Abstract
Neonicotinoids are systemic insecticides that can have biostimulant effect in various cultures. The aim of this study was to evaluate if neonicotinoids have stimulant effects on the growth and tolerance of sugarcane to drought stress. The experiment was performed in pots, in a fully randomized design, laid out in a 2 × 4 factorial scheme, with five replicates, with two irrigation schemes, irrigated and with water deficit, and three neonicotinoid insecticides: imidacloprid, thiamethoxam, and clothianidin and a witness. Neonicotinoids had beneficial effects on the initial growth of sugarcane, increasing the total weight of plants, resulting in greater plant height, and higher mass of root system, especially, which clothianidin increased production of 42 % of the stem mass, and 20 % of the root mass, resulting in a 37 % increase in the total mass relative to the non-stressed plant controls, 121 days after planting. Thiamethoxam and clothianidin were the most efficient to minimize the effects of drought stress, attested by the lower accumulation of proline, and improvement in the photochemistry and photosynthesis activity of the drought stressed plants.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Sugarcane has an annual cycle of at least 12 months, and it needs good water supply 80 % of that time, until at least 2 months before harvest, when the maturation process begins, i.e., when the concentration of sugar increases in the stems (Moore and Botha 2013). However, in most producing regions of the world, rainfall is poorly distributed throughout the year, impairing the productivity of the crop.
Sugarcane is propagated asexually, planted by sprouting of axillary buds of culms (Bonnett 2013). This process requires moisture in the soil. Therefore, a rapid germination of gems and their rooting are crucial for the establishment of the culture.
Some neonicotinoids, such as imidacloprid, clothianidin and thiamethoxam, have been tested for presenting, in addition to its insecticide efficiency, a tonic or stimulant effect, with increased production and vigor in the treated plants, (Chiriboga et al. 2008; Thielert 2006; Gonias et al. 2008; Macedo and Castro 2011). In addition, they can increase the tolerance of plants to water stress (Chiriboga et al. 2008; Geissler and Wessjohann 2011). One of the observed effects is the greater development of the plant root system (Macedo and Castro 2011; Chiriboga et al. 2008).
Imidacloprid is a first generation neonicotinoid insecticide and nowadays is the most widely used and widespread neonicotinoid in the world. Imidacloprid has antioxidant properties similar to nicotine and nicotinic acid, because they have a structure similar to chloropyridine (Gonias et al. 2008). In cotton, imidacloprid improved carboxylation efficiency in drought stressed plants (Thielert 2006). In Populus nigra, it increased plant growth both in optimum conditions as well as in situations of water deficit (Chiriboga et al. 2008).
Thiamethoxam is a second-generation neonicotinoid insecticide, widely used to control sucking insects, it presents bioactivator effect, since, even in the absence of pests, it promotes an increase in vigor and development of treated plants (Macedo et al. 2013a; Pereira 2010). In soybeans, thiamethoxam stimulated increase of leaf area, root dry mass, and aerial parts, resulting in greater productivity and, in addition, increased tolerance to stress (Tavares et al. 2007; Ford et al. 2011). It has also been observed that thiamethoxam increases the vigor of corn seed in germination (Horii et al. 2007).
Clothianidin is also a second-generation neonicotinoid insecticide, and, in addition to the insecticide effect, it has shown positive results increasing leaf area in soybeans and corn (Cox et al. 2007).
Our working hypothesis is that neonicotinoid biostimulants can have positive effect on the initial growth of sugarcane, mitigating the effects of water deficit on plants. Our aim was to study the morpho-physiological behavior of sugarcane under water deficit, subjected to treatments with three neonicotinoid insecticides.
2 Methods
The experiment was conducted in a greenhouse, (9°27′55″S, 35°49′31″W, 130 m). The experimental design was fully randomized, laid out in a 2 × 4 factorial scheme, comprising two irrigation schemes (hydric deficit and irrigated) and three neonicotinoid insecticides (imidacloprid, clothianidin, and thiamethoxam), and one more as witness, with five replicates (pots).
The plants were grown in 20 L pots, with 20 kg of dry soil. Soil fertility was corrected by nutrient analysis. Field capacity (CC: 20.3 % v/v), permanent wilting point (PWP: 15.3 % v/v) and the available water capacity (AWC) (v/v) were determined based on the water retention curve.
Five single-bud cuttings were planted in each pot. They were sprayed with neonicotinoids, dosed according to technical recommendation: imidacloprid, 0.0120 g pot−1; thiamethoxam, 0.0241 g vase−1; and clothianidin, 0.0151 g vase−1. Distilled water was used for spraying the control treatment. Application was made with manual spray using constant volume in all treatments.
After applying treatments, the stems were covered with a 20 mm layer of soil. Plants were thinned out 10 days after emergence leaving only one plant per pot.
Irrigated treatment was maintained near field capacity (FC) with 80 % of the available water capacity (AWC) during the entire experimental period. Water deficit treatment was applied 88 days after planting (DAP), keeping the pots close to PWP with 20 % of AWC during the last 32 days of the experiment.
After 120 DAP, measurements of gas exchange analyses, photochemical efficiency, SPAD index and leaf water potential (ψw) were performed. All evaluations were made on the middle third of leaf +1, except water potential, which was measured on leaf +2. The numbering of the leaves was based on using previous classification (Van Dillewijn 1952).
Gas exchange measurements were made with an infrared gas analyzer (IRGA; LI-6400xt, LI-COR Inc., Lincoln, Nebraska, USA), through which was quantified: net CO2 assimilation (A), stomatal conductance (gs), transpiration (E) and intrinsic water use efficiency (IEUA). Gas exchange was measured between 07:00 am and 08:00 am and between 11:00 am and 12:00 pm.
Maximum photosystem II (PSII) photochemical efficiency (Fv/Fm) was measured at predawn, at 4:30 am, and at noon, with a pulse amplitude modulation fluorometer (PAM 2500, Walz, Effeltrich, Germany; Kalaji et al. 2014). Effective photochemical efficiency (ΦPSII) was quantified on the same leaves at 10:00 am (Baker 2008). Total chlorophyll content was estimated by SPAD index (leaf greenness) measured with a chlorophyllometer (SPAD-502, Minolta, Osaka, Japan) and determined by organic solvent extraction method, according to Hendry and Grime (1993) on the same leaf in which gas exchanges were made.
Water potential (ψw) was quantified at noon with a Scholander pressure pump (Soil-moisture Equipment, Santa Barbara, USA; Tyree and Hammel 1972).
At 121 DAP the plants were harvested and plant height, soil up to leaf insertion +1; culm diameter, measured at 1 cm of the soil, and the number of tillers were measured. Then the plants were collected, separated into leaves, stalks, and roots. Leaf area was quantified with a leaf area meter (LI—3100, LI-COR Inc., Lincoln, Nebraska, USA). Dry mass was quantified after drying in an oven with forced air circulation at 65 °C.
The determination of proline in the leaves was made according to the methodology described by Bates et al. (1973).
Statistical analysis was performed with the Sisvar program (Ferreira 2000), where the data was analyzed by a normality test, analysis of variance, and average test.
3 Results
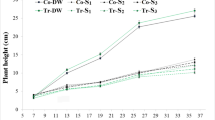
Plant height was increased by clothianidin only in non-stressed plants and was inhibited by thiamethoxam when under water deficit (Fig. 1a). The leaf area was severely affected by water deficit (Fig. 1b) and clothianidin showed remedial effect on the process, maintaining leaf area 21.4 % higher compared to plants stressed and untreated (control). While thiamethoxam had negative effect on leaf area in irrigated plants. Culm diameter did not varied among treatments of irrigated plants, but was influenced negatively by imidacloprid and thiamethoxam in plants under water stress (Fig. 1c). Tiller number increased with the application of imidacloprid and clothianidin in irrigated plants and inhibited by imidacloprid and thiamethoxam in plants under water stress (Fig. 1d).
Changes after 120 DAP with treatment in sugarcane plants for the last 32 days under water deficit in: a plant height; b leaf area; c stem diameter; and d mass of tillers. Imid, imidacloprid; Thia, thiamethoxam; Colt, clothianidin. Gray bars irrigated plants; white bars plants under water deficit. Different letters on bars with the same legend indicate significant differences after Tukey’s test (p < 0.05)
Stem mass increased 42.9 % in irrigated plants and 30.1 % in plants under water deficit relative to control plants when clothianidin was applied (Fig. 2a). Leaf mass was inhibited by neonicotinoids in plants under water deficit (Fig. 2b). The mass of tiller increased with the application of imidacloprid (51.5 %), thiamethoxam (75.4 %), and clothianidin (102.6 %) in irrigated plants. While plants under water stress, only clothianidin had a positive effect on tiller mass (67.8 %) relative to control plants (Fig. 2c). Also, only clothianidin increased root dry mass in irrigated plants (Fig. 2d). The total dry mass increased with the application of clothianidin (37.2 %) in irrigated plants and decreased with imidacloprid in plants under water deficit (Fig. 2e).
Changes after 120 DAP with treatment in sugarcane plants for the last 32 days under water deficit in: a Stem mass; b leaf mass; c tiller mass; d root mass; and E, plant mass. Imid, imidacloprid; Thia, thiamethoxam; Colt, clothianidin. Gray bars irrigated plants; white bars plants under water deficit. Different letters on bars with the same legend indicate significant differences after Tukey’s test (p < 0.05)
SPAD index and total chlorophyll content were not influenced by treatments with neonicotinoids, (Fig. 3a, b). Midday water potential remained above −0.7 in all treatments of plants under irrigation (Fig. 3c). Water potential of plants under water stress reached −1.3 MPa when no neonicotinoid was applied; with the application of clothianidin it was higher, −0.6 MPa at noon. Proline content remained constant in irrigated plants. In plants under water stress proline content was 238.5 % larger, on average, compared to plants under irrigation. Thiamethoxam and clothianidin minimized this effect (Fig. 3d).
Changes after 120 DAP with treatment in sugarcane plants for the last 32 days under water deficit in: a SPAD index; b chlorophyll; c proline; and d leaf water potential. Imid, imidacloprid; Thia, thiamethoxam; Colt, clothianidin. Gray bars irrigated plants; white bars plants under water deficit. Different letters on bars with the same legend indicate significant differences after Tukey’s test (p < 0.05)
Early morning net photosynthesis rate was reduced dramatically (50 %) by water deficit, and increased with thiamethoxam (21.1 %) and clothianidin (29.9 %) in irrigated plants. However, it was not influenced by neonicotinoids in plants under water deficit (Fig. 4a). Net photosynthesis rate at noon was further inhibited by water deficit (93 %) approaching zero, at this time. This effect was minimized by applying thiamethoxam (Fig. 4b). Stomatal conductance decreased between 7:00 am and 11:00 am only in irrigated plants (Fig. 4c, d). The application of neonicotinoids had varying effects on the transpiration and water use efficiency (Fig. 4e, f, g, h).
Changes after 120 DAP with treatment in sugarcane plants for the last 32 days under water deficit in: a, b Photosynthesis; c, d stomatal conductance; e, f transpiration; g, h intrinsic water use efficiency; at a, c, e, g 08:00 am; and b, d, f, h 12:00 pm. Imid, imidacloprid; Thia, thiamethoxam; Colt, clothianidin. Gray bars irrigated plants; white bars plants under water deficit. Different letters on bars with the same legend indicate significant differences after Tukey’s test (p < 0.05)
The Fv/Fm remained above 0.7 in all treatments at dawn (Fig. 5a), but was dramatically reduced with water deficit at noon; however, it was not influenced by neonicotinoids at this time (Fig. 5b). Meanwhile, ФPSII was not influenced by neonicotinoids in irrigated plants; however, it was higher with thiamethoxam (85.7 %) and clothianidin (80.1 %) applied to plants under water deficit (Fig. 5c).
Changes after 120 DAP with treatment in sugarcane plants for the last 32 days under water deficit in: a, b maximum photosystem II photochemical efficiency; at a 4:30 am; and b at 12:00 pm; and c effective photochemical efficiency. Imid, imidacloprid; Thia, thiamethoxam; Colt, clothianidin. Gray bars irrigated plants; white bars plants under water deficit. Different letters on bars with the same legend indicate significant differences after Tukey’s test (p < 0.05)
4 Discussion
Application of clothianidin induced growth of sugarcane, especially in plants under irrigation that remained in optimum moisture condition longer (121 DAP). Changes in plant growth by applying neonicotinoids has been observed in various cultures (Macedo et al. 2013a; Acevedo and Clavijo 2008; Preetha and Stanley 2012). This seems to be one of the most notable effects of neonicotinoids in plants such as wheat (Macedo and Castro 2011; Cox et al. 2007), cotton (Preetha and Stanley 2012), and woody plants (Chiriboga 2009).
In our experiment, leaf area had a slight reduction with the application of thiamethoxam in irrigated plants, and increased with the application of clothianidin in stressed plants. Neonicotinoids generally induce increased leaf area in plants such as soybeans, (Cox et al. 2007), cotton (Gonias et al. 2006, 2008), sorghum (Mote et al. 1993), and in several woody plants (Chiriboga 2009).
Clothianidin also induced an increase of 21 % of the leaf area of stressed plants, compared with non-treated plants. This may indicate that clothianidin had the effect of minimizing the stress on the plant, because one of the first morphological changes due to the drought stress is reduction in leaf area (Jaleel et al. 2009). Similar effects were found in plants under water stress (Thielert 2006) and thermal stress (Gonias et al. 2008).
The production of tillers is important for the production of biomass from sugarcane and is closely linked with its productivity (Bonnett 2013). The application of neonicotinoids increased 37 % the number of tillers, and up to 100 % its weight, when was considered the application of clothianidin in non-stressed plants.
Clothianidin was more effective in inducing biomass production when compared to other neonicotinoids, imidacloprid and thiamethoxam, providing an increase in production of 42 % in culm mass, and 20 % in root mass. This resulted in a 37 % increase in total mass relative to control in plants without stress. Similar results were found in vines (Herbert et al. 2008), wheat (Macedo and Castro 2011), and brachiaria (Macedo et al. 2013b). In sugarcane, 130 days after application, thiamethoxam increased by up to 72 % the root dry mass (Pereira et al. 2010). The increase of the root system is associated with increased tolerance to water deficit in various cultures (Thielert 2006; Geissler and Wessjohann 2011). It can be suggested that the increase of the root system caused by clothianidin application is related to the smaller effect of water stress on these plants.
Neonicotinoids did not influence the levels of chlorophyll and SPAD index in this experiment. Similar results were found in cotton, by Gonias et al. (2008), when imidacloprid was applied, and in wheat, by Macedo and Castro (2011), when thiamethoxam was applied. On the other hand, in stressed plants, proline content was reduced by applying thiamethoxam and clothianidin. Proline is an important osmoprotector in many plants (Delauney and Verma 1993), and its increase is related to protection against drought stress (Kishor et al. 1995). Proline constitutes less than 5 % of the total free amino acids of plants under normal conditions. However, under various forms of stress, the concentration of proline can reach 80 % of the total pool of amino acids (Kishor et al. 2005). Consequently, these results suggest that thiamethoxam and clothianidin application reduced the stress on sugarcane. This can also be confirmed by the smaller reduction in leaf water potential in stressed plants treated with these neonicotinoids.
The application of thiamethoxam and clothianidin increased photosynthetic activity of sugarcane in non-stressed plants, and effective quantum efficiency in stressed plants. Similar results were found by Stamm et al. (2014) in soybeans, in which an increase in the gene expression related to photosynthetic activity with thiamethoxam application was noted. On the other hand, the application of imidacloprid had no effect on sugarcane that could be noted in our results. However, in cotton, the application of imidacloprid increased photosynthetic activity and effective photochemical efficiency (Gonias et al. 2008).
A decrease in Fv/Fm reflects damage to the photosystem II; this damage can be permanent, that is, the PS II does not recover overnight; or it can be temporary, in that Fv/Fm decreases in the hottest hours of the day and recovers at the end of the day (Kalaji et al. 2014). In sugarcane, it was observed that there was a more intense photoinhibition in plants under stress during the day, but the plants were able to recover overnight, even in plants reaching water potential of −1.3 MPa at noon. This shows that the species is well adapted to tropical conditions in which sporadic droughts associated with high solar radiation and temperature are common phenomena.
While neonicotinoids had not effect on the integrity of the photosystem II (Fv/Fm), thiamethoxam and clothianidin improved effective quantum efficiency of PS II in plants under water stress. This means that a larger part of the energy absorbed by chlorophylls of PSII were used for photosynthesis, i.e. it may indicate a better transport of electrons by the photosystem, consequently a better net photosynthesis rate (Maxwell and Johnson 2000).
5 Conclusion
The results showed that neonicotinoids have beneficial effects on initial growth of sugarcane by increasing the total plant weight, resulting from a greater plant height and improvement of the root system. Thiamethoxam and clothianidin were the most efficient in minimizing the effects of drought stress, which could be verified by a smaller accumulation of proline and improvement on the photochemical activity and photosynthesis of the plants.
References
Acevedo JC, Clavijo J (2008) Investigación agronômica em Colombia. In: Clavijo J (ed) Tiametoxam: Un nuevo concepto em vigor y productivad. Arte Litográfico, Bogotá, pp 41–91
Baker NR (2008) Chlorophyll fluorescence: a probe of photosynthesis in vivo. Annu Rev Plant Biol 59:89–113. doi:10.1146/annurev.arplant.59.032607.092759
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207
Bonnett GD (2013) Developmental stages (phenology). In: Moore PH, Botha FC (eds) Sugarcane: physiology, biochemistry, and functional biology. Wiley Blackwell, Ames, pp 35–53
Chiriboga A (2009) Physiological responses of woody plants to imidacloprid formulations. Doctoral Thesis, The Ohio State University, Columbus
Chiriboga A, Herms DA, Royalty RN (2008) Imidaclopride (Merit®2F) increases the growth of the poplar (Populus nigra) clone NC5271 by increasing total leaf area. Department of Entomology, The Ohio State University, Columbus. http://hdl.handle.net/1811/31872. Accessed 25 Oct 2015
Cox WJ, Shields E, Cherney JH (2007) The effect of clothianidin seed treatments on corn growth following soybean. Crop Sci 47:2482–2485. doi:10.2135/cropsci2006.12.0810
Delauney AJ, Verma DPS (1993) Proline biosynthesis and osmoregulation in plants. Plant J 4:215–223. doi:10.1046/j.1365-313x.1993.04020215.x
Ferreira DF (2000) Análises estatísticas por meio do Sisvar para Windows versão 4.0. Reunião anual da região brasileira da sociedade internacional de biometria 45:235
Ford KA, Gulevich AG, Swenson TL, Casida JE (2011) Neonicotinoid insecticides: oxidative stress in planta and metallo-oxidase inhibition. J Agric Food Chem 59:4860–4867. doi:10.1021/jf200485k
Geissler T, Wessjohann LA (2011) A whole-plant microtiter plate assay for drought stress tolerance-inducing effects. J Plant Growth Regul 30:504–511. doi:10.1007/s00344-011-9212-1
Gonias ED, Oosterhuis DM, Bibi AC (2006) How the insecticide Trimax TM improves the growth and yield of cotton. In: Proceedings of the Beltwide Cotton Conference, San Antonio
Gonias ED, Oosterhuis DM, Bibi AC (2008) Physiologic response of cotton to the insecticide imidacloprid under high-temperature stress. J Plant Growth Regul 27:77–82. doi:10.1007/s00344-007-9033-4
Hendry GAF, Grime JP (1993) Methods in comparative plant ecology, a laboratory manual. Chapman & Hall, London
Herbert KS, Hoffmann AA, Powell KS (2008) Assaying the potential benefits of thiamethoxam and imidacloprid for phylloxera suppression and improvements to grapevine vigour. Crop Prot 27:1229–1236. doi:10.1016/j.cropro.2008.03.006
Horii A, McCue P, Shetty K (2007) Enhancement of seed vigour following insecticide and phenolic elicitor treatment. Bioresour Technol 98:623–632. doi:10.1016/j.biortech.2006.02.028
Jaleel CA, Manivannan P, Wahid A, Farooq M, Al-Juburi HJ, Somasundaram R, Panneerselvam R (2009) Drought stress in plants: a review on morphological characteristics and pigments composition. Int J Agric Biol 11:100–105. http://www.fspublishers.org
Kalaji HM, Schansker G, Ladle RJ, Goltsev V, Bosa K, Allakhverdiev SI, Zivcak M (2014) Frequently asked questions about in vivo chlorophyll fluorescence: practical issues. Photosynth Res 122:121–158. doi:10.1007/s11120-040-0024-6
Kishor PK, Hong Z, Miao GH, Hu CAA, Verma DPS (1995) Overexpression of [delta]-pyrroline-5-carboxylate synthetase increases proline production and confers osmotolerance in transgenic plants. Plant Physiol 108:1387–1394. doi:10.1104/pp.108.4.1387
Kishor PK, Sangam S, Amrutha RN, Laxmi PS, Naidu KR, Rao KRSS, Sreenivasulu N (2005) Regulation of proline biosynthesis, degradation, uptake and transport in higher plants: its implications in plant growth and abiotic stress tolerance. Curr Sci 88:424–438
Macedo WR, Castro PRC (2011) Thiamethoxam: molecule moderator of growth, metabolism and production of spring wheat. Pestic Biochem Physiol 100:299–304. doi:10.1016/j.pestbp.2011.05.003
Macedo WR, Araújo DK, Castro PRDC (2013a) Unravelling the physiologic and metabolic action of thiamethoxam on rice plants. Pestic Biochem Physiol 107:244–249. doi:10.1016/j.pestbp.2013.08.001
Macedo WR, Fernandes GM, Possenti RA, Lambais GR, Castro PRDC (2013b) Responses in root growth, nitrogen metabolism and nutritional quality in Brachiaria with the use of thiamethoxam. Acta Physiol Plant 35:205–211. doi:10.1007/s11738-012-1064-1
Maxwell K, Johnson GN (2000) Chlorophyll fluorescence—a practical guide. J Exp Bot 51:659–668. doi:10.1093/jexbot/51.345.659
Moore PH, Botha FC (2013) Sugarcane: physiology, biochemistry & functional biology. Wiley Blackwell, Ames
Mote UN, Datkhile R, Pawar SA (1993) A new insecticide, Imidacloprid as a seed dresser for the control of sucking pests of cotton. Pestology 27:23–26
Pereira MA (2010) Tiametoxam em plantas de Cana-de-acucar, feijoeiro, soja, laranjeira e cafeeiro: parâmetros de desenvolvimento e aspectos bioquímicos. Tese, ESALQ, Piracicaba
Pereira JM, Fernandes PM, Veloso VRS (2010) Efeito fisiológico do inseticida thiamethoxam na cultura da cana-de-açúcar. Arquivos do Instituto de Biologia 77:159–164
Preetha G, Stanley J (2012) Influence of neonicotinoid insecticides on the plant growth attributes of cotton and okra. J Plant Nutr 35:1234–1245. doi:10.1080/01904167.2012.676134
Stamm MD, Enders LS, Donze-Reiner TJ, Baxendale FP, Siegfried BD, Heng-Moss TM (2014) Transcriptional response of soybean to thiamethoxam seed treatment in the presence and absence of drought stress. BMC Genom 15:1055–1068. doi:10.1186/1471-2164-15-1055
Tavares S, Castro PRC, Ribeiro RV, Aramaki PH (2007) Avaliação dos efeitos fisiológicos de tiametoxam no tratamento de sementes de soja. Revista da Agricultura 82:47–54
Thielert W (2006) A unique product: the story of the imidacloprid stress shield. Bayer Crop Sci 59:73–86
Tyree MT, Hammel HT (1972) The measurement of the turgor pressure and the water relations of plants by the pressure-bomb technique. J Exp Bot 23:267–282. doi:10.1093/jxb/23.1.267
Van Dillewijn C (1952) Botany of sugarcane. Chronica Botanica, Waltham
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary materials.
Rights and permissions
About this article
Cite this article
Endres, L., Oliveira, N.G., Ferreira, V.M. et al. Morphological and physiological response of sugarcane under abiotic stress to neonicotinoid insecticides. Theor. Exp. Plant Physiol. 28, 347–355 (2016). https://doi.org/10.1007/s40626-016-0056-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40626-016-0056-8