Abstract
Purpose
Psychiatric disorders could affect the patients’ abilities to cope with diabetes. The objectives of this study were to assess the prevalence of depression and alexithymia among type 2 diabetic patients and investigate the possible correlations between these psychopathological phenomena and glycaemic control assessed through glycated hemoglobin (HbA1c).
Methods
All the patients were evaluated through 20-item Toronto Alexithymia Scale (TAS-20), Hamilton rating scale for depression and Quality of Life Index. HbA1c values, diabetes duration, therapy and socio-demographic characteristics were recorded.
Results
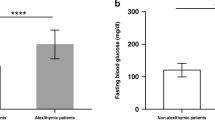
One hundred and twenty-eight patients (75 males and 53 female, mean age 64.7 ± 11.2 years) were enrolled. Alexithymic patients, compared to non-alexithymic ones, presented a significantly higher HbA1c (7.7 ± 1.5 vs. 7 ± 1.5, p = 0.016). No statistically significant difference was found when comparing the HbA1c of depressed versus non-depressed patients. Considering the raw values of HbA1c, the higher percentage was recorded among patients suffering from depression plus alexithymia (comorbidity group) followed by patients presenting alexithymia only, patients with neither depression nor alexithymia (control group) and, finally, those presenting depression only. The comorbidity group presented a significantly higher value of HbA1c (7.7 ± 1.2) than the control group (7 ± 1.6, p < 0.04) and the depressed patients (6.9 ± 1.3, p = 0.04). At the logistic regression, the HbA1c was found to be significantly associated only with alexithymia (TAS-20 total score) and insulin therapy.
Conclusions
Alexithymia more than depression influences glycaemic control. When evaluating a diabetic patient, a rapid screening for psychopathological alterations would guarantee a more accurate management. The treatment of any associated psychiatric disorders would improve the patients’ quality of life.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Diabetes represents a major public health problem and implies huge consequences on patient’s physical and mental health as well as on the quality of life [1–3].
Being diabetic requires special efforts and a considerable compliance in order to achieve the goal of a good glycaemic control [4]. The presence of a psychiatric disorder could sensitively affect the patient’s ability to cope with diabetes and to correctly follow the strict and complex therapeutic protocols. A strong link between diabetes and psychiatric disorders has been in fact demonstrated [2, 5]. If the presence of diabetes doubles the odds of developing depression [6], on the other hand depressive symptoms could represent an independent risk factor for diabetes [7]. The comorbidity of depression and diabetes leads to metabolic imbalance (in terms of higher blood glucose levels and increased risk of hypoglycaemic episodes) [8–10], worse quality of life [11], more diabetic complications and higher mortality [12, 13]. Conversely, the treatment of depression ameliorates the glycaemic control and, vice versa, the latter exerts positive effects on the patient’s mental health [14]. Another psychopathological phenomenon which has been frequently reported among diabetic patients is represented by alexithymia, a multidimensional concept that associates an emotional component, focused on the difficulty in identifying and describing feelings, with a cognitive one, centered on the use of a concrete and poorly introspective way of thinking [15].
It goes without saying that the presence of alexithymia could deeply affect the patients’ compliance and their self-regulation. This psychological construct is highly represented among diabetic patients and exerts a negative effect on their glycaemic control [4].
It has been reported that patients with ‘difficulty in describing feelings’ were more likely to present higher values of glycated hemoglobin (HbA1c) over the time and took advantage from treatments performed in a hospital setting [5]. The findings since here reported are not exempt from criticism and the supposed link between diabetes, depression and alexithymia needs to be confirmed. Some authors, in fact, have questioned the role of depression and alexithymia on glycaemic control [16, 17]. In order to provide further evidence in this field, the objectives of this study were to: (a) assess the prevalence of depression and alexithymia in a sample of type 2 diabetic patients; (b) investigate the possible correlations between these psychopathological phenomena and patient’s glycaemic control, assessed through HbA1c.
Methods
Subjects suffering from type 2 diabetes referring to the Andrology and Endocrinology Unit of our University Hospital for a first or control visit have been consecutively enrolled. No form of compensation was given to subjects who accepted to participate to the study. The study was approved by the Hospital Ethics Committee and each individual signed a written informed consent. Each enrolled subject, during one visit, underwent the following questionnaires:
-
The Italian version of the 20-item Toronto Alexithymia Scale (TAS-20) [18, 19] investigating the presence of alexithymia. It consists of three subscales: subscale I (assessing the “difficulty in identifying feelings”); subscale II (assessing the “difficulty in describing feelings”); subscale III (assessing the “externally oriented thinking”). Each item provides answers from a Likert-scale specifying levels of “strong disagreement” or “strong agreement”. Total scores of 61 and above indicate an alexithymic state. This self-rating questionnaire presents a good internal consistency and an acceptable relative stability [20].
-
The Italian version of the Hamilton rating scale for depression (HAM-D) [21] investigating the presence of depressive symptoms. It consists of 21 items, each of which provides answers with a 5-, 4-, or a 3-point rating scale. A score >7 indicates the presence of a depressive condition. Six factors can be isolated from the HAM-D: factor I (anxiety/somatization), factor II (weight), factor III (cognitive disturbance), factor IV (diurnal variation), factor V (psychomotor retardation) and factor VI (sleep).
-
The Italian version of the Quality of Life Index (QL) [22] evaluating the individual’s quality of life. It consists of five items pertaining to activity, daily life, health, support and mood. Each item provides answers on a 3-point rating scale. There is not a defined cut-off; the higher the total score, the better the quality of life.
-
A questionnaire on socio-demographic characteristics, created ad hoc. The questions pertain to the following information: sex, age, diabetes duration, type of work, marital status, presence of a living alone condition, presence of children and frequency of contact with them, presence of grandchildren, the patient’s definition of family relationships (excellent/good/bad), the patient’s self-sufficiency (self-sufficient/not self-sufficient). The latter was interpreted as the ability to care for themselves independently (everyday activities, personal hygiene, etc.).
In addition, the values of glycated hemoglobin (HbA1c) exhibited by the patients were recorded. The analysis had been carried out by independent laboratories and only those performed within 3 months from the beginning of the study were considered.
HbA1c is a reliable biomarker of average blood glucose concentrations over the preceding 2–3 months [23] universally used for the assessment of glycaemic control; as reported in literature, a value of HbA1c <7 % (measured with a blood sample) indicates a good glucose balance, both in healthy and diabetic individuals, while a value ≥7 % is considered “pathological” as indicative of a bad glucose balance [24].
Moreover, therapy [diet, oral therapy, or insulin (the latter group including also the cases of oral therapy + insulin)] and diabetes duration have been recorded for each patient. All the variables above mentioned have been collected for each patient and stored in a database created ad hoc.
Based on the HAM-D total score, the TAS-20 total score and the HbA1c value, the patients were divided into depressed and non-depressed, alexithymic and non-alexithymic, and subjects with normal or pathological HbA1c. Differences in terms of age, disease duration, HbA1c, therapy of diabetes and psychiatric variables were analyzed for each group (depressed versus non-depressed, alexithymic versus non-alexithymic, subjects with normal versus subjects with pathological HbA1c). Since psychiatric disorders could exist in comorbidity (i.e. among depressed patients there could be alexithymic subjects), a further analysis has been performed after dividing the whole sample into four groups: control group (no depression–no alexithymia); depressed patients (depression only); alexithymic patients (alexithymia only); comorbidity group (depression+alexithymia). This was done in order to eliminate the risk of confounding variables.
Statistical analysis
The collected data have been recorded in an Excel database and have been analyzed using Statistical Package for Social Science (SPSS). Data not implying a numerical ordering (qualitative data, e.g. presence or absence of depression/alexithymia) are expressed as absolute and percentage frequency while quantitative data (e.g. age, duration of disease) are expressed as mean ± standard deviation (SD). The comparisons between the groups have been performed through t test for mean ± SD and Chi-square and z test for percentages.
Furthermore, a multivariate approach, trough unconditional regression analysis, was performed in order to evaluate the relationship between psychiatric and demographical variables, therapy of diabetes and HbA1c. In particular, the odds ratio (OR), confidence interval (CI) 95 % and p value were calculated for each clinical variable (depression, alexithymia and therapy). Possible correlations between psychiatric variables and glycaemic control have been investigated through the Spearman’s correlation coefficient. The level of significance was kept at 0.05.
Results
General characteristics of the sample
One hundred and twenty-eight patients (75 males and 53 females) suffering from type 2 diabetes have been included in the study. The mean age was 64.8 ± 11.2 years, the mean diabetes duration was 11.9 ± 9.9 years, the mean value of HbA1c was 7.3 ± 1.5. The TAS-20 total mean score was 55.9 ± 14.0, that of HAM-D was 8.8 ± 6.5 and that of QL was 8.7 ± 1.3. For more details on the general characteristics of the sample, see Table 1.
Socio-demographic characteristics of the sample
No statistically significant differences were recorded in terms of socio-demographic characteristics when comparing patients with normal to those with pathological HbA1c values. See Table 2.
Depressed versus non-depressed patients: glycaemic control and psychosocial variables
The whole sample was divided into two groups (considering the HAM-D total score): depressed patients and non-depressed patients.
Sixty-three (49.2 %) patients had a HAM-D total score ≤7 (non-depressed patients) while 65 (50.8 %) patients had a HAM-D total score >7 (depressed patients). Depressed patients presented a longer even if not significant diabetes duration (13.5 ± 11.0) than non-depressed patients (10.3 ± 8.6, p = 0.064). TAS-20 total score was higher in depressed patients (60.6 ± 13.1) than in non-depressed patients (51 ± 13.1, p < 0.001); each TAS-20 subscale was higher among depressed patients than non-depressed patients. QL was lower in depressed patients (8.1 ± 1.4) than in non-depressed patients (9.3 ± 0.9, p < 0.001). No difference in terms of HbA1c value between depressed patients and non-depressed patients was found. For more details see Table 3.
Alexithymic versus non-alexithymic patients: glycaemic control and psychosocial variables
Based on the TAS-20 total score, the whole sample was divided into two groups: alexithymic patients and non-alexithymic patients.
Sixty-four (50 %) patients had a TAS-20 total score ≥61 (alexithymic patients). Alexithymic patients presented a significantly higher (11.0 ± 6.5) HAM-D total score than non-alexithymic patients (6.6 ± 5.6, p < 0.001). In addition, a positive correlation between HbA1c values and TAS-20 factor I has been recorded (r = 0.304; p < 0.05).
QL was lower (8.2 ± 1.3) in alexithymic patients than in non-alexithymic patients (9.1 ± 1.2, p < 0.001). HbA1c value was higher in alexithymic patients (7.7 ± 1.5) than non-alexithymic patients (7 ± 1.5, p = 0.016). For more details, see Table 3.
Psychosocial variables in subjects with normal versus subjects with pathological subjects
Based on the HbA1c values, patients were divided into two previously reported groups: subjects with normal HbA1c and subjects with pathological HbA1c. Subjects with pathological HbA1c presented a significantly higher scores than subjects with normal HbA1c at both HAM-D (10.8 ± 6.8 vs. 6.6 ± 5.3, p < 0.001) and TAS-20 (60.3 ± 13.1 vs. 51.2 ± 13.3, p < 0.001). For more details see Table 4.
Impact of alone/comorbid psychiatric variables on glycaemic control
In order to clarify the role of depression and alexithymia (alone or comorbid) on glycaemic control, a further analysis was performed after dividing the whole sample into four groups according to HAM-D and TAS-20 total scores: control group (no depression, no alexithymia); depression group (depression only); alexithymia group (alexithymia only); comorbidity group (depression+alexithymia). The 32.8 % (33 M; 9 F) of patients had neither depression nor alexithymia; the 17.2 % (13 M; 9 F) had depression only; the 16.4 % (12 M; 9 F) had alexithymia only; the 33.6 % (17 M; 26 F) had both depression and alexithymia.
Considering the raw values of HbA1c, the higher value was recorded in the comorbidity group (7.7 ± 1.2 %) followed by the alexithymia group (7.6 ± 2), the control group (7 ± 1.6 %) and depression group (6.9 ± 1.3 %). The comorbidity group presented a significantly higher value of HbA1c than the control group (p = 0.04) and the depression group (p = 0.02).
Multivariate analysis
A logistic regression has been performed using the value of the HbA1c as dependent variable and age, gender, HAM-D total score, TAS-20 total score and therapy of diabetes as independent variables. The HbA1c was found to be significantly associated with age (OR 0.9, CI 0.91–0.99), alexithymia (OR 3.1, CI 1.28–7.48) and insulin therapy (OR 24.3, CI 2.64–223.98). For more details, see Table 5.
Discussion
Interesting findings have arisen from the statistical analysis performed on our sample of type 2 diabetic patients.
One of the objectives of the present study was to assess the prevalence of depression and alexithymia. We found that more than the 17 % and the 16 % of the patients were depressed and alexithymic, respectively. Nearly the 34 % had both depression and alexithymia.
Unfortunately, the impact of depression and alexithymia on glycaemic control is still controversial [16, 17, 25]. Previous researches highlighted the bidirectional relationship between depression and diabetes. A recent cohort study demonstrated a certain association between weight cycling (common phenomenon in diabetes) and depression in type 2 diabetic patients [26]. Moreover, it seems that the association between diabetes and depression could be due to the somatic-affective component of the latter [27]; even the depressive temperament seems to negatively affect the glycaemic control adjustment to diabetes [28]. On the other hand, recent evidence suggests that the worse glycaemic control could be linked, more than to depression, to the diabetes-related distress [29], a psychosocial issue independently associated with self-management and perceived burden of diabetes [30].
Another research topic lately interesting the Scholars is the link between diabetes and alexithymia. In our study, alexithymia more than depression seems to exert a certain role on glycaemic control. More specifically, alexithymic patients compared to non-alexithymic patients presented a significantly higher HbA1c value while no significant difference in terms of HbA1c value was found between depressed patients and non-depressed patients. This association was confirmed at the multivariate analysis: in our sample alexithymia triples the risk of a worse glycaemic control. Our results are consistent with previous studies hypothesizing that alexithymia could even represent an independent risk factor for diabetes, as well as for high triglyceride levels and elevated blood pressure, thus determining a worsening of the metabolic syndrome [31, 32].
Nevertheless, even though in our study no difference between depressed patients and non-depressed patients was found in terms of HbA1c value, subjects with pathological HbA1c presented a higher HAM-D total score when compared to subjects with normal HbA1c: patients with a worse glucose balance are then more likely to present a depressive condition. Speculating, in this case, depression could be probably secondary to a worse glycaemic control. Diabetic patients with alexithymia, and even more those with depression+alexithymia, presented a higher HbA1c if compared to the other groups (control and depression group); therefore, it seems reasonable to hypothesize a certain impact of depression on worsening glycaemic control among alexithymic patients. Another interesting data is the positive correlation between TAS-20 subscale I (“difficulty in identifying feelings”) and HbA1c values. Since alexithymia is associated with unhealthy behaviors (such as compulsive eating or dieting) and a biased perception and reporting of somatic symptoms [33, 34], it is easy to imagine how diabetic patients with a difficulty in identifying feelings could scarcely understand their health condition, misinterpret their somatic symptoms and hardly cope with the complex management of diabetes. It has been previously demonstrated, in fact, that alexithymic patients are likely to show impaired ability of self-care and disease coping strategies [4]; hence, it would be risky to underestimate the impact of psychopathology on physical health. It is, in fact, universally recognized that patients with severe psychiatric disorders present worse systemic health and shortened life expectancy [35].
This study presents some limitations. Firstly, the small sample size limits the possibility to generalize our findings to the diabetic population. Secondly, the cross-sectional design of the study does not allow us to infer a causality role of depression and alexithymia on glycaemic control.
Thirdly, we did not consider other variables (e.g. stressful life events) that, according to literature data [36], could affect glucose balance. Our findings shed light to the controversial role of depression and alexithymia on glycaemic control: alexithymia, more than depression, influences glycaemic control of type 2 diabetic patients. Other studies are necessary to confirm or disconfirm the results here reported.
In conclusion, when evaluating a diabetic patient, a rapid screening for psychopathological alterations would guarantee a more accurate management. The treatment of any associated psychiatric disorders and the choice of more acceptable therapeutic approaches for special subpopulations of diabetic patients (e.g. alexithymic ones), thus favoring compliance and reducing the risk of diabetic complications, would improve the patients’ quality of life.
References
Cade WT (2008) Diabetes-related microvascular and macrovascular diseases in the physical therapy setting. Phys Ther 88:1322–1335. doi:10.2522/ptj.20080008
Manoudi F, Chagh R, Benhima I, Asri F, Diouri A, Tazi I (2012) Depressive disorders in diabetic patients. Encephale 38:404–410. doi:10.1016/j.encep.2012.01.010 (Article in French)
Kazemi-Galougahi MH, Ghaziani HN, Ardebili HE, Mahmoudi M (2012) Quality of life in type 2 diabetic patients and related effective factors. Indian J Med Sci 66:230–237. doi:10.4103/0019-5359.115216
Topsever P, Filiz TM, Salman S et al (2006) Alexithymia in diabetes mellitus. Scott Med J 51:15–20. doi:10.1258/rsmsmj.51.3.15
Luminet O, de Timary P, Buysschaert M, Luts A (2006) The role of alexithymia factors in glucose control of persons with type 1 diabetes: a pilot study. Diabetes Metab 32:417–424 (DM-11-2006-32-5-1262-3636-101019-200519822)
Anderson RJ, Freedland KE, Clouse RE, Lustman PJ (2001) The prevalence of comorbid depression in adults with diabetes: a meta-analysis. Diabetes Care 24:1069–1078. doi:10.2337/diacare.24.6.1069
Carnethon MR, Kinder LS, Fair JM, Stafford RS, Fortmann SP (2003) Symptoms of depression as a risk factor for incident diabetes: findings from the National Health and Nutrition Examination Epidemiologic Follow-up Study, 1971–1992. Am J Epidemiol 158:416–423. doi:10.1093/aje/kwg172
De la Roca-Chiapas JM, Hernández-González M, Candelario M et al (2013) Association between depression and higher glucose levels in middle-aged Mexican patients with diabetes. Rev Invest Clin 65:209–213 (23877807)
Lustman PJ, Anderson RJ, Freedland KE, de Groot M, Carney RM, Clouse RE (2000) Depression and poor glycemic control: a meta-analytic review of the literature. Diabetes Care 23:934–942. doi:10.2337/diacare.23.7.934
Katon WJ, Young BA, Russo J et al (2013) Association of depression with increased risk of severe hypoglycemic episodes in patients with diabetes. Ann Fam Med 11:245–250. doi:10.1370/afm.1501
Eren I, Erdi O, Sahin M (2008) The effect of depression on quality of life of patients with type II diabetes mellitus. Depress Anxiety 25:98–106. doi:10.1002/da.20288
de Groot M, Anderson R, Freedland KE, Clouse RE, Lustman PJ (2001) Association of depression and diabetes complications: a meta-analysis. Psychosom Med 63:619–630. doi:10.1097/00006842-200107000-00015
Coleman SM, Katon W, Lin E, Von Korff M (2013) Depression and death in diabetes; 10-year follow-up of all-cause and cause-specific mortality in a diabetic cohort. Psychosom 54:428–436. doi:10.1016/j.psym.2013.02.015
Lustman PJ, Clouse RE (2005) Depression in diabetic patients: the relationship between mood and glycemic control. J Diabetes Complicat 19:113–122. doi:10.1016/j.jdiacomp.2004.01.002
Farges F, Corcos M, Speranza M et al (2004) Alexithymie et toxicomanie: lien avec la depression [Alexithymia, depression and drug addiction]. Enceph 30:201–211. doi:10.1016/S0013-7006(04)95431-0 (Article in French)
Tsujii S, Hayashino Y, Ishii H, Diabetes Distress and Care Registry at Tenri Study Group (2012) Diabetes distress, but not depressive symptoms, is associated with glycaemic control among Japanese patients with type 2 diabetes: Diabetes Distress and Care Registry at Tenri (DDCRT 1). Diabet Med 29:1451–1455. doi:10.1111/j.1464-5491.2012.03647.x
Friedman S, Vila G, Even C et al (2003) Alexithymia in insulin-dependent diabetes mellitus is related to depression and not to somatic variables or compliance. J Psychosom Res 55:285–287. doi:10.1016/S0022-3999(02)00636-0
Bagby RM, Parker JDA, Taylor GJ (1994) The twenty-item Toronto Alexithymia Scale: I. Item selection and cross-validation of the factor structure. J Psychosom Res 38:23–32. doi:10.1016/0022-3999(94)90005-1
Taylor GJ, Bagby RM, Parker JDA (1997) Disorders of affect regulation: alexithymia in medical and psychiatric illness. Cambridge University Press, Cambridge
Picardi A, Caroppo E, Porcelli P, Di Maria G, Munittola G, Martinotti G (2012) Alexithymia and severe psychopathology: a study on psychiatric inpatients. Psychopathol 45:159–166. doi:10.1159/000330261
Hamilton M (1960) A rating scale for depression. J Neurol Neurosurg Psychiat 23:56–62. doi:10.1136/jnnp.23.1.56
Spitzer WO, Dobson AJ, Hall J et al (1981) Measuring the quality of life of cancer patients: a concise QL-index for use by physicians. J Chronic Dis 34:585–597. doi:10.1016/0021-9681(81)90058-8
The American Diabetes Association (2010) Diagnosis and classification of diabetes mellitus. Diabetes Care 33:S62–S69. doi:10.2337/dc10-S062
Sacks DB, Bruns DE, Goldstein DE (2002) Guidelines and recommendations for laboratory analysis in the diagnosis and management of diabetes mellitus. Clin Chem 48:436–472. doi:10.2337/dc11-9998
Alonso-Moràn E, Satylganova A, Orueta JF, Nuno-Solinis R (2014) Prevalence of depression in adults with type 2 diabetes in Basque Country: relationship with glycaemic control and health care costs. BMC Public Health 14:769. doi:10.1186/1471-2458-14-769
Messier L, Elisha B, Schmitz N et al (2014) Weight cycling and depressive symptoms in diabetes: a community-based study of adults with type 2 diabetes mellitus in Quebec. Can J Diabetes 38:456–460. doi:10.1016/j.jcjd.2014.01.005
Wiltink J, Michal M, Wild PS et al (2014) Association between depression and diabetes in the community: do symptoms dimensions matter ? Results from the Gutenberg Health Study. PLoS One 9:e105499. doi:10.1371/journal.pone.0105499
Gois C, Barbosa A, Ferro A et al (2011) The role of affective temperaments in metabolic control in patients with type 2 diabetes. J Affect Disord 134:52–58. doi:10.1016/j.jad.2011.05.021
Fisher L, Glasgow RE, Strycker LA (2010) The relationship between diabetes distress and clinical depression with glycemic control among patients with type 2 diabetes. Diabetes Care 33:1034–1036. doi:10.2337/dc09-2175
Aikens JE (2012) Prospective associations between emotional distress and poor outcomes in type 2 diabetes. Diabetes Care 35:2472–2478. doi:10.2337/dc12-0181
Lemche AV, Chaban OS, Lemche E (2010) Alexithymia predicts triglyceride level, systolic blood pressure, and diabetic status in metabolic syndrome. Eur Psychiatry 25:1019. doi:10.1016/S0924-9338(10)71009-9
Lemche AV, Chaban OS, Lemche E (2014) Alexithymia as a risk factor for type 2 diabetes mellitus in the metabolic syndrome: a cross-sectional study. Psychiatry Res 215:438–443. doi:10.1016/j.psychres.2013.12.004
Lumley MA, Neely LC, Burger AJ (2007) The assessment of alexithymia in medical settings: implications for understanding and treating health problems. J Pers Assess 89:230–246. doi:10.1080/00223890701629698
Lumley MA, Stettner L, Wehmer F (1996) How are alexithymia and physical illness linked? A review and critique of pathways. J Psychosom Res 41:505–518. doi:10.1016/S0022-3999(96)00222-X
De Hert M, Dekker JM, Wood D, Kahl KG, Holt RIG, Möller H-J (2009) Cardiovascular disease and diabetes in people with severe mental illness position statement from the European Psychiatric Association (EPA), supported by the European Association for the Study of Diabetes (EASD) and the European Society of Cardiology (ESC) (Short Survey). Eur Psychiatry 24:412–424. doi:10.1016/j.eurpsy.2009.01.005
Fisher L, Mullan JT, Arean P, Glasgow RE, Hessler D, Masharani U (2010) Diabetes distress but not clinical depression or depressive symptoms is associated with glycaemic control in both cross-sectional and longitudinal analyses. Diabetes Care 33:23–28. doi:10.2337/dc09-1238
Conflict of interest
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Additional information
A. Luca and M. Luca contributed equally to this work.
Rights and permissions
About this article
Cite this article
Luca, A., Luca, M., Di Mauro, M. et al. Alexithymia, more than depression, influences glycaemic control of type 2 diabetic patients. J Endocrinol Invest 38, 653–660 (2015). https://doi.org/10.1007/s40618-015-0238-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-015-0238-2