Opinion statement
Dengue fever is a viral disease with no curative treatment. However, symptomatic treatment exists to alleviate the fever and provide analgesia to the multiple types of pain mainly headache, arthralgia, myalgia, and retro-ocular pain. Antiviral drugs to interfere with the process of viral replication are still under investigation. In the past, homeopathic treatment has been tried as well, with no success. In the case of severe dengue, hospitalization with strict monitoring of vital signs, parenteral liquids, and electrolytes replacement and, if needed, blood transfusion, are the most adequate courses of management. Most of the scientific work on the topic is dedicated to the development of a vaccine that may prevent the infection by any of the four serotypes of dengue, a tetravalent vaccine. Different research groups are searching for vaccine candidates among the different stages of viral development. One such group has completed a phase III trial and their vaccine has been licensed in different countries. However, more work is required to enhance this vaccine effectiveness beyond the current 60% protection. Moreover, new vaccine candidates are needed to provide safe immunization against the four dengue serotypes, whether preventing infection or reinfections.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Dengue fever is the most important arthropod-borne viral infection worldwide. Currently, three billion people live at risk of infection, of which 390 million contract it every year [1]. This review provides a summary of dengue’s transmission cycle, the disease’s clinical significance, and treatment as well as an update of the advances in the fight against the infection, specifically vaccine development.
Biology and transmission
Dengue’s causal agent is a member of the Flaviviridae family, dengue virus (DENV), which includes four serotypes, DENV 1–4, and according to recent findings, a fifth serotype as well which is under further research [2,3,4]. DENV shares characteristics with other Flavivirus such as Yellow Fever and Zika virus. An important common feature is the transmission by mosquitoes of the genus Aedes, primarily Ae aegypti with Ae. albopictus playing a role in transmission to a lesser extent in some geographic areas.
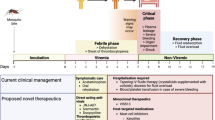
Humans are the only natural host for DENV. In them, the incubation period (called intrinsic incubation period) varies from 3 to14 days; it entails [2, 3, 5] the period from inoculation of the virus during the mosquito bite to the beginning of clinical manifestations. In the vector, the incubation period (also called extrinsic incubation period) ranges from 8 to 12 days. This period extends since the female mosquito takes a blood meal containing the virus until the viral replication is completed, and the pathogen can be transmitted during another blood meal from the vector to a susceptible human host [2, 5, 6]. The vector is an urban insect with a preference for human blood (anthropophilic). Its preferred habitat is in urban and per urban areas where it may breed in and around human dwellings, taking advantage of any man-made receptacle able to collect clean water (especially discarded plastic bottles and similar containers as well as discarded tires). In developing countries located in tropical and subtropical regions, water shortage is common. This forces the population to store water in cement tanks, cisterns, drums, etc.), which, if left without maintenance become ideal breeding sites for the vector [7]. Chemical and biological vector control methods with larvicide, ovicide, and adulticide effects are available. [8,9,10,11,12,13,14].
Clinical presentation and treatment
Dengue has a broad range of clinical manifestations going from an asymptomatic infection in around 85% of infected hosts and only 15% will develop clinical symptoms. In 5% of dengue infections, there will be clinical symptomatology classified as severe [1, 2]. Asymptomatic carriers play a role of monumental importance in the epidemiology of dengue as they maintain the transmission cycle infecting new mosquito cohorts who will continue infecting new susceptible human hosts [2].
Patients with clinical dengue are classified according to the recent WHO classification in dengue without warning signs, dengue with warning signs, and severe dengue; it is particularly important to remark that warning signs such as abdominal pain, vomiting, clinical fluid accumulation, mucosal bleed, lethargy, restlessness, liver enlargement >2 cm, and an increase in hematocrit concentration while there is a rapid decrease in the platelet count are a guide to decide the hospitalization of patients and moving from a symptomatic treatment to a more monitored management which includes fluids replacement as a key component foreseeing the passage of the clinical condition to a severe dengue which will be characterized by one or more of the following changes: severe plasma leakage, severe hemorrhages, and severe organ impairment (heart, liver, and central nervous system) [1].
It is important to mention that genetic studies show a differential risk for developing dengue’s clinical manifestations, in particular, for severe dengue fever. These human genetic factors can be relevant in the development of vaccines and in the selection of antiviral treatments [15].
Current treatments
Symptomatic or palliative treatment
Dengue is a febrile illness without a curative treatment, but analgesics and antipyretics have been used to alleviate symptoms (control fever and pain relief, especially joint pain, myalgia, ocular pain, and headache).
Currently acetaminophen (paracetamol) is the drug of choice to reduce symptoms. It should be known that doses should not exceed 1 g per dose and a maximum of 4 g per day. In adults, the administration route is oral (available in tablets and suspensions). Patients with hepatic disease or with history of chronic alcohol consumptions should be careful since acetaminophen can be hepatotoxic [1].
Acetyl salicylic acid (Aspirin) is a drug with analgesic and antipyretic properties but is contraindicated for dengue because it can precipitate hemorrhagic manifestations and gastrointestinal bleeding due to its inhibition of platelets adhesion [1, 16, 17].
Severe dengue cases with hemorrhagic and extravasation manifestations need hospitalization with close monitoring of vital signs, liquids, and electrolytes input and outputs [18] [Administration of intravenous fluids to any patient vomiting or with a high or rapidly rising hematocrit, in the case of severe dengue, is recommended the use of isotonic intravenous fluids like 0.9 saline, or Ringer’s lactate [1]. Once the hemodynamic status is stable, fluid therapy must be gradually reduced, replaced with oral liquids, and eventually suspended.
The survival rate in the absence of hospitalization can vary from 20 to a high of 50% according to different authors. With proper treatment, the mortality reduces to less than 3%. [1, 19].
Antiviral treatment
Several studies have explored the possibility to use antiviral treatment to interfere with the disease process caused by the dengue virus.
Two main treatment groups have been identified:
-
i.
Direct acting antivirals with mechanism of action on the dengue virus itself,
-
ii.
Host modulators whose mechanism of action are based on the fact that DENV relies on several host factors for replications [20].
A third group of treatment includes natural products (e.g., medicinal plants) that are used traditionally but require further studies to analyze their mechanism of action [21••].
Most drug studies have been done in animal models, and few molecules have been evaluated in dengue virus-infected patients: Some of the most important are balapiravir (a direct acting antiviral) and some under study such as Adefovir dipivoxil (direct acting antiviral) that are going through in vitro tests but without a firm evidence of a therapeutic effect on patients [22, 23]; some host modulators are chloroquine, lovastatin, prednisolone, and celgosivir [22].
The similarity of dengue with Hepatititis C virus, another member of the Flaviviridae family, makes very likely that antivirals in use in the latter specifically Celgosivir can have clinical effects on dengue disease. Clinical trials are ongoing and no concluding results are available yet. [22].
Products of the statin group (lovastatin) are actually under study to identify their role stabilizing the endothelial membrane where many of the pathogenic changes occur in dengue, showing no therapeutic effects at the used doses [24].
Natural products
Research teams have investigated the effects of different natural products originated in different plant families that have been used by different cultures using whole plants or part of them; most of these studies are in a stage of in vitro observation using different extracts (aqueous, ethanol, methanol flavonoid, polysaccharides, essential oils, and others); these trials are still in process of providing conclusive evidence. [25•] Several groups are currently researching the effects of papaya (Carica papaya) leaves infusions especially in the thrombocytopenia caused by the dengue infection as well as the observed stabilizing effect on leucocytes but not concluding results are available yet [26, 27•].
In recent years, homeopathic approaches have been tested through clinical trials with no success in Central America in the management of febrile symptoms and dengue disease duration; results were similar to the control group [28, 29].
Vaccine development and current status
There has been is an intense effort to develop a dengue vaccine in the last decades. The challenge has been to develop a tetravalent vaccine (preventive for infections caused by any of the four dengue serotypes). The development of a dengue vaccine had to follow any of the potential five types of vaccine design: Live attenuated, chimeric live attenuated, inactivated vaccine, subunit vaccine, or nucleic acid-based (see Table 1).
There are some vaccines already approved for public health trials but still with sub-optimum results [32••] The current effort to develop vaccines is having the positive effect to increase the understanding of the immune response and the role of CD4 cells in the process triggered by different serotypes but to get improvement in the vaccine protection we need to continue gathering a deeper understanding of this response [33].
A vaccine now licensed in several countries and developed by Sanofi Pasteur (CYD-TDV, named Dengvaxia) was able to protect, in the first 25 months of the two Phase III trial, 66% of a subset of 9–16 year old participants. However, a significantly lower efficacy (including negative vaccine efficacy) was noted for children younger than 9 years of age.
This vaccine corresponds to the live attenuated vaccine type and has been licensed in six countries being of more benefit to certain age groups (usually children >9 years and adults) in areas with more intense transmission and to individuals with previous infections. In areas with lower transmission can constitute a risk to predispose to subsequent infections and an increase in hospitalization of severe cases [34,35,36••, 37].
The development of the dengue vaccine has been accompanied by modeling of its impact in the population and its cost/effectiveness. In a recent study in the Philippines, it was identified that vaccinating children older than 9 years at a cost under $70 can still have a good impact in the protection in the population and being cost effective; similar analysis have been done in Southeast Asia and some South American countries [38,39,40, 41•].
Some other research teams are working in dengue vaccines which are in earlier stages of testing, and their results will take several years to be known; some of them are dengue vaccine candidates that contain truncated, recombinant, dengue virus envelope protein from all four dengue virus serotypes (DEN-80E) [42].
Different approaches are being used in some other vaccines, some are live attenuated chimeric dengue vaccines (such as DENVax and LAV Delta 30), DEN protein subunit V180 (deni-80E), and also DEN DNA vaccines. These vaccines are not yet in the Phase III stage and are pending to prove their clinical efficacy for dengue in humans [43].
The expectation is that in a 5-year period or longer, there will be access to different vaccine formulations. Even though new vaccines are planned to be developed and improved, once implemented, we would see an improvement in the control of dengue and a reduction in the number of cases; to fulfill this goal, innovative ideas such as mosquito-based immunization strategies should be considered reaching also a reduction in the vectorial capacity to transmit the virus [44, 45].
On the other hand, the development and implementation of vaccines should not be in exchange for the weakening of current programs and prevention of vector borne control and other control efforts for mosquito-borne control diseases like dengue. It is realistic to assume that dengue prevention through vaccines will require to be reinforced by field operations to keep low densities of the vector [46, 47].
One of the issues to be addressed is the differential level of protection provided by the vaccine depending on the serotype and the need of several years to get an increase in the protection of the general population going from 24 to 54% [45].
Referring again to Dengvaxia, it has been considered to include three doses, and it is use concomitantly (simultaneously) or sequentially after the license pentavalent vaccine booster, and recent studies with toddlers in Mexico has shown no issues on safety or immune interference which was a particular concern in the scheduling of the vaccine [48].
Reviews of the results of the studies of this licensed vaccine demonstrated better immunogenicity for DENV4 and worse for DENV1. The vaccine efficacy was studied in participants aged 2–16 years old with DENV4 with the best result and DENV2 with the worst (The best and worst immunogenicity results were for DENV4 and DENV1, respectively). Vaccine efficacy of 60% was derived from studies with participants aged 2–16 years old, with DENV4 and DENV2 presenting the best and worst results [49, 50].
Dengvaxia was first registered in Mexico in December, 2015, and it has a 0/6/12-month schedule in Phase III clinical studies. It has been registered for use in individuals 9–45 years of age living in endemic areas [49]. In other countries, the efficacy has been variable ranging from 31.3% (95% CI 1.3–51.9%) in Mexico to 79.0% (95% CI 52.3–91.5%) in Malaysia. This variability in efficacy likely reflects at least in part the baseline seropositivity and circulating serotypes, both of which affect the performance of the vaccine. [51••, 52].
During the following years, different research teams constituted by health institutions and those of pharmaceutical companies will continue updating their results in the clinical trials leading to the marketing of successful new vaccines.
Summary and conclusions
Dengue is a disease that because of its vector-borne transmission requires to be tackled initially through control of breeding sites containing developmental stages of Aedes aegypti mosquito; thus breaking the chain of transmission. Since continued transmission is favored by a lack of control in the peridomestic surroundings, individuals, families, and communities must work together to contain transmission.
Once the disease is manifested, it has to be treated symptomatically and monitored closely.
Antiviral therapies are not available at this time. Natural medicine has been utilized by populations without evidence, but there is increasing interest to study some products with scientific rigor. The commercial availability of a dengue vaccine is real. It is important to notice that the most advanced dengue vaccine (Dengvaxia) is recommended by most researchers for areas with hyper-endemicity and to reduce severe cases and hospitalization, and the level of efficacy is still under 70% but as a measure of public health, such vaccine can indeed reduce the burden of disease. New developments are expected to improve the protection of patients.
In a holistic perspective, we need to continue a triple approach to dengue: prevention through mosquito breeding sites control, preparedness for cases of severe dengue, and vaccine development; as it has been stressed by disease modelers, the protection of the populations will be enhanced through the combined approach to the epidemiological cycle.
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
World Health Organization (2009) Dengue: guidelines for diagnosis, treatment, prevention and control. New edition.
WHO. Dengue hemorrhagic fever. Diagnosis, treatment, prevention and control. Geneva: World Health Organization; 1997.
Guzman MG, Alvarez M, Halstead SB. Secondary infection as a risk factor for dengue hemorrhagic fever/dengue shock syndrome: an historical perspective and role of antibody-dependent enhancement of infection. Arch Virol. 2013;158:1445. doi:10.1007/s00705-013-1645-3.
Mustafa MS, Rasotgi V, Jain S, Gupta V. Discovery of fifth serotype of dengue virus (DENV-5): a new public health dilemma in dengue control. Med J Armed Forces India. 2015;71:67–70.
Chan M, Johansson MA. The incubation periods of dengue viruses. PLoS One. 2012;7(11):e50972. doi:10.1371/journal.pone.0050972.
Gubler DJ. Dengue viruses. In: BWJ M, Van Regenmortel MHV, editors. Desk encyclopedia of human and medical virology. Boston: Academic Press; 2010. p. 372–82. ISBN 0-12-375147-0.
Sherman C, Fernández EA, Chan AS, Lozano RC, Leontsini E, Winch PJ. La Untadita: a procedure for maintaining washbasins and drums free of Aedes aegypti based on modification of existing practices. Am J Trop Med Hyg. 1998;58(2):257–62.
Kraemer MUG, Sinka ME, Duda KA, et al. The global compendium of Aedes aegypti and Ae. albopictus occurrence. Scientific Data. 2015;2:150035. doi:10.1038/sdata.2015.35.
Marten GG, Borjas G, Cush M, Fernández EA, Reid JW. Control of larval Aedes aegypti (Diptera: Culicidate) by ciclopoid copepods in peridomestic breeding containers. J Medical Entomology. 1994;31(1):36–44.
Sherman C, Fernandez EA, Chan AST, Lozano RC, Leontsini E, Winch Pl. La Untadita: a procedure for maintaining washbasins and drums free of Aedes aegypti based on modification of existing practices. Am J Tropical Med Hygiene. 1998;58(2):257–62.
Mackay AJ, Amador M, Felix G, Acevedo V, Barrera R. Evaluation of household bleach as an ovicide for the control of Aedes aegypti. J Am Mosq Control Assoc. 2015;31(1):77–84.
Fernández E, Martínez M, Sherman C. Social mobilization for dengue control in Honduras. Dengue Bulletin. 2004;28(Supplement):30–4.
Tapia-Conyer R, Méndez-Galván J, Burciaga-Zúñiga P. Community participation in the prevention and control of dengue: the patio limpio strategy in Mexico. Paediatrics and international child health. 2012;32(sup1):10–3.
Quintero J, García-Betancourt T, Cortés S, García D, Alcalá L, González-Uribe C, et al. Effectiveness and feasibility of long-lasting insecticide-treated curtains and water container covers for dengue vector control in Colombia: a cluster randomised trial. Trans R Soc Trop Med Hyg. 2015;109(2):116–25.
Loeb M. Genetic susceptibility to West Nile virus and dengue. Publ Health Genom. 2013;16:4–8.
Valerio L, de Balanzo X, Jimenez O, et al. Haemorrhagic exanthema due to dengue virus induced by acetylsalicylic acid. An Sist Sanit Navar. 2006;29:439–42.
Ramirez-Ronda CH, Garcia CD. Dengue in the western hemisphere. Infect Dis Clin N Am. 1994 Mar;8(1):107–28. Review
Harris E, et al. Fluid intake and decreased risk for hospitalization for dengue fever, Nicaragua. Emerg Infect Dis. 2003;9:1003–6.
Prachee KD, Patil ST, Pawar SP. A review on dengue. Int J Pharmaceut Biol Arch. 2013;4(6):1075–85.
Krishnan MN, Garcia-Blanco MA. Targeting host factors to treat West Nile and dengue viral infections. Viruses. 2014;6(2):683–708. doi:10.3390/v6020683.
•• Fusco DN, Chung RT. Review of current dengue treatment and therapeutics in development. J Bioanal Biomed. 2014;1(8):1. This is a very complete and comprehensive review of the antiviral therapy for dengue and the current status of the research on their use fesibility
Kaptein SJ, Neyts J. Towards antiviral therapies for treating dengue virus infections. Curr Opin Pharmacol. 2016 Oct 31;30:1–7.
Sinha SN, Kar PK, Perugu S, RamaKrishna UV, Thakur CP. Adefovir dipivoxil—a possible regimen for the treatment of dengue virus (DENV) infection. Chemom Intell Lab Syst. 2016;155:120–7.
Whitehorn J, Nguyen CV, Khanh LP, et al.: Lovastatin for the treatment of adult patients with dengue: a randomized, double-blind, placebo-controlled trial. Clin Infect Dis. 2015; pii: civ949.
• Kadir SL, Yaakob H, Zulkijflj RM. Potential antidengue medicinal plants: a review. J Nat Med. 2013;67(4):677–89. A good inventory of candidates for antidengue medicinal plants
Charan J, Saxena D, Goyal JP, Yasobant S. Efficacy and safety of Carica papaya leaf extract in the dengue: a systematic review and meta-analysis. Int J Appl Basic Med Res. 2016 Oct;6(4):249.
• Chinnappan S, Shettikothanuru Ramachandrappa V, Tamilarasu K, Krishnan UM, Balakrishna Pillai AK, Rajendiran S. Inhibition of platelet aggregation by the leaf extract of Carica papaya during dengue infection: an in vitro study. Viral Immunol. 2016;29(3):164–8. It is an important paper because it goes beyond what is known about a natural product and explores its mechanisms of action to reduce dengue symptomatology
Jacobs J, Fernandez EA, Merizalde B, Avila-Montes GA, Crothers D. The use of homeopathic combination remedy for dengue fever symptoms: a pilot RCT in Honduras. Homeopathy. 2007 Jan 31;96(1):22–6.
Martinez EZ, Nunes AA. A homeopatia na prevenção e tratamento da dengue: uma revisão. Cad saúde colet,(Rio J). 2014;22(4):321–8.
Whitehead SS, Blaney JE, Durbin AP, Murphy BR. Prospects for a dengue virus vaccine. Nat Rev Microbiol. 2007;5(7):518–28.
Centers for Diseases Control and Prevention. Understanding how vaccines work. 2013 http://www.cdc.gov/vaccinesFrom CDC.
•• Villar L, Dayan GH, Arredondo-García JL, Rivera DM, Cunha R, Deseda C, et al. Efficacy of a tetravalent dengue vaccine in children in Latin America. N Engl J Med. 2015;372(2):113–23. This paper provides an insight in the process of testing the efficacy of the most advanced dengue vaccine—Dengvaxia—and the results obtained in Latin America
Angelo MA, Grifoni A, O'Rourke PH, Sidney J, Paul S, Peters B, et al. Human CD4+ T cell responses to an attenuated tetravalent dengue vaccine parallel those induced by natural infection in magnitude, HLA restriction, and antigen specificity. J Virol. 2017;91(5):e02147–16.
Ferguson NM, Rodríguez-Barraquer I, Dorigatti I, Mier-y-Teran-Romero L, Laydon DJ, Cummings DA. Benefits and risks of the Sanofi-Pasteur dengue vaccine: modeling optimal deployment. Science. 2016;353(6303):1033–6.
Flasche S, Jit M, Rodríguez-Barraquer I, Coudeville L, Recker M, Koelle K, et al. Comparative modelling of dengue vaccine public health impact (CMDVI). Geneva: World Health Organization; 2016.
•• Aguiar M, Stollenwerk N, Halstead SB. The risks behind Dengvaxia recommendation. Lancet Infect Dis. 2016;16(8):882. A critical analysis of the strengths and shortcomings of the vaccine Dengvaccia
Deen J. The dengue vaccine dilemma: balancing the individual and population risks and benefits. PLoS Med. 2016 Nov 29;13(11):e1002182.
Shim E. Dengue dynamics and vaccine cost-effectiveness analysis in the Philippines. Am j trop med hyg. 2016;95(5):1137–47.
González Morales NL, Núñez-López M, Ramos-Castañeda J, Velasco-Hernández JX. Transmission dynamics of two dengue serotypes with vaccination scenarios. Math Biosci. 2017;287:54–71. doi:10.1016/j.mbs.2016.10.001.Epub2016Oct 20.
Harapan H, Anwar S, Setiawan AM, Sasmono RT. Dengue vaccine acceptance and associated factors in Indonesia: a community-based cross-sectional survey in Aceh. Vaccine. 2016 Jul 12;34(32):3670–5.
• Godói IP, Santos AS, Reis EA, Lemos LL, Brandão CM, Alvares J, et al. Consumer willingness to pay for dengue vaccine CYD-TDV, Dengvaccia ® in Brazil; implications for future pricing considerations. Front Pharmacol. 2017;8:41. doi:10.3389/fphar.2017.00041.eCollection 2017. The issue of accessibility to the vaccine is analized in this paper in terms of costs and prices of Dengvaxia in Brazil
Swaminathan G, Thoryk EA, Cox KS, Smith JS, Wolf JJ, Gindy ME, et al. A tetravalent sub-unit dengue vaccine formulated with ionizable cationic lipid nanoparticle induces significant immune responses in rodents and non-human primates. Sci Rep. 2016;6
Torresi J, Ebert G, Pellegrini M. Vaccines licensed and in clinical trial for the prevention of dengue. Hum Vaccin Immunother. 2017;14:1–14. doi:10.1080/21645515.2016.1261770.
Liu Y, Liu J, Cheng G. Vaccines and immunization strategies for dengue prevention. Emerging Microbes & Infections. 2016;5(7):e77.
Coudeville L, Baurin N, L’Azou M, Guy B. Potential impact of dengue vaccination: insights from two large-scale phase III trials with a tetravalent dengue vaccine. Vaccine. 2016;34(50):6426–35.
López-Gatell H, Alpuche-Aranda CM, Santos-Preciadob JI, Hernández-Ávila M. Dengue vaccine: local decisions, global consequences. Bull World Health Organ. 2016 Nov 1;94(11):850.
World Health Organization. Global strategy for dengue prevention and control 2012–2020.
Scott LJ. Tetravalent dengue vaccine: a review in the prevention of dengue disease. Drugs. 2016 Sep 1;76(13):1301–12.
Shim E. Cost-effectiveness of dengue vaccination in Yucatán, Mexico using a dynamic dengue transmission model. Hozbor DF, ed. PLoS One. 2017; 12(4):e0175020. doi:10.1371/journal.pone.0175020.
Bentsi-Enchill AD, Schmitz J, Edelman R, Durbin A, Roehrig JT, Smith PG, et al. Live dengue vaccines technical consultation reporting group. Long-term safety assessment of live attenuated tetravalent dengue vaccines: deliberations from a WHO technical consultation. Vaccine. 2013;31(23):2603–9.
•• World Health Organization. Dengue vaccine: WHO position paper—July 2016. Wkly Epidemiol Rec. 2016;30:349–64. This document is important because it provides information about the implications of introducing the vaccine to the countries and discuss aspects of health policies at the level of national governments and World Health Organization that will influence the adoption of the Dengue vaccine
Flasche S, Jit M, Rodríguez-Barraquer I, Coudeville L, Recker M, Koelle K, et al. The long-term safety, public health impact, and cost-effectiveness of routine vaccination with a recombinant, live-attenuated dengue vaccine (Dengvaxia): a model comparison study. PLoS Med. 2016;13(11):e1002181.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Eduardo A. Fernandez declares that he has no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Neglected Tropical Diseases
Rights and permissions
About this article
Cite this article
Fernandez, E.A. Moving to a Dengue Preventive Treatment Through New Vaccines. Curr Treat Options Infect Dis 9, 347–355 (2017). https://doi.org/10.1007/s40506-017-0132-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40506-017-0132-x