Abstract
Background
Falling is a significant concern for many elderly adults but identifying individuals at risk of falling is difficult, and it is not clear how elderly adults adapt to challenging walking.
Aims
The aim of the current study was to determine the effects of walking at non-preferred speeds on the coordination between foot and trunk acceleration variability in healthy elderly adults with and without fall history compared to healthy young adults.
Methods
Subjects walked on a treadmill at 80%–120% of their preferred walking speed while trunk and foot accelerations were recorded with wireless inertial sensors. Variability of accelerations was measured by root mean square, range, sample entropy, and Lyapunov exponent. The gait stability index was calculated using each variability metric in the frontal and sagittal plane by taking the ratio of trunk acceleration variability divided by foot acceleration variability.
Results
Healthy young adults demonstrated larger trunk accelerations relative to foot accelerations at faster walking speeds compared to elderly adults, but both young and elderly adults show similar adaption to their acceleration regularity. Between group differences showed that elderly adult fallers coordinate acceleration variability between the trunk and feet differently compared to elderly non-fallers and young adults.
Discussion
The current results indicate that during gait, elderly fallers demonstrate more constrained, less adaptable trunk movement relative to their foot movement and this pattern is different compared to elderly non-fallers and healthy young.
Conclusions
Coordination between trunk and foot acceleration variability plays an important role in maintaining stability during gait.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Falling is a significant concern for many elderly adults, with approximately one-third of elderly adults experiencing at least one fall per year [1]. Falls experienced by elderly adults can severely impact quality of life through injury, hospitalization, and death [2]. This increased risk of falls may arise from any combination of common physiological characteristics of aging including decreased muscle strength [3], reduced sensory feedback [4, 5], and reduced cognitive functions [6]. However, even if an individual demonstrates normal capacity in these physiological domains, elderly adults may still have difficulty maintaining stability when walking under challenging conditions [7, 8]. Simple functional tests are often used to screen persons for fall risk [9], but previous studies have shown that these functional tests may not be adequate to predict fall risk [10]. There is a need for sensitive, objective measures of fall risk as it relates to gait stability that can be employed in a clinical setting or used to monitor walking function at home in daily life [11]. Such objective measures need to capture features of an individual’s walking pattern which may contribute to loss of stability during walking, but it is not currently clear what features of walking are behind increased fall risk in elderly adults. Previous studies have identified trunk acceleration variability measures such as Lyapunov exponents to relate to fall risk [12, 13], while other studies using the same measures do not find any relation to fall risk [14]. Studies have also identified numerous gait characteristics that significantly relate to fall risk when combined in regression models [14], but the individual measures themselves may not be representative of actual walking performance or may not have a clear mechanistic basis for why they would specifically relate to fall risk in older adults. Therefore, it remains unclear what specific gait characteristics are demonstrated in older adults that ultimately lead to decreased stability and an increased risk of falling.
Stability during walking can be defined as the ability to maintain functional upright gait without falling [15]. Maintaining upright, stable gait requires a carefully controlled interaction between the base of support (BoS) and center of mass (CoM) [16]. During walking, this relationship is dynamically maintained from step to step, with the sensorimotor system controlling trunk sway and step placement across each step [17, 18]. In healthy adults, altering or constraining movement of the trunk segment results in altered foot movement to compensate [19], and similarly constraining foot movement results in altered trunk movement [20]. In a study by Arvin et al. when step placement was constrained to a narrower step width, the peak mediolateral CoM displacement also decreased, however, the variability of the mediolateral CoM velocity subsequently increased in elderly adults, suggesting that elderly adults’ trunk movement was more actively controlled in response to the altered step placement [20]. These studies highlight the underlying coordination that must exist between the trunk and the feet during healthy, stable walking. However, it is currently not clear how this coordination between segments directly relates to stability or fall risk during walking. If this coordination is directly related to fall risk, then one would expect to observe altered segment coordination in elderly adults with a history of fall risk compared to those without a history of falls. Additionally, walking under challenging conditions which make walking inherently unstable (i.e., non-preferred walking speed) would likely require a specific adaptation to maintain the coordination between the trunk and feet, with inappropriate adaptations potentially underlying increased fall risk.
Elderly adults tend to walk with shorter and wider steps, demonstrating a conservative gait pattern [21]. This conservative gait pattern is demonstrated in measures of margin of stability, where elderly adult fallers demonstrate a larger margin of stability compared to healthy young adults, keeping their CoM well within their BoS [18]. However, the margin of stability generally requires a gait lab with a motion capture system to measure subjects’ gait, which limits its clinical or real-world utility for measuring fall risk. To monitor movement outside of a laboratory setting, many previous studies have identified measures of acceleration variability to be related to fall risk in aging individuals [13, 22]. Measuring variability of movement at a particular segment provides an understanding of how movement at that segment is being controlled through the underlying sensorimotor system [23]. Healthy adults demonstrate an optimal gait pattern, with optimal levels of variability of movement of their individual segments to allow for stable and adaptable gait [24]. Previous studies have shown that elderly adults demonstrate higher standard deviations of step width and mediolateral margin of stability [20, 25]. These are linear measures of variability which provide information about the magnitude of variability around an average [26]. Nonlinear measures of variability provide information on the temporal structure of variability in a time series, which is of significant interest in measuring the control systems driving the movement being recorded [24]. Structure of trunk acceleration variability has also been associated with fall history in elderly adults, where elderly adults who have a history of falls also have more unpredictable trunk accelerations [13, 27]. All of these previous studies have examined movement variability of the trunk or feet independently, but it is likely that the coordination between the trunk and feet may actually be more important for maintaining stability during walking compared to movement of the trunk or feet independently [17]. For example, altered trunk movement could stabilize center of mass motion to compensate for altered foot movement, thus maintaining whole body stability even though motion of an individual segment is abnormal. It is common for elderly individuals to have weakness or decreased range of motion, which could give rise to altered segmental relationships and ultimately lead to a higher risk for falls [28]. Since the optimal walking pattern is demonstrated at a person’s normal walking speed [24, 29], it is important to understand how elderly adults and elderly adults at risk for falls adapt their gait to changing walking speeds since this is common during walking a real-world environment.
The aim of the current study was to determine the effects of walking at non-preferred speeds on the coordination between foot and trunk acceleration variability in healthy young adults compared to healthy elderly adults with and without fall history. Specifically, we expect that elderly adults with a history of falls may maintain coordination between their trunk and foot segments differently compared to elderly adults who do not fall and compared to the optimal gait of healthy young adults. We hypothesized that (1) the trunk and foot segment coordination will be different between healthy young, elderly fallers and elderly non-fallers, and (2) the segment coordination will not change in response to changing gait speeds.
Methods
Participants
Twenty-five healthy young (HY) adults (mean 23, range 20–30), 25 healthy elderly (HE) adults (mean 73, range 67–85) and 15 elderly adult fallers (EF) with two or more falls in the previous 12 months (mean 74, range 65–85) were enrolled in the current study (Table 1). The University of Kansas Medical Center Human Research Committee approved this study and all participants gave informed written consent prior to testing. All subjects were free of any known musculoskeletal or neurological disorders that would negatively affect their gait or balance.
Data collection
Subjects’ preferred walking speed (PWS) was determined following previously used protocol where treadmill speed was increased and decreased until subjects reported the speed was faster or slower than preferred [29]. Subjects wore two wireless inertial sensors (Opal, APDM, Portland, OR, USA; 128 Hz), one lumbar accelerometer and one foot accelerometer [30, 31]. The lumbar sensor was placed over the posterior surface of the lumbar spine at approximately the level of L5. The foot sensor was placed on the lateral surface of the distal shank, superior to the ankle joint such that subjects’ footwear would not come into contact with the sensor during walking. A treadmill (Woodway Bari-Mill, Eugene, OR, USA) was used to collect sufficiently long samples of walking [32, 33] and to sensitively control the speeds at which subjects walked for all trials [29, 34]. Accelerations from both sensors were recorded while subjects walked for 3 min at five different speeds: 80%, 90%, 100%, 110%, and 120% of their own PWS. The speed conditions were presented in random order.
Data analysis
The raw three-dimensional acceleration time series were exported to Matlab (Matlab version R2013b, The MathWorks Inc., Natick, Massachusetts, USA) and were transformed to resultant frontal and sagittal plane time series local to the individual sensors. The frontal and sagittal planes were examined separately since gait is laterally unstable, and control of movement in this plane requires active control compared to passive control in the sagittal plane [35]. Since subjects walked at different speeds, the middle 60 strides were used for consistent analysis across subjects and speeds. A custom Matlab program was used to calculate all variability measures. All subsequent analyses were performed on the resultant sagittal and frontal plane time series. Data were left unfiltered for appropriate analysis of time series characteristics [36].
Linear variability measures root mean square (RMS) and range were calculated from the frontal and sagittal plane acceleration time series for the foot and lumbar sensors [30]. RMS was used to measure the absolute dispersion of accelerations in each time series. Range was calculated as the difference between the maximum and minimum peaks in the acceleration time series.
Nonlinear variability measures were used to quantify the temporal structure of variability within the time series, which provides information about how movement of the foot and trunk segments is controlled [37]. As the gait cycle repeats, patterns in the time series repeat, and nonlinear measures provide information about how tightly controlled the patterns are over the entire length of the time series [24]. Sample entropy (SaEn) and Lyapunov exponents (LyE) were calculated from the foot and lumbar time series in the frontal and sagittal planes. A thorough explanation of sample entropy can be found in the previous literature [32, 37,38,39]. Methods for all variability calculations have been outlined in detail previously [30].
As the primary outcomes, gait stability index (GSI) metrics were calculated as the ratio of lumbar acceleration (ACC) variability divided by foot acceleration (ACC) variability, using each of the four variability metrics (RMS, range, SaEn, LyE) in the frontal and sagittal planes [40].
Four GSI metrics were calculated in the frontal and sagittal planes: GSIRMS, GSIRange, GSISaEn, GSILyE, resulting in eight GSI metrics total used in the statistical analysis.
The GSI metrics are unitless measures to examine lumbar acceleration variability relative to foot acceleration variability within an individual subject. A GSI equal to 1 indicates that acceleration variability at the two segments is exactly equal while a GSI greater than one indicates more lumbar acceleration variability relative to foot acceleration variability and a GSI of less than one indicates less lumbar acceleration variability relative to foot acceleration variability [40].
Statistical analysis
The Shapiro–Wilk normality test indicated that all data were normally distributed. Three Group (HY, HE, and EF) by 5 Speed (80%–120% PWS) analysis of variance was used to determine whether the GSI metrics differed across Groups or Speeds. Post hoc paired comparisons were performed to investigate any significant interactions.
Results
Speed effects
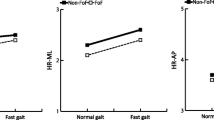
The GSIRMS showed a main effect of Speed in the sagittal (F = 5.055, p = 0.001) and frontal (F = 5.158, p = 0.001) planes. GSIRMS was the only variable to demonstrate a significant interaction between Group and Speed in the sagittal (F = 3.384, p = 0.001) and frontal (F = 5.817, p < 0.001) planes. The HY group showed adaptations to Speed where the GSIRMS at the two fastest walking speeds was greater than at the three slower speeds for both the sagittal (p < 0.001) and frontal (p < 0.001) planes, while the HE and EF groups did not show adaptations to Speed (Fig. 1a, b). The GSISaEn showed a main effect of Speed in the sagittal (F = 5.912, p < 0.001) and frontal (F = 2.253, p = 0.041) planes (Fig. 1c), where the GSISaEn at the two slowest speeds was significantly higher compared to the two highest speeds in the sagittal (p < 0.040) and frontal (p < 0.021) planes.
Group effects
The GSIRMS in the sagittal plane showed a main effect of Group (F = 4.905, p = 0.011) where EF had lower GSIRMS values across all speeds compared to HE (p < 0.04) and compared to HY (p < 0.030) (Fig. 1a). The GSISaEn showed a main effect of Group in the sagittal (F = 12.525, p < 0.001) and frontal (F = 3.198, p = 0.049). In the sagittal plane, HY had lower GSISaEn values across all speeds compared to HE (p < 0.026) and EF (p < 0.013). In the frontal plane, HY had lower GSISaEn values across all speeds compared to HE (p < 0.048) and EF (p < 0.047). The GSILyE in the frontal plane also demonstrated a main effect of Group (F = 4.424, p = 0.017) where EF had lower GSILyE values across all speeds compared to HE (p < 0.045) and at preferred walking speed in HY (p = 0.019) (Fig. 1e).
Discussion
The purpose of the current study was to determine how elderly fall-prone adults control their trunk acceleration variability and foot acceleration variability during walking under normal and challenging conditions when compared to healthy young and elderly non-faller subjects. Our first hypothesis regarding effects of Speed was partially supported, as we found that the GSIRMS (a measure of amount of lumbar acceleration relative to foot acceleration) increased with walking speed in healthy young adults, but elderly adults did not show similar adaptations to speed. However, our results also indicate that younger and elderly adult fallers and non-fallers similarly adapt their GSISaEn (a measure of the regularity of lumbar acceleration relative to foot acceleration) to increasing walking speed. Our second hypothesis regarding differences between groups was also partially supported, as the elderly adult fallers walked with lower GSIRMS and GSILyE compared to elderly non-fallers and healthy young adults, and healthy young adults walked with lower GSISaEn compared to both elderly adult groups.
The GSIRMS results in the sagittal and frontal planes indicated that both elderly fallers and non-fallers adapt their lumbar acceleration relative to their foot acceleration differently compared to healthy young adults. As healthy young adults walked faster, their lumbar accelerations increased more than their foot accelerations (Fig. 2). Previous studies have shown that lumbar accelerations increase in all directions as walking speed increases [41], however, no previous studies have examined how walking speed effects lumbar accelerations relative to foot accelerations. The results from the current study indicate that while elderly fallers, elderly non-fallers, and healthy young adults increased lumbar and foot accelerations with increasing walking speed, the healthy young adults increase their lumbar accelerations relative to their foot accelerations more than is seen in both of the elderly groups. These results indicate that the elderly fallers and elderly non-fallers constrain their lumbar accelerations to minimize motion of their head [22] and center of mass [18], while healthy young subjects are able to safely allow for more lumbar accelerations during walking. One possibility is that healthy young adults can safely use the momentum of the lumbar segment in forward propulsion at faster walking speeds more than elderly adults who constrain their lumbar motion more within their base of support to maintain a stable consistent gait. Future studies should further explore how lumbar motion is used in forward propulsion at different walking speeds. Elderly fallers and elderly non-fallers demonstrated the lowest GSIRMS across all speeds compared to healthy young subjects. This result seems to parallel the conservative gait phenomena observed in previous studies, where elderly adults walk with a more conservative gait pattern, minimizing trunk motion to keep their CoM more within the boundaries of their BoS [18].
Top: Sagittal plane trunk (black) and foot (gray) acceleration RMS values at 80–120% preferred walking speed in healthy young adults. Bottom: Sagittal plane GSIRMS 80%–120% preferred walking speed in healthy young adults—the resulting ratio of trunk RMS divided by foot RMS. Note how the trunk acceleration increases more than the foot acceleration, driving the GSIRMS increase at faster walking speeds
While the GSIRMS showed different adaptations to Speed across the three groups, the GSISaEn in the sagittal plane showed that all three groups adapted the regularity of their foot and lumbar accelerations similarly across speeds, with lumbar acceleration becoming more regular relative to foot acceleration as walking speed increased. Previous studies have shown that more irregular lumbar accelerations at slow walking speeds may represent more active control of lumbar movement when walking at slower speeds, whereas lumbar movement becomes more passively controlled when walking at faster speeds [41]. While the sagittal plane GSISaEn revealed similar adaptations to walking speed in the three groups, there were differences in the GSISaEn between the three groups across walking speeds. Elderly fallers had the lowest GSISaEn of the three groups, which indicates that elderly fallers walked with more regular lumbar accelerations relative to foot accelerations than did elderly non-fallers and healthy young. Previous studies have showed that elderly fallers demonstrate more regular accelerations at the trunk compared to elderly non-fallers, which may be indicative of decreased adaptability leading to gait instability [42]. Therefore, in the current study, increased regularity of lumbar accelerations relative to foot accelerations in elderly fallers compared to elderly non-fallers and healthy young adults would indicate a lack of stability that is maintained across slow and fast walking speeds.
Group differences in GSI were also evident in the frontal plane, which is considered to be controlled by more active feedback during walking [35]. Elderly fallers had the lowest GSILyE across speeds compared to elderly non-fallers and healthy young adults. The lower GSILyE in elderly fallers compared to indicates that elderly fallers have more predictable accelerations at the lumbar relative to the feet across all walking speeds. In relation to the loss of complexity hypothesis, a lower LyE at the lumbar relative to the feet compared to healthy young adults indicates a less complex and more predictable gait pattern in the elderly fallers [24]. The increased predictability of lumbar accelerations in the elderly faller group may further indicate a lack of adaptability or lack of flexibility to appropriately react to small perturbations from step to step, as has been shown in previous studies of elderly adults with fall history [13].
The GSIRange did not demonstrate any effect of Speed or Group in the frontal or sagittal plane. Range of acceleration is a linear measure of variability which provides an indication of the absolute spread of the maximum and minimum accelerations in the time series. In practice, the extreme acceleration values that are quantified in the range metric are likely the points at which an individual experienced a slight stumble or otherwise deviated their gait. Compared to RMS which provides information about the average spread of accelerations around the mean over the entire time series, the range metric is an indicator of extreme values at two points in the time series (one minimum value, one maximum value). While range of accelerations may be useful as a global value, or as a single segment acceleration measure [22], it may not be a good metric to use when examining relationships between the lumbar and foot accelerations.
A limitation of the current study is that the three groups walked with different preferred walking speeds, which could have an effect on the accelerations of the lumbar and the feet. However, since we examined subjects’ gait using a ratio of lumbar acceleration variability to foot acceleration variability, any normalization of the lumbar and foot acceleration metrics would cancel out, and any effect due to different preferred walking speeds would be negated. Therefore, the GSI metrics are not dependent on subjects’ preferred walking speed. A second limitation of the current study is that a treadmill was used to collect the walking data, and individuals can demonstrate altered muscle activation when walking on a treadmill [43, 44]. However, previous work has also shown that kinematics and kinetics of treadmill gait is largely similar to that of overground walking [45, 46]. In the current study, whole body stability was examined, which has been shown to be similar between treadmill and overground walking when examining margin of stability [47]. The treadmill was necessary for use in the current study to collect a sufficiently long time series to appropriately analyze gait variability [32], and to accurately control the speeds at which subjects walked for each of the five trials. A third limitation of the current study is that the elderly subjects with and without falls had no comorbidities that would significantly affect their walking or balance. Therefore, caution should be taken when generalizing the results of the current study to the wider population of elderly adults who may have comorbidities such as neuropathy or loss of vision which could additionally affect their walking and balance function.
The coordination between trunk and foot acceleration variability appears to play an important role in maintaining stability during gait. The current results indicate that during gait, elderly fallers specifically demonstrate more constrained, less adaptable trunk movement relative to foot movement and this pattern is different compared to elderly non-fallers and healthy young adults. Additionally, we showed that healthy young adults adapt to speed differently compared to elderly adults, with healthy young adults increasing their lumbar acceleration relative to their foot accelerations when walking at faster speeds. These findings demonstrate the importance of interpreting the control of lumbar and foot movement in context of the whole body, as the movement of these segments must be coordinated with each other to maintain stable gait under challenging conditions. The GSI metrics used in this study quantified these cautious gait characteristics using portable wireless sensors which could be used for examining a range of fall-risk populations in clinical and at-home settings without the need for a treadmill. Future studies will examine what GSI metrics are most appropriate for identifying fall risk, and how much overground walking is necessary for appropriate calculation of the GSI metrics to determine feasibility for use in clinical and at-home settings.
Data availability
The datasets generated during and/or analyzed during the current study may be available from the corresponding author on reasonable request.
References
Tromp AM, Pluijm SM, Smit JH et al (2001) Fall-risk screening test: a prospective study on predictors for falls in community-dwelling elderly. J Clin Epidemiol 54:837–844
Salva A, Bolibar I, Pera G et al (2004) Incidence and consequences of falls among elderly people living in the community. Med clin 122:172–176
Goodpaster BH, Park SW, Harris TB et al (2006) The loss of skeletal muscle strength, mass, and quality in older adults: the health, aging and body composition study. J Gerontol Ser A Biol Sci Med Sci 61:1059–1064
Kaplan FS, Nixon JE, Reitz M et al (1985) Age-related changes in proprioception and sensation of joint position. Acta orthop Scand 56:72–74
Sloane PD, Baloh RW, Honrubia V (1989) The vestibular system in the elderly: clinical implications. Am J Otolaryngol 10:422–429
Springer S, Giladi N, Peretz C et al (2006) Dual-tasking effects on gait variability: the role of aging, falls, and executive function. Mov Disord Off J Mov Disord Soc 21:950–957. https://doi.org/10.1002/mds.20848
Francis CA, Franz JR, O’Connor SM et al (2015) Gait variability in healthy old adults is more affected by a visual perturbation than by a cognitive or narrow step placement demand. Gait Posture 42:380–385. https://doi.org/10.1016/j.gaitpost.2015.07.006
Kang HG, Dingwell JB (2016) Differential changes with age in multiscale entropy of electromyography signals from leg muscles during treadmill walking. PLoS ONE 11:e0162034. https://doi.org/10.1371/journal.pone.0162034
Palumbo P, Palmerini L, Bandinelli S et al (2015) Fall risk assessment tools for elderly living in the community: can we do better? PLoS ONE 10:e0146247. https://doi.org/10.1371/journal.pone.0146247
Barry E, Galvin R, Keogh C et al (2014) Is the Timed Up and Go test a useful predictor of risk of falls in community dwelling older adults: a systematic review and meta- analysis. BMC Geriatr 14:14–14. https://doi.org/10.1186/1471-2318-14-14
Muir SW, Berg K, Chesworth B et al (2010) Balance impairment as a risk factor for falls in community-dwelling older adults who are high functioning: a prospective study. Phys Ther 90:338–347. https://doi.org/10.2522/ptj.20090163
Lockhart TE, Liu J (2008) Differentiating fall-prone and healthy adults using local dynamic stability. Ergonomics 51:1860–1872. https://doi.org/10.1080/00140130802567079
Toebes MJ, Hoozemans MJ, Furrer R et al (2012) Local dynamic stability and variability of gait are associated with fall history in elderly subjects. Gait Posture 36:527–531. https://doi.org/10.1016/j.gaitpost.2012.05.016
Rispens SM, van Schooten KS, Pijnappels M et al (2014) Identification of fall risk predictors in daily life measurements: gait characteristics’ reliability and association with self-reported fall history. Neurorehabil Neural Repair. https://doi.org/10.1177/1545968314532031
Bruijn SM, Meijer OG, Beek PJ et al (2013) Assessing the stability of human locomotion: a review of current measures. J R Soc Interface R Soc 10:20120999. https://doi.org/10.1098/rsif.2012.0999
Winter DA (1995) Human balance and posture control during standing and walking. Gait Posture 3:193–214. https://doi.org/10.1016/0966-6362(96)82849-9
Hurt CP, Rosenblatt N, Crenshaw JR et al (2010) Variation in trunk kinematics influences variation in step width during treadmill walking by older and younger adults. Gait Posture 31:461–464. https://doi.org/10.1016/j.gaitpost.2010.02.001
Lugade V, Lin V, Chou LS (2011) Center of mass and base of support interaction during gait. Gait Posture 33:406–411. https://doi.org/10.1016/j.gaitpost.2010.12.013
Arvin M, van Dieen JH, Bruijn SM (2016) Effects of constrained trunk movement on frontal plane gait kinematics. J Biomech 49:3085–3089. https://doi.org/10.1016/j.jbiomech.2016.07.015
Arvin M, Mazaheri M, Hoozemans MJ et al (2016) Effects of narrow base gait on mediolateral balance control in young and older adults. J Biomech 49:1264–1267. https://doi.org/10.1016/j.jbiomech.2016.03.011
Maki BE (1997) Gait changes in older adults: predictors of falls or indicators of fear. J Am Geriatr Soc 45(3):313–320
Kavanagh JJ, Barrett RS, Morrison S (2004) Upper body accelerations during walking in healthy young and elderly men. Gait Posture 20:291–298. https://doi.org/10.1016/j.gaitpost.2003.10.004
Harbourne RT, Stergiou N (2009) Movement variability and the use of nonlinear tools: principles to guide physical therapist practice. Phys Ther 89:267–282. https://doi.org/10.2522/ptj.20080130
Stergiou N, Harbourne R, Cavanaugh J (2006) Optimal movement variability: a new theoretical perspective for neurologic physical therapy. J Neurol Phys Ther JNPT 30:120–129
Dean JC, Alexander NB, Kuo AD (2007) The effect of lateral stabilization on walking in young and old adults. IEEE Trans Biomed Eng 54:1919–1926. https://doi.org/10.1109/tbme.2007.901031
Terrier P, Reynard F (2015) Effect of age on the variability and stability of gait: a cross-sectional treadmill study in healthy individuals between 20 and 69 years of age. Gait Posture 41:170–174. https://doi.org/10.1016/j.gaitpost.2014.09.024
Hamacher D, Singh NB, Van Dieen JH et al (2011) Kinematic measures for assessing gait stability in elderly individuals: a systematic review. J R Soc Interface R Soc 8:1682–1698. https://doi.org/10.1098/rsif.2011.0416
Kerrigan DC, Lee LW, Collins JJ et al (2001) Reduced hip extension during walking: healthy elderly and fallers versus young adults. Arch Phys Med Rehabil 82:26–30. https://doi.org/10.1053/apmr.2001.18584
Kang HG, Dingwell JB (2008) Effects of walking speed, strength and range of motion on gait stability in healthy older adults. J Biomech 41:2899–2905. https://doi.org/10.1016/j.jbiomech.2008.08.002
Craig JJ, Bruetsch AP, Lynch SG et al (2017) The relationship between trunk and foot acceleration variability during walking shows minor changes in persons with multiple sclerosis. Clin Biomech 49:16–21. https://doi.org/10.1016/j.clinbiomech.2017.07.011
Huisinga JM, Mancini M, St George RJ et al (2013) Accelerometry reveals differences in gait variability between patients with multiple sclerosis and healthy controls. Ann Biomed Eng 41:1670–1679. https://doi.org/10.1007/s10439-012-0697-y
Yentes J, Hunt N, Schmid K et al (2013) The appropriate use of approximate entropy and sample entropy with short data sets. Ann Biomed Eng 41:349–365. https://doi.org/10.1007/s10439-012-0668-3
Mehdizadeh S, Sanjari MA (2017) Effect of noise and filtering on largest Lyapunov exponent of time series associated with human walking. J Biomech. https://doi.org/10.1016/j.jbiomech.2017.09.009
Dingwell JB, Marin LC (2006) Kinematic variability and local dynamic stability of upper body motions when walking at different speeds. J Biomech 39:444–452. https://doi.org/10.1016/j.jbiomech.2004.12.014
O’Connor SM, Kuo AD (2009) Direction-dependent control of balance during walking and standing. J Neurophys 102:1411–1419. https://doi.org/10.1152/jn.00131.2009
Mees AI, Judd K (1993) Dangers of geometric filtering. Physica D 68(3–4):427–436. https://doi.org/10.1016/0167-2789(93)90135-N
Stergiou N (2016) Nonlinear analysis for human movement variability. CRC Press, Boca Raton
Richman JS, Moorman JR (2000) Physiological time-series analysis using approximate entropy and sample entropy. Am J Phys Heart Circ Phys 278:H2039–H2049
Wolf A, Swift JB, Swinney HL et al (1985) Determining Lyapunov exponents from a time series. Phys D 16:285–317. https://doi.org/10.1016/0167-2789(85)90011-9
Craig J, Bruetsch A, Horak F et al (2017) Movement variability of the trunk and feet during walking is altered in persons with multiple sclerosis compared to healthy controls. Paper presented at the 5th international symposium on gait and balance in multiple sclerosis, Portland, OR
Kavanagh JJ (2009) Lower trunk motion and speed-dependence during walking. J Neuroeng Rehabil 6:9. https://doi.org/10.1186/1743-0003-6-9
Ihlen EAF, Weiss A, Bourke A et al (2016) The complexity of daily life walking in older adult community-dwelling fallers and non-fallers. J Biomech 49:1420–1428. https://doi.org/10.1016/j.jbiomech.2016.02.055
Watt JR, Franz JR, Jackson K et al (2010) A three-dimensional kinematic and kinetic comparison of overground and treadmill walking in healthy elderly subjects. Clin Biomech 25:444–449. https://doi.org/10.1016/j.clinbiomech.2009.09.002
Khademi-Kalantari K, Rahimi F, Hosseini SM et al (2017) Lower limb muscular activity during walking at different speeds: over-ground versus treadmill walking: A voluntary response evaluation. J Bodywork Mov Ther 21:605–611. https://doi.org/10.1016/j.jbmt.2016.09.009
Riley PO, Paolini G, Della Croce U et al (2007) A kinematic and kinetic comparison of overground and treadmill walking in healthy subjects. Gait Posture 26:17–24. https://doi.org/10.1016/j.gaitpost.2006.07.003
Hollman JH, Watkins MK, Imhoff AC et al (2016) A comparison of variability in spatiotemporal gait parameters between treadmill and overground walking conditions. Gait Posture 43:204–209. https://doi.org/10.1016/j.gaitpost.2015.09.024
Rosenblatt NJ, Grabiner MD (2010) Measures of frontal plane stability during treadmill and overground walking. Gait Posture 31(3):380–384. https://doi.org/10.1016/j.gaitpost.2010.01.002
Funding
This work was supported by the American Society of Biomechanics Grant-in-Aid, the National Institute of Health Ruth L. Kirschstein National Research Service Award T32 HD057850 from the National Institute of Child Health and Human Development, the Frontiers Pilot and Collaborative Studies Funding Program UL1TR000001, and University of Kansas Medical Center—the School of Health Professions Pilot Research Grant.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Craig, J.J., Bruetsch, A.P. & Huisinga, J.M. Coordination of trunk and foot acceleration during gait is affected by walking velocity and fall history in elderly adults. Aging Clin Exp Res 31, 943–950 (2019). https://doi.org/10.1007/s40520-018-1036-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40520-018-1036-4