Abstract
Background/Aims
Serum free light chain (FLC) levels are correlated with chronic kidney disease (CKD) stages and are highest in patients on hemodialysis (HD). Aim of this study was to assess the FLC removal efficiency of Elisio™-210H dialyzer using either high-flux HD or on line high efficiency hemodiafiltration (HDF) modalities in CKD-5D patients.
Methods
In this prospective and comparative study, 20 CKD-5D patients free from multiple myeloma were randomized in two groups: HD versus on line HDF. All patients were dialyzed with Elisio™-210H dialyzer. Serum samples were collected before and after the midweek dialysis session, before randomization and at the end of the study to measure κ and λ FLC concentrations. Reduction ratios were corrected for net ultrafiltration.
Results
For both HD and HDF mode, κ and λ FLC concentrations were significantly lower after dialysis than before but median reductions in κ and λ FLC levels were significantly higher in HDF versus HD groups (κ 73.5 vs. 65.5 %, p = 0.04 and λ 51.0 vs. 36.6 %, p = 0.07). After dialysis, all κ/λ ratio values were between 0.26 and 1.65 which is the reference range described in subjects with normal kidney function, for both HD and HDF groups (median κ/λ ratios were 0.80 [0.47–1.22] and 0.67 [0.50–0.79] respectively).
Conclusion
This study shows the superiority of on line HDF compared with HD to remove both κ and λ FLC. Moreover, all post-dialysis κ/λ ratios reached normal reference range.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The circulating level of κ and λ free immunoglobulin light chains (FLC) is dependent on an equilibrium between production and clearance. Production of κ FLC is double that of λ FLC. Their renal clearance depends on their molecular weight: monomeric κ chains (22 kDa) are cleared about three times faster than dimeric λ chains (45 kDa) [1]. In chronic kidney disease (CKD) patients, polyclonal FLC increase exponentially as the glomerular filtration rate (GFR) falls and can be up to 20–30 times the normal values in dialysis. Elevated concentrations of polyclonal FLC have been reported in CKD dialysis patients (CKD-5D), at higher levels than in pre-dialysis patients [2, 3]. In addition, classical dialyzers have revealed difficulties in efficiently removing FLC.
Through the development of sensitive serum FLC immunoassay for diagnostics [4] and the introduction of protein-leaking membranes [5, 6] as an add-on treatment for protecting the kidney in multiple myeloma, clearance and circulating FLC concentrations have been greatly improved. Two diagnostic ranges have been proposed to assess the risk of monoclonal gammopathies for the κ/λ ratio depending on the renal function: a “normal reference range” and an “extended renal failure reference range”. In people with normal renal function, the two combined mechanisms of production/clearance lead to a median κ/λ ratio of 0.58 [0.26–1.65] [7]. In patients with advanced renal failure, removal of FLC from the circulation occurs through pinocytosis [1] and the median κ/λ FLC ratio rises to 1.12 [0.37–3.10] [8].
However, to date, in dialysis patients free from multiple myeloma the effect of renal replacement therapy on the κ/λ FLC ratio using new generations of dialysis membranes has not been clarified. We recently showed the superiority of high efficiency hemodiafiltration (HDF) over high-flux hemodialysis (HD) in FLC removal in a population of multiple myeloma patients [9]. The aim of the present study was to evaluate, in CKD-5D patients free from multiple myeloma, the FLC removal ability of a new design generation of dialyzers, ELISIO™-H series, which possess fibers of a greater internal length, using either HD or online HDF modality.
Methods
Patients
Twenty CKD-5D patients undergoing maintenance HD at a single Montpellier dialysis facility were recruited for this study. Only patients older than 18 years, undergoing HD 3 times/week for more than 3 months, with a stable anticoagulation scheme, a hemoglobin level >10.5 g/dl and vascular access allowing a minimum blood flow of 300 ml/min during treatment were eligible for the study, after giving written informed consent. Patients were excluded if they had malignancy, or symptoms/signs of acute/chronic inflammatory or infectious diseases. The study was conducted in accordance with the principles of the Declaration of Helsinki and in compliance with the International Conference on Harmonization/Good Clinical Practice regulations. In accordance with French Law, the study was approved by our Institution’s ethical committee with the following number 2008-A00852-53. The study was also registered at clinicaltrials.gov (NCT01653808).
Study design, membrane and dialysis conditions
This was a prospective, randomized, comparative study with two parallel arms, HD versus online HDF. All patients were dialyzed with the Elisio™-210H dialyzer (polynephron™ membrane based on polyethersulfone polymer, effective membrane surface area: 2.1 m2; Nipro Europe, Zaventem, Belgium) for the entire study. Briefly, after a 1-month washout period in HD mode (Elisio™-210H dialyzer), patients were randomly assigned to two treatment groups, HD (n = 10) and HDF (n = 10) for 4 months.
Dialysis conditions remained unchanged for each patient: three sessions/week, 3–4 h/session, with a blood flow (QB) of 350–400 ml/min, ultrapure bicarbonate buffered dialysate, and dialysate flow (QD) of 500 ml/min. Online production of clean dialysate and ultrapure substitution fluid was performed in both HD and online HDF modality using the 5008 machine system (Fresenius Medical Care, Bad Homburg, Germany). HDF was performed in postdilution mode at a substitution flow rate (Qs) set at 100 ml/min (18–24 l/session). The ultrafiltration flow rate (QUF) was set according to each patient’s interdialytic weight gain (total ultrafiltration 20–26 l/session). The routine anticoagulation protocol was unchanged and consisted of a single bolus intravenous administration of low molecular weight heparin 2–3 min before launching the dialysis session.
Sample collection and FLC assays
Blood samples were drawn from the arterial line before and after the midweek dialysis session. First sampling was performed at baseline before randomization (M0, corresponding to the end of the washout period) when all patients were still dialyzed in HD mode. Thereafter, samples were drawn at the end of the study period (after 4 months of treatment from baseline) before and after the midweek dialysis session.
Serum κ and λ FLC concentrations were measured by nephelometry on a Siemens BNII analyzer using the FREELITE immunoassay (The Binding Site, Birmingham, UK).
Calculations
FLC reduction ratios were calculated using the usual equation:
where Cpre and Cpost are serum FLC concentrations pre- and post-treatment respectively. Reduction percentages were calculated by multiplying the reduction ratio by 100 %.
As previously described [9], we utilized a single-compartment kinetic model already published for β2-microgobulin [10] to correct post-dialysis FLC measurements for net ultrafiltration:
where Cpost C denotes the concentration of FLC after dialysis corrected for net ultrafiltration; BWpost and ∆BW are, respectively, the body weight after dialysis and the difference between pre- and post-treatment body weights.
Effective κ- and λ-FLC clearances (Kd) were calculated as follows, according to Leypoldt et al. [11]:
where QUF is the ultrafiltration flow rate, T, the session length, and V(T) the extracellular volume at the end of the dialysis session.
Statistical analysis
Data were expressed as median and range for quantitative variables with skewed distribution, mean and standard deviation (SD) for quantitative variables with normal distribution, and proportion for categorical variables. The comparisons of variables between periods of treatment and between pre- and post-dialysis evaluations were performed using the Mann–Whitney-U/Wilcoxon test. Values were considered statistically significant at p < 0.05. Spearman correlation coefficient (rho) was used to determine the relationships between quantitative variables. All analyses were carried out with R software, version 2.15.1 (2012-06-22).
Results
Characteristics of the CKD-5D patients are summarized in Table 1a.
Before the end of the treatment period, 3 patients withdrew from the study (1 in the HDF group who moved to another dialysis center, and 2 in the HD group: 1 transplantation and 1 for personal decision) leaving 17 patients. One patient, who had primitive amyloidosis, was excluded from the statistical analysis.
At M0, κ and λ FLC concentrations did not differ significantly between the two groups, HD and HDF, either before or after dialysis sessions. Likewise, no significant differences were observed between HD and HDF for both κ and λ FLC clearances at M0.
Κ and λ FLC median concentrations before and after dialysis at M4 are reported in Table 1b. Both κ and λ FLC concentrations were significantly lower after the dialysis session than before, whatever the dialysis mode used. Treatment for 4 months with HDF compared to HD led to significantly weaker post-dialysis concentrations in κ (25.6 mg/l [18.0–54.6] vs. 52.3 mg/l [21.7–103.2] in HDF and HD, respectively; p = 0.02) and in λ FLC (38.1 mg/l [29.5–77.8] vs. 65.5 mg/l [43.6–149.6] in HDF and HD, respectively; p = 0.02). FLC clearances were higher in HDF than HD both for κ (50.4 ml/min [30.9–59.9] vs. 65.7 ml/min [48.1–81.0]; p = 0.012) and for λ (23.8 ml/min [17.6–33.2] vs. 37.8 ml/min [26.1–49.1]; p = 0.006).
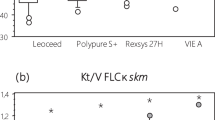
After correction of the post-treatment measurements for fluid removal using Eq. 2, median reductions in serum FLC levels at M4 were significantly higher in the HDF than HD group both for κ (73.5 vs. 65.5 %; p = 0.004), and for λ (51.0 vs. 36.5 %; p = 0.007) (Fig. 1a). Median reductions in serum κ and λ FLC levels at M4 in the HDF group were also significantly higher than baseline levels (M0).
Percentage of reduction (a) and κ/λ ratio (b) in serum FLC. a Effect of dialysis modality and time on κ and λ FLC removal efficiency. No significant difference was observed between M0 and M4 HD in either κ or λ FLC percentage of reduction. b Effect of dialysis with HD or online HDF technique using Elisio™-210H dialyzer on κ/λ ratio at M4 and two trial “reference intervals”. Continuos lines normal reference range; dashed lines extended renal failure reference range. HD hemodialysis, HDF online hemodiafiltration. ***p < 0.001; **p < 0.01; *p < 0.05
A statistically significant linear correlation between FLC reduction percentages and substitution volumes in the online HDF group was observed for κ FLC (p = 0.008) but also for λ FLC (p = 0.008). Results concerning the κ/λ FLC ratio are presented in Fig. 1b. Before dialysis, median κ/λ FLC ratios were 1.52 [0.80–2.02] and 1.33 [0.90–1.69] in the HD and HDF groups, respectively. All κ/λ ratio values were between 0.37 and 3.10, the reference range described in CKD patients. After dialysis, median κ/λ ratios were 0.80 [0.47–1.22] and 0.67 [0.50–0.79] in the HD and HDF groups, respectively. All κ/λ ratio values were between 0.26 and 1.65, the reference range described in subjects with normal kidney function.
HDF treatment resulted also in a significant improvement of FLC depuration comparing the M4 HDF reduction value against the HD baseline value. In the HDF group, only one patient had a κ/λ ratio modestly higher than the upper limit (1.69 vs. 1.65) observed in healthy individuals. By contrast, 3/8 (37.5 %) had a κ/λ ratio ≥the reference range (1.82, 1.86 and 2.02) in the HD group.
Discussion
In the present work, we investigated the effect of HD and online HDF using an Elisio™-210H dialyzer on serum FLC levels and the κ/λ FLC ratio in CKD-5D patients. Results concerning raw serum FLC levels attest Elisio™-210H dialyzer’s capacity to remove κ and λ FLC both with the HD and online HDF mode. While Haynes et al. [12] did not find evidence of an association between polyclonal excess of FLC and mortality, the benefit of an efficient polyclonal FLC removal to prevent renal toxicity can be argued quite readily. Moreover, while some authors consider FLC as uremic toxins [3], others suggest that polyclonal FLC levels can be used for monitoring removal of middle molecular weight uremic toxins [13]. Recently, Desjardins et al. [14] found that elevated levels of κ and λ FLC were associated with inflammation, vascular calcifications and other uremic toxins. And according to Hutchison et al. [15], measurement of serum polyclonal FLC, which may identify global immune activation, could also represent a biomarker for risk assessment and stratification in CKD patients.
FLC reduction percentages confirm the established superiority of HDF treatment over HD in middle-sized molecules either in acute or chronic treatment. Nevertheless, our median reduction values were higher than previously described with routine dialyzers in multiple myeloma patients [9]. This is partly due to the nature of the Elisio™-210H dialyzer which allows internal filtration thus improving removal of middle-size molecules (with median beta-2 microglobulin reduction ratios in HD modality ≥75 %, data not shown) compared with routine dialyzers in conventional HD. Despite huge reduction ratios obtained with Elisio™-210H in HD mode (control group), the differences still observed with Elisio™-210H in HDF compared to conventional HD may be attributable to the HDF technique and convective volume. In a recent prospective, observational, non-randomized, before and after study comparing FLC removal between online high efficiency HDF and conventional high-flux HD in chronic dialysis patients without monoclonal gammopathy, Lamy et al. [16] found that κ FLC removal was better in HDF than HD. Contrary to our results, no difference in λ FLC removal was reported in their study. The reduction values with HDF in our study were higher, particularly for λ-FLC levels. Furthermore, their pre-dialysis raw serum κ and λ FLC levels increased over time with HDF. Even though there was no significant difference in pre-dialysis serum FLC levels at M4 in our study between HD and HDF mode, neither was there a significant difference in pre-dialysis serum FLC levels between M0 and M4 for either dialysis modality. Nevertheless, post-dialysis raw serum κ and λ FLC levels were significantly lower at M4 (compared with M0) for patients using HDF mode (data not shown), suggesting a lower exposure to polyclonal FLC excess in the HDF group. It is interesting to note that supra-hemodiafiltration with endogenous reinfusion investigated by Pasquali et al. [17] also offers a promising track regarding FLC removal.
Despite the small number of patients, our results regarding κ/λ FLC ratio corroborate the recommendations of both Katzmann et al. [7] and Hutchison et al. [18]. Indeed, all κ/λ ratios fitted into “the extended renal failure reference range” before dialysis and into “the normal reference range” after dialysis for both HD and HDF groups. Thus, before the dialysis session the extended reference range should be used, while after the dialysis session the normal reference range can be applied. Only a few patients (3 in HD and 1 in HDF) had pre-dialysis κ/λ FLC ratios above 1.65, which may be explained by the long-term effect of Elisio™-210H dialyzer on serum FLC concentrations, with weaker predialytic ratios in HDF.
The major limitation of this study is the small number of patients and the relatively limited follow-up time, which precluded analysis on whether internal filtration-enhanced HD was equivalent to HDF in terms of clinical outcomes. Moreover, due to the absence of data at intermediate points of the study and a few hours after the end of the dialysis sessions, we were unable to provide a more precise analysis of the pharmacodynamics and pharmacokinetics of FLC.
Finally, this work offers promising results as regards the management of polyclonal FLC excess in CKD-5D patients. But larger studies with a longer follow-up are needed to evaluate the long-term effects of new generation dialyzers on serum FLC levels and patient morbidity.
References
Bradwell AR (2008) Serum free light chain analysis, 5th edn. The Binding Site Ltd, Birmingham
Wakasugi K, Sasaki M, Suzuki M, Azuma N, Nobuto T (1991) Increased concentrations of free light chain lambda in sera from chronic hemodialysis patients. Biomat Artif Cell Im 19:97–109
Cohen G, Rudnicki M, Schmaldienst S, Hörl WH (2002) Effect of dialysis on serum/plasma levels of free immunoglobulin light chains in end-stage renal disease patients. Nephrol Dial Transplant 17:879–883
Bradwell AR, Carr-Smith HD, Mead GP, Tang LX, Showell PJ, Drayson MT, Drew R (2001) Highly sensitive, automated immunoassay for immunoglobulin free light chains in serum and urine. Clin Chem 47:673–680
Ward RA (2005) Protein-leaking membranes for hemodialysis: a new class of membranes in search of an application? J Am Soc Nephrol 16:2421–2430
Hutchison CA, Cockwell P, Reid S, Chandler K, Mead GP, Harrison J, Hattersley J, Evans ND, Chappell MJ, Cook M, Goehl H, Storr M, Bradwell AR (2007) Efficient removal of immunoglobulin free light chains by hemodialysis for multiple myeloma: in vitro and in vivo studies. J Am Soc Nephrol 18:886–895
Katzmann JA, Clark RJ, Abraham RS, Bryant S, Lymp JF, Bradwell AR, Kyle RA (2002) Serum reference intervals and diagnostic ranges for free kappa and free lambda immunoglobulin light chains: relative sensitivity for detection of monoclonal light chains. Clin Chem 48:1437–1444
Hutchison CA, Plant T, Drayson M, Cockwell P, Kountouri M, Basnayake K, Harding S, Bradwell AR, Mead G (2008) Serum free light chain measurement aids the diagnosis of myeloma in patients with severe renal failure. BMC Nephrol 9:11
Granger Vallée A, Chenine L, Leray-Moragues H, Patrier L, Cognot C, Cartron G, Cristol J-P, Canaud B (2011) Online high-efficiency haemodiafiltration achieves higher serum free light chain removal than high-flux haemodialysis in multiple myeloma patients: preliminary quantitative study. Nephrol Dial Transplant 26:3627–3633
Bergström J, Wehle B (1987) No change in corrected beta 2-microglobulin concentration after cuprophane haemodialysis. Lancet 1:628–629
Leypoldt JK, Cheung AK (1992) Deeter RB (1997) Single compartment models for evaluating beta 2-microglobulin clearance during hemodialysis. ASAIO J 43:904–909
Haynes R, Hutchison CA, Emberson J, Dasgupta T, Wheeler DC, Townend JN, Landray MJ, Cockwell P (2011) Serum free light chains and the risk of ESRD and death in CKD. Clin J Am Soc Nephrol 6:2829–2837
Gondouin B, Scherberich JE, Bevins A, Cockwell P, Hutchison CA (2011) A potential use of polyclonal free light chain levels for monitoring in a chronic dialysis population. Poster presented at the 48th ERA-EDTA congress. Prague, Czech Republic
Desjardins L, Liabeuf S, Lenglet A, Lemke H-D, Vanholder R, Choukroun G, Massy ZA, Group EUTEW (2013) Association between free light chain levels, and disease progression and mortality in chronic kidney disease. Toxins 5:2058–2073
Hutchison CA, Burmeister A, Harding SJ, Basnayake K, Church H, Jesky MD, White K, Green CE, Stringer SJ, Bassett P, Ferro CJ, Cockwell P (2014) Serum polyclonal immunoglobulin free light chain levels predict mortality in people with chronic kidney disease. Mayo Clin Proc 89:615–622
Lamy T, Henri P, Lobbedez T, Comby E, Ryckelynck J-P, Ficheux M (2014) Comparison between on-line high-efficiency hemodiafiltration and conventional high-flux hemodialysis for polyclonal free light chain removal. Blood Purif 37:93–98
Pasquali S, Iannuzzella F, Corradini M, Mattei S, Bovino A, Stefani A, Palladino G, Caiazzo M (2015) A novel option for reducing free light chains in myeloma kidney: supra-hemodiafiltration with endogenous reinfusion (HFR). J Nephrol 28:251–254
Hutchison CA, Harding S, Hewins P, Mead GP, Townsend J, Bradwell AR, Cockwell P (2008) Quantitative assessment of serum and urinary polyclonal free light chains in patients with chronic kidney disease. Clin J Am Soc Nephrol 3:1684–1690
Acknowledgments
FREELITE immunoassay kits were provided with the compliments of The Binding Site (Birmingham, UK). This study was supported by Nipro Corporation, Japan.
Conflict of interest
Chloé Bourguignon, Leïla Chenine, Anne Sophie Bargnoux, Hélène Leray-Moragues, Jean-Paul Cristol and Marion Morena have no conflicts of interest to declare. Bernard Canaud is a full-time employee of Fresenius Medical Care. The research conducted was sponsored by Nipro Corporation.
Ethical approval
The study was conducted in accordance with the principles of the Declaration of Helsinki and in compliance with the International Conference on Harmonization/Good Clinical Practice regulations. In accordance with French Law, the study was approved by our Institution’s ethical committee with the following number 2008-A00852-53. The study was also registered at clinicaltrials.gov (NCT01653808).
Informed consent
Informed consent was obtained from all individual participants included in the study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bourguignon, C., Chenine, L., Bargnoux, A.S. et al. Hemodiafiltration improves free light chain removal and normalizes κ/λ ratio in hemodialysis patients. J Nephrol 29, 251–257 (2016). https://doi.org/10.1007/s40620-015-0207-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40620-015-0207-z