Abstract
Background
Heart rate variability (HRV) is a method to assess the autonomic nervous system and reflects possibly central brain states. HRV has previously not been examined in patients with hip fracture and delirium.
Aims
To explore HRV parameters in hip fracture patients with and without delirium.
Methods
Patients admitted to Oslo University Hospital with hip fracture and sinus rhythm in electrocardiogram (ECG) were included. Delirium was diagnosed using the confusion assessment method. HRV was assessed preoperatively after a relaxing period of five minutes, by measuring an ECG signal over 5 min. Parameters in time domain (the standard deviation of the QRS distances—SDNN) and frequency domain (total power (TP), low frequency (LF), high frequency (HF) and LF/HF ratio) were calculated.
Results
Seventy-five patients were included in the study, and 21 of them had subsyndromal delirium and were excluded from the analysis. Fifty-four patients with a mean age of 83.5 years (SD 8.6, 78% females) were included. Twenty-six patients (48%) had preoperative delirium, 11 (20%) developed delirium postoperatively, whereas 17 (31%) never developed delirium. SDNN, TP and HF values were significantly higher in patients with delirium compared to patients without delirium, and LF and LF/HF were lower. Patients developing postoperative delirium had decreased LF and increased HF before symptom onset.
Discussion
Increased SDNN, TP and HF and decreased LF values might reflect an abnormal stress response in delirium.
Conclusion
HRV measurements in patients with hip fractures provide additional information beyond heart rate and might be used to identify relevant pathophysiological factors in delirium.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Older patients admitted to hospitals often develop symptoms of acute cognitive and attentional deterioration, diagnosed as delirium [1]. Delirium is diagnosed based on a formal cognitive assessment and history of acute onset of symptoms, and it has to be distinguished from other syndromes sharing some of the same symptoms, like dementia [2]. Up to 50% of patients with hip fracture experience delirium during their hospital stay [3].
Delirium is preventable in 30–40% of cases [4], and early identification of patients at risk is crucial [2]. The development of delirium occurs as an interplay between predisposing and precipitating factors, and older patients with already existing cognitive impairment and multi-morbidity are at highest risk of developing delirium even in the presence of a mild acute insult. In contrast, in a young healthy person several noxious factors are necessary to move the brain out of balance [2].
The underlying pathophysiology of delirium is still poorly understood [5]. Among other theories, aberrant stress responses and alterations in autonomic nervous system (ANS) activity have been suggested [6]. ANS activity can indirectly be assessed by measuring heart rate variability (HRV) [7]. HRV is determined by an electrocardiogram (ECG), where the distances of normal QRS complexes are measured as a time series of consecutive values in milliseconds [8]. Linear parameters in the time domain like the standard deviation of the QRS distances (SDNN) and the square root of the mean squared differences of successive NN intervals (RMSSD) are most frequently used [9]. Both parameters have been mostly used for risk stratification in cardiovascular diseases [10] but also to identify autonomic neuropathy [11] and general mortality [12, 13]. SDNN is usually reduced in patients with serious diseases like myocardial infarction or sepsis [10, 14] and frequently increased after successful health interventions [15, 16].
The frequency domain is often calculated with a fast Fourier transformation, and three spectral parameters are distinguished: very-low-frequency (VLF), low-frequency (LF) and high-frequency (HF) components [17]. In addition, total power (TP) represents the variance of all QRS intervals. For the determination of HF and LF (and linear parameters), short-term recording of 5 min is sufficient, whereas VLF needs recordings at least of 10 min [8, 18]. For the characterization of the ANS activity, LF is often considered as a proxy for the activity of the sympathetic nervous system (SNS), HF to the activity of the parasympathetic nervous system (PNS) and the LF/HF ratio as reflecting the balance between SNS and PNS [19, 20].
Newer research suggests that HRV also reflects central brain states, in particular the central autonomic network [21]. Investigations with HRV have been conducted in acute conditions like sepsis [14, 22] and trauma [23], but very few studies have been done in patients with delirium [24, 25].
Aims
We aimed to explore possible changes in HRV parameters in hip fracture patients with delirium. We were particularly interested in: (1) HRV differences in patients with ongoing delirium compared to patients without delirium and (2) if alterations in HRV measured preoperatively were associated with development of postoperative delirium.
Methods
Participants in this study were recruited from a study of patients with acute hip fracture, at Oslo University Hospital, Norway, the Oslo Orthogeriatric Trial. Methods and results from this trial are published elsewhere [3, 26]. All consecutive patients admitted to this large level-3 university hospital with hip fracture were invited to the trial in the emergency department. Patients were not included if the hip fracture occurred because of a high-energy trauma or if they were moribund on admission. This was based on a clinical judgment by the attending orthopaedic surgeon. Only patients with sinus rhythm in their ECG were included in the present study.
All patients were assessed for delirium once daily, using the short version of the confusion assessment method (CAM) [27] preoperatively and until the fifth postoperative day. CAM was applied by the study geriatrician or a study nurse, based on a 10–30-min interview with the patient, together with information from close relatives, nurses and hospital records.
For the purpose of the analyses, the following delirium categories were distinguished:
-
Preoperative delirium—those who had delirium before surgery.
-
Postoperative delirium—those who were free from delirium when first examined but developed delirium after surgery.
-
Never delirium—those who never experienced any symptoms of delirium during their hospital stay.
We did not include patients with subsyndromal delirium in the analysis, i.e. those who at any point (preoperatively or postoperatively) experienced some delirious symptoms (≥ 2 positive CAM features, but never fulfilled the criteria for full-scale delirium).
The patients were also assessed for dementia. An experienced geriatrician and an experienced specialist in old age psychiatry independently assessed whether a patient fulfilled the criteria for dementia. In the case of disagreement, a consensus diagnosis was made. They applied the International Classification of Diseases, version 10 (ICD-10), research criteria for dementia [28]. In addition, depression was assessed using the Cornell scale [29]. Patients with a score of 8 or more were considered as having depression.
A 5-minute ECG signal was recorded within 24 h after arrival preoperatively by a Biocom 4000 ECG recorder and digitalized. The Biocom 4000 ECG interface unit uses dry silver/silver chloride ECG electrodes attached to two fingers of the right and the left hands, respectively. Patients were asked to relax for 5 min. Afterwards, a continuous ECG signal was measured over 5 min. The signals were processed according to international recommendations [17].
Time-domain parameters (SDNN, RMSSD) and frequency-domain parameters (TP, HF, LF, LF/HF ratio) were calculated by the Heart Rhythm Scanner, version 2.0 (Biocom Technologies, USA).
We present all descriptive data for continuous variables as means and standard errors of the mean (SEM) and for dichotomous variables as percentages. Normally distributed continuous data were analysed using Student’s t-tests. Categorical data were analysed using Chi-square tests. We used SPSS, version 25 (IBM, Armonk, NY), for statistical analysis.
Results
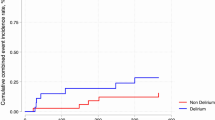
A total of 75 patients were included during the study period between 2010 and 2012. Mean age was 82.6 years (SD 9.1) and 76% were females. Twenty-one patients had subsyndromal delirium and were not included in the analysis. This left 54 patients for the statistical analysis with a mean age of 83.5 years (SD 8.6). All variables were normally distributed. Twenty-six (48%) patients had preoperative delirium, 11 (20%) developed postoperative delirium, and 17 (31%) never developed delirium during their hospital stay. First, we compared all patients with delirium (n = 37) with patients who never developed delirium (n = 17) (Table 1). Patients with delirium were older and had more often dementia. Patients without delirium more often used angiotensin-2 blockers (p = 0.03) than patients with delirium, but for the other drugs, there were no statistically significant differences between the groups. All HRV indices were higher in the delirium group, and these differences were significant for SDNN (p < 0.02) and TP (p = 0.02) (Fig. 1), but not for HF and LF in absolute values. Delirious patients had a significantly lower LF/HF ratio than those with no delirium (p = 0.03) (Fig. 2).
Intake of most medical drugs was not associated with differences in HRV parameters, with the exceptions that digitalis usage was associated with lower HF (p = 0.01) and lower LF/HF (p = < 0.01) and that the use of diuretics was associated with lower LFnorm (p = 0.04) and higher HFnorm (p = 0.04).
When we compared patients with preoperative delirium (n = 26), i.e. those who had ongoing delirium when the HRV data were obtained, with those who never developed delirium (n = 17), we found similar results (Table 2; Fig. 3) of significantly higher SDNN and TP and lower LF/HF ratio in the delirium group.
Finally, we compared patients who were free from delirium when the ECG recording was done, but who developed delirium postoperatively (n = 11), with patients who never developed delirium (n = 17, Table 3). All HRV indices were higher in those who developed postoperative delirium, but the differences did not reach statistical significance, except for HF and LF in normalized units.
Discussion
In this study, we explored possible differences in heart rate variability in hip fracture patients with and without delirium. Our main findings were as follows:
Firstly, in patients with delirium the values of HRV variables, in both time (SDNN) and frequency domains (TP), were increased compared to patients without delirium. This indicates that patients with delirium had a stronger autonomic modulation of the sinus node. Patients with delirium had higher HRV indices indicating parasympathetic control (such as HF abs and RMSSD), but sympathetic modulation might also be larger, as LF is a result of both sympathetic and parasympathetic activity. This results in higher values of SDNN and TP, reflecting total variability in the group with delirium.
Secondly, the findings of higher HF norm, lower LF norm and thus lower LF/HF ratio in patients with delirium were similar in both the group with preoperative delirium and postoperative delirium.
A usual ECG investigation returns heart rate and possibly heart rhythm disturbances. In our study, as in many previous studies, the heart rate is not different between the groups despite profound differences in HRV parameters. Also in our study, HRV revealed hidden characteristics in ECG which otherwise are missed [8].
Increased SDNN and TP are generally interpreted as increased heart rate variability, and SDNN is usually highly correlated with TP [30]. SDNN is an established risk factor for cardiovascular events and general health in 24-h measurements [10, 31], but also shorter measurements have shown similar results [32, 33].
Differences in HFnorm, LFnorm and LF/HF are usually discussed in terms of ANS. Increased activity of SNS should result in increased LFnorm and increased activity of PNS in increased HFnorm. Based on the findings that increased stress and serious disease would increase LF and decrease HF, we would have expected patients with delirium to have an elevated LF/HF ratio, as well as reduced SDNN. However, this was not the case in our findings. Stress, however, does not necessarily lead to increased LF or decreased SDNN [34]. Several experimental stress studies also observed a decreased LF in (experimental) stress situations [35,36,37]. SDNN values are also usually decreased during mental stress [38], but increased values have been reported [39]. According to the classical stress response paradigm, the stress response is divided into a first phase where primarily the SNS is activated, and PNS suppressed, and a second phase, when the PNS is involved in restoring physiological functions [40]. The increased SDNN in delirium patients could be based on an abnormal stress response.
Although no unified explanations of the pathophysiological mechanisms of delirium are presented until now, the upregulation of adrenergic pathways and consecutively the sympathetic nervous system (SNS) has been proposed [41]. Other models explain delirium as a consequence of neuroimmunological changes caused by the release of different proinflammatory mediators [42].
The vagal nerve not only is involved in heart rate regulation, but also plays a role in respiration, inflammation and metabolic control [40, 43, 44]. When parasympathetic nerves sense increased interleukin-1, afferent fibres signal to the brain and cause eventually increased vagal efferent activity which triggers the release of acetylcholine, dampening local inflammatory reactions [45] as countermeasure. Both increased SDNN, TP and HFnorm and decreased LFnorm might reflect this effect on the vagal function.
In the last years, HRV has been increasingly discussed as a proxy for adaptivity of the brain to internal or external perturbations [46]. In our sample, increased heart rate variability might not reflect adaptivity, but might indicate that in acute situations like delirium HRV rather mirrors the occurring physiological changes.
We are not aware of previous studies investigating HRV in relation to delirium in hip fracture patients. There are only a few previous studies that have focused on the association between delirium and HRV in other patient groups. In one study comparing patients with and without delirium in an intensive care unit (ICU), no significant differences in HFnu or LF/HF were found [24]. The study investigated, however, only 13 patients with, and 12 without delirium which might indicate that the study was statistically underpowered. The direction of the reported results was that mean HF norm was higher in delirium group and the LF/HF ratio was lower in delirium. Thus, the direction of the differences was the same as in our study.
In another investigation including 60 ICU patients, patients in a delirium state had higher SDNN (like in our study), RMSSD and entropy values, but no differences in frequency-domain parameters [47]. Finally, a study investigated 14 geriatric patients with an infection and measured HRV in supine and head-up tilt position [25]. In patients with delirium, SDNN and RMSSD values were higher and LF lower, similar as in our study.
Our study has some strengths. The delirium assessments were conducted by the study geriatrician (LOW) or by trained study nurses using a standardized instrument. We used a standardized approach in obtaining HRV data early after the arrival in the hospital.
There are limitations to this work that should be noted. The sample size is too small to be able to adjust for drug use or dementia status in the statistical analyses. Thus, we cannot rule out that the observed HRV differences could be explained largely by dementia. A recent meta-analysis, however, concluded that in most previous investigations, HRV values were reduced in dementia [48], except in one study, where LF and HF (but not SDNN, or TP) were elevated [49]. We therefore interpret the observed increased HRV values in the present study to be associated with delirium.
Other possible confounders that effect HRV variables are respiratory rate and heart rate, as well as medication. We did not measure respiratory rate during ECG recording. There were no significant differences in mean heart rate between the groups. This lack of heart rate differences can be an indirect indication that respiratory rate was not affected significantly. Usually, respiratory rates between 8 and 20 should not have major effects on HRV and HRV measurements [50].
Conclusion
In this explorative study on HRV in hip fracture patients, we found associations between delirium and increased values of SDNN and TP, as well as a decreased LF/HF ratio. HRV measurements provide additional information beyond heart rate and might be used to identify relevant pathophysiological factors in delirium.
Data availability
The datasets generated and analysed during the current study are available from the corresponding author on reasonable request.
References
Benhamou D, Brouquet A (2016) Postoperative cerebral dysfunction in the elderly: diagnosis and prophylaxis. J Visc Surg 153:S27–s32. https://doi.org/10.1016/j.jviscsurg.2016.09.015
Inouye SK, Westendorp RG, Saczynski JS (2014) Delirium in elderly people. Lancet 383:911–922. https://doi.org/10.1016/s0140-6736(13)60688-1
Watne LO, Torbergsen AC, Conroy S et al (2014) The effect of a pre- and postoperative orthogeriatric service on cognitive function in patients with hip fracture: randomized controlled trial (Oslo Orthogeriatric Trial). BMC Med 12:63. https://doi.org/10.1186/1741-7015-12-63
Marcantonio ER, Flacker JM, Wright RJ et al (2001) Reducing delirium after hip fracture: a randomized trial. J Am Geriatr Soc 49:516–522
Maldonado JR (2013) Neuropathogenesis of delirium: review of current etiologic theories and common pathways. Am J Geriatr Psychiatry 21:1190–1222. https://doi.org/10.1016/j.jagp.2013.09.005
Maclullich AM, Ferguson KJ, Miller T et al (2008) Unravelling the pathophysiology of delirium: a focus on the role of aberrant stress responses. J Psychosom Res 65:229–238. https://doi.org/10.1016/j.jpsychores.2008.05.019
Shaffer F, McCraty R, Zerr CL (2014) A healthy heart is not a metronome: an integrative review of the heart's anatomy and heart rate variability. Front Psychol 5:1040. https://doi.org/10.3389/fpsyg.2014.01040
Ernst G (2017) Hidden signals-the history and methods of heart rate variability. Front Public Health 5:265. https://doi.org/10.3389/fpubh.2017.00265
Sassi R, Cerutti S, Lombardi F et al (2015) Advances in heart rate variability signal analysis: joint position statement by the e-Cardiology ESC Working Group and the European Heart Rhythm Association co-endorsed by the Asia Pacific Heart Rhythm Society. Europace 17:1341–1353. https://doi.org/10.1093/europace/euv015
Huikuri HV, Stein PK (2013) Heart rate variability in risk stratification of cardiac patients. Prog Cardiovasc Dis 56:153–159. https://doi.org/10.1016/j.pcad.2013.07.003
Lin K, Wei L, Huang Z et al (2017) Combination of Ewing test, heart rate variability, and heart rate turbulence analysis for early diagnosis of diabetic cardiac autonomic neuropathy. Medicine 96:e8296. https://doi.org/10.1097/md.0000000000008296
Tsuji H, Venditti FJ Jr, Manders ES et al (1994) Reduced heart rate variability and mortality risk in an elderly cohort The Framingham Heart Study. Circulation 90:878–883. https://doi.org/10.1161/01.cir.90.2.878
Huikuri HV, Makikallio TH, Airaksinen KE et al (1998) Power-law relationship of heart rate variability as a predictor of mortality in the elderly. Circulation 97:2031–2036
de Castilho FM, Ribeiro ALP, Nobre V et al (2018) Heart rate variability as predictor of mortality in sepsis: a systematic review. PLoS One 13:e0203487. https://doi.org/10.1371/journal.pone.0203487
Nagendra H, Kumar V, Mukherjee S (2015) Cognitive behavior evaluation based on physiological parameters among young healthy subjects with yoga as intervention. Comput Math Methods Med 2015:821061. https://doi.org/10.1155/2015/821061
Kaikkonen KM, Korpelainen RI, Tulppo MP et al (2014) Physical activity and aerobic fitness are positively associated with heart rate variability in obese adults. J Phys Act Health 11:1614–1621. https://doi.org/10.1123/jpah.2012-0405
Heart rate variability (1996) Standards of measurement, physiological interpretation, and clinical use. Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Eur Heart J 17:354–381
McCraty R, Shaffer F (2015) Heart rate variability: new perspectives on physiological mechanisms, assessment of self-regulatory capacity, and health risk. Glob Adv Health Med 4:46–61. https://doi.org/10.7453/gahmj.2014.073
Kleiger RE, Stein PK, Bigger JT Jr (2005) Heart rate variability: measurement and clinical utility. Ann Noninvasive Electrocardiol 10:88–101. https://doi.org/10.1111/j.1542-474X.2005.10101.x
Ernst G (2014) Heart rate variability. Springer, London
Smith R, Thayer JF, Khalsa SS et al (2017) The hierarchical basis of neurovisceral integration. Neurosci Biobehav Rev 75:274–296. https://doi.org/10.1016/j.neubiorev.2017.02.003
Pontet J, Contreras P, Curbelo A et al (2003) Heart rate variability as early marker of multiple organ dysfunction syndrome in septic patients. J Crit Care 18:156–163
Ryan ML, Thorson CM, Otero CA et al (2011) Clinical applications of heart rate variability in the triage and assessment of traumatically injured patients. Anesthesiol Res Pract 2011:416590. https://doi.org/10.1155/2011/416590
Zaal IJ, van der Kooi AW, van Schelven LJ et al (2015) Heart rate variability in intensive care unit patients with delirium. J Neuropsychiatr Clin Neurosci 27:e112–e116. https://doi.org/10.1176/appi.neuropsych.13090213
Neerland BE, Wyller TB, Wyller VBB (2019) Autonomic cardiovascular control in older patients with acute infection and delirium: a pilot study of orthostatic stress responses. BMC Geriatr 19:23. https://doi.org/10.1186/s12877-019-1035-0
Wyller TB, Watne LO, Torbergsen A et al (2012) The effect of a pre- and post-operative orthogeriatric service on cognitive function in patients with hip fracture. The protocol of the Oslo Orthogeriatrics Trial. BMC Geriatr. https://doi.org/10.1186/1471-231812-36
Inouye SK, van Dyck CH, Alessi CA et al (1990) Clarifying confusion: the confusion assessment method. A new method for detection of delirium. Ann Inter Med 113:941–948. https://doi.org/10.7326/0003-4819-113-12-941
Organization WH The ICD-10 Classification of mental and behavioural disorders. Diagnostic criteria for research. World Health Organization (Available from: www.who.int/classifications/icd/en/GRNBOOK.pdf), Geneva. Accessed 26 Dec 2019
Alexopoulos GS, Abrams RC, Young RC et al (1988) Cornell scale for depression in dementia. Biol Psychiatry 23:271–284. https://doi.org/10.1016/0006-3223(88)90038-8
Wittling WW, Wittling RA (2012) Herzschlagvariabilität: Frühwarnsystem, Stress- und Fitnessindikator. Eichsfeld-Verlag, Heiligenstadt
Hillebrand S, Gast KB, de Mutsert R et al (2013) Heart rate variability and first cardiovascular event in populations without known cardiovascular disease: meta-analysis and dose-response meta-regression. Europace 15:742–749. https://doi.org/10.1093/europace/eus341
de Bruyne MC, Kors JA, Hoes AW et al (1999) Both decreased and increased heart rate variability on the standard 10-second electrocardiogram predict cardiac mortality in the elderly: the Rotterdam Study. Am J Epidemiol 150:1282–1288. https://doi.org/10.1093/oxfordjournals.aje.a009959
Gerritsen J, Dekker JM, TenVoorde BJ et al (2001) Impaired autonomic function is associated with increased mortality, especially in subjects with diabetes, hypertension, or a history of cardiovascular disease: the Hoorn Study. Diabetes Care 24:1793–1798
Berntson G, Cacioppo J (2004) Heart rate variability: stress and psychiatric conditions. Dynamic Electrocardiography. Futura, New York
Hjortskov N, Rissen D, Blangsted AK et al (2004) The effect of mental stress on heart rate variability and blood pressure during computer work. Eur J Appl Physiol 92:84–89. https://doi.org/10.1007/s00421-004-1055-z
Tharion E, Parthasarathy S, Neelakantan N (2009) Short-term heart rate variability measures in students during examinations. Natl Med J India 22:63–66
Taelman J, Vandeput S, Vlemincx E et al (2011) Instantaneous changes in heart rate regulation due to mental load in simulated office work. Eur J Appl Physiol 111:1497–1505. https://doi.org/10.1007/s00421-010-1776-0
Castaldo R, Melillo P, Bracale U et al (2015) Acute mental stress assessment via short term HRV analysis in healthy adults: A systematic review with meta-analysis. Biomed Signal Proc Control 18:370–377. https://doi.org/10.1016/j.bspc.2015.02.012
Schubert C, Lambertz M, Nelesen RA et al (2009) Effects of stress on heart rate complexity—a comparison between short-term and chronic stress. Biol Psychol 80:325–332. https://doi.org/10.1016/j.biopsycho.2008.11.005
Molina PE (2005) Neurobiology of the stress response: contribution of the sympathetic nervous system to the neuroimmune axis in traumatic injury. Shock 24:3–10
Maldonado JR (2018) Delirium pathophysiology: an updated hypothesis of the etiology of acute brain failure. Int J Geriatr Psychiatry 33:1428–1457. https://doi.org/10.1002/gps.4823
de Rooij SE, van Munster BC, Korevaar JC et al (2007) Cytokines and acute phase response in delirium. J Psychosom Res 62:521–525. https://doi.org/10.1016/j.jpsychores.2006.11.013
Thayer JF, Loerbroks A, Sternberg EM (2011) Inflammation and cardiorespiratory control: the role of the vagus nerve. Respir Physiol Neurobiol 178:387–394. https://doi.org/10.1016/j.resp.2011.05.016
Groves DA, Brown VJ (2005) Vagal nerve stimulation: a review of its applications and potential mechanisms that mediate its clinical effects. Neurosci Biobehav Rev 29:493–500. https://doi.org/10.1016/j.neubiorev.2005.01.004
Sternberg EM (2006) Neural regulation of innate immunity: a coordinated nonspecific host response to pathogens. Nat Rev Immunol 6:318–328. https://doi.org/10.1038/nri1810
Kemp AH, Quintana DS (2013) The relationship between mental and physical health: insights from the study of heart rate variability. Int J Psychophysiol 89:288–296. https://doi.org/10.1016/j.ijpsycho.2013.06.018
Jooyoung O, Dongrae C, Jongin K et al (2017) Changes in heart rate variability of patients with delirium in intensive care unit. Conference proceedings : Annual International Conference of the IEEE Engineering in Medicine and Biology Society IEEE Engineering in Medicine and Biology Society Annual Conference 2017:3118–3121. https://doi.org/10.1109/embc.2017.8037517
da Silva VP, Ramalho Oliveira BR, Tavares Mello RG et al (2018) Heart Rate Variability Indexes in Dementia: a systematic review with a quantitative analysis. Curr Alzheimer Res 15:80–88. https://doi.org/10.2174/1567205014666170531082352
Kasanuki K, Iseki E, Fujishiro H et al (2015) Impaired heart rate variability in patients with dementia with Lewy bodies: efficacy of electrocardiogram as a supporting diagnostic marker. Parkinsonism Relat Dis 21:749–754. https://doi.org/10.1016/j.parkreldis.2015.04.024
Frenneaux MP (2004) Autonomic changes in patients with heart failure and in post-myocardial infarction patients. Heart 90:1248–1255. https://doi.org/10.1136/hrt.2003.026146
Funding
The study was funded by the Research Council of Norway and the Norwegian Association for Public Health. The sponsors had no role in the design and conduct of the study; collection, management, analysis and interpretation of the data; and preparation, review or approval of the manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Gernot Ernst and Leiv Otto Watne. The first draft of the manuscript was written by Gernot Ernst, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The study protocol was reviewed and accepted by the Regional Committee for Medical and Health Research Ethic in South-East Norway (REK 2009/450).
Informed consent
Informed consent was obtained from the patients or substitute decision-makers if the patients did not have the capacity to consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ernst, G., Watne, L.O., Rostrup, M. et al. Delirium in patients with hip fracture is associated with increased heart rate variability. Aging Clin Exp Res 32, 2311–2318 (2020). https://doi.org/10.1007/s40520-019-01447-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40520-019-01447-5