Abstract
Purpose of review
This review aimed to compile the main studies on the Mayaro virus (MAYV), addressing the clinical manifestations of Mayaro fever, in addition to assessing the possibility of epidemic events and a possible pandemic on a global scale.
Recent findings
Recent studies using animal models and the evaluation of infection in humans demonstrate that MAYV induces a strong inflammatory response that plays an important role in the development and progression of tissue damage and in the induction of myalgia and arthralgia common to the infection. Due to the similarity of symptoms with other co-circulating arboviruses, differential diagnosis is difficult, which contributes to underestimates of the actual number of cases, effecting epidemiological studies. The development of new methods for molecular diagnosis, faster and more sensitive, is extremely important for overcoming this complication. As there is still no specific treatment or vaccine for MAYV, measures for the prevention and containment of the vector are the best alternatives for disease control.
Summary
The increase in the number of possible vectors and their geographic distribution require worldwide health services to dedicate more attention to MAYV, especially in countries in tropical and subtropical regions. Its potential expansion in urbanized areas signals an alert for greater efforts to be made in the treatment and control of the virus.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Mayaro virus and epidemiology
The Mayaro virus (MAYV) is an enveloped RNA virus, belonging to the Togaviridae family and to the alphavirus genus. MAYV, together with the Chikungunya (CHIKV), Ross River (RRV), Sindbis (SINV), Barmah Forest (BFV), and O’nyong-nyong viruses, is part of the group of arthritogenic alphaviruses, which are responsible for causing debilitating chronic disease, such as persistent arthralgia and myalgia [1].
MAYV is endemic of South American countries, having been isolated for the first time in Trinidad and Tobago, 1954. Since the first report, it has been responsible for sporadic outbreaks of Mayaro fever (MF), particularly in regions around the Amazon basin. The MAYV virus is maintained in the wild in a continuous wild cycle of transmission, which primarily involves the Haemagogus janthinomys mosquito as vector and non-human primates as a host [2]. However, many other genera of mosquitoes have been proposed as potential vectors for MAYV, including urban vectors. The potential of MAYV to adapt to new vectors and hosts has favored its urbanization and increased its geographical distribution, making this pathogen an emergent threat to public health [3, 4].
Pathogenesis and prevention and treatment strategies
One of the most striking features of infection by arthritogenic alphaviruses is persistent and debilitating arthritis/arthralgia. This persistent joint pain has a major socioeconomic impact [5]. The cellular and molecular mechanisms of alphaviral arthritis are still understood, but some studies in animal models have shown that the host’s immune response plays an important role in tissue damage [6, 7]. The evaluation of the inflammatory response in the pathogenesis induced by CHIKV, RRV, and MAYV has highlighted mediators IL-6, TNF, INF-γ, and CCL2 and macrophages, as the main factors associated with tissue destruction and severe long-duration arthralgia [8••, 9, 10].
Currently, there is no specific therapy for MAYV, being the treatment based only on symptom control, using analgesics and non-steroidal anti-inflammatory drugs [11]. In addition, there is no licensed vaccine for MAYV infection control and spread. However, there are different proposed vaccine candidates, including different strategies as subunit, DNA, and live attenuated vaccines. In preclinical tests, DNA and live attenuated vaccines were able to induce an efficient protective response [12,13,14,15]. Therefore, although it has been shown to be inefficient in controlling other arboviruses, the only available approach to contain MAYV spread is vector control [11].
Clinical manifestations
MAYV is the etiologic agent of MF, a neglected acute febrile illness, that represents an important public health challenge in South and Central America [16, 17]. In addition, Mayaro fever symptoms are similar to other arboviruses, which make the disease often clinically indistinguishable from Chikungunya, dengue, and Zika fever. Thus, accurate diagnosis of MAYV infections is difficult in places where these viruses co-circulate [3, 17]).
Mayaro fever is a mild, self-limiting, non-specific illness and is characterized by the presence of two phases [18]. After infection, an incubation period of 7 to 12 days can occur [19]. The acute phase of the disease usually lasts for 2–5 days and starts abruptly with high fever (usually between 39 and 40 °C), associated with arthralgia/arthritis, rash, headache, myalgia, retro-ocular pain, and swelling in the joints. In addition, in some cases, less frequent symptoms may occur, including nausea, vomiting, diarrhea, swollen lymph nodes, abdominal pain, and hemorrhagic phenomena such as petechiae, gingival bleeding, and epistaxis [18, 20,21,22]. Maculopapular rash is a common clinical manifestation, occurring in 90% of children and 50% of adults. It usually appears on the fifth day and persists for 3 to 4 days and can be followed by desquamation [18, 23].
Retro-ocular pain and photophobia are clinical signs frequently reported in patients with MF, but no study has demonstrated whether there are other ocular complications associated with MAYV infection. In patients infected with CHIKV, it has been observed that these ocular manifestations in the acute phase of the disease are associated with inflammatory complications, such as anterior uveitis (the most frequent), posterior uveitis, and optic neuropathy. These reports, together with the observation made by Santos et al. [24] in which animals infected with MAYV show signs of irritation in the eyes and of the virus replicating in these tissues, demonstrate the importance of assessing more carefully patients who present these clinical signs during a MAYV infection [25,26,27].
The convalescent phase can last for a few weeks, during which time the patients experience weakness, arthralgia/arthritis, and myalgia [2]. Disabling arthralgia is the most striking clinical feature in MAYV infection and other arthritogenic alphaviruses. Joint pain starts in the acute phase, and in more than 50% of MAYV infections, it progresses to the chronic form (Fig. 1) [5, 28•]. It is considered the chronic stage of the disease when pain and arthritis persist for more than 3 months after the onset of symptoms [7]. Many studies have reported that polyarthralgia can last from 2 to 12 months, but there is no evidence of permanent joint damage [5, 29, 30]. This phase of the disease is characterized by the maintenance of the inflammatory response in the joints, and there is no evidence until the moment of viral persistence in these tissues. Among the affected joints, the main ones are the ankles, wrists, hands, feet, elbows, and knees [28•].
Time line of Mayaro fever. After infection, an incubation period of 7–12 days may occur. Mayaro fever is divided into two phases, acute and convalescent, but in 50% of cases, the disease can progress to a chronic form. The acute phase starts abruptly with high fever and other general symptoms. In this phase, the production of many inflammatory mediators has been observed. Then, the convalescent phase begins, which includes joint and muscle pain. The chronic phase is characterized mainly by persistent arthralgia. IL-7 and VEGF have been considered markers (highlighted in red letters) of disease severity, as they are elevated throughout the analyzed clinical course. Red arrow (infection). Black arrow (brutal onset of symptoms). Inflammatory mediators include data from studies with patients and animal models.
Immune mediators of MAYV infection and serological analyses
MAYV infection elicits a strong inflammatory immune response caused by secretion of a variety of mediators. A study cohort analyzed blood samples from patients who developed persistent arthralgia and who fully recovered from infection. The data collected revealed that various immune mediators were significantly elevated during the acute phase of viral infection. The chemokine MCP-1, related to regulation of monocyte/macrophage migration and infiltration, peaked during the acute phase and remained elevated for up to 6 months post-infection in patients with persistent arthralgia. Levels of the cytokines IL-2 and IL-9, involved in cell proliferation, stood out during the convalescent phase and for up to 3 months post-infection. Also, levels of the cytokines IL-7 and IL-13 were found to be elevated during this phase. Another pro-inflammatory cytokine, IL-17, showed a significant increase at all time points analyzed. This mediator has been associated with both joint inflammation and joint destruction. Vascular endothelial growth factor (VEGF), a chemokine related to stimulation of angiogenesis, showed a significant increase after 3 and 6 months post-infection. Other immune mediators were found to be elevated in subjects who developed persistent arthralgia, as well as those who fully recovered from the disease, regardless of the phase of infection, compared with a healthy control group, including IL-1Ra, IL-8, G-CSF, IFN-γ, PDGF-BB, and TNF-α. Furthermore, the study also demonstrated that although MAYV infection elicited a robust anti-MAYV immune response, the magnitude of response showed no significant difference between patients with persistent arthralgia and those who fully recovered. The authors speculated that antibodies were not able to reach and neutralize the virus at the infection points and thus protect individuals from developing persistent arthralgia [31••].
Similarly, Tappe et al. [5] reported sustained cytokine levels in blood samples from six patients during the recovery phase (> 30 days post-infection). In this study, patients reported prolonged arthralgia from MAYV infection for 2 to 12 months post-infection, in accordance with the pro-inflammatory cytokine and chemokine levels observed. Mediators such as IL-5, IL-6, IL-7, IL-8, IL-9, IL-10, IL-13, IL-17, IP-10, RANTES, IFN-γ, GM-CSF, MIP-1α, and MIP-1β were significantly higher during the recovery phase, suggesting virus persistence and replication and maintenance of a local inflammatory process.
In an arthritis-myositis murine model, Santos et al. [24] have demonstrated a similar outcome for MAYV infection. A panel of pro-inflammatory immune mediators has shown significant increase, parallel to the observation of histopathological changes after viral infection. Histological analyses revealed damage to skeletal muscle tissue, joints, and bones, characterized by inflammatory infiltrates, degenerating fibers, collagen deposition, and vasculitis, more prominent on days 7 and 10 post-infection. The chemokine MCP-1 and the cytokines IL-6, INF-γ, and TNF peaked during days 3 and 10 post-infection, leading to the inflammatory state observed in those tissues. The regeneration process started on day 10 post-infection, in which the levels of the anti-inflammatory cytokines IL-4 and IL-10 peaked and continued, with IL-4 levels remaining elevated for up to 20 days post-infection. Based on these findings, MAYV infection results in a strong local inflammatory process similar to CHIKV infection, affecting muscle and joint-associated tissues.
As part of the alphavirus serological group, termed the Semliki Complex, MAYV shares antigenic sites with other alphaviruses, which can lead to cross-reactivity in serological tests and impair correct diagnosis [30]. Viral isolation from blood in cell cultures has been proved a reliable and efficient method for alphavirus identification; however, MAYV presents a short viremia period of only 2 to 3 days, which can complicate its application [32]. In this manner, alphavirus diagnosis is usually determined by serum analyses measuring the immune response against viral infection, such as IgM and IgG antibody levels. IgM levels normally elevate in 3 days post-infection and continue for up to 3 months. Their detection is essential for diagnosis and disease surveillance, whereas IgG levels can persist for years and are useful for analyses of the convalescent phase and epidemiologic studies [33]. The plaque reduction neutralization test (PRNT) is considered to be the gold standard serological method for alphavirus infection. This test relies on the neutralizing ability of antibodies against viral particles, quantifying the amount of antibodies necessary to reduce lysis plaque formation. Although sensitive and specific, this technique is expensive and time-consuming [32].
One of the most common techniques for alphavirus diagnosis is the enzyme-linked immunosorbent assay (ELISA). Other methods based on hemagglutination inhibition (HI), complement fixation (CF), and immunofluorescence assays are also useful for anti-MAYV antibody detection, although the HI and CF methods may require paired sera due to their inability to differentiate IgM from IgG [2].
Wang et al. [34] have developed a combined method using RT-PCR and ELISA for alphavirus detection and differentiation as an effort to overcome misdiagnosis. Viral RNA is amplified and converted into complementary DNA through RT-PCR using specific primers for the nsP1 gene, followed by an ELISA using biotin-labeled probes targeting MAYV amplicons. As an effort to improve sensitivity in diagnostic tests, Fumagalli et al. [35] have developed an ELISA for detecting antibodies against recombinant envelope protein 2 from MAYV. IgG detection showed a 100% sensitivity and 78% specificity compared with standard test MAYV-specific PRNT50.
Fischer et al. [36] have proposed combining ELISAs for MAYV and CHIKV detection in order to reduce misdiagnosis between them, an especially interesting approach for geographic regions with overlapping transmission.
Mechanism associated with pathogenesis
The pathogenic mechanism of MAYV is still poorly understood. But similar to that observed for CHIKV and RRV, MAYV also induces a robust inflammatory response, which has been described as having an important role in the development and progression of musculoskeletal damage [5, 6, 24, 31••]. Interestingly, from studies with animal models performed mainly with RRV and CHIKV, many factors of the host’s immune and inflammatory response already have their roles well described in the alphaviral pathogenesis [6, 7].
The severity and persistence of the joint pains are associated with two factors: the replication of the virus in target tissues (muscle tissue and tissues associated with the joints) and the inflammatory host response. The replication of alphaviruses in target tissues induces the influx of immune cells and the potent production of inflammatory cytokines and chemokines [6, 7]).
The analysis of the inflammatory infiltrate of the muscles and joints of animal models of RRV and CHIKV has shown that macrophages are the predominant cells, but there is also the presence of neutrophils, T lymphocytes, and NK cells [37, 38]. These same cells were also seen in the synovial fluid, blood serum, and muscle biopsy of patients [39,40,41]. In addition, macrophages and their pro-inflammatory products, CCL2, TNF-α, and INF-γ, are considered to be the main factors associated with tissue damage induced by RRV and CHKV in animal models. This was demonstrated in studies in which the depletion of macrophages resulted in a reduction in the severity of the disease [8••, 9, 42]. Macrophages are also the target cells for the replication of RRV, CHIKV, and SINV and can maintain a persistent infection of these viruses in patients with long-lasting arthralgia [43]. Cavalheiro et al. [44] demonstrated that MAYV is also capable of replicating in macrophages and inducing TNF and reactive oxygen species production.
Studies in animal models for RRV, CHIKV, and MAYV, investigating the role of the adaptive immune response mediated mainly by TCD4+ and TCD8+ cells, have shown that these cells act differently against infection by these viruses. For RRV, it was demonstrated that T lymphocytes do not play a critical role in the development of tissue damage, but TCD8+ proved to be important in controlling the viral load in muscle tissue. During the course of CHIKV infection, TCD4+ cells mediate joint swelling and damage to joint tissues, but the depletion of TCD4+ and TCD8+ had no effect on viremia [37, 45, 46]. Figueiredo et al. [47] demonstrated that acquired immunity is important both in the control of replication and in tissue damage induced by MAYV.
MAYV, similar to CHIKV and RRV, induces high levels of IL-6, CCL2, and TNF, which are important inflammatory mediators in the alphaviral pathogenesis, acting on tissue destruction and severe and long-lasting arthralgia [10]. These mediators have been detected in the serum of patients and in animal models [24, 31••, 38]. The cytokine IL-6 and the chemokines MCP-1/CCL2, MCP-2/CCL8, and MCP-3/CCL7 have been described as being associated with the osteoclastogenesis process induced by CHIKV and RRV. In vitro studies with human osteoblasts and in animal models have shown that alphaviruses can infect osteoblasts and induce the production of IL-6, which in turn leads to changes in the RANKL:OPG ratio and increases the expression of CCL2. These changes in the joint environment are responsible for bone pathologies [48,49,50].
MAYV also has the potential to replicate in human osteoblasts (HOB), as well as in human chondrocytes (HC) and fibroblast-like synoviocytes (HFLS), the main cells involved in osteoarthritis from MAYV infection. HC infection resulted in the expression of matrix metalloproteinases (MMP), including MMP1, MMP7, MMP8, MMP10, MMP13, MMP14, and MMP15, which may be involved in the destruction of articular cartilage and may also contribute to MAYV-induced bone loss [51].
The CCL2-CCR2 axis is primarily responsible for the recruitment of monocytes/macrophages during alphavirus infection. The CCL2 blockade with Bindarit in animal models for CHIKV and RRV showed that this chemokine is a pathogenic factor in arthritis, since its absence was accompanied by a significant reduction in tissue damage [9, 42]. CCL2 levels are elevated in the serum of patients during the acute phase of the diseases and also in the target tissues in animal models for RRV, CHIKV, and MAYV [8••, 24•, 31••, 52, 53].
Advances in vaccines and treatment
To date, there are no vaccines available against MAYV. However, several studies have been reported on this subject. Weise et al. [12] developed a live-attenuated vaccine candidate employing a modified IRES region from the encephalomyocarditis virus (ECMV) in the MAYV genome. This strategy reduced translation of viral structural proteins and inhibited viral replication in a C6/36 Aedes albopictus cell culture. Chuong et al. [54] reported the construction and characterization of two infectious clones of two strains of MAYV. Both clones managed to infect and replicate in mammalian and mosquito cell cultures, a valid approach that can assist in the development of DNA vaccines. In addition, Choi et al. [14] have developed a novel DNA vaccine containing a synthetic full-length envelope gene sequence. This construction elicited a strong neutralizing immune response against E1 and E2 proteins in immunized mice and induced cellular response by activating CD4+ and CD8+ T cells. Also, it protected interferon α/β knockout mice from morbidity and mortality in a lethal MAYV challenge.
On the other hand, Khan et al. [13] applied immunoinformatics to propose a subunit vaccine model containing five structural protein epitopes against MAYV. In silico validation for protein cloning and expression in Escherichia coli was successful, but further experimental validation is necessary.
Concerning treatment options, only palliative measures are used against MAYV infection since there are no specific antiviral drugs. Several studies have been conducted in the past few years in the search for new therapeutic alternatives. Weak bases, such as ammonium chloride and chloroquine, have shown significant reduction in the formation of new MAYV particles in mammalian cell culture experiments, although further testing is needed [55].
The antiviral effect of seven thienopyridine derivatives has been demonstrated by Amorim et al. [56]. The compounds showed inhibitory capacity in a non-toxic concentration for Vero cell culture. The most active compound was able to inhibit precociously in vitro MAYV replication in pretreated Vero cells. Late stages of replication were also affected through interference in the formation of new viral particles. Molecular modeling predictions revealed low toxicity risk and good oral bioavailability for these potential antiviral candidates.
Additionally, Ferreira et al. [57] have identified a flavonoid compound, epicatechin, from Salacia crassifolia, as a potent antiviral candidate against a constructed three-dimensional model of MAYV C protein. Bioassays confirmed its antiviral action with a 50% effective concentration (EC50) of 0.247 μmol/mL and selectivity index of 7. Epicatechin displayed a potent inhibition effect on the MAYV cycle, reducing viral load.
Camini et al. [58] have demonstrated the anti-MAYV effect of silymarin, a natural complex extracted from Silybum marianum. Silymarin compromised viral replication and reduced cell damage from oxidative stress caused by the virus. Another therapeutic candidate has been proposed by Ferraz et al. [59]. Proanthocyanidin, a flavonoid isolated from Maytenus imbricate, showed a virucidal effect against MAYV-infected Vero cells with low cytotoxicity and high selectivity index, blocking viral adsorption and internalization.
Mayaro spread: local or global threat?
In recent years, some works have been published about the risk of a local and/or global epidemic of MAYV [11, 32, 60••]. To start an arboviral epidemic, the vertebrate vector and/or human host need be continuously exposed in a permissive environment [61], making these risks mainly related to host availability, vector genotypic improvement, and geographic distribution.
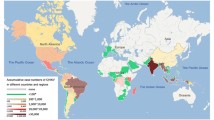
Arboviruses are considered to be emergent and re-emergent pathogens causing disease around the word [62]. Although some arboviruses, such as DENV, are restricted to tropical areas, in a few decades, this distribution may include Europe and the USA [60••] (Fig. 2). Originally isolated in Trinidad and Tobago more than 60 years ago from 5 symptomatic infected humans [11, 32], today, MAYV occupies a prominent place in Latin America, with a broad distribution, extending from Argentina (positive for Una virus, a subtype of Mayaro, in serological assay) to Mexico, more precisely in Trinidad and Tobago, French Guiana, Brazil, Ecuador, Venezuela, Haiti, Bolivia, Surinam, Costa Rica, Guatemala, Panama, Peru, and sporadically in Colombia (autochthonous, isolated from mosquitoes). Besides this local incidence, several cases have been reported in European countries (France, Netherlands, Germany, and Switzerland) and in the USA. All these cases were imported from Latin America [32, 63]. In times of asymptomatic and highly contagious viral diseases [64], arboviral-like illness has a great advantage in terms of control, i.e., the need of a susceptible arthropod vector to transmit between humans [65]. This requirement normally restricts arboviral infections to regional incidence, corresponding to wild host and vector distribution [61]. However, this virus can use diverse arthropod vectors, and these vectors can change their original distribution, which can increase the original viral spread [3, 66].
Mayaro virus spread. The square shows the main region with MAYV incidence. Several countries of South and Central America, more Mexico, are places with major MAYV cases described. Besides the entire tropical region (between the solid lines) being susceptible to viral spread, due to the presence of probable vectors, some cases have been reported in Argentina, which is in the subtropical region. The dotted line shows the area in which the DENV was hypothesized to spread (imported and presumptive cases). Considering that MAYV can be carried by the same vectors as DENV and Anopheles, this area can be the next focus of a MAYV epidemic.
Several mosquitoes of different genera are described as possible vectors for MAYV: Aedes aegypti, Hg. janthinomys, Mansonia venezuelensis, Culex quinquefasciatus, Ae. scapularis, Psorophora ferox, and Cx. vomerifer, and maybe, under some conditions, Aedes albopictus seems be a vector [3]. Anopheles freeborni, An. gambiae, An. quadrimaculatus, and An. stephensi showed a capacity for transmitting MAYV in laboratory experiments, and Ae. aegypti and An. stephensi present distinct infectivity and body dissemination capabilities, depending on the viral strain used. Although Cx. quinquefasciatus was proposed as a vector of MAYV cases during a DENV outbreak that occurred in the city of Cuiabá (Mato Grosso state, Brazil) in 2013, another research performed using a laboratory strain showed that Cx. quinquefasciatus was able to maintain the MAYV infection, but not to transmit it [4, 67]. The adaptability of some strains to different vectors and hosts, combined with the capability of Anopheles to drive an invasion and transmission of Mayaro virus across geographically diverse regions [4], represents an alert to public health services around the world regarding the possibility of major epidemic events.
To diversify its host range, a virus needs to meet some conditions, one of which is genotype change [61]. For CHIKV, it is known that one residue modification is enough to improve viral replication in a different vector than primarily described [68]. MAYV are differentiated into three genotypes, related to spatial and temporal aspects. These are the following: genotype D—dispersed; genotype L—limited to the north central region of Brazil; and the third and lesser variety, genotype N—a new, single-sequence genotype isolated from samples taken in Puerto Maldonado, Peru [32, 69]. Recently, a fourth genotype was isolated from the Amazon basin, which is a hybrid between the D and L genotypes, giving rise to a D/L genotype [70•].
Conclusions
Mayaro fever is a neglected arbovirosis that can result in serious public health issues for affected countries, particularly in the Americas, where co-circulation with other arboviruses can impair correct diagnosis and cause underestimation of infection rates. Despite the clinical course of MAYV infection and its inflammatory response showing similarities with other alphaviruses, further studies are needed to fully characterize the pathogenesis of MAYV and, consequently, to develop specific therapies and vaccines. Combining different serological tests can improve differential diagnosis for MAYV, since cross-reactivity is an important factor that must be considered in endemic areas. The potential emergence of MAYV indicates a constant demand for new therapeutic strategies. The possible diversity in arthropod vectors, which MAYV can use to spread, the change in the geographic area of the incidence of the known hosts, and the rise of new virus genotypes demonstrate the high potential for a new outbreak in traditional endemic areas in the short term or a pandemic with global reach in the long term.
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Suhrbier A, Jaffar-Bandjee MC, Gasque P. Arthritogenic alphaviruses--an overview. Nat Rev Rheumatol. 2012;8(7):420–9.
Mota MT, et al. Complete genome sequence of Mayaro virus imported from the Amazon Basin to Sao Paulo State, Brazil. Genome Announc. 2015;3(6):e01341–15.
Mackay IM, Arden KE. Mayaro virus: a forest virus primed for a trip to the city? Microbes Infect. 2016;18(12):724–34.
Brustolin M, Pujhari S, Henderson CA, Rasgon JL. Anopheles mosquitoes may drive invasion and transmission of Mayaro virus across geographically diverse regions. PLoS Negl Trop Dis. 2018;12(11):e0006895.
Tappe D, Pérez-Girón JV, Just-Nübling G, Schuster G, Gómez-Medina S, Günther S, et al. Sustained elevated cytokine levels during recovery phase of Mayaro virus infection. Emerg Infect Dis. 2016;22(4):750–2.
Assuncao-Miranda I, Cruz-Oliveira C, Da Poian AT. Molecular mechanisms involved in the pathogenesis of alphavirus-induced arthritis. Biomed Res Int. 2013;2013:973516.
Mostafavi H, et al. Arthritogenic alphavirus-induced immunopathology and targeting host inflammation as a therapeutic strategy for alphaviral disease. Viruses. 2019;11(3):290.
•• Lidbury BA, et al. Macrophage-derived proinflammatory factors contribute to the development of arthritis and myositis after infection with an arthrogenic alphavirus. J Infect Dis. 2008;197(11):1585–93 This work demonstrated the role of macrophages and their mediators in alphaviral pathogenesis and guided further studies to understand how these factors work and in the development of therapies using these factors as targets. In addition, studies with different alphaviruses, including MAYV, have shown that these immune factors act similarly in the diseases induced by these alphaviruses.
Rulli NE, Guglielmotti A, Mangano G, Rolph MS, Apicella C, Zaid A, et al. Amelioration of alphavirus-induced arthritis and myositis in a mouse model by treatment with bindarit, an inhibitor of monocyte chemotactic proteins. Arthritis Rheum. 2009;60(8):2513–23.
Ng LFP. Immunopathology of Chikungunya virus infection: lessons learned from patients and animal models. Annu Rev Virol. 2017;4(1):413–27.
Esposito DLA, Fonseca B. Will Mayaro virus be responsible for the next outbreak of an arthropod-borne virus in Brazil? Braz J Infect Dis. 2017;21(5):540–4.
Weise WJ, Hermance ME, Forrester N, Adams AP, Langsjoen R, Gorchakov R, et al. A novel live-attenuated vaccine candidate for Mayaro fever. PLoS Negl Trop Dis. 2014;8(8):e2969.
Khan S, Khan A, Rehman AU, Ahmad I, Ullah S, Khan AA, et al. Immunoinformatics and structural vaccinology driven prediction of multi-epitope vaccine against Mayaro virus and validation through in-silico expression. Infect Genet Evol. 2019;73:390–400.
Choi H, Kudchodkar SB, Reuschel EL, Asija K, Borole P, Ho M, et al. Protective immunity by an engineered DNA vaccine for Mayaro virus. PLoS Negl Trop Dis. 2019;13(2):e0007042.
Mota MTO, et al. In-depth characterization of a novel live-attenuated Mayaro virus vaccine candidate using an immunocompetent mouse model of Mayaro disease. Sci Rep. 2020;10(1):5306.
Izurieta RO, DeLacure DA, Izurieta A, Hoare IA, Reina Ortiz M. Mayaro virus: the jungle flu. Virus Adaptation and Treatment. 2018;10:9–17.
Aguilar-Luis MA, del Valle-Mendoza J, Silva-Caso W, Gil-Ramirez T, Levy-Blitchtein S, Bazán-Mayra J, et al. An emerging public health threat: Mayaro virus increases its distribution in Peru. Int J Infect Dis. 2020;92:253–8.
Pinheiro FP, et al. An outbreak of Mayaro virus disease in Belterra, Brazil. I. Clinical and virological findings. Am J Trop Med Hyg. 1981;30(3):674–81.
Izurieta RO, Macaluso M, Watts DM, Tesh RB, Guerra B, Cruz LM, et al. Hunting in the rainforest and Mayaro virus infection: an emerging alphavirus in Ecuador. J Glob Infect Dis. 2011;3(4):317–23.
Azevedo RS, et al. Mayaro fever virus, Brazilian Amazon. Emerg Infect Dis. 2009;15(11):1830–2.
Mourao MP, et al. Mayaro fever in the city of Manaus, Brazil, 2007-2008. Vector Borne Zoonotic Dis. 2012;12(1):42–6.
Neumayr A, Gabriel M, Fritz J, Günther S, Hatz C, Schmidt-Chanasit J, et al. Mayaro virus infection in traveler returning from Amazon Basin, northern Peru. Emerg Infect Dis. 2012;18(4):695–6.
Torres JR, Russell KL, Vasquez C, Barrera R, Tesh RB, Salas R, et al. Family cluster of Mayaro fever, Venezuela. Emerg Infect Dis. 2004;10(7):1304–6.
• Santos FM, et al. Animal model of arthritis and myositis induced by the Mayaro virus. PLoS Negl Trop Dis. 2019;13(5):e0007375 This paper describes a novel animal model designed for studying arthritis and myositis induced by Mayaro virus. In this study, it was possible to observe disease development and the main immune mediators related to pathogenesis.
Mahendradas P, Ranganna SK, Shetty R, Balu R, Narayana KM, Babu RB, et al. Ocular manifestations associated with chikungunya. Ophthalmology. 2008;115(2):287–91.
Lalitha P, Rathinam S, Banushree K, Maheshkumar S, Vijayakumar R, Sathe P. Ocular involvement associated with an epidemic outbreak of chikungunya virus infection. Am J Ophthalmol. 2007;144(4):552–6.
Merle H, Donnio A, Jean-Charles A, Guyomarch J, Hage R, Najioullah F, et al. Ocular manifestations of emerging arboviruses: dengue fever, Chikungunya, Zika virus, West Nile virus, and yellow fever. J Fr Ophtalmol. 2018;41(6):e235–43.
• Halsey ES, et al. Mayaro virus infection, Amazon Basin region, Peru, 2010–2013. Emerg Infect Dis. 2013;19(11):1839–42 This work comprises a cohort study, in which patients infected by MAYV are followed for a long time. In this study, authors demonstrate that arthralgia can persist for a year and has a major impact on people’s life quality. It also shows the main affected joints.
Theilacker C, Held J, Allering L, Emmerich P, Schmidt-Chanasit J, Kern WV, et al. Prolonged polyarthralgia in a German traveller with Mayaro virus infection without inflammatory correlates. BMC Infect Dis. 2013;13:369.
Hassing RJ, et al. Cross-reactivity of antibodies to viruses belonging to the Semliki forest serocomplex. Euro Surveill. 2010;15(23):19588.
•• Santiago FW, et al. Long-term arthralgia after Mayaro virus infection correlates with sustained pro-inflammatory cytokine response. PLoS Negl Trop Dis. 2015;9(10):e0004104 This cohort presents a complete panel of immune mediators elicited during Mayaro fever as well as a comparison between groups with different post-infection states.
Acosta-Ampudia Y, Monsalve DM, Rodríguez Y, Pacheco Y, Anaya JM, Ramírez-Santana C. Mayaro: an emerging viral threat? Emerg Microbes Infect. 2018;7(1):163.
Figueiredo ML, Figueiredo LT. Emerging alphaviruses in the Americas: Chikungunya and Mayaro. Rev Soc Bras Med Trop. 2014;47(6):677–83.
Wang E, Paessler S, Aguilar PV, Carrara AS, Ni H, Greene IP, et al. Reverse transcription-PCR-enzyme-linked immunosorbent assay for rapid detection and differentiation of alphavirus infections. J Clin Microbiol. 2006;44(11):4000–8.
Fumagalli MJ, et al. Development of an enzyme-linked immunosorbent assay to detect antibodies targeting recombinant envelope protein 2 of Mayaro virus. J Clin Microbiol. 2019;57(5):e01892–18.
Fischer C, et al. Robustness of serologic investigations for Chikungunya and Mayaro viruses following coemergence. mSphere. 2020;5(1)e00915–19.
Morrison TE, Whitmore AC, Shabman RS, Lidbury BA, Mahalingam S, Heise MT. Characterization of Ross River virus tropism and virus-induced inflammation in a mouse model of viral arthritis and myositis. J Virol. 2006;80(2):737–49.
Gardner J, Anraku I, le TT, Larcher T, Major L, Roques P, et al. Chikungunya virus arthritis in adult wild-type mice. J Virol. 2010;84(16):8021–32.
Soden M, Vasudevan H, Roberts B, Coelen R, Hamlin G, Vasudevan S, et al. Detection of viral ribonucleic acid and histologic analysis of inflamed synovium in Ross River virus infection. Arthritis Rheum. 2000;43(2):365–9.
Ozden S, Huerre M, Riviere JP, Coffey LL, Afonso PV, Mouly V, et al. Human muscle satellite cells as targets of Chikungunya virus infection. PLoS One. 2007;2(6):e527.
Wauquier N, Becquart P, Nkoghe D, Padilla C, Ndjoyi-Mbiguino A, Leroy EM. The acute phase of Chikungunya virus infection in humans is associated with strong innate immunity and T CD8 cell activation. J Infect Dis. 2011;204(1):115–23.
Rulli NE, Rolph MS, Srikiatkhachorn A, Anantapreecha S, Guglielmotti A, Mahalingam S. Protection from arthritis and myositis in a mouse model of acute chikungunya virus disease by bindarit, an inhibitor of monocyte chemotactic protein-1 synthesis. J Infect Dis. 2011;204(7):1026–30.
Kumar S, Jaffar-Bandjee MC, Giry C, Connen de Kerillis L, Merits A, Gasque P, et al. Mouse macrophage innate immune response to Chikungunya virus infection. Virol J. 2012;9:313.
Cavalheiro MG, et al. Macrophages as target cells for Mayaro virus infection: involvement of reactive oxygen species in the inflammatory response during virus replication. An Acad Bras Cienc. 2016;88(3):1485–99.
Teo TH, Lum FM, Claser C, Lulla V, Lulla A, Merits A, et al. A pathogenic role for CD4+ T cells during Chikungunya virus infection in mice. J Immunol. 2013;190(1):259–69.
Burrack KS, Montgomery SA, Homann D, Morrison TE. CD8+ T cells control Ross River virus infection in musculoskeletal tissues of infected mice. J Immunol. 2015;194(2):678–89.
Figueiredo CM, Neris RLS, Gavino-Leopoldino D, da Silva MOL, Almeida JS, dos-Santos JS, et al. Mayaro virus replication restriction and induction of muscular inflammation in mice are dependent on age, type-I interferon response, and adaptive immunity. Front Microbiol. 2019;10:2246.
Noret M, et al. Interleukin 6, RANKL, and osteoprotegerin expression by chikungunya virus-infected human osteoblasts. J Infect Dis. 2012;206(3):455–7 457–9.
Chen W, Foo SS, Rulli NE, Taylor A, Sheng KC, Herrero LJ, et al. Arthritogenic alphaviral infection perturbs osteoblast function and triggers pathologic bone loss. Proc Natl Acad Sci U S A. 2014;111(16):6040–5.
Chen W, Foo SS, Taylor A, Lulla A, Merits A, Hueston L, et al. Bindarit, an inhibitor of monocyte chemotactic protein synthesis, protects against bone loss induced by chikungunya virus infection. J Virol. 2015;89(1):581–93.
Bengue M, et al. Mayaro virus infects human chondrocytes and induces the expression of arthritis-related genes associated with joint degradation. Viruses. 2019;11(9):797.
Kelvin AA, Banner D, Silvi G, Moro ML, Spataro N, Gaibani P, et al. Inflammatory cytokine expression is associated with chikungunya virus resolution and symptom severity. PLoS Negl Trop Dis. 2011;5(8):e1279.
Chaaitanya IK, et al. Role of proinflammatory cytokines and chemokines in chronic arthropathy in CHIKV infection. Viral Immunol. 2011;24(4):265–71.
Chuong C, Bates TA, Weger-Lucarelli J. Infectious cDNA clones of two strains of Mayaro virus for studies on viral pathogenesis and vaccine development. Virology. 2019;535:227–31.
Ferreira DF, et al. Weak bases affect late stages of Mayaro virus replication cycle in vertebrate cells. J Med Microbiol. 2000;49(4):313–8.
Amorim R, de Meneses MDF, Borges JC, da Silva Pinheiro LC, Caldas LA, Cirne-Santos CC, et al. Thieno[2,3-b]pyridine derivatives: a new class of antiviral drugs against Mayaro virus. Arch Virol. 2017;162(6):1577–87.
Ferreira PG, Ferraz AC, Figueiredo JE, Lima CF, Rodrigues VG, Taranto AG, et al. Detection of the antiviral activity of epicatechin isolated from Salacia crassifolia (Celastraceae) against Mayaro virus based on protein C homology modelling and virtual screening. Arch Virol. 2018;163(6):1567–76.
Camini FC, da Silva TF, da Silva Caetano CC, Almeida LT, Ferraz AC, Alves Vitoreti VM, et al. Antiviral activity of silymarin against Mayaro virus and protective effect in virus-induced oxidative stress. Antivir Res. 2018;158:8–12.
Ferraz AC, Moraes TFS, Nizer WSC, Santos M, Tótola AH, Ferreira JMS, et al. Virucidal activity of proanthocyanidin against Mayaro virus. Antivir Res. 2019;168:76–81.
•• Mota MTdO, Avilla CM, Nogueira ML. Mayaro virus: a neglected threat could cause the next worldwide viral epidemic. Future Virology. 2019;14(6):375–7 First paper to consider the possibility of a worldwide Mayaro virus epidemic.
Weaver SC, Reisen WK. Present and future arboviral threats. Antivir Res. 2010;85(2):328–45.
Gubler DJ. The global threat of emergent/re-emergent vector-borne diseases. In: Atkinson PW, editor. Vector biology, ecology and control. Dordrecht: Springer Netherlands; 2010. p. 39–62.
Ganjian N, Riviere-Cinnamond A. Mayaro virus in Latin America and the Caribbean. Rev Panam Salud Publica. 2020;44:e14.
Li R, et al. Substantial undocumented infection facilitates the rapid dissemination of novel coronavirus (SARS-CoV2). Science. 2020. p. 489–93.
Marchi S, Trombetta CM, Montomoli E. Emerging and re-emerging arboviral diseases as a global health problem. Public Health: Emerging and Re-emerging Issues. 2018:25.
Ali R, Mohammed A, Jayaraman J, Nandram N, Feng RS, Lezcano RD, et al. Changing patterns in the distribution of the Mayaro virus vector Haemagogus species in Trinidad, West Indies. Acta Trop. 2019;199:105108.
Serra OP, Cardoso BF, Ribeiro ALM, Santos FAL, Slhessarenko RD. Mayaro virus and dengue virus 1 and 4 natural infection in culicids from Cuiaba, state of Mato Grosso, Brazil. Mem Inst Oswaldo Cruz. 2016;111(1):20–9.
Tsetsarkin KA, Vanlandingham DL, McGee CE, Higgs S. A single mutation in chikungunya virus affects vector specificity and epidemic potential. PLoS Pathog. 2007;3(12):e201.
Auguste AJ, Liria J, Forrester NL, Giambalvo D, Moncada M, Long KC, et al. Evolutionary and ecological characterization of Mayaro virus strains isolated during an outbreak, Venezuela, 2010. Emerg Infect Dis. 2015;21(10):1742–50.
• Mavian C, et al. Emergence of recombinant Mayaro virus strains from the Amazon basin. Sci Rep. 2017;7(1):8718 Reports of a hybrid genotype in Amazon basin.
Acknowledgments
CAPES, FAPEMIG, and CNPq for scholarship support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Franciele Martins Santos declares that there is no conflict of interest. Roberto Sousa Dias declares that there is no conflict of interest. Luciana de Souza Fernandes declares that there is no conflict of interest. Cynthia Canedo da Silva declares that there is no conflict of interest. Sérgio Oliveira de Paula declares that there is no conflict of interest.
Human/animal study statement
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Viral Infections
Rights and permissions
About this article
Cite this article
Santos, F.M., Dias, R.S., de Souza Fernandes, L. et al. Mayaro Virus Infection: Clinical Features and Global Threat. Curr Treat Options Infect Dis 12, 387–397 (2020). https://doi.org/10.1007/s40506-020-00240-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40506-020-00240-7