Abstract
Available methods using image analysis (IA) are expensive and evaluated only partally in rice plants in controlled and field conditions. The objectives of this study were to: (i) estimate green area rate per seedling (GAR) in various rice cultivars and lines using new IA software, and (ii) elucidate relationship between GAR and some quantitative traits in seedlings grown at nursery. After taken randomly seedlings grown for 45 days at nursery, digital image of individual seedling was taken using a digital camera. Digital images were processed with IA software using k-means algorithm. After obtained R (red), G (green) and B (blue) values of each pixel from seedling image, these values were transformed into HSB color system. Pixel counts were obtained from HSB data, and GAR was estimated as rate of pixel counts of green area per seedling and pixel counts of input image. Results indicated that GAR was quantitative index and phenotyping trait that can be measured simply by non-destructive method using IA software without any significant alteration of plant morphology. GARs were significantly different among genotypes. GAR was significantly and positively correlated with number of tillers per seedling (NTS), leaf number per seedling (LNS), seedling dry mass (SDM) and aboveground total dry mass (TDM), i. e., aerial biomass in seedling and wasn’t significantly correlated with seedling height. Significant level for linear regression of GAR and SDM, TDM related to seedling was shown with high determination coefficients of 0.796–0.8762. GAR had a stronger relationship with SDM and TDM related to biomass than NTS and LNS.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Data for quantitative traits such as nitrogen content in plant, biomass, leaf area index (LAI), the number of tillers and plant height in the experimental fields, are widely collected to compare the characteristics of cultivars or lines, mutants and transgenic plants, i.e. differences in growth and development (Lubis et al. 2003), fertilization response (Nakano et al. 2008), response to biotic or abiotic stress (Sarangi et al. 2015), and soil fertility conditions (Jearakongman et al. 2003) in crops. Producing high quality seedlings requires periodically collecting seedling growth data to control growth and development of seedlings at nursery. Moreover, it is indispensable to measure many seedlings with different genetic backgrounds to collect data for quantitative traits related to their growth and development (Abdelkahalik et al. 2005).
Morphological traits provide a feasible way to assess plant growth, development and impacts of biotic or abiotic stress and yield (Fahlgren et al. 2015). In the conventional methods for analyzing the whole-plant phenotype in rice, plant architecture (PA) is measured with a ruler or just by counting the the number of leaves and tillers that are well known parameters to indicate the growth and development (Ishizuka et al. 2005). Hand-held measuring of quantitative seedling traits under field conditions may introduce fluctuation into measurements. Moreover, since the conventional measurement of quantitative traits such as plant height, length and width of leaf blade, the number of tillers and leaves in rice is often time-consuming and labor-intensive work (Morishima et al. 1967; Abdelkahalik et al. 2005), the establishment of more efficient methods for data collection from the experimental fields would facilitate the precise statistical analyses, model building/validation, database construction, and selection of lines for breeding of high-yielding rice cultivars (Shibayama et al. 2011).
Attempts have been made to improve the method using image analysis (IA) in rice for estimation of stratified leaf area (Oka and Hinata 1986), characterization of plant architecture (PA) (Suzuki et al. 2011), comparison of PA between new and old cultivars (Oka and Hinata 1989), kinetic measurement of growth (Ishizuka et al. 2005), and evaluation of the degree of sprout leaf bending (Zheng et al. 2008).
Some IA methods evaluated only part of the rice plant in controlled and field environments (Suzuki et al. 2011; Duan et al. 2015; Gong et al. 2018). In addition, many IA methods for phenotyping are used on the complex camera apparatus (Ishizuka et al. 2005), 3D scanalyzer system (Hairmansis et al. 2014), multispectral and hyperspectral remote sensing (Huang et al. 2012; Golzarian et al. 2017), thermal infrared imaging (Jones et al. 2009), laser imaging (Paulus et al. 2014), fluorescence imaging (Harbinson et al. 2012), 3D imaging (Klose et al. 2009) and nuclear magnetic resonance imaging (Hillnhütter et al. 2012), positron emission tomography (Poorter et al. 2012) and computed tomography (Karunakaran et al. 2004). Since these approaches are expensive and complex, these seem to be difficult to apply low-cost and simple methods to many plants and populations in controlled and field environments.
In general, the phenotyping of quantitative traits in crops is labor intensive and expensive (Polania et al. 2017). Thus, the development of highly accurate and efficient morphological data acquisition and processing approaches is of great significance for plant phenotyping, plant physiology, and further plant breeding (Tardieu et al. 2017).
In order to compare the early growth and development in hybrid plants, mutants and transgenic plants with the high-yielding potential, quantitative traits such as plant height, number of tillers, number of leaves and plant dry mass have been used (Abdelkahalik et al. 2005). Leaves are the primary structure for photosynthesis (Yan et al. 2012). Ideotypes with high-yielding potential should exhibit a large leaf area in rice (Hu et al. 2013). Thus, to investigate a set of plants grown at nursery, it is necessary to establish the measurement method that not only reflects some quantitative traits but also is easy to apply to elevated seedling number. Although the study on quantitative evaluation of PA using P-type Fourier descriptors in seedling (Suzuki et al. 2011) was performed, no data to elucidate the relationship between green area rate per seedling (GAR) and quantitative traits in various genotypes have been conduced; GAR might show any relationship with some quantitative PA and biomass traits in seedlings grown at nursery.
The objectives of this study were to: (i) estimate GAR in various rice cultivars and lines using IA software, and (ii) elucidate the relationship between GAR and some quantitative PA and biomass traits of seedlings grown at nursery.
2 Material and methods
2.1 Material
In present experiment, we used four japonica rice (Oryza sativa L. ssp. japonica) cultivars: cv. Pyongyang 53, cv. Phyongbuk 18, cv. Sohae 8, cv. Sohaechal 16, two japonica inbred lines: 5-39-1 and 10-21-1, and an indica rice (O. sativa L. ssp. indica) cultivar: cv. Tongsung 1. The former four cultivars and the last cultivar are the leading cultivars in the western regions of Democratic People’s Republic of Korea (DPR Korea). Seeds of genotypes were provided by the Rice Research Institute, the Academy of Agricultural Sciences (AAS) of DPR Korea.
2.2 Site description
The experiment for growth of rice seedling was carried out in trial field (lat 39° 01′10″N, long 125° 44′44″E, alt 30 m a.s.l.) of life science faculty of Kim Il Sung University in 2019.
2.3 Experimentation and data collection from nursery in paddy field
A randomized split-plot design (a plot size of 1.0 m × 1.5 m with three replicates) was used in the present experiment. The seeds were sown manually in each plot at a rate of 9 g of seeds m−2 on April 6, 2019. Chemical fertilizers of N, P2O5 and K2O were applied at a concentration of 30 g m−2 each as a basal manure and 30 g m−2 each as an additional manure. Plots were regularly hand-weeded and pesticides were used to prevent pest damage. Plant height and tillering in rice are crucial factors determining rice PA and influencing grain production. Hence we measured some quantitative traits involving the seedling height (SH) and number of tillers per seedling (NTS). After taken randomly for each genotypes (except the border seedling at plots) grown for 45 days at nursery after sowing, seedlings were washed with water, and then SH, NTS and LNS (leaf number per seedling) were hand-held measured, respectively.
Seedling dry mass (SDM) was weighed after 48 h in an oven at 80 °C to a constant mass, and after cutted root in seedling dried in the oven, aboveground total dry mass (TDM), i. e., aerial biomass was determined.
2.4 Image capture and image analysis procedure
After taken randomly ten seedlings, photograph of individual seedling profile was taken using the digital camera (SONY, Japan, 5.1 Mega Pixels) at a height of 25 cm on white background (Fig. 1a).
The Original side-view digital images of photographs taken from seedling were processed with the IA software (Golden Field 1.0) developed by authors using k-means algorithm (Hartigan and Wong 1979) (Fig. 1b). After obtained R (red), G (green) and B (blue) values of each pixel from the corresponding side view RGB images, these values were transformed into HSB color system. The pixel counts were be obtained from the HSB data.
GAS (green part area including leaves and stem except root in seedling) was estimated from the side-view digital image of seedling. It was the projection of a seedling body including leaves and stem except root to two-dimensional (2D) plane.
GAR, relative index, was calculated as the rate of pixel counts of GAS and total pixel counts of input image.
2.5 Data analysis
Statistical analyses were performed using the SPSS 21.0 (SPSS Inc. Chicago, IL, USA). Means were compared based on the Fisher’s least significant difference (LSD) test at the 0.05 level. Pearson correlation analysis with two-tailed test was used to determine whether a significant relationship existed between GAR and some quantitative PA and biomass traits in seedling. The regression model was built in SPSS using curve estimation with the least square method.
3 Results
3.1 Differences in some quantitative traits at nursery
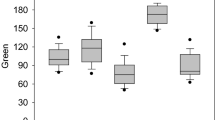
Various genotypes were visually discriminated by PA in seedling (right of Fig. 2). Some quantitative traits such as SH, NTS and LNS were significantly different among genotypes (Table 1). In addition, TDM and SDM were also significantly different among genotypes. TDM and SDM of cv. Pyongbuk 18 were largest as 0.34 g and 0.52 g among genotypes, respectively. TDM of line 10-21-1 was least as 0.20 g among genotypes, and SDM of cv. Sohae 8 was least as 0.24 g among genotypes.
Seedlings grown during 45 days after sowing at nursery in paddy field (left), Original side-view digital images of 45 days old seedlings after sowing of accessions (middle), and corresponding side view RGB images of processed with IA software (right) (online color). a Japonica rice cv. Pyongyang 53, b Japonica rice cv. Phyongbuk 18, c Japonica rice cv. Sohae 8, d Japonica rice cv. Sohaechal 16, e Indica rice cv. Tongsung 1, f Japonica rice line 5–39-1, g Japonica rice line 10–21-1
3.2 GAR at nursery
Leaves of plants did not overlap in 45 days old seedlings. When the original side-view digital images (middle of Fig. 2) were processed with IA software, plant body including leaves and stem except the root in seedling was changed the corresponding side view RGB images (right of Fig. 2). GARs were significantly different among genotypes (Table 2). Especially, GAR of cv. Pyongyang 53 was largest as 3.62 among genotypes, whereas one of cv. Sohae 8 was least as 2.12.
3.3 Relationship between GAR and some quantitative PA and biomass traits
GAR was significantly and positively correlated with NTS, LNS, SDM and TDM (r = 0.83, p < 0.05), and was not significantly correlated with SH (Table 3). Correlation coefficient (r) between GAR and TDM was highest as 0.94 (p < 0.01).
Positive correlation among GAR and some quantitative traits was established with a high level of significance (Figs. 3, 4). The linear regression models showed the relationships between GAR, NTS, LNS with the determination coefficient (R2) of 0.6958 and 0.7789. However, GAR had poor linear relationship with SH (R2 = 0.0365). Significant level for the linear regression models of GAR and SDM, TDM were shown with the high determination coefficients of 0.796 and 0.8762. GAR had a stronger relationships with SDM and TDM than NTS and LNS.
4 Discussion
4.1 Differences in GAR and some quantitative PA and biomass traits among genotypes
In previous studies using IA, PA in rice seedling has been not expressed comprehensively (Zheng et al. 2008; Suzuki et al. 2011). 3D PA is spatial arrangement of the aboveground parts in plants and canopies (Yan et al. 2012). Architecture of leaves and stem is essential for elevating the efficiency of the light harvest in plant (Hayashi 1972). Thus, PA is one of the most important types of phenotyping traits (Wang et al. 2019).
SH, LNS, seedling biomass, leaf area per seedling are well known parameters to indicate the growth and development of seedling (Sarangi et al. 2015).
SH, NTS, LNS, TDM and SDM were significantly different among genotypes (Table 1).
GAR in this study is the relative value and image-based phenotyping trait that can be measured simply by the non-destructive method using IA software without any significant alteration of plant morphology (Table 1; Fig. 2). GAR is conceptually different with LAI (total one-side area of leaf tissue per unit ground surface area (Watson 1947)). Although LAI is a major index of photosynthetic production in crops, the measurement requires equipments such as laser leaf area meter, plant canopy analyzer and scanner, and data collection for the determination of LAI is time-consuming and labor-intesive (Shibayama et al. 2011; Hu et al. 2013). However, estimation of GAR using IA software can be proposed as one of the simple, non-destructive, and repeatable assessment methods that don’t introduce principally fluctuation into measurement.
4.2 Relationships between GAR and some quantitative PA and biomass traits
The elucidation of relationship among quantitative traits in crop may facilitate the selection of the most important characteristic in breeding programs (Sabesan et al. 2009; Polania et al. 2017). However, no data to elucidate the relationship between image-based phenotype and quantitative traits have been conduced in rice.
Since the measurement for quantitative PA traits takes much time and labor (Morishima et al. 1967; Abdelkahalik et al. 2005), PA in rice has been estimated visually and empirically in the pratical breeding (Yang and Hwa 2008). By using IA, some researchers have been investigated PA of rice seedling grown at pot (Oka and Hinata 1988; Suzuki et al. 2011). However, because the distribution of leaves and tillers in plant is affected by the environmental conditions such as temperature (Huang et al. 2001), nitrogen supply (Amin et al. 2002), photoperiod (Goto 2003), especially plant stand density (Ariyaratna et al. 2011; Zand and Shakiba 2013), PA of seedling grown at nursery may be different with that grown at pot.
Tillering in rice is not only one of the key factors relating to PA but also a central subject concerning grain yield (Zhao et al. 2006; Liang et al. 2014).
In practice, the evaluation of PA in cultivars and growing conditions has been carried out by the experience and impression (Yang and Hwa 2008).
Although the present method had a shortcoming that the processing image was 2D projection of spatially arranged leaves and tillers, PA in seedling was well characterized (Fig. 2) than results of computer IA in rice (Oka and Hinata 1988, 1989; Suzuki et al. 2011).
Based on the estimation of GAR using IA software, we elucidated the relationship between it and some quantitative PA and biomass traits in seedling grown at nursery. GAR was significantly and positively correlated with NTS, LNS, SDM and TDM, and wasn’t significantly correlated with SH (Table 3). In addition, The linear regression model could be used to describe the relationship between GAR and NTS, LNS, and SDM, TDM (Figs. 3, 4). The results showed that GAR increased linearly with increasing SDM and TDM. Based on the relations, it seems to be possible to present the biomass properties of seedling from GAR.
This survey approach using IA seems to be not only simpler, faster and more repeatable but also to be time- and labor-saving than the other methods (Oka and Hinata 1988; van Hees and Mead 2000; Suzuki et al. 2011).
In general, the condition and management of the nursery define the quality of rice seedlings, and producing high-quality seedlings mostly depends on the management of nurseries (Lampayan et al. 2015). Thus, GAR estimated using IA software seems to be an index that can carry out assessment of not only the condition and management of the nursery but also the quality of growth and development of seedling.
Based on the concept of ideotype (Peng et al. 2008), in the future study we intend to elucidate relatiobship between GAR and some quantitative PA and biomass traits from the seedling stage to the mature stage, and finally to evaluate yield components from GAR in cultivars with high-yielding potential.
We intend to make available the present method without charge to anyone in the world who is interested in image-based phenotyping in rice.
The results from this study suggested that GAR estimated using IA software from digital image of seedling grown at nursery is the image-based phenotype, i.e., phenotyping trait. GAR was significantly and positively correlated with NTS and LNS, and SDM and TDM to related biomass, respectively. The linear regression model showed the relationship between SDM, TDM, NTS and LNS with GAR in seedling. It seems to be possible to present the biomass and PA properties of seedling from GAR. The assessment on GAR estimated using IA software seems to be the new method for the quantitative selection index of seedling at nursery.
Data availability
Seeds of rice cultivars used in this study were provided by the Rice Research Institute (RRI), the Academy of Agricultural Sciences (AAS) of DPR Korea.
Code availability. Authors have not been used any code.
References
Abdelkahalik AF, Shishido R, Nomura K, Ikehashi H (2005) QTL-based analysis of heteorosis for grain shape traits and seedling characteristics in an indica-japonica Hybrid in Rice (Oryza sativa L.). Breed Sci 55:41–48
Amin SMN, Uchida N, Hatanaka T, Azuma T, Yasuda T, Tsugawa H (2002) Varietal differences of rice (Oryza sativa L.) growth to low nitrogen supply. Environ Control Biol 40:195–200
Ariyaratna M, Takamure I, Kato K (2011) Plant architecture and its responses to high planting density and low fertilizer of reduced culm number mutants in rice (Oryza sativa L.). J Plant Breed Crop Sci 3:114–119
Duan LF, Huang CL, Chen GX, Yang WN (2015) Determination of rice panicle numbers during heading by multi-angle imaging. Crop J 3:211–219
Fahlgren N, Gehan MA, Baxter I (2015) Lights, camera, action: high-throughput plant phenotyping is ready for a close-up. Curr Opin Plant Biol 24:93–99
Golzarian MR, Frick RA, Rajendran K, Berger B, Roy S, Tester M, Lun DS (2017) Accurate inference of shoot biomass from high-throughput images of cereal plants. Plant Methods 7:1–11
Gong L, Lin K, Wang T, Liu CL, Yuan Z, Zhang DB (2018) Image-based on-panicle rice [Oryza sativa L.] grain counting with a prior edge wavelet correction model. Agronomy 8:91–105
Goto E (2003) Effects of light quality on growth of crop plants under artificial lighting. Environ Control Biol 41:121–132
Hairmansis A, Berger B, Tester M, Roy SJ (2014) Image-based phenotyping for non-destructive screening of different salinity tolerance traits in rice. Rice 7:16–26
Harbinson J, Prinzenberg AE, Kruijer W, Aarts MG (2012) High throughput screening with chlorophyll fluorescence imaging and its use in crop improvement. Curr Opin Biotechnol 23:221–226
Hartigan JA, Wong MA (1979) Algorithm as 136: A k-means clustering algorithm. J R Stat Soc Ser C 28:100–108
Hayashi K (1972) Efficiencies of solar energy conversion in rice varieties. Bull Nat Inst Agric Sci Ser D 23(23):1–67
Hillnhütter C, Sikora R, Oerke EC, van Dusschoten D (2012) Nuclear magnetic resonance: a tool for imaging belowground damage caused by Heterodera schachtii and Rhizoctonia solani on sugar beet. J Exp Bot 63:319–327
Hu N, Lu CG, Yao KM, Chen J, Zhang XC (2013) Analysis and simulation of plant type on canopy structure and radiation transmission in rice. Rice Sci 20:229–237
Huang JZ, Shrestha A, Tollenaar M, Deen W, Rahimian H, Swanton CJ (2001) Effect of temperature and photoperiod on the phenological development of common lambsquarters. Weed Sci 49:500–508
Huang J, Liao H, Zhu Y, Sun J, Sun Q, Liu X (2012) Hyperspectral detection of rice damaged by rice leaf folder (Cnaphalocrocis medinalis). Comput Electr Agric 82:100–107
Ishizuka T, Tanabata T, Takano M, Shinomura T (2005) Kinetic measuring method of rice growth in tillering stage using automatic digital imaging system. Environ Control Biol 43:83–96
Jearakongman S, Immark S, Noenplub A, Fukai S, Cooper M (2003) Effect of plot size on accuracy of yield estimation of rainfed lowland rice genotypes with different plant heights and grown under different soil fertility conditions. Plant Prod Sci 6:95–102
Jones HG, Serraj R, Loveys BR, Xiong L, Wheaton A, Price AH (2009) Thermal infrared imaging of crop canopies for the remote diagnosis and quantification of plant responses to water stress in the field. Funct Plant Biol 36:978–989
Karunakaran C, Jayas D, White N (2004) Detection of internal wheat seed infestation by Rhyzopertha dominica using x-ray imaging. J Stored Prod Res 40:507–516
Klose R, Penlington J, Ruckelshausen A (2009) Usability study of 3D time-of-flight cameras for automatic plant phenotyping. Bornimer Agrartech Ber 69:93–105
Lampayan RM, Faronilo JE, Tuong TP, Espiritu AJ, de Dios JL, Bayot RS, Bueno CS, Hosen Y (2015) Effects of seedbed management and delayed transplanting of rice seedlings on crop performance, grain yield, and water productivity. For Crop Res 183:303–314
Liang WH, Shang F, Lin QT, Lou C, Zhang J (2014) Tillering and panicle branching genes in rice. Gene 537:1–5
Lubis I, Shiraiwa T, Ohnishi M, Horie T, Inoue N (2003) Contribution of sink and source size to yield variation among rice varieties. Plant Prod Sci 6:119–125
Morishima H, Oka H, Chang TT (1967) Analysis of genetic variations in plant type of rice. Estimation of indices showing genetic plant type and their correlation with yielding capacity in a segregating population. Jpn J Breed 17:73–84
Nakano H, Morita S, Kusuda O (2008) Effect of nitrogen application rate and timing on grain yield and protein content of the bread wheat cultivar ‘Minaminokaori’ in southwestern Japan. Plant Prod Sci 11:151–157
Oka M, Hinata K (1986) Estimation of stratified leaf area in rice plant by means of image analysis. Jpn J Breed 36(suppl.1):280–281
Oka M, Hinata K (1988) An application of computer image analysis for characterization of plant type in rice varieties. Jpn J Breed 38:449–458
Oka M, Hinata K (1989) Comparison of plant type between new and old rice varieties using computer image analysis. Jpn J Crop Sci 58:232–239
Paulus S, Behmann J, Mahlein AK, Plümer L, Kuhlmann H (2014) Low-cost 3D systems: suitable tools for plant phenotyping. Sensors 14:3001–3018
Peng SB, Khush GS, Virk P, Tang QY, Zou YB (2008) Progress in ideotype breeding to increase rice yield potential. For Crop Res 108:32–38
Polania J, Poschenrieder C, Rao I, Beebe S (2017) Root traits and their potential links to plant ideotypes to improve drought resistance in common bean. Theor Exp Plant Physiol 29:143–154
Poorter H, Bühler J, van Dusschoten D, Climent J, Postma JA (2012) Pot size matters: a meta-analysis of the effects of rooting volume on plant growth. Funct Plant Biol 39:839–850
Sabesan T, Suresh R, Saravanan K (2009) Genetic variability and correlation for yield and grain quality characters of rice grown in coastal saline lowland of Tamilnadu. Electr J Plant Breed 1:56–59
Sarangi SK, Maji B, Singh S, Burman D, Mandal S, Sharma DK, Singh US, Ismail AM, Haefele SM (2015) Improved nursery management further enhances the productivity of stress-tolerant rice varieties in coastal rainfed lowlands. For Crop Res 174:61–70
Shibayama M, Sakamoto T, Takada E, Inoue A, Morita K, Yamaguchi T, Takahashi W, Kimura A (2011) Regression-based models to predict rice leaf area index using biennial fixed point continuous observations of near infrared digital images. Plant Prod Sci 14:365–376
Suzuki K, Zheng ZY, Tamura Y, Hirata Y (2011) Establishment of a quantitative evaluation method of rice plant type using P–type fourier descriptors. Plant Prod Sci 14:105–110
Tardieu F, Cabrera-Bosquet L, Pridmore T, Bennett M (2017) Plant phenomics, from sensors to knowledge. Curr Biol 27:R770–R783
van Hees W, Mead BR (2000) Ocular estimates of understory vegetation structure in a closed Picea glauca / Betula papyrifera forest. J Veg Sci 11:195–200
Wang YJ, Wen WL, Wu S, Wang CY, Yu ZT, Guo XY, Zhao CJ (2019) Maize plant phenotyping: comparing 3D laser scanning, multi-view stereo reconstruction, and 3D digitizing estimates. Remote Sens 11:63–80
Watson DJ (1947) Comparative physiological studies in the growth of field crops. I. Variation in net assimilation rate and leaf area between species and varieties, and within and between years. Ann Bot 11:41–76
Yan WG, HuBL ZQJ, Jia LM, Jackson A, Pan XH, Huang BH, Yan ZB, Deren C (2012) Short and erect rice (ser) mutant from ‘Khao Dawk Mali 105’ improves plant architecture. Plant Breed 131:282–285
Yang XC, Hwa CM (2008) Genetic modification of plant architecture and variety improvement in rice. Heredity 101:396–404
Zand N, Shakiba MR (2013) Effect of plant density and nitrogen fertilizer on some attribute of grain sorghum (Sorghum bicolor (L.) moench). Int J Adv Biol Biomed Res 1:1577–1582
Zhao DL, Atlin GN, Bastiaans L, Spiertz JHJ (2006) Developing selection protocols for weed competitiveness in aerobic rice. For Crop Res 97:272–285
Zheng ZY, Iwata H, Hirata Y, Tamura Y (2008) Quantitative evaluation of the degree of sprout leaf bending of rice cultivars using P-type Fourier descriptors and principal component analysis. Euphytica 163:259–266
Acknowledgements
We thank Yong-Hyon Pae in the State Commission of Science and technology of Democratic People's Republic of Korea for technical support in the field trial.
Funding
This research received no specific grant from any funding agency,commercial or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
Kwang-O Jong conceived and designed this study and wrote the manuscript, Kwang-Myong Han and Kwang-Phil Kim conducted data gathering, Son-Il Kwak and Yu-Jin Jang developed IA software (Golden Field 1.0) and performed IA and Chol Ho performed statistical analyses. All authors read and commented on the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical approval
This manuscript has not been published before, and is not under consideration for publication anywhere else. Required Statements Rice researchers can use the results in this study for the survey of rice seedling at nursery.
Informed consent
All co-authors have been participated equally in this research. This manuscript publication has been approved by all co-authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jong, KO., Han, KM., Kwak, SI. et al. Simple estimation of green area rate using image analysis and quantitative traits related to plant architecture and biomass in rice seedling. Theor. Exp. Plant Physiol. 33, 225–234 (2021). https://doi.org/10.1007/s40626-021-00207-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40626-021-00207-z