Abstract
Purpose
Nutritional insufficiency (NI) is a potential consequence of restrictive eating disorders. NI patients often require hospitalization for refeeding to restore medical stability and prevent complications such as refeeding syndrome. Limited information is available on the optimal approach to refeeding. In this study, we describe an inpatient NI care path and compare treatment outcomes at an academic medical center and a community hospital.
Methods
A retrospective chart review was conducted on inpatients treated using a standardized NI care path at either the academic site, from August 2012 to July 2013 (n = 51), or the community site, from August 2013 to July 2014 (n = 39). Demographic information, eating disorder history, and treatment variables were recorded for each patient. Data were compared using the Kruskal–Wallis test and Fisher’s exact test.
Results
Patients admitted to the community site had shorter hospital stays than patients admitted to the academic site (IQR 2–4 vs. 2–7 days, p = 0.03). All patients were discharged in <14 days with a median stay of 3 days. The median initial calorie prescription was 2200 calories for both groups. No clinical cases of refeeding syndrome occurred, with only one patient developing hypophosphatemia during refeeding.
Conclusions
A standardized care path with a higher-calorie intervention allows for short-term hospitalization of NI patients without increasing the risk of refeeding syndrome, regardless of treatment site. This study demonstrates the efficiency and safety of treating NI patients on a regular medical floor at a community hospital.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Clinical care paths have been increasingly recognized as useful tools to improve both the quality and efficiency of care in specific patient populations. The purpose of care paths is to provide a structured multidisciplinary plan of care based on a patient’s medical presentation or diagnosis [1, 2]. This approach can reduce variability in care and, by implementing evidence-based practices, lead to better patient outcomes. Clinical care paths exist for a growing number of conditions, from asthma in children [3–5] to stroke in elderly adults [6, 7]. Few clinical care paths, however, have been published for psychiatric disorders, including eating disorders [8].
Nutritional insufficiency (NI), defined as inadequate nutrition to meet metabolic needs, often occurs in the context of a restrictive eating disorder, such as anorexia nervosa [9, 10]. Initial treatment of NI emphasizes nutritional rehabilitation, or “refeeding,” to stabilize vitals and increase body weight [9, 11]. Many of these patients require hospitalization to monitor vitals and detect signs of “refeeding syndrome,” a life-threatening condition that can occur following electrolyte shifts during refeeding [12, 13]. Safe rehabilitation requires skilled providers familiar with the complications associated with refeeding. In addition, given the psychological component of eating disorders [14], special precautions must be taken to ensure that patients comply with treatment and do not engage in risky behaviors, such as purging meals or inflicting self-harm. A team of trained caregivers, including physicians, dietitians, nurses, mental health providers, and social workers, creates the optimal environment for successful inpatient treatment. This multidisciplinary approach, along with the high cost of inpatient care [15], makes NI an ideal candidate for a clinical care path [16, 17].
Several guidelines and position papers have been published on the treatment of eating disorders [18–20], but these papers do not provide specific protocols for treatment or standards for discharge. As a result, physician and institutional practices vary widely [21]. Although a handful of hospital protocols and care paths have been published [16, 17, 22, 23], these typically describe treatment plans that require several weeks of inpatient care in a specialty or psychiatric unit.
At the Cleveland Clinic Children’s Hospital, providers have utilized a clinical care path to treat patients with NI for over 20 years. This care path is designed to stabilize NI patients during a short-term hospital stay on a regular medical floor, while providing patients and families with resources for comprehensive care after discharge. Initially, the NI care path was implemented at the Cleveland Clinic Main Campus, an academic center where inpatients have access to subspecialty and intensive care. In August 2013, the care path was implemented at a community hospital within the Cleveland Clinic Health System, a site with fewer inpatient services and no prior experience treating patients with NI. This paper outlines important elements of the NI care path as well as outcomes of patients treated at both the academic center and the community hospital, demonstrating the effectiveness of the care path in multiple healthcare environments.
Methods
Nutritional insufficiency care path
Order set
Table 1 lists the criteria for hospital admission for patients with NI. When a patient is admitted, the physician selects the NI order set in the electronic medical record (Supplement 1). This order set alerts other practitioners that the patient is an NI patient. It includes instructions for nursing, such as restrictions on activity, vital sign specifications, and mealtime procedures, that are also included in a nursing protocol and checklist (discussed below). The order set also prompts dietary and adolescent medicine consultation (mandatory for all patients) as well as consults as needed to psychiatry, behavioral medicine, and case management.
A recommended panel of laboratories is included within the order set to evaluate electrolytes, liver function, kidney function, hormonal function, and blood counts, unless these laboratories were already obtained on the day of admission. Urinalysis with specific gravity is obtained daily prior to breakfast to assess hydration status and detect water loading. A complete metabolic panel, phosphorus level, and magnesium level are obtained as needed during hospitalization, typically daily if abnormal or every other day if normal. Patients also receive phosphorus supplementation with Neutra-Phos®, 500 mg orally twice daily for 5 days to reduce the risk of hypophosphatemia, one of the primary electrolyte disturbances observed in refeeding syndrome.
Nursing protocol and checklist
A nursing protocol (Supplement 2) provides a brief rationale for the protocol along with specific procedures for initial and daily assessment of the patient and his or her room environment. A corresponding nursing checklist presents the daily assessment in a practical manner, allowing for more consistent care regardless of the familiarity of the nursing staff with caring for NI patients. Instructions for documentation, parent education, and patient privileges are also included in the protocol. The care path was designed under the premise of good behavior with rewards, such as cell phone use and a daily wheelchair ride, unless a patient exhibits self-sabotaging behavior. This approach allows nurses and other caregivers to partner with patients and avoid developing antagonistic relationships.
Dietitian guidelines
Dietitians meet with each patient and family within 24 h of admission and develop a dietary plan based on his or her diet history. Patients are started on diets with a minimum of 1500 kcal/day, with a majority of patients started on 2200 kcal/day. Dietitians use an exchange system with a specified number of servings from each food group to ensure a balanced diet (Table 2). Calories are increased by about 250 calories daily with adjustments to meet a weight gain goal of 0.2 kg/day. Patients are required to finish their meals in 30 min and their snacks in 20 min. If they consume <100 % of their food in the allotted time, the calories are replaced by liquid supplements, and if they refuse the supplements, a nasogastric tube is placed for enteral feeding. Water intake is limited to 8 oz a day to prevent water loading within the first 24 h, with additional caloric fluid intake to meet needs. After the first 24 h, fluids are liberalized assuming normal renal function and no evidence of significant third spacing.
The Maudsley approach, or family-based refeeding, was used during admission for patients under 18 years of age and in some patients over 18 years of age with family available to provide support. Over the course of admission, the dietitian partnered with parents to help them plan meals and understand the energy needs of their child. Patients participated by expressing their likes and dislikes, beginning to work on fear foods, and learning to trust parental choices regarding meal planning.
Discharge and aftercare
While discharge criteria are based on medical stabilization (Table 1), connecting patients and families with resources for ongoing treatment is essential for long-term recovery. Physicians with training in eating disorder treatment educate patients and families on the medical complications of disordered eating and available therapies. The overall goal of these conversations is to work with families to develop a discharge plan that meets the current needs of the patient and leads to not only physical, but emotional and psychological, health. Many patients begin outpatient treatment programs at a local eating disorder treatment center following discharge. For most patients under 18 years of age, family-based therapy (the Maudsley approach) is recommended [24], although some patients and families require day treatment or residential programs to help the initial recovery process.
Study population
We identified all patients ages 8–23 years admitted for the treatment of NI at either the Cleveland Clinic Main Campus, a multispecialty academic medical center, from August 2012 to July 2013 or the Cleveland Clinic Hillcrest Hospital, a community hospital, from August 2013 to July 2014. Patients were included if they were hospitalized for more than 24 h and treated using the NI care path. Patients treated at the academic center who would not qualify for treatment at a community hospital due to major comorbidities, such as postoperative patients or patients with renal failure, were excluded from the study population. No patients refused treatment. For patients readmitted during the study period, only the first admission was included for analysis.
Study design
Electronic records for all eligible patients were retrospectively reviewed. Demographic information and eating disorder history were obtained from semi-structured interviews conducted by providers involved in patient care during admission. Eating disorder diagnoses were determined retrospectively by a trained researcher using Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5), criteria and confirmed by an adolescent medicine specialist [25]. DSM-5 diagnoses included anorexia nervosa, bulimia nervosa, atypical anorexia nervosa (a disorder with features of anorexia nervosa but normal body weight), atypical bulimia nervosa (a disorder characterized by binge eating and purging that occurs less than once a week or for fewer than 3 months), and avoidant/restrictive food intake disorder (ARFID, a disorder characterized by nutritional insufficiency without evidence of a body image disturbance or lack of access to food). Duration of illness and weight loss were estimated from the history obtained by adolescent medicine specialists consulted during admission or the admitting physician. Treatment variables, including length of stay (LOS), daily weights, daily caloric intake, daily vital signs, electrocardiogram findings, and electrolyte abnormalities, were recorded for each patient. Electrolyte abnormalities were based on the normal range used in the hospital laboratory and included hypophosphatemia (<2.5 mg/dL), hypomagnesemia (<1.7 mg/dL), and hypokalemia (<3.5 meq/L).
Baseline weight was defined as the weight obtained at 0600 on the first full day of admission. Rate of weight gain was determined by dividing the change in weight from baseline to the morning of discharge by LOS. Baseline caloric intake was defined as the initial calories prescribed at the first dietitian consult, usually the first full day of admission. Final caloric intake was defined as the caloric intake for the last day of admission when the patient received all three meals in the hospital. Rate of caloric intake was calculated by dividing the change in calories by the days between initial and final caloric prescriptions.
Body mass index (BMI) was calculated by dividing the patient weight in kilograms by the height in meters squared. Median BMI, the 50th percentile BMI based on age and gender, was determined using the 2000 Centers for Disease Control and Prevention BMI-for-age growth charts for patients aged 2–20 years. For patients over 20 years of age, a median BMI of 21.8 and 23.04 was used for females and males, respectively. Percent expected body weight (%EBW) was calculated by dividing the patient BMI by the median BMI for his or her age and multiplying by 100.
This study was approved by the Cleveland Clinic Institutional Review Board.
Statistical analysis
Data were described using medians and interquartile ranges (IQR) for continuous variables and counts and percentages for categorical variables. Patients admitted to the academic center and patients admitted to the community hospital were compared using the Mann–Whitney U test for continuous variables and Fisher’s exact test for categorical variables. Differences in LOS were also assessed using the Kaplan–Meier method and log-rank test. The association between weight gain during hospitalization and LOS was assessed for the total population using linear regression analysis. All analyses were two-tailed and performed at a significance level of 0.05. Results were analyzed using JMP 9.0 (SAS Institute Inc, Cary, NC, USA).
Results
Between August 2012 and July 2013, 56 patients were placed on the NI care path at a multidisciplinary academic medical center. Of these patients, two were excluded because they had admissions that were <24 h and three were excluded because they required services not routinely provided at a community hospital, resulting in a total of 51 academic center patients. Following implementation of the NI care path at a community hospital, 41 patients were placed on the NI care path between August 2013 and July 2014. Of these patients, one was excluded because she had an admission of less than 24 h and one was excluded because he was transferred from the academic medical center, resulting in a total of 39 community hospital patients. No NI patients were treated at the community hospital prior to implementation of the care path.
The patients included in the analysis were predominantly female (90 %, n = 81) and non-Hispanic white (92 %, n = 83). Age ranged from 8 to 23 years, with a median of 17 years. Most patients met the DSM-5 criteria for anorexia nervosa (70 %, n = 63), with other patients meeting the criteria for atypical anorexia nervosa (14 %, n = 14), bulimia nervosa (8 %, n = 7), and ARFID (8 %, n = 7). Most patients, regardless of diagnosis, were bradycardic (71 %, n = 64) upon admission, with a large percentage experiencing hypotension (40 %, n = 36) and/or a prolonged QT interval (>450 ms, 20 %, n = 18). As summarized in Table 3, baseline characteristics, including demographics and eating disorder history, did not differ between the two treatment sites.
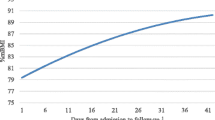
Length of hospital stay differed significantly between patients admitted to the academic center and patients admitted to the community hospital on Kaplan–Meier analysis (Fig. 1, p = 0.03). While the median LOS was 3 days for both groups, the interquartile range was larger for patients admitted to the academic center than for patients admitted to the community hospital (IQR 2–7 vs. 2–4 days, p = 0.03, Table 4). The initial calorie prescriptions, rate of caloric increase, and rate of weight gain did not significantly differ for the two treatment sites.
No clinical cases of refeeding syndrome were observed. Electrolyte abnormalities on admission included hypokalemia (n = 13), hypophosphatemia (n = 2), and hypomagnesemia (n = 1). All patients with low phosphorus or magnesium and 12 patients with low potassium levels had normal levels within 24 h of initiating refeeding or by their next laboratory study. Two patients with borderline hypokalemia (K = 3.4 meq/L) did not have repeat laboratory studies during admission. One patient admitted to the community hospital developed hypophosphatemia during admission. A majority of patients, however, did not have repeat laboratory studies during admission, including 61 % (n = 55) without repeat phosphorus, 64 % (n = 58) without repeat magnesium level, and 51 % (n = 46) without repeat potassium levels. These absent studies prevent accurate estimates of the frequency of electrolyte shifts among patients included in the study. There was no significant difference in the occurrence of electrolyte abnormalities or the percentage of missing laboratory data between the treatment sites.
Discussion
This study describes a care path designed to safely and efficiently stabilize adolescents hospitalized for NI. This care path has been used to treat patients at both a multispecialty academic center and, more recently, a community hospital. Comparing these two sites of care, patients received similar dietary interventions and experienced similar rates of weight gain, regardless of healthcare environment. Patients treated at the community hospital, however, had shorter hospitalizations compared to those treated at the academic site.
Duration of hospitalization in our study population was shorter than that typically reported in the literature, with all of our patients discharged in <14 days [26–29]. This difference in LOS likely reflects both individual aspects of our care path, such as the use of higher-calorie diets and the absence of weight criteria for discharge, as well as the unified approach to care achieved through the use of a clinical care path. Practices vary widely among individual physicians and institutions with regard to refeeding and treatment goals [21], potentially leading to longer stays. To our knowledge, however, no studies have compared outcomes of NI patients treated with and without a care path. Reductions in LOS for other conditions, on the other hand, have been demonstrated following implementation of clinical care paths [2].
Interestingly, in our study, patients treated at the community hospital were often discharged earlier than those treated at the academic center. Reasons for this difference are unclear, but may reflect differences in team training and care environment. At the community hospital, team members participated in care path training prior to implementation as well as quarterly meetings to address any problems or concerns with the care path. At the academic center, in contrast, formal training occurred decades prior to the study and team meetings were infrequent for caregivers other than rotating residents. The benefit of regular multidisciplinary sessions was apparent in the growing confidence and skill displayed by team members at the community hospital, many of whom initially questioned their ability to adequately treat these patients. In addition to more frequent training, providers at the community site cared for patients on the same hospital floor, potentially improving communication among team members. The academic site, on the other hand, admitted NI patients to four different floors, resulting in a greater number of personnel coordinating their care and discharge.
Many inpatient treatment programs include a target weight as part of their discharge criteria for patients with eating disorders [30]. Although weight restoration plays an important role in eating disorder recovery, it is not necessary to achieve acute medical stabilization, including resolution of vital sign instability and electrolyte imbalance. Our approach emphasizes the short-term value of inpatient stabilization and the greater value of ongoing outpatient support [31]. In our region, we partner with an accredited outpatient center that offers a spectrum of services, including family-based therapy and day treatment programs. Since 2006, the National Eating Disorder Quality Improvement Collaborative has been tracking outcomes of adolescent medicine eating disorder programs across the nation [32, 33], including the Cleveland Clinic Children’s Hospital. Their results have found no difference between our approach and that of other programs, including longer inpatient programs, with respect to weight restoration at 1 year [32, 33].
Concerns about refeeding syndrome have led to conservative guidelines for nutritional rehabilitation without strong evidence to support their assumptions [34]. During prolonged periods of caloric deficit, blood glucose levels decline, resulting in catabolism of fat and protein stores and depletion of intracellular electrolytes. During refeeding, insulin stimulates anabolic processes that require electrolytes, further decreasing levels of phosphorus, magnesium, and potassium in the blood and, consequently, leading to widespread cellular and organ dysfunction or “refeeding syndrome” [13]. Traditional approaches assume that slow caloric replenishment can prevent refeeding syndrome [20, 35]. Case reports have shown, however, that refeeding syndrome can occur with intakes as low as 500–800 kcal/day [36, 37]. Conversely, increasing evidence suggests that higher-calorie diets may have benefits without increasing the risk of refeeding syndrome. In 2010, Whitelaw et al. [38] published a study demonstrating the safety of initiating refeeding with diets of 1900 kcal or higher. Two recent studies by Garber et al. [28] and Golden et al. [29] also demonstrated that higher-calorie interventions (about 1400–2400 kcal) decreased LOS compared to lower-calorie interventions (about 800–1300 kcal) without increasing the incidence of hypophosphatemia in adolescents with anorexia nervosa.
Consistent with this emergent understanding of refeeding syndrome, our NI care path uses relatively high-calorie diets (median 2200 calories) to initiate refeeding. No clinical cases of refeeding syndrome occurred in our study population, and only one patient had documented hypophosphatemia during refeeding. This patient had one of the most severe presentations in the study, with a low %EBW (68 %), substantial weight loss (45 kg), and elevated liver function tests at admission. These results are consistent with other studies that report an association between low body weight and medical complications during refeeding, including death [28, 38–40]. Recent studies, however, have reported similar rates of medical complications among eating disorder patients regardless of weight status at admission [41, 42], highlighting the need for additional research on risk factors for medical complications during refeeding.
We recognize several limitations to this study. First, because patients with NI were not previously treated at the community hospital, we were unable to compare patient outcomes before and after implementation of the NI care path. Another major constraint is the retrospective design, which inherently limits the information available for analysis. The reported number of electrolyte abnormalities during refeeding is likely underestimated since many patients did not have laboratory studies performed after admission. Refeeding syndrome, however, should not be identified solely on the basis of electrolyte disturbances [43]. In addition, the study population was predominantly white females with moderate malnutrition, potentially limiting its generalizability to other groups of patients. Lastly, we do not have follow-up information in this data set to assess patient outcomes after discharge and determine whether short-term hospitalization is an effective first step in the treatment of NI, although other studies provide reassuring results [31].
This study describes the successful implementation of an NI care path at both an academic center and a community hospital. While treating patients with eating disorders is complex, often requiring a skilled multidisciplinary team, treatment does not have to be initiated within a specialized facility or on a specialized floor. A standardized care path, along with care path training by experienced adolescent medicine physicians, can allow for short-term medical stabilization of NI patients on a regular medical floor at a community site. Our results demonstrate that a higher-calorie intervention with prophylactic phosphorus supplementation minimizes hospital stay without placing NI patients at a higher risk of refeeding syndrome.
References
Kinsman L, Rotter T, James E, Snow P, Willis J (2010) What is a clinical pathway? Development of a definition to inform the debate. BMC Med 8:31. doi:10.1186/1741-7015-8-31
Rotter T, Kinsman L, James E et al (2010) Clinical pathways: effects on professional practice, patient outcomes, length of stay and hospital costs. Cochr Datab Syst Rev 3:CD006632. doi:10.1002/14651858.CD006632.pub2
Cunningham S, Logan C, Lockerbie L, Dunn MJG, McMurray A, Prescott RJ (2008) Effect of an integrated care pathway on acute asthma/wheeze in children attending hospital: cluster randomized trial. J Pediatr 152(3):315–320. doi:10.1016/j.jpeds.2007.09.033
Kelly CS, Andersen CL, Pestian JP et al (2000) Improved outcomes for hospitalized asthmatic children using a clinical pathway. Ann Allergy Asthma Immunol Off Publ Am Coll Allergy Asthma Immunol 84(5):509–516. doi:10.1016/S1081-1206(10)62514-8
Wazeka A, Valacer DJ, Cooper M, Caplan DW, DiMaio M (2001) Impact of a pediatric asthma clinical pathway on hospital cost and length of stay. Pediatr Pulmonol 32(3):211–216. doi:10.1002/ppul.1110
Kwan J, Hand P, Dennis M, Sandercock P (2004) Effects of introducing an integrated care pathway in an acute stroke unit. Age Ageing 33(4):362–367. doi:10.1093/ageing/afh104
Sulch D, Perez I, Melbourn A, Kalra L (2000) Randomized controlled trial of integrated (managed) care pathway for stroke rehabilitation. Stroke J Cereb Circ 31(8):1929–1934. doi:10.1161/01.STR.31.8.1929
Evans-Lacko SE, Jarrett M, McCrone P, Thornicroft G (2008) Clinical pathways in psychiatry. Br J Psychiatry J Ment Sci 193(1):4–5. doi:10.1192/bjp.bp.107.048926
Auron M, Rome E (2011) Anorexia nervosa and bulimia nervosa: what the hospitalist needs to know about CPT 269.9, or nutritional insufficiency. Am Coll Physicians Hosp 2011:38–45
Ornstein RM, Rosen DS, Mammel KA et al (2013) Distribution of eating disorders in children and adolescents using the proposed DSM-5 criteria for feeding and eating disorders. J Adolesc Health Off Publ Soc Adolesc Med 53(2):303–305. doi:10.1016/j.jadohealth.2013.03.025
Katzman DK (2005) Medical complications in adolescents with anorexia nervosa: a review of the literature. Int J Eat Disord 37(Suppl):S52–S59. doi:10.1002/eat.20118 (discussion S87–S89)
Hearing SD (2004) Refeeding syndrome. BMJ 328(7445):908–909. doi:10.1136/bmj.328.7445.908
Mehanna HM, Moledina J, Travis J (2008) Refeeding syndrome: what it is, and how to prevent and treat it. BMJ 336(7659):1495–1498. doi:10.1136/bmj.a301
Herzog DB, Keller MB, Sacks NR, Yeh CJ, Lavori PW (1992) Psychiatric comorbidity in treatment-seeking anorexics and bulimics. J Am Acad Child Adolesc Psychiatry 31(5):810–818. doi:10.1097/00004583-199209000-00006
Toulany A, Wong M, Katzman DK et al (2015) Cost analysis of inpatient treatment of anorexia nervosa in adolescents: hospital and caregiver perspectives. CMAJ Open 3(2):E192–E197. doi:10.9778/cmajo.20140086
Lock J (1999) How clinical pathways can be useful: an example of a clinical pathway for the treatment of anorexia nervosa in adolescents. Clin Child Psychol Psychiatry 4(3):331–340. doi:10.1177/1359104599004003004
Sylvester CJ, Forman SF (2008) Clinical practice guidelines for treating restrictive eating disorder patients during medical hospitalization. Curr Opin Pediatr 20(4):390–397. doi:10.1097/MOP.0b013e32830504ae
Society for Adolescent Health and Medicine, Golden NH, Katzman DK et al (2015) Position Paper of the Society for Adolescent Health and Medicine: medical management of restrictive eating disorders in adolescents and young adults. J Adolesc Health Off Publ Soc Adolesc Med 56(1):121–125. doi:10.1016/j.jadohealth.2014.10.259
Rosen DS, American Academy of Pediatrics Committee on Adolescence (2010) Identification and management of eating disorders in children and adolescents. Pediatrics 126(6):1240–1253. doi:10.1542/peds.2010-2821
American Psychiatric Association (2006) Treatment of patients with eating disorders, third edition. American Psychiatric Association. Am J Psychiatry 163(7 Suppl):4–54
Schwartz BI, Mansbach JM, Marion JG, Katzman DK, Forman SF (2008) Variations in admission practices for adolescents with anorexia nervosa: a North American sample. J Adolesc Health Off Publ Soc Adolesc Med 43(5):425–431. doi:10.1016/j.jadohealth.2008.04.010
Wagner RA, MacCaughelty CR (2013) A track-based approach for treating eating disorders. Bull Menninger Clin 77(3):222–232. doi:10.1521/bumc.2013.77.3.222
Treat TA, Gaskill JA, McCabe EB, Ghinassi FA, Luczak AD, Marcus MD (2005) Short-term outcome of psychiatric inpatients with anorexia nervosa in the current care environment. Int J Eat Disord 38(2):123–133. doi:10.1002/eat.20160
Lock J, le Grange D (2005) Family-based treatment of eating disorders. Int J Eat Disord 37(S1):S64–S67. doi:10.1002/eat.20122
American Psychiatric Association (2013) Diagnostic and statistical manual of mental disorders, 5th edn. American Psychiatric Association, Washington, DC
Garber AK, Michihata N, Hetnal K, Shafer M-A, Moscicki A-B (2012) A prospective examination of weight gain in hospitalized adolescents with anorexia nervosa on a recommended refeeding protocol. J Adolesc Health Off Publ Soc Adolesc Med 50(1):24–29. doi:10.1016/j.jadohealth.2011.06.011
Wiseman CV, Sunday SR, Klapper F, Harris WA, Halmi KA (2001) Changing patterns of hospitalization in eating disorder patients. Int J Eat Disord 30(1):69–74. doi:10.1002/eat.1055
Garber AK, Mauldin K, Michihata N, Buckelew SM, Shafer M-A, Moscicki A-B (2013) Higher calorie diets increase rate of weight gain and shorten hospital stay in hospitalized adolescents with anorexia nervosa. J Adolesc Health Off Publ Soc Adolesc Med 53(5):579–584. doi:10.1016/j.jadohealth.2013.07.014
Golden NH, Keane-Miller C, Sainani KL, Kapphahn CJ (2013) Higher caloric intake in hospitalized adolescents with anorexia nervosa is associated with reduced length of stay and no increased rate of refeeding syndrome. J Adolesc Health Off Publ Soc Adolesc Med 53(5):573–578. doi:10.1016/j.jadohealth.2013.05.014
Peter Roots JH (2006) The use of target weights in the inpatient treatment of adolescent anorexia nervosa. Eur Eat Disord Rev 14(5):323–328. doi:10.1002/erv.723
Herpertz-Dahlmann B, Schwarte R, Krei M et al (2014) Day-patient treatment after short inpatient care versus continued inpatient treatment in adolescents with anorexia nervosa (ANDI): a multicentre, randomised, open-label, non-inferiority trial. Lancet 383(9924):1222–1229. doi:10.1016/S0140-6736(13)62411-3
Forman SF, Grodin LF, Graham DA et al (2011) An eleven site national quality improvement evaluation of adolescent medicine-based eating disorder programs: predictors of weight outcomes at one year and risk adjustment analyses. J Adolesc Health Off Publ Soc Adolesc Med 49(6):594–600. doi:10.1016/j.jadohealth.2011.04.023
Forman SF, McKenzie N, Hehn R et al (2014) Predictors of outcome at 1 year in adolescents with DSM-5 restrictive eating disorders: report of the national eating disorders quality improvement collaborative. J Adolesc Health Off Publ Soc Adolesc Med 55(6):750–756. doi:10.1016/j.jadohealth.2014.06.014
Katzman DK (2012) Refeeding hospitalized adolescents with anorexia nervosa: is “start low, advance slow” urban legend or evidence based? J Adolesc Health 50(1):1–2. doi:10.1016/j.jadohealth.2011.10.003
American Dietetic Association (2006) Position of the American Dietetic Association: nutrition intervention in the treatment of anorexia nervosa, bulimia nervosa, and other eating disorders. J Am Diet Assoc 106(12):2073–2082. doi:10.1016/j.jada.2006.09.007
Norris ML, Pinhas L, Nadeau P-O, Katzman DK (2012) Delirium and refeeding syndrome in anorexia nervosa. Int J Eat Disord 45(3):439–442. doi:10.1002/eat.20963
Kohn MR, Golden NH, Shenker IR (1998) Cardiac arrest and delirium: presentations of the refeeding syndrome in severely malnourished adolescents with anorexia nervosa. J Adolesc Health Off Publ Soc Adolesc Med 22(3):239–243. doi:10.1016/S1054-139X(97)00163-8
Whitelaw M, Gilbertson H, Lam P-Y, Sawyer SM (2010) Does aggressive refeeding in hospitalized adolescents with anorexia nervosa result in increased hypophosphatemia? J Adolesc Health Off Publ Soc Adolesc Med 46(6):577–582. doi:10.1016/j.jadohealth.2009.11.207
Ornstein RM, Golden NH, Jacobson MS, Shenker IR (2003) Hypophosphatemia during nutritional rehabilitation in anorexia nervosa: implications for refeeding and monitoring. J Adolesc Health Off Publ Soc Adolesc Med 32(1):83–88
Nakamura M, Yasunaga H, Shimada T, Horiguchi H, Matsuda S, Fushimi K (2013) Body mass index and in-hospital mortality in anorexia nervosa: data from the Japanese Diagnosis Procedure Combination database. Eat Weight Disord 18(4):437–439. doi:10.1007/s40519-013-0051-x
Whitelaw M, Gilbertson H, Lee KJ, Sawyer SM (2014) Restrictive eating disorders among adolescent inpatients. Pediatrics 134(3):e758–e764. doi:10.1542/peds.2014-0070
Peebles R, Hardy KK, Wilson JL, Lock JD (2010) Are diagnostic criteria for eating disorders markers of medical severity? Pediatrics 125(5):e1193–e1201. doi:10.1542/peds.2008-1777
Tasci I, Bulucu MF, Saglam K (2015) Acid-base disturbances may complicate the diagnosis of refeeding syndrome. Eur J Clin Nutr 69(8):975. doi:10.1038/ejcn.2015.34
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Ethical approval
This study was approved by the Cleveland Clinic Institutional Review Board.
Informed consent
Informed consent was waived due to the retrospective design of the study and the minimal risk to subjects.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplement 1
Epic order set for nutritional insufficiency care path (DOCX 183 kb)
Supplement 2
Nutritional insufficiency care path nursing protocol (DOC 35 kb)
Rights and permissions
About this article
Cite this article
Strandjord, S.E., Sieke, E.H., Richmond, M. et al. Medical stabilization of adolescents with nutritional insufficiency: a clinical care path. Eat Weight Disord 21, 403–410 (2016). https://doi.org/10.1007/s40519-015-0245-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40519-015-0245-5