Abstract
Purpose of the review
Perioperative anaphylaxis is a severe, systemic hypersensitivity reaction of rapid onset, that is generally unpredictable, may be life-threatening, and occurs after the use of different drugs, substances, or stimuli during an anesthetic or surgical procedure.
Recent findings
The potential causes of reactions during procedures depend on the moment it occurs. In the induction phase, neuromuscular blockers, intravenous antibiotics, and opioids should be considered. During the procedure, along with antibiotics and opioids, other causes include local anesthetics, nonsteroidal anti-inflammatory agents (NSAIDs), contrast media, clorhexidin, and latex. In the recovery phase, the most common agents are latex and medications which reverse anesthesia effects. Recently, sugammadex has been discovered to be an important causal agent.
Summary
Early onset of treatment is essential in anaphylaxis and could prevent complications or death. The management is the same in all ages following the accepted protocols for the management of anaphylaxis. Adrenaline is the treatment of choice. A complete review of the clinical and perioperative history is essential before any procedure in patients with prior anaphylaxis. It is recommended that surgeons and anesthesiologists communicate each other their concerns and ask the allergist if there is a doubt about an allergic reaction.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Perioperative anaphylaxis is a severe, systemic hypersensitivity reaction of rapid onset, that is generally unpredictable, may be life-threatening, and occurs after the use of different drugs, substances, or stimuli during an anesthetic or surgical procedure [1]. Perioperative anaphylaxis has a higher mortality rate than anaphylaxis in other settings. This is mostly due to impairment of early recognition factors of anaphylaxis. The known etiology may be difficult to acknowledge because the patient receives multiple medications during the same period of time [2••].
Epidemiology
The estimated incidence of perioperative anaphylaxis varies from 1:25,00 to 1:20,000 surgeries, with a mortality rate ranging from 3 to 9% [3]. Around the world, it accounts for 9–19% of all surgical complications and 5–7% of all deaths during anesthesia, occuring equally in girls and boys. It is more common in adult women than men. The proportion of IgE-mediated allergic reactions is around 60% of all reactions, and it appears to be similar between countries [2••].
Types of reactions
Immunoglobulin E-mediated anaphylaxis
During immediate IgE-mediated hypersensitivity systemic reactions, proinflammatory mediators are released from mast cells and basophils. Histamine, triptase, cytokines, mediators derived from phospholipids such as prostaglandin D2, leukotrienes, thromboxane A2, and platelet-activating factor are other mediators released. Affected organs include skin, mucous membranes, respiratory, cardiovascular, and gastrointestinal systems [4, 5].
Non-IgE-mediated-anaphylaxis
Nonallergic anaphylaxis occurs without specific immune response and includes several mechanisms, such as activation of the complement system with production of anaphylatoxins, dysfunction of arachidonic acid metabolism, and direct activation of mast cells [3].
Causal agents of perioperative anaphylaxis
The potential causes of reactions during procedures depend on the moment it ocurrs. In the induction phase, neuromuscular blockers, intavenous antibiotics, and opioids should be considered. During the procedure, along with antibiotics and opioids, other causes include local anesthetics, nonsteroidal anti-inflammatory agents (NSAIDs), contrast media, clorhexidin, and latex. In the recovery phase, the most common agents are latex and medications which reverse anestesia effects [6•].
The risk of anaphylaxis increases with frequency, intravenous route of administration, and the specific antigen exposure time. Chronic lung disease, recent intracranial surgery, and hyperthyroidism are comorbidities that enhance the severity of the symptoms and decrease the response to treatment.
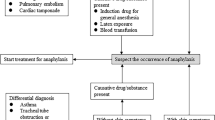
There are numerous variables that can impact the most common causes of intraoperative anaphylaxis (Fig. 1) from country to country. These variables include the ability to identify intraoperative anaphylaxis and initiate referral, the severity of the reactions that are included, the type of neuromuscular blockers, the understanding of the evaluation, sensitizing substances available, specific testing to medications used, and whether or not skin prick or intradermal tests are used for diagnosis [7].
Antibiotics or dyes are reported to cause allergic reactions. Reactions from neuromuscular blocking agents (NMBAs) remain an important cause in various countries but are not always reported. Other causal agents are chlorhexidine and recently sugammadex [8]. On the other hand, reactions to local anesthetics are uncommon [9].
Population with higher risk factors
-
Patients with history of allergic reacton in previous procedures or anesthesia.
-
Patients with drug allergy which has been diagnosed previously and may be used in the procedure.
-
Patients with high latex exposure due to multiple procedures (children with spina bifida).
-
Patients with suspicion of latex allergy due to symptoms or those who present oral or generalized symptoms when in contact with banana, kiwi, avocado, pineapple, papaya, or chestnut mainly because of cross-reactivity between latex and mentioned foods [3].
Local anesthetics
Local anesthetics stabilize neuronal membranes by inhibiting the ionic movement required for the trasmission of neural impulses. They are used in dentistry, ophthalmology, minor surgery, endoscopies, and obstetrics.
Local anesthetics are available in multiple forms including sprays, ointments, gels, solutions, and injectable forms. The topical anesthetics seem to be safe. Allergic contact dermatitis can occur. True allergy to local anesthetics represents less than 1% of all reactions.
Types of local anesthetics
Local anesthetics may be classified into two groups (Table 1), i.e., benzoic acid esters (group 1) or primary amides (group 2) based on the linking chain between the hydrophilic amine group and the lipophilic aromatic ring.
The commonly used benzoid acid ester is procaine (Novocain). The benzoid acid esters often cross-react with each other but do not cross-react with primary amides [9].
Lidocaine is the prototype for amide local anesthetics, and it is characterized by a rapid onset and intermediate duration of action. Mepivacaine has a rapid onset and medium duration of efficacy. Bupivacaine is 4 times more potent than lidocaine and mepivacaine, and has longer duration and action. This agent has potential higher cardiotoxicity and should be used with caution in patients taking B-blockers or digoxin [10].
The para-amino group (procaine, tetracaine) has a greater ability to cause allergic reactions than the amide group (lidocaine, bupivacaine, mepivacaine) [11].
Neuromuscular blockers
Neuromuscular blockers (NMBs) have a major role in causing perioperative anaphylaxis (POP). Both depolarizing (suxamethonium) and nondepolarizing NMBs (benzylisoquinolines and aminosteroids) are capable of triggering POP via IgE-mediated or nonIgE mediated pathways [12]. According to various studies, the most common cause of anaphylaxis during anesthesia was reportedly NMBs (50–70%). Succinylcholine (60.6%) and atracurium (19.6%) were the major causative drugs, whereas anaphylaxis to cisatracurium (5.9%), vecuronium (4.6%), rocuronium (4.3%), pancuronium (2.7%), and mivacurium (1.9%) was also reported [13].
Due to ammonium structures, sensitization may occur to these agents after exposure to materials containing tertiary and quaternary ammonium groups including over-the-counter drugs, cosmetics, disinfectants, and products found in food.
Women have a 3-fold risk of developing anaphylaxis than males do. Hairdressers through exposure from their work products have been found to have a higher rate of sensitization to NMBs. Because there is no sex difference in the incidence of anaphylaxis before puberty, another hypothesis supports hormonal involvement [14••].
Cross-sensitivity is approximately 60 to 70% among the neuromuscular blockers. In general, succinylcholine and rocuronium are thought to be associated with increased allergic risk, and atracurium, cisatracurium, and pancuronium with less risk. However, atracurium is associated with histamine release, which may lead to bronchospasm and hypotension [15].
Sugammadex
Sugammadex is a reversal agent for neuromuscular blockers and a newest cause of perioperative anaphylaxis (POP). Japan has been considered the highest user of sugammadex, being used in 10% of anesthetic cases, and the incidence of perioperative reactions is 1 in 2500 cases [16].
Reactions to sugammadex occur late in the surgery because they are given to reverse neuromuscular blockade [13]. The sensitizing trigger is not definitively known. Cyclodextrin is found in food additives and cosmetics, and this potentially could be the causal agent [17••].
Antibiotics
Penicillins and cephalosporins are responsible for the majority of antibiotic-induced perioperative anaphylactic reactions. The incidence has been increasing due to a widespread use of perioperative antibiotic prophylaxis with beta-lactams [3].
Amoxicillin or amoxicillin/clavulanic acid are the leading antibiotic trigger for perioperative anaphylaxis, but has recently been substituted in some regions by teicoplanin [9].
Confirmation of teicoplanin allergy is challenging because of the limited sensitivity and lack of standardization of skin test [18].
The common practice of administering antibiotics after induction of anesthesia runs the risk of antibiotic-induced anaphylaxis being aggravated by general or neuraxial anesthesia [19].
Latex
Latex allergy is a major cause of perioperative anaphylaxis worldwide. Latex is derived from the Hevea brasiliensis tree. Among its allergens of clinical relevance are Hev b 1, Hev b 3, Hev b 5, Hev b 6.01, and Hev b 7.
Patients at risk of developing latex allergy include atopic patients who experience high latex exposure, medical interventions, and several surgical procedures. Symptoms associated include skin rash, bronchospasm, and cardiovascular collapse. They tend to appear in the maintenance phase of anesthesia.
Diagnosis is clinically supported by skin prick test (SPT) or serum-specific IgE to Hev b 1. Challenge test, such as glove use test, should be applied if there is a highly suggestive history of latex allergy, but the diagnostic tests are negative [3].
Chlorhexidine
Chlorhexidine is now well-recognized as a cause of perioperative anaphylaxis occurring between 9.3 and 9.6% [20].While intravascular (via coated central venous catheters) or intramucosal exposure (via lubricating gel for urethral catheters) remains the predominant route of exposure, pure cutaneous contact may also trigger anaphylaxis [21, 22]. Skin prick test and specific IgE are reliable tools exhibiting high levels of sensitivity and specificity, exceeding 90% [23].
Dyes
Blue dye, used for sentinel node biopsy during cancer surgery, was identified in 6% of cases of perioperative anaphylaxis. It can be potentially missed as a culprit since it is administered intradermally by surgeons and not routinely listed on drug charts by anesthetists [24].
Morphinomimetics
Anaphylaxis during the perioperative period due to opioids is very rare [25]. Direct activation of dermal mast cells by narcotics stimulates histamine release [26, 27]. Codeine, morphine, and hydrocodone are the opioids that most frequently cause allergic reactions [28]. Morphine is a tertiary amine that causes nonimmunological histamine release, and meperidine is the opioid that most frequently causes nonimmunological histamine release rather than other opioids. Besides, IgE-mediated anaphylaxis related to fentanyl has been reported. IgE-mediated reactions have been described [29]. Mostly, these reactions include redness, urticaria, pruritus, and mild hipotension [25]. Several risk factors have been described to present morphinomimetics reactions, such as previous drug sensitization, history of opioid allergy, drug addiction (heroine), advanced age (> 65 years), obesity, obstructive pulmonary disease, sleep apnea, and anaphylaxis history. CYP2D6*41 polymorphism increases the risk of presenting adverse events to oxycodone and hydrocodone, and 1846G > A and 2549 > A variants to codeine. Anaphylaxis can be mortal during anesthesia in 9% [28].
For the diagnosis, prick tests and intradermal tests can be performed, in which phenylpiperidines are used (Table 2), cross-reactivity is uncommon between phenylpiperidines.
Hypnotics
Thiopental anaphylaxis is the most frequent among barbiturates. Its incidence is estimated in 1 in every 30,000 administrations, followed by nonbarbiturates. Propofol has an incidence of 1 in every 60,000 [30]. The mechanism of action in these types of reactions is by direct histamine release. In addition, IgE-mediated hypersensitivity has been described. Reactions are more frequent in women [25]. Patients allergic to egg and soy have not been shown to present more risk to propofol anaphylaxis. Due to its formulation characteristics, 10% soy, 2.25% glicerol, and 1.2% egg lectin, reactions that involve propofol are mostly related to aliphenol that carries two isopropyl groups that act as antigen epitopes, which are found in dermatological products and can explain anaphylaxis on the first exposition to propofol. Histamine release can be directly stimulated and this effect can be greater when it is administrated with muscle relaxants [26].
Allergic reactions to benzodiazepines are rare. The solvent Cremophor has been described as the responsible of the majority of the reactions. Drugs related to anaphylaxis to benzodiazepines are diazepam and midazolam. Allergic reactions to intravenous diazepam are caused by propylene glycol and can be found in cosmetic drugs, vaccines, and occasionally in foods, and can present cross-reactivity with other benzodiazepines caused by the metabolite demethyldiazepam. Midazolam is a safe drug, since it does not contain metabolites. Also, it has been used safely to induce anesthesia in patients with food allergy. [25, 29, 30].
Hypnotics, including barbiturates (thiopental) and no barbiturates (propofol) can induce IgE-mediated hypersensitivity reactions, as well as nonIgE-mediated reactions [31].
In order to diagnose allergy to hypnotics, skin tests can be done with the concentrations shown in Table 3.
Neuroleptics
Neuroleptics are drugs that act on the dopaminergic system. Several classes exist according to their biochemical structure. Reactions to haloperidol are rare and are caused by an IgE-mediated hypersensitivity. Droperidol, commonly used as antiemetic and sedative on postoperative period, is very unusual to unleash reactions, but the risk is greater if phenotiazin hypersensitivity is present. Symptoms more frequently present in allergic reactions are pruritus, urticaria, bronchospasm and angioedema, as well as anaphylaxis. Prick tests can be performed to establish a diagnosis [28].
The neuroleptic malignant syndrome has been described as a rare and sometimes fatal adverse reaction, which is produced by the administration of antipsychotics and dopamine antagonists or by the rapid withdrawal of antiparksonian drugs. It is characterized by presenting hyperthermia and muscular stiffness, as well as autonomous instability, diaphoresis, alteration of the state of consciousness, CPK elevation, and leukocytosis. [32].
Colloids
Plasma-derivated products or colloids can produce immunological and nonimmunological reactions during anesthesia [33]. Albumin, dextran, hydroxyethylamidon, and gelatin are colloids that are frequently used in the operating room and cause around 4% of perioperatory anaphylaxis [25]. They can cause severe allergic reactions occurring during the first 20 min of the infusion in 20% of patients [34].
It has been described that people with a history of allergy to multiple drugs and men are more at risk to develop an allergic reaction to these agents. Egg allergy is not a contraindication to use albumin since it is different to human albumin [29]. For the diagnosis, pure undiluted solution can be used to perform prick tests using a 1/10 pure solution, and in case that those result negative, intradermal tests can be done (1/1000 to 1/10) [30, 35].
Diagnosis
The diagnosis of anaphylaxis is mainly clinical and is based on the appearance and severity of symptoms, as well as the time elapsed between exposure to the medication used in surgery and the onset of the condition. The lack of experience, not having the patient’s body in sight and the varied use of medications during anesthesia makes it difficult to establish a timely diagnosis [36].
The time elapsed between the administration of the suspect medication and the clinical signs and symptoms is very useful. In general, when symptoms occur within the first 30 min of anesthesia, the main culprits are neuromuscular blockers, antibiotics, and hypnotics. When symptoms begin after 30 min of anesthesia, the causes include chlorhexidine, latex, dyes, plasma expanders, and blood products [14••].
In the perioperative environment, classic anaphylaxis symptoms such as dyspnea or pruritus are not regularly present, and it is common to find alterations in patient ventilation, oxygen saturation, heart rate, and blood pressure. The absence of skin symptoms does not exclude the diagnosis of anaphylaxis. It is common that, given these symptoms, an incorrect diagnosis is made due to the various factors associated in a surgery and the previous medical condition of the patient; frequently, an evaluation by the allergist is not possible, and this fact can lead to a potentially fatal future reexposure [37].
Anaphylaxis can occur at any time during anesthesia. The reactions associated with neural plaque blockers or antibiotics commonly occur in up to 90% of cases in the induction phase, while those related to latex appear in later stages of anesthesia [38].
Histamine and tryptase are two mediators released by basophils and mast cells during allergic reactions. Both can be measured in the patient’s serum, and their utility has been demonstrated in the diagnosis of perioperative anaphylaxis. However, they should be taken in the first hours of the reactions since the half-lives of histamine and tryptase are 20 and 120 min, respectively.
For tryptase, a sensitivity of 64% and specificity of 89% is reported with a positive predictive value of 92% and a negative predictive value of 54%. In relation to histamine, sensitivity is reported at 75%, specificity at 51% [39].
Skin tests can identify the causative agent but are usually performed after the month of presentation of anaphylaxis, so its usefulness lies in avoiding subsequent cases. Serum detection of medications related to perioperative anaphylaxis is a useful tool to complement the patient’s approach. Tests for specific IgE of neuromuscular plaque blockers, thiopental, morphine, propofol, and latex are also available.
Skin tests along with the medical history remain the basis of diagnosis for an IgE-mediated reaction. Skin tests are usually performed 4 to 6 weeks after the reaction. The tests should be performed by a professional with experience in performing and interpreting tests with medications used in anesthesia. However, asymptomatic sensitization is common. A positive skin test or an elevated level of agent-specific IgE only indicates that the patient is sensitized to that agent, but does not verify that it is the cause of anaphylaxis [14••].
Prick and intradermal skin tests with dilutions of commercially available medications are recommended. Standardized procedures and dilutions must be precisely defined for each medication used to avoid false positive results [40].
The indications for the challenge tests are limited. They should be performed only when obtaining a negative skin test and the evidence recommend those for local anesthetics, beta-lactam antibiotics, and latex [41].
Treatment
Early onset of treatment is essential in anaphylaxis and could prevent hypoxic complications or death. The management is basically the same in all ages following the accepted protocols for the management of anaphylaxis. Adrenaline is the treatment of choice in anaphylaxis due to its alpha and beta-agonist properties, which result in vasoconstriction, increased peripheral vascular resistance, decreased mucosal edema, inotropism, chronotropism, and bronchodilation. Other recommended actions are the suspension of the administration of the drug. The administration of anesthetic drugs and the surgical intervention should be suspended when the reaction occurs in the anesthetic induction. Other than the administration of adrenaline, 100% oxygen, fluid replacement, and place the patient in Trendelenburg position are recommended [5, 42].
Prevention
Evidence has shown that a complete review of the clinical and perioperative history is essential before any procedure in patients with prior anaphylaxis because they have a higher risk of recurrence during subsequent anesthetics.
It is highly recommended that surgeons and anesthesiologists communicate each other their concerns and ask the allergist if there is a doubt about an allergic reaction. An allergy evaluation should then be performed based on the clinical and surgical history to identify the drug or substance responsible [14••]. Allergy tests should be performed on all high-risk patients before any future anesthesia. Recently, recommendations have been proposed to identify the population at risk of perioperative anaphylaxis that could benefit from a more precise preoperative evaluation to investigate sensitization to drugs and anesthetic agents, provided that the opportunity for surgery permits. There are some risk factors that predispose to anaphylaxis during the perioperative period, which are the following: patients allergic to any of the drugs that can be administered during anesthesia; patients who have presented symptoms that suggest an allergic reaction in previous anesthesia; patients who presented an allergy-suggestive clinic after exposure to latex; children who have undergone multiple surgical interventions, especially those suffering from spina bifida; patients who had an allergy to avocado, kiwi, banana, chestnut; and patients who report clinical symptoms before exposure to the cold [43].
It is very important to completely avoid contact with latex from the first surgical procedure in children with spina bifida or who will undergo multiple interventions. Latex particles can be dispersed in the air with the use of powder gloves, assuming a risk factor for sensitization by inhalation. The incidence of sensitization in healthcare professionals can be reduced by wearing gloves without powder [44]. In patients where there is no available alternative to the one that triggered anaphylaxis, desensitization could be proposed considering factors such as type of immunological reaction, risk benefit of the procedure, and availability of personnel and facilities necessary to perform desensitization.
Premedication schemes have shown that they do not effectively prevent perioperative anaphylaxis. The only prevention is to avoid contact with the medication responsible for the reaction [14••].
Conclusions
Perioperative anaphylaxis is a severe, systemic hypersensitivity reaction that occurs of rapid onset. In most patients, it is generally unpredictable and presents after exposure to different drugs, substances, or stimuli during an anesthetic or surgical procedure. Early onset of treatment is essential in anaphylaxis and could prevent complications or death. The management is established according to anaphylaxis protocols, being adrenaline the treatment of choice. A complete review of the clinical and perioperative history is essential before any procedure in patients with prior anaphylaxis. It is recommended that surgeons and anesthesiologists communicate each other their concerns and request a patient evaluation by an allergist when needed.
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
López González JM, García Paz V, Gómez Jiménez M, et al. Perioperative anaphylaxis. Cir May Amb. 2013, 2;18.
•• Hsu K, Hepner DL. Current knowledge and management of hypersensitivity to perioperative drugs and radiocontrast media. J Allergy Clin Immunol Pract. 2017;5(3):587–92. https://doi.org/10.1016/j.jaip.2017.03.016 Excellent paper on the management approach of perioperative anaphylaxis.
Régnier Galvão V, Giavina-Bianchi P, Castells M. Perioperative anaphylaxis. Curr Allergy Asthma Rep. 2014;14(8):452–10. https://doi.org/10.1007/s11882-014-0452-6.
Simons FE, Ardusso LR, Bilò MB, World Allergy Organization, et al. 2012 update: World Allergy Organization guidelines for the assessment and management of anaphylaxis. Curr Opin Allergy Clin Immunol. 2012;12:389–99.
Dhami S, Panesar SS, Roberts G, Muraro A, Worm M, Bilò MB, et al. Management of anaphylaxis: a systematic review. Allergy. 2014;69(2):168–75.
• Jares E. Reactions to drugs used in anesthesiology. In: Drug Hypersensitivity. Editorial Médica Panamericana. 2017, pp.18–19. This reference provides a complete and comprehensive review of important periopeative hypersensitivity causal agents.
Mertes PM, et al. Epidemiology of perioperative anaphylaxis. Presse Med. 2016Sep;45(9):758–67. https://doi.org/10.1016/j.lpm.2016.02.024.
Dong SW, Mertes PM, Petitpain N, Hasdenteufel F, Malinovsky JM, Gerap. Hypersensitivity reactions during anesthesia. Results from the ninth French survey (2005–2007). Minerva Anestesiol. 2012;78:868–78.
Volcheck GW, Mertes PM. Local and general anesthetics immediate hypersensitivity reactions. Immunol Allergy Clin N Am. 2014;34:525–46.
Ogle OE, Mahjoubi G. Local anesthesia: agents, techniques and complications. Dent Clin N Am. 2012;56(1):133–48.
Michavila Gomez AV, Belver Gonzalez MT, Alvarez NC, Giner Muñoz MT, Hernando Sastre V, Porto Arceo JA, et al. Perioperative anaphylactic reactions. Review and procedure protocol in paediatrics. Allergol Immunopathol (Madr). 2015;43(2):203–14.
McNeil BD, Pundir P, Meeker S, Han L, Undem BJ, Kulka M, et al. Identification of a mast cell-specific receptor crucial for pseudo-allergic drug reactions. Nature. 2015;519:237–41. https://doi.org/10.1038/nature14022.
Takazawa T, Mitsuhata H, Mertes PM. Sugammadex and rocuronium-induced anaphylaxis. J Anesth. 2015;30(2):290–7. https://doi.org/10.1007/s00540-015-2105-x.
•• Volcheck GW, Hepner DL. Identification and management of perioperative anaphylaxis. J Allergy Clin Immunol Pract. 2019;7(7):2134–42. https://doi.org/10.1016/j.jaip.2019.05.033 This article provides valuable up to date information regarding awareness and optimal treatment for perioperative anaphylaxis.
Mertes PM, Volcheck GW. Anaphylaxis to neuromuscular blocking drugs: all neuromuscular blocking drugs are not the same. Anesthesiology. 2015;122(1):5–7.
Miyazaki Y, Sunaga H, Kida K, Hobo S, Inoue N, Muto M, et al. Incidence of anaphylaxis associated with sugammadex. Anesth Analg. 2018;126:1505–8.
•• Min KC, Woo T, Assaid C, McCrea J, et al. Incidence of hypersensitivity and anaphylaxis with sugammadex. J Clin Anesth. 2018;47:67–73 This article describes up and coming hypersensitivity reactions to recently identifed agent, sugammadex.
Savic LC, Garcez T, Hopkins PM, Harper NJN, Savic S. Teicoplanin allergy— an emerging problem in the anaesthetic allergy clinic. Br J Anaesth. 2015;115:595–600. https://doi.org/10.1093/bja/aev307.
Misbah SA, Krishna MT. Perioperative anaphylaxis- an investigational challenge. Front Immunol. 2019;10:1117.
Opstrup MS, Malling HJ, Kroigaard M, et al. Standardised testing with chlorhexidine in perioperative allergy—a large single-centre evaluation. Allergy. 2014;69:1390–6. https://doi.org/10.1111/all.12466.
Spoerl D, Jandus P, Harr T. Pitfalls and peculiarities in chlorhexidine allergy. J Allergy Clin Immunol Pract. 2016;4:991–2. https://doi.org/10.1016/j.jaip.2016.03.017.
Lasa EM, Gonzalez C, Garcia-Lirio E, Martinez S, Arroabarren E, Gamboa PM. Anaphylaxis caused by immediate hypersensitivity to topical chlorhexidine in children. Ann Allergy Asthma Immunol. 2017;118:118–9. https://doi.org/10.1016/j.anai.2016.10.013.
Anderson J, Rose M, Green S, Fernando SL. The utility of specific IgE testing to chlorhexidine in the investigation of peri-operative adverse reactions. Ann Allergy Asthma Immunol. 2015;114:417–32. https://doi.org/10.1016/j.anai.2015.02.002.
Krishna MT, York M, Chin T, Gnanakumaran G, Heslegrave J, Derbridge C, et al. Multicentre retrospective analysis of anaphylaxis during anaesthesia in the United Kingdom: aetiology and diagnostic performance of acute serum tryptase. Clin Exp Immunol. 2014;178:399–404. https://doi.org/10.1111/cei.12424.
Kannan JA, Bernstein JA. Perioperative anaphylaxis diagnosis, evaluation, and management. Immunol Allergy Clin N Am. 2015;35(2):321–34. https://doi.org/10.1016/j.iac.2015.01.002.
Chacko T, Ledford D. Peri-anesthetic anaphylaxis. Immunol Allergy Clin N Am. 2007;27(2):213–30. https://doi.org/10.1016/j.iac.2007.03.006.
Adriaensens I, Vercauteren I, Soetens M, et al. Allergic reactions during labour analgesia and caesarean section anaesthesia. Int J Obstet Anesth. 2013;22:231–42. https://doi.org/10.1016/j.ijoa.2013.04.010.
González-Díaz SN, Arias Cruz A, Monge Ortega OP. Reacciones adversas inmunitarias y no inmunitarias a fármacos de uso perioperatorio. Rev Alerg Mex. 2019;66(1):99–114.
Hepner D, Castells M. Anaphylaxis during the perioperative period. Anesth Analg. 2003;97(5):1381–95. https://doi.org/10.1213/01.ANE.0000082993.84883.7D.
Dewachter P, Mouton-Faivre C, Emala CW. Anaphylaxis and anesthesia: controversies and new insights. Anesthesiology. 2009;111:1141–50. https://doi.org/10.1097/ALN.0b013e3181bbd443.
Nel L, Efrem E. Peri-operative anaphylaxis. Br J Clin Pharmacol. 2011;71(5):647–58. https://doi.org/10.1111/j.1365-2125.2011.03913.x PMID: 21235622; PMCID: PMC3093071.
Nisijima K, Shioda K. A rare case of neuroleptic malignant syndrome without elevated serum creatine kinasa. Nueropsychiatr Dis Treat. 2014;10:403–7.
Norred C. Anesthetic-induced anaphylaxis. Update for Nurse Anesthetists. AANA J. 2012;80(2):129–40.
Mali S. Anaphylaxis during the perioperative period. Anesth Essays Res. 2012;6(2):124–33. https://doi.org/10.4103/0259-1162.108286 PMID: 25885604; PMCID: PMC4173455.
Dewachter P, Mouton-Faivre C. What investigation after an anaphylactic reaction during anaesthesia? Curr Opin Anaesthesiol. 2008;21(3):363–8.
Rocha MJF. Cómo hacer frente a una reacción alérgica en el perioperatorio: del rash a la anafilaxia. Rev Mex Anest. 2013;36(S1).
Mertes PM, Laxenaire MC. Allergic reactions occurring during anaesthesia. Eur J Anaesthesiol. 2002;19(4):240–62.
Kroigaard M, Garvey LH, Menne T, et al. Allergic reactions in anaesthesia: are suspected causes confirmed on subsequent testing? Br J Anaesth. 2005;95(4):468–71.
Mertes PM, Laxenaire MC, Alla F. Anaphylactic and anaphylactoid reactions occurring during anesthesia in France in 1999–2000. Anesthesiology. 2003;99(3):536–45.
Mertes PM, Tajima K, Regnier-kimmoun MA, et al. Perioperative anaphylaxis. Med Clin N Am. 2010;94(4):761–89 xi.
Demoly P, Romano A, Botelho C, Bousquet-Rouanet L, Gaeta F, Silva R, et al. Determining the negative predictive value of provocation tests with beta-lactams. Allergy. 2010;65(3):327–32.
Berrio Valencia MI. Perioperative anaphylaxis. Braz J Anesthesiol. 2015;65(4).
Mertes PM, Malinovsky JM, Jouffroy L, Aberer W, Terreehorst I, Brockow K. Reducing the risk of anaphylaxis during anesthesia: 2011 updated guidelines for clinical practice. J Investig Allergol Clin Immunol. 2011;21:442–53.
Mertes PM, Lambert M, Gunt's Rodriguez RM, Aimone Gestin I, Mouton Faivre C, Moneret Vautrin DA, et al. Perioperative anaphylaxis. Immunol Allergy Clin North Am. 2009;29:429–5.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Sandra Nora González-Díaz, Cindy Elizabeth de Lira-Quezada, Rosalaura Virginia Villarreal-González, Rosa Ivett Guzmán-Avilán, Carlos Macouzet-Sanchez and Gabriela Galindo Rodríguez declare no conflicts of interest relevant to this manuscript.
Human and animal rights and informed consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Anaphylaxis
Rights and permissions
About this article
Cite this article
Gonzalez-Diaz, S.N., de Lira-Quezada, C.E., Villarreal-Gonzalez, R.V. et al. Perioperative Anaphylaxis. Curr Treat Options Allergy 7, 198–210 (2020). https://doi.org/10.1007/s40521-020-00250-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40521-020-00250-2