGE handbook of personality theory and assessment, vol 2: personality measurement and testing (pp. 179–198). Sage Publications, Inc. https://doi.org/10.4135/9781849200479.n9.
Crewther, B. T., Hecht, M., Potts, N., Kilduff, L. P., Drawer, S., & Cook, C. J. (2020). A longitudinal investigation of bidirectional and time-dependent interrelationships between testosterone and training motivation in an elite rugby environment. Hormones and Behavior, 126, 104866. https://doi.org/10.1016/j.yhbeh.2020.104866.
Daly, M., & Wilson, M. (1988). Homicide. Aldine de Gruyter.
Dedovic, K., Duchesne, A., Andrews, J., Engert, V., & Pruessner, J. C. (2009). The brain and the stress axis: the neural correlates of cortisol regulation in response to stress. Neuroimage, 47(3), 864–871. https://doi.org/10.1016/j.neuroimage.2009.05.074.
Dekkers, T. J., van Rentergem, J. A. A., Meijer, B., Popma, A., Wagemaker, E., & Huizenga, H. M. (2019). A meta-analytical evaluation of the dual-hormone hypothesis: does cortisol moderate the relationship between testosterone and status, dominance, risk taking, aggression, and psychopathy? Neuroscience & Biobehavioral Reviews, 96, 250–271. https://doi.org/10.1016/j.neubiorev.2018.12.004.
Denson, T. F., Capper, M. M., Oaten, M., Friese, M., & Schofield, T. P. (2011). Self-control training decreases aggression in response to provocation in aggressive individuals. Journal of Research in Personality, 45(2), 252–256. https://doi.org/10.1016/j.jrp.2011.02.001.
Denson, T. F., Mehta, P. H., & Ho Tan, D. (2013). Endogenous testosterone and cortisol jointly influence reactive aggression in women. Psychoneuroendocrinology, 38(3), 416–424. https://doi.org/10.1016/j.psyneuen.2012.07.003.
Denson, T. F., von Hippel, W., Kemp, R. I., & Teo, L. S. (2010). Glucose consumption decreases impulsive aggression in response to provocation in aggressive individuals. Journal of Experimental Social Psychology, 46(6), 1023–1028. https://doi.org/10.1016/j.jesp.2010.05.023.
Dickerson, S. S., Gruenewald, T. L., & Kemeny, M. E. (2009). Psychobiological responses to Social Self threat: functional or detrimental? Self and Identity, 8(2–3), 270–285. https://doi.org/10.1080/15298860802505186.
Duell, N., van Hoorn, J., McCormick, E. M., Prinstein, M. J., & Telzer, E. H. (2021). Hormonal and neural correlates of prosocial conformity in adolescents. Developmental Cognitive Neuroscience, 48, 100936. https://doi.org/10.1016/j.dcn.2021.100936.
Eisenegger, C., Haushofer, J., & Fehr, E. (2011). The role of testosterone in social interaction. Trends in Cognitive Sciences, 15(6), 263–271. https://doi.org/10.1016/j.tics.2011.04.008.
Ellis, B. J., Giudice, D., Dishion, M., Figueredo, T. J., Gray, A. J., Griskevicius, P., Hawley, V., Jacobs, P. H., James, W. J., Volk, J., A. A., & Wilson, D. S. (2012). The evolutionary basis of risky adolescent behavior: implications for science, policy, and practice. Developmental Psychology, 48, 598–623. https://doi.org/10.1037/a0026220.
Frazier, P. A., Tix, A. P., & Barron, K. E. (2004). Testing Moderator and Mediator Effects in Counseling psychology research. Journal of Counseling Psychology, 51, 115–134. https://doi.org/10.1037/0022-0167.51.1.115.
Gelman, A. (2008). Scaling regression inputs by dividing by two standard deviations. Statistics in Medicine, 27(15), 2865–2873. https://doi.org/10.1002/sim.3107.
Geniole, S. N., Bird, B. M., McVittie, J. S., Purcell, R. B., Archer, J., & Carré, J. M. (2020). Is testosterone linked to human aggression? A meta-analytic examination of the relationship between baseline, dynamic, and manipulated testosterone on human aggression. Hormones and Behavior, 123, 104644. https://doi.org/10.1016/j.yhbeh.2019.104644.
Geniole, S. N., Busseri, M. A., & McCormick, C. M. (2013). Testosterone dynamics and psychopathic personality traits independently predict antagonistic behavior towards the perceived loser of a competitive interaction. Hormones and Behavior, 64(5), 790–798. https://doi.org/10.1016/j.yhbeh.2013.09.005.
Geniole, S. N., & Carré, J. M. (2018). Human social neuroendocrinology: review of the rapid effects of testosterone. Hormones and Behavior, 104, 192–205. https://doi.org/10.1016/j.yhbeh.2018.06.001.
Geniole, S. N., Procyshyn, T. L., Marley, N., Ortiz, T. L., Bird, B. M., Marcellus, A. L., Welker, K. M., Bonin, P. L., Goldfarb, B., Watson, N. V., & Carré, J. M. (2019). Using a Psychopharmacogenetic Approach to identify the Pathways through which—and the people for whom—testosterone promotes aggression. Psychological Science, 30(4), 481–494. https://doi.org/10.1177/0956797619826970.
Gidlow, C. J., Randall, J., Gillman, J., Silk, S., & Jones, M. V. (2016). Hair cortisol and self-reported stress in healthy, working adults. Psychoneuroendocrinology, 63, 163–169. https://doi.org/10.1016/j.psyneuen.2015.09.022.
Goldberg, L. R., Johnson, J. A., Eber, H. W., Hogan, R., Ashton, M. C., Cloninger, C. R., & Gough, H. G. (2006). The international personality item pool and the future of public-domain personality measures. Journal of Research in Personality, 40(1), 84–96. https://doi.org/10.1016/j.jrp.2005.08.007.
Gosling, S. D., Rentfrow, P. J., & Swann, W. B. (2003). A very brief measure of the big-five personality domains. Journal of Research in Personality, 37(6), 504–528. https://doi.org/10.1016/S0092-6566(03)00046-1.
Gray, P. B., Straftis, A. A., Bird, B. M., McHale, T. S., & Zilioli, S. (2020). Human reproductive behavior, life history, and the challenge hypothesis: a 30-year review, retrospective and future directions. Hormones and Behavior, 123, 104530. https://doi.org/10.1016/j.yhbeh.2019.04.017.
Grebe, N. M., Giudice, D., Emery Thompson, M., Nickels, M., Ponzi, N., Zilioli, D., Maestripieri, S., D., & Gangestad, S. W. (2019a). Testosterone, cortisol, and status-striving personality features: a review and empirical evaluation of the dual hormone hypothesis. Hormones and Behavior, 109, 25–37. https://doi.org/10.1016/j.yhbeh.2019a.01.006.
Grebe, N. M., Sarafin, R. E., Strenth, C. R., & Zilioli, S. (2019b). Pair-bonding, fatherhood, and the role of testosterone: a meta-analytic review. Neuroscience & Biobehavioral Reviews, 98, 221–233. https://doi.org/10.1016/j.neubiorev.2019b.01.010.
Greff, M. J. E., Levine, J. M., Abuzgaia, A. M., Elzagallaai, A. A., Rieder, M. J., & van Uum, S. H. M (2019). Hair cortisol analysis: an update on methodological considerations and clinical applications. Clinical Biochemistry, 63, 1–9. https://doi.org/10.1016/j.clinbiochem.2018.09.010.
Grotzinger, A. D., Mann, F. D., Patterson, M. W., Tackett, J. L., Tucker-Drob, E. M., & Harden, K. P. (2018). Hair and salivary testosterone, Hair Cortisol, and externalizing behaviors in adolescents. Psychological Science, 29(5), 688–699. https://doi.org/10.1177/0956797617742981.
Hayes, A. F. (2020). Introduction to Mediation, Moderation, and conditional process analysis, Second Edition: a regression-based Approach. Guilford Publications.
Hayes, A. F., & Scharkow, M. (2013). The relative trustworthiness of Inferential tests of the Indirect Effect in Statistical Mediation Analysis: does Method really Matter? Psychological Science, 24(10), 1918–1927. https://doi.org/10.1177/0956797613480187.
Houston, J., Harris, P., McIntire, S., & Francis, D. (2002). Revising the Competitiveness Index using factor analysis. Psychological Reports, 90(1), 31–34. https://doi.org/10.2466/pr0.2002.90.1.31.
Jackson, D. N. (1974). Personality research form manual (3rd ed.). Research Psychologists Press.
Jones, A. C., & Josephs, R. A. (2006). Interspecies hormonal interactions between man and the domestic dog (Canis familiaris). Hormones and Behavior, 50(3), 393–400. https://doi.org/10.1016/j.yhbeh.2006.04.007.
Jones, D. N., & Paulhus, D. L. (2014). Introducing the short Dark Triad (SD3): a brief measure of Dark personality traits. Assessment, 21(1), 28–41. https://doi.org/10.1177/1073191113514105.
Josephs, R. A., Sellers, J. G., Newman, M. L., & Mehta, P. H. (2006). The mismatch effect: when testosterone and status are at odds. Journal of Personality and Social Psychology, 90(6), 999–1013. https://doi.org/10.1037/0022-3514.90.6.999.
Judd, C. M., & Kenny, D. A. (1981). Process analysis: estimating mediation in treatment evaluations. Evaluation Review, 5(5), 602–619. https://doi.org/10.1177/0193841X8100500502.
Kemeny, M. E. (2009). Psychobiological responses to social threat: evolution of a psychological model in psychoneuroimmunology. Brain Behavior and Immunity, 23(1), 1–9. https://doi.org/10.1016/j.bbi.2008.08.008.
Knight, E. L., Christian, C. B., Morales, P. J., Harbaugh, W. T., Mayr, U., & Mehta, P. H. (2017). Exogenous testosterone enhances cortisol and affective responses to social-evaluative stress in dominant men. Psychoneuroendocrinology, 85, 151–157. https://doi.org/10.1016/j.psyneuen.2017.08.014.
Knight, E. L., McShane, B. B., Kutlikova, H. H., Morales, P. J., Christian, C. B., Harbaugh, W. T., Mayr, U., Ortiz, T. L., Gilbert, K., Ma-Kellams, C., Riečanský, I., Watson, N. V., Eisenegger, C., Lamm, C., Mehta, P. H., & Carré, J. M. (2020a). Weak and Variable Effects of Exogenous Testosterone on Cognitive Reflection Test Performance in Three Experiments: Commentary on Nave, Nadler, Zava, and Camerer (2017). Psychological Science, 31(7), 890–897. https://doi.org/10.1177/0956797619885607
Knight, E. L., Morales, P. J., Christian, C. B., Prasad, S., Harbaugh, W. T., Mehta, P. H., & Mayr, U. (2022). The causal effect of testosterone on men’s competitive behavior is moderated by basal cortisol and cues to an opponent’s status: Evidence for a context-dependent dual-hormone hypothesis. Journal of Personality and Social Psychology, No Pagination Specified-No Pagination Specified. https://doi.org/10.1037/pspa0000305
Knight, E. L., Sarkar, A., Prasad, S., & Mehta, P. H. (2020b). Beyond the challenge hypothesis: the emergence of the dual-hormone hypothesis and recommendations for future research. Hormones and Behavior, 123, 104657. https://doi.org/10.1016/j.yhbeh.2019.104657.
Köllner, M. G., Janson, K. T., & Bleck, K. (2018). The social biopsychology of implicit motive development. Mehta (eds.) Routledge International Handbook of Social Neuroendocrinology. Routledge. O. C. Schultheiss.
Kordsmeyer, T. L., & Penke, L. (2019). Effects of male testosterone and its interaction with cortisol on self- and observer-rated personality states in a competitive mating context. Journal of Research in Personality, 78, 76–92. https://doi.org/10.1016/j.jrp.2018.11.001.
Krackhardt, D. (1987). Cognitive social structures. Social Networks, 9(2), 109–134. https://doi.org/10.1016/0378-8733(87)90009-8.
Kurath, J., & Mata, R. (2018). Individual differences in risk taking and endogeneous levels of testosterone, estradiol, and cortisol: a systematic literature search and three independent meta-analyses. Neuroscience & Biobehavioral Reviews, 90, 428–446. https://doi.org/10.1016/j.neubiorev.2018.05.003.
Lee, J. J., Gino, F., Jin, E. S., Rice, L. K., & Josephs, R. A. (2015). Hormones and ethics: understanding the biological basis of unethical conduct. Journal of Experimental Psychology: General, 144, 891–897. https://doi.org/10.1037/xge0000099.
Lejuez, C. W., Aklin, W. M., Zvolensky, M. J., & Pedulla, C. M. (2003). Evaluation of the Balloon Analogue Risk Task (BART) as a predictor of adolescent real-world risk-taking behaviours. Journal of Adolescence, 26(4), 475–479. https://doi.org/10.1016/S0140-1971(03)00036-8.
Liening, S. H., Stanton, S. J., Saini, E. K., & Schultheiss, O. C. (2010). Salivary testosterone, cortisol, and progesterone: two-week stability, interhormone correlations, and effects of time of day, menstrual cycle, and oral contraceptive use on steroid hormone levels. Physiology & Behavior, 99(1), 8–16. https://doi.org/10.1016/j.physbeh.2009.10.001.
Lombardo, G., Enache, D., Gianotti, L., Schatzberg, A. F., Young, A. H., Pariante, C. M., & Mondelli, V. (2019). Baseline cortisol and the efficacy of antiglucocorticoid treatment in mood disorders: a meta-analysis. Psychoneuroendocrinology, 110, 104420. https://doi.org/10.1016/j.psyneuen.2019.104420.
Long, J. A. (2021). interactions: Comprehensive, User-Friendly Toolkit for Probing Interactions (1.1.5). https://CRAN.R-project.org/package=interactions
Losecaat Vermeer, A. B., Krol, I., Gausterer, C., Wagner, B., Eisenegger, C., & Lamm, C. (2020). Exogenous testosterone increases status-seeking motivation in men with unstable low social status. Psychoneuroendocrinology, 113, 104552. https://doi.org/10.1016/j.psyneuen.2019.104552.
Losecaat Vermeer, A. B., Riečanský, I., & Eisenegger, C. (2016). Chapter 9—Competition, testosterone, and adult neurobehavioral plasticity. In B. Studer & S. Knecht (Eds.), Progress in Brain Research (Vol. 229, pp. 213–238). Elsevier. https://doi.org/10.1016/bs.pbr.2016.05.004
Lüdecke, D., Bartel, A., Schwemmer, C., Powell, C., Djalovski, A., & Titz, J. (2021). sjPlot: Data Visualization for Statistics in Social Science (2.8.10). https://CRAN.R-project.org/package=sjPlot
Mazur, A., & Booth, A. (1998). Testosterone and dominance in men. Behavioral and Brain Sciences, 21(3), 353–363. https://doi.org/10.1017/S0140525X98001228.
McEwen, B. S. (2019). What is the confusion with cortisol? Chronic Stress, 3, 2470547019833647. https://doi.org/10.1177/2470547019833647.
Mehta, P. H., & Beer, J. (2010). Neural mechanisms of the testosterone–aggression relation: the role of Orbitofrontal Cortex. Journal of Cognitive Neuroscience, 22(10), 2357–2368. https://doi.org/10.1162/jocn.2009.21389.
Mehta, P. H., Jones, A. C., & Josephs, R. A. (2008). The social endocrinology of dominance: basal testosterone predicts cortisol changes and behavior following victory and defeat. Journal of Personality and Social Psychology, 94, 1078–1093. https://doi.org/10.1037/0022-3514.94.6.1078.
Mehta, P. H., & Josephs, R. A. (2010). Testosterone and cortisol jointly regulate dominance: evidence for a dual-hormone hypothesis. Hormones and Behavior, 58(5), 898–906. https://doi.org/10.1016/j.yhbeh.2010.08.020.
Mehta, P. H., Lawless DesJardins, N. M., van Vugt, M., & Josephs, R. A. (2017). Hormonal underpinnings of status conflict: testosterone and cortisol are related to decisions and satisfaction in the hawk-dove game. Hormones and Behavior, 92, 141–154. https://doi.org/10.1016/j.yhbeh.2017.03.009.
Mehta, P. H., Mor, S., Yap, A. J., & Prasad, S. (2015a). Dual-hormone changes are related to bargaining performance. Psychological Science, 26(6), 866–876. https://doi.org/10.1177/0956797615572905.
Mehta, P. H., & Prasad, S. (2015). The dual-hormone hypothesis: a brief review and future research agenda. Current Opinion in Behavioral Sciences, 3, 163–168. https://doi.org/10.1016/j.cobeha.2015.04.008.
Mehta, P. H., Snyder, N. A., Knight, E. L., & Lassetter, B. (2015b). Close versus decisive victory moderates the effect of testosterone change on competitive decisions and task enjoyment. Adaptive Human Behavior and Physiology, 1(3), 291–311. https://doi.org/10.1007/s40750-014-0014-0.
Mehta, P. H., van Son, V., Welker, K. M., Prasad, S., Sanfey, A. G., Smidts, A., & Roelofs, K. (2015c). Exogenous testosterone in women enhances and inhibits competitive decision-making depending on victory–defeat experience and trait dominance. Psychoneuroendocrinology, 60, 224–236. https://doi.org/10.1016/j.psyneuen.2015c.07.004.
Mehta, P. H., Welker, K. M., Zilioli, S., & Carré, J. M. (2015d). Testosterone and cortisol jointly modulate risk-taking. Psychoneuroendocrinology, 56, 88–99. https://doi.org/10.1016/j.psyneuen.2015d.02.023.
Mossink, J. C. L., Verkuil, B., Burger, A. M., Tollenaar, M. S., & Brosschot, J. F. (2015). Ambulatory assessed implicit affect is associated with salivary cortisol. Frontiers in Psychology, 6. https://www.frontiersin.org/article/https://doi.org/10.3389/fpsyg.2015.00111
Muller, M. N. (2017). Testosterone and reproductive effort in male primates. Hormones and Behavior, 91, 36–51. https://doi.org/10.1016/j.yhbeh.2016.09.001.
Murphy, B. A., Casto, K. V., Watts, A. L., Costello, T. H., Jolink, T. A., Verona, E., & Algoe, S. B. (2022). “Feeling Powerful” Versus “Desiring Power”: a pervasive and problematic conflation in Personality Assessment? Journal of Research in Personality, 104305. https://doi.org/10.1016/j.jrp.2022.104305.
Newman, M. L., Sellers, J. G., & Josephs, R. A. (2005). Testosterone, cognition, and social status. Hormones and Behavior, 47(2), 205–211. https://doi.org/10.1016/j.yhbeh.2004.09.008.
Pfattheicher, S. (2016). Testosterone, cortisol and the Dark Triad: narcissism (but not Machiavellianism or psychopathy) is positively related to basal testosterone and cortisol. Personality and Individual Differences, 97, 115–119. https://doi.org/10.1016/j.paid.2016.03.015.
Ponzi, D., Zilioli, S., Mehta, P. H., Maslov, A., & Watson, N. V. (2016). Social network centrality and hormones: the interaction of testosterone and cortisol. Psychoneuroendocrinology, 68, 6–13. https://doi.org/10.1016/j.psyneuen.2016.02.014.
Popma, A., Vermeiren, R., Geluk, C. A. M. L., Rinne, T., van den Brink, W., Knol, D. L., Jansen, L. M. C., van Engeland, H., & Doreleijers, T. A. H. (2007). Cortisol Moderates the relationship between Testosterone and Aggression in Delinquent Male Adolescents. Biological Psychiatry, 61(3), 405–411. https://doi.org/10.1016/j.biopsych.2006.06.006.
Prasad, S., Lassetter, B., Welker, K. M., & Mehta, P. H. (2019). Unstable correspondence between salivary testosterone measured with enzyme immunoassays and tandem mass spectrometry. Psychoneuroendocrinology, 109, 104373. https://doi.org/10.1016/j.psyneuen.2019.104373.
Prasad, S., Narayanan, J., Lim, V. K. G., Koh, G. C. H., Koh, D. S. Q., & Mehta, P. H. (2017). Preliminary evidence that acute stress moderates basal testosterone’s association with retaliatory behavior. Hormones and Behavior, 92, 128–140. https://doi.org/10.1016/j.yhbeh.2016.10.020.
Putman, P., Hermans, E. J., Koppeschaar, H., van Schijndel, A., & van Honk, J. (2007). A single administration of cortisol acutely reduces preconscious attention for fear in anxious young men. Psychoneuroendocrinology, 32(7), 793–802. https://doi.org/10.1016/j.psyneuen.2007.05.009.
Putman, P., & Roelofs, K. (2011). Effects of single cortisol administrations on human affect reviewed: coping with stress through adaptive regulation of automatic cognitive processing. Psychoneuroendocrinology, 36(4), 439–448. https://doi.org/10.1016/j.psyneuen.2010.12.001.
Quirin, M., Kazén, M., Rohrmann, S., & Kuhl, J. (2009). Implicit but not explicit Affectivity predicts circadian and reactive cortisol: using the implicit positive and negative affect test. Journal of Personality, 77(2), 401–426. https://doi.org/10.1111/j.1467-6494.2008.00552.x.
Radke, S., Volman, I., Mehta, P., van Son, V., Enter, D., Sanfey, A., Toni, I., de Bruijn, E. R. A., & Roelofs, K. (2015). Testosterone biases the amygdala toward social threat approach. Science Advances, 1(5), e1400074. https://doi.org/10.1126/sciadv.1400074.
Roberts, B. W., Luo, J., Briley, D. A., Chow, P. I., Su, R., & Hill, P. L. (2017). A systematic review of personality trait change through intervention. Psychological Bulletin, 143, 117–141. https://doi.org/10.1037/bul0000088.
Ronay, R., van der Meij, L., Oostrom, J. K., & Pollet, T. V. (2018). No Evidence for a Relationship Between Hair Testosterone Concentrations and 2D:4D Ratio or Risk Taking. Frontiers in Behavioral Neuroscience, 12. https://doi.org/10.3389/fnbeh.2018.00030
Roney, J. R. (2016). Theoretical frameworks for human behavioral endocrinology. Hormones and Behavior, 84, 97–110. https://doi.org/10.1016/j.yhbeh.2016.06.004.
Rosenberg, M. (1979). Conceiving the self. Basic Books.
Rosvall, K. A., Bentz, A. B., & George, E. M. (2020). How research on female vertebrates contributes to an expanded challenge hypothesis. Hormones and Behavior, 123, 104565. https://doi.org/10.1016/j.yhbeh.2019.104565.
Sarkar, A., Mehta, P. H., & Josephs, R. A. (2018). The dual-hormone approach to dominance and status-seeking. In Routledge International Handbook of Social Neuroendocrinology, Oliver C. Schultheiss, Pranjal H. Mehta (Eds.). Routledge. https://doi.org/10.4324/9781315200439
Schmidt, K., Enge, S., & Miller, R. (2020). Reconsidering the construct validity of self-reported chronic stress: a multidimensional item response theory approach. Psychological Assessment, 32(11), 997–1014. https://doi.org/10.1037/pas0000829.
Schultheiss, O. C. (2013). The Hormonal Correlates of Implicit Motives. Social and Personality Psychology Compass, 7(1), 52–65. https://doi.org/10.1111/spc3.12008.
Schultheiss, O. C., Dlugash, G., & Mehta, P. H. (2019). Hormone measurement in social neuroendocrinology: a comparison of immunoassay and mass spectrometry methods. Mehta (eds.), Routledge international handbook of social neuroendocrinology. Routledge. InO.C. Schultheiss.
Sellers, J. G., Mehl, M. R., & Josephs, R. A. (2007). Hormones and personality: Testosterone as a marker of individual differences. Journal of Research in Personality, 41(1), 126–138. https://doi.org/10.1016/j.jrp.2006.02.004.
Sherman, G. D., & Mehta, P. H. (2020). Stress, cortisol, and social hierarchy. Current Opinion in Psychology, 33, 227–232. https://doi.org/10.1016/j.copsyc.2019.09.013.
Sherman, G. D., Rice, L. K., Jin, E. S., Jones, A. C., & Josephs, R. A. (2017). Sex differences in cortisol’s regulation of affiliative behavior. Hormones and Behavior, 92, 20–28. https://doi.org/10.1016/j.yhbeh.2016.12.005.
Shields, A. N., Brandes, C. M., Reardon, K. W., España, R. A., & Tackett, J. L. (2021). Do Testosterone and Cortisol jointly relate to adolescent dominance? A pre-registered multi-method interrogation of the dual-hormone hypothesis. Adaptive Human Behavior and Physiology, 7(2), 183–208. https://doi.org/10.1007/s40750-021-00167-3.
Slatcher, R. B., Mehta, P. H., & Josephs, R. A. (2011). Testosterone and self-reported Dominance Interact to Influence Human mating Behavior. Social Psychological and Personality Science, 2(5), 531–539. https://doi.org/10.1177/1948550611400099.
Smeets-Janssen, M. M. J., Roelofs, K., van Pelt, J., Spinhoven, P., Zitman, F. G., Penninx, B. W. J. H., & Giltay, E. J. (2015). Salivary testosterone is consistently and positively Associated with Extraversion: results from the Netherlands study of depression and anxiety. Neuropsychobiology, 71(2), 76–84. https://doi.org/10.1159/000369024.
Smither, R. D., & Houston, J. M. (1992). The nature of competitiveness: the Development and Validation of the Competitiveness Index. Educational and Psychological Measurement, 52(2), 407–418. https://doi.org/10.1177/0013164492052002016.
Stanton, S. J., & Schultheiss, O. C. (2009). The hormonal correlates of implicit power motivation. Journal of Research in Personality, 43(5), 942–949. https://doi.org/10.1016/j.jrp.2009.04.001.
Staufenbiel, S. M., Penninx, B. W. J. H., Spijker, A. T., Elzinga, B. M., & van Rossum, E. F. C. (2013). Hair cortisol, stress exposure, and mental health in humans: a systematic review. Psychoneuroendocrinology, 38(8), 1220–1235. https://doi.org/10.1016/j.psyneuen.2012.11.015.
Sundin, Z. W., Chopik, W. J., Welker, K. M., Ascigil, E., Brandes, C. M., Chin, K., Ketay, S., Knight, E. L., Kordsmeyer, T. L., McLarney-Vesotski, A. R., Prasad, S., Reese, Z. A., Roy, A. R. K., Sim, L., Stern, J., Carré, J. M., Edelstein, R. S., Mehta, P. H., Penke, L., & Tackett, J. L. (2021). Estimating the Associations between Big five personality traits, testosterone, and Cortisol. Adaptive Human Behavior and Physiology, 7(3), 307–340. https://doi.org/10.1007/s40750-020-00159-9.
Taieb, J., Mathian, B., Millot, F., Patricot, M. C., Mathieu, E., Queyrel, N., Lacroix, I., Somma-Delpero, C., & Boudou, P. (2003). Testosterone measured by 10 Immunoassays and by isotope-dilution gas Chromatography–Mass Spectrometry in Sera from 116 men, women, and children. Clinical Chemistry, 49(8), 1381–1395. https://doi.org/10.1373/49.8.1381.
Terburg, D., Aarts, H., & van Honk, J. (2012). Testosterone affects Gaze Aversion from Angry faces outside of conscious awareness. Psychological Science, 23(5), 459–463. https://doi.org/10.1177/0956797611433336.
Terburg, D., & van Honk, J. (2013). Approach–Avoidance versus Dominance–Submissiveness: a multilevel neural Framework on how Testosterone promotes Social Status. Emotion Review, 5(3), 296–302. https://doi.org/10.1177/1754073913477510.
Tibbe, T. D., & Montoya, A. K. (2022). Correcting the Bias correction for the bootstrap confidence interval in Mediation Analysis. Frontiers in Psychology, 13, 810258. https://doi.org/10.3389/fpsyg.2022.810258.
Torrance, J. S., Hahn, A. C., Kandrik, M., DeBruine, L. M., & Jones, B. C. (2018). No evidence for Associations between men’s salivary testosterone and responses on the Intrasexual competitiveness scale. Adaptive Human Behavior and Physiology, 4(3), 321–327. https://doi.org/10.1007/s40750-018-0095-2.
van der Meij, L., Schaveling, J., & van Vugt, M. (2016). Basal testosterone, leadership and dominance: a field study and meta-analysis. Psychoneuroendocrinology, 72, 72–79. https://doi.org/10.1016/j.psyneuen.2016.06.005.
van Honk, J., Schutter, D. J. L. G., Hermans, E. J., Putman, P., Tuiten, A., & Koppeschaar, H. (2004). Testosterone shifts the balance between sensitivity for punishment and reward in healthy young women. Psychoneuroendocrinology, 29(7), 937–943. https://doi.org/10.1016/j.psyneuen.2003.08.007.
van Peer, J. M., Enter, D., van Steenbergen, H., Spinhoven, P., & Roelofs, K. (2017). Exogenous testosterone affects early threat processing in socially anxious and healthy women. Biological Psychology, 129, 82–89. https://doi.org/10.1016/j.biopsycho.2017.08.003.
Vazire, S. (2010). Who knows what about a person? The self–other knowledge asymmetry (SOKA) model. Journal of Personality and Social Psychology, 98(2), 281–300.
Volman, I., Toni, I., Verhagen, L., & Roelofs, K. (2011). Endogenous testosterone modulates prefrontal–amygdala connectivity during Social Emotional Behavior. Cerebral Cortex, 21(10), 2282–2290. https://doi.org/10.1093/cercor/bhr001.
Wagner, J., Lüdtke, O., & Robitzsch, A. (2019). Does personality become more stable with age? Disentangling state and trait effects for the big five across the life span using local structural equation modeling. Journal of Personality and Social Psychology, 116, 666–680. https://doi.org/10.1037/pspp0000203.
Wegner, M., Schüler, J., Scheuermann, S., Machado, K., S., & Budde, H. (2015). The implicit power motive and adolescents’ salivary cortisol responses to acute psychosocial stress and exercise in school. CNS & Neurological Disorders - Drug Targets- CNS & Neurological Disorders), 14(9), 1219–1224.
Welker, K. M., & Carré, J. M. (2015). Individual differences in Testosterone Predict Persistence in Men. European Journal of Personality, 29(1), 83–89. https://doi.org/10.1002/per.1958.
Welker, K. M., Goetz, S. M. M., & Carré, J. M. (2015). Perceived and experimentally manipulated status moderates the relationship between facial structure and risk-taking. Evolution and Human Behavior, 36(6), 423–429. https://doi.org/10.1016/j.evolhumbehav.2015.03.006.
Welker, K. M., Lozoya, E., Campbell, J. A., Neumann, C. S., & Carré, J. M. (2014). Testosterone, cortisol, and psychopathic traits in men and women. Physiology & Behavior, 129, 230–236. https://doi.org/10.1016/j.physbeh.2014.02.057.
Welker, K. M., Roy, A. R. K., Geniole, S., Kitayama, S., & Carré, J. M. (2019). Taking risks for personal gain: an investigation of self-construal and testosterone responses to competition. Social Neuroscience, 14(1), 99–113. https://doi.org/10.1080/17470919.2017.1407822.
Wells, S., Tremblay, P. F., Flynn, A., Russell, E., Kennedy, J., Rehm, J., Van Uum, S., Koren, G., & Graham, K. (2014). Associations of hair cortisol concentration with self-reported measures of stress and mental health-related factors in a pooled database of diverse community samples. Stress (Amsterdam, Netherlands), 17(4), 334–342. https://doi.org/10.3109/10253890.2014.930432.
Wingfield, J. C., Hegner, R. E., Dufty, A. M., & Ball, G. F. (1990). The “Challenge Hypothesis”: theoretical implications for patterns of testosterone secretion, mating Systems, and breeding strategies. The American Naturalist, 136(6), 829–846. https://doi.org/10.1086/285134.
Wingfield, J. C., Lynn, S. E., & Soma, K. K. (2001). Avoiding the ‘Costs’ of Testosterone: ecological bases of hormone-behavior interactions. Brain Behavior and Evolution, 57(5), 239–251. https://doi.org/10.1159/000047243.
Wirth, M. M., & Schultheiss, O. C. (2007). Basal testosterone moderates responses to anger faces in humans. Physiology & Behavior, 90(2), 496–505. https://doi.org/10.1016/j.physbeh.2006.10.016.
Zilioli, S., & Bird, B. M. (2017). Functional significance of men’s testosterone reactivity to social stimuli. Frontiers in Neuroendocrinology, 47, 1–18. https://doi.org/10.1016/j.yfrne.2017.06.002.
Acknowledgements
We would like to thank Tom Denson, Justin Carré, Richard Ronay, Thomas Pollet, Davide Ponzi, and Neil Watson for kindly sharing their data from prior studies.
Funding
This research was supported by various funding sources awarded to the authors of the original studies. These sources include National Science Foundation Grant# BCS0423405 awarded to Robert Josephs (co-author on this manuscript); Hay Group David C. McClelland Fellowship awarded to Pranjal Mehta (co-author on this manuscript); National Science Foundation Grant# 1451848 awarded to Pranjal Mehta; a National Research Council Fellowship with the U.S. Army Natick Soldier Research, Development and Engineering Center, Research, Development and Engineering Center (NSRDEC) awarded to Kathleen Casto; Discovery Grant# 0194522 from the Natural Sciences and Engineering Research Council of Canada (NSERC) awarded to Neil Watson; an Australian Research Council’s (ARC) Discovery Projects grant to Tom Denson. These funding entities had no further role in study design; in the collection, analysis and interpretation of data; in the writing of the report; and in the decision to submit the paper for publication. Any opinions, findings, and conclusions or recommendations expressed in this material are those of the author(s) and do not necessarily reflect the views of the National Science Foundation or the Hay Group.
Author information
Authors and Affiliations
Contributions
KC, SP, and PM: Study conceptualization, writing, data collection and analysis. RJ: Study conceptualization and writing. SZ, KW, AM: data collection and analyses and writing. AM: data collection.
Corresponding author
Ethics declarations
Ethics Approval
All study procedures were approved by the respective university Institutional Review Boards. All persons gave their informed consent prior to their inclusion in the study.
Conflict of interest
The authors declare that they have no conflict of interest.
Competing Interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: The article note that can be found in the first page of the published article that says ?These authors contributed equally? should be changed to ?The first two authors contributed equally to this work.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Casto, K.V., Prasad, S., Josephs, R.A. et al. No Compelling Evidence that Self-Reported Personality Traits Explain Basal Testosterone and Cortisol’s Associations with Status-Relevant Behavior. Adaptive Human Behavior and Physiology 9, 88–122 (2023). https://doi.org/10.1007/s40750-023-00210-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40750-023-00210-5