Abstract
Grain legumes including common bean are mostly grown in rainfed production systems and invariably suffer from water stress at critical growth stages leading to significant yield reductions. Drought is one of the most importantstress limiting common bean productivity. The yield has been the primary breeding objective and has been improved by either targeting yield per se as well as yield components. However, yield is a highly complex trait with low heritability and screening for drought resilience based on yieldand/or secondary traits is increasingly appreciated as better approach. Drought tolerance breeding is relatively complex on account of quantitative inheritance and greater environmental influence therefore, it is imperative to identify less complex traits related to the drought that will improve upon selection for drought tolerance and separate these traits into major components that may help further understanding of the genetic basis. In most of the grain legumes breeders have concentrated on above-ground components. However, increasing experimental evidences have reinforced the premise that the hidden half of the plant including its structural and functional attributes and associated microbiome can be an effective tool for improving stress resilience. However, an improved understanding of the relationship between below and above-ground traits will contribute to improved productivity under drought stress. Among the above ground traits that have emerged as most effective surrogates are canopy temperature depression, leaf water status, stay green trait, phostosynthetic efficiency, membrane stability, limited transpiration and reproductive success and have contributed to our mechanistic understanding of plant response to stress. This paper discusses some of the aspects of root architecture and physiological traits to highlight mechanistic and applied perspectives from a plant breeding standpoint in common bean.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The world population is projected to be around 9.8 billion toward the year 2050, to feed them the global agriculture production had to be doubled. Conversely, the natural resources especially land and water are fast shrinking and degrading. Historical observations and model simulations suggested a high risk of drought across the globe (Dai 2013; Mishra and Liu 2014; Mallya et al. 2016). Out of the various types of drought, meteorological and agricultural droughts that occur due to the reduced precipitation and soil moisture respectively are important in the Indian perspective (Prakash 2018). Drought in India occurring on account of the failure of summer monsoon has led to substantial loss of food crops, and future prediction of rainfall indicate a high chance of below-normal rainfall or drought on the all-India scale (Kulkarni et al. 2016). Under predicted climate change scenario, water constraints are expected to increase. However, greater research was focused on identification and development of drought-tolerant cereals, however there is a lack of improvement of drought tolerance of grain legumes except for chickpea, soybean and pigeon pea (Serraj et al. 2004a, b; Ganeshan et al. 2012).
Grain legumes play a significant role in the traditional diets of people across the world and grown for its immature and dry seeds, as it contains high protein, oil, and micronutrients concentration (Broughton et al. 2003; Vaz Patto et al. 2015). The protein sourced from grain legumes often costs one-fifth as much as protein from milk. Hence it is named as “poor people’s meat.” Apart from this, it also contributes to the sustainable improvement of the environment by its biological nitrogen fixation process and weeds suppression ability (Rubiales and Mikic 2015). The important grain legumes grown in India are chickpea (Cicer arietinum L.), pigeon pea (Cajanus cajan), black gram [Vigna mungo (L.) R. Wilczek], common bean (Phaseolus vulgaris L.), green gram [Vigna radiata (L.) R. Wilczek], and soybean [Glycine max (L.) Merr.]. Grain legumes are mostly grown in rainfed production systems, and estimate shows that crop yields would decline from 30 to 82% under climate change by the end of twenty-first century, despite the beneficial effects of increased levels of carbon dioxide (Hatfield et al. 2011). In specific, failure of summer monsoon has led to substantial yield loss in pulses (42% for pigeon pea, 71% for green gram, and 74% for black gram) in India (Kulkarni et al. 2016). Plant responses to drought stress are complicated due to variation in timing of occurrence, duration, and intensity (Rosenzweig and Colls 2015), and the impact of the droughtis aggravated under semi-arid tropical conditions because of erratic and unpredictable rainfall, and occurrence of high temperatures during various crop phenophases, and poor soil characteristics.
This review focuses on common bean, in the specific, improvement of drought tolerance through root traits and physiological approaches. Globally, ~ 18 million metric tons of common beans are produced annually, and India is the second largest producer followed by Brazil and the United States (FAOSTAT 2016). Common bean is cultivated across five continents and spans from 52°N to 32°S latitude, and between near sea level and 3000 m above mean sea level (Lioi and Piergiovanni 2013). Common bean performs best in moderate growing temperatures (> 10 and < 30 °C) with about 400 mm of precipitation during growing season. The term bean is widely used to represent different species of large-seeded legumes including dry, kidney, French, navy, pinto, garden, snap, green, wax and string bean, and these names refers to specific types or uses of common bean. In another way, common beans can be classified into dry edible beans (mature seeds are used for consumption) and garden beans (immature pods are used as a vegetable). The production of common bean is constrained by various environmental stresses (Vyas 2014), of which drought (water deficit) is one of the most important stress limiting common bean productivity (Fang et al. 2010). About 60% of the common bean growing areas in the world face drought stress during its crop phenophases (Wortmann et al. 1998). However, this estimation might increase due to climate change (Chaves et al. 2003). Tolerance to drought is a result of various morphological, physiological and phenological processes that vary with each target environment (Chaves et al. 2003). Due to high variability in drought stress, it is difficult in characterizing the traits associated with drought tolerance, leading to less improvement of drought stress breeding.
Definition of the problem
Water is becoming a scarce resource. To adapt to this situation harnessing the genetic resources and exploitation of natural variation for drought associated traits in crops may be considered as a feasible solution. The yield has been the primary breeding objective and has been improved either by targeting yield per se as well as yield components. However, yield being a highly complex trait with low heritability, screening for drought resilience based on yield and secondary traits will be a better approach (Monneveux et al. 2008). Therefore, it is critical to identify high throughput and reliable indirect selection criteria for large-scale screening of germplasm. Both below (root) and above ground (shoot) plant parts are associated with drought stress tolerance, the various shoot, androot-related traits are presented below.
Breeding for drought tolerance
Common bean is invariably not noted for its tolerance to drought. Recent reports suggest that only 7% of the bean area receives adequate rainfall and 60% of the area is under severe water stress (White and Singh 1991; Rao 2014). Drought is a major production constraint of common bean and has been reported to cause yield reduction ranging from 60 to 100% (Porch and Jahn 2001; Beebe et al. 2008; Polania et al. 2016). Moisture stress, especially, during flowering and grain filling periods reduces seed yield through decreased seed weight and accelerated maturity of common bean (Singh 1995). In common bean, Khalid (2017) reported a decline of more than 30% in seed yield, due to a reduction in pods per plant (31%) on account of flower abortion, as well as pod length (17%), and seeds per pod (14.5%) under drought stress. Singh (2007) reported as high as 88% reduction in bean yields across different cultivars and stress severities. In Latin America and Africa, 73% and 40% of area respectively experiences moderate to severe drought stress (van Schoonhoven and Voysest 1989), while as in Eastern and Southern Africa, about 75% reduction in common bean yields under drought have been reported (Katungi et al. 2010). Recently, Smith et al (2019) reported significant decline in common bean yield and other parameters such as harvest index, plant biomass, leaf area and root exploration, with a slight increase in rooting depth.
Development of drought tolerant cultivars is relatively difficult, on account of quantitative inheritance and significant effect of environmental (Mir et al. 2012). Studies on genetic basis of drought tolerance in common bean have revealed that both additive and non-additive gene action was involved in drought tolerance (Makunde et al. 2007; Asadi et al. 2010; Blair et al. 2010; Amongi et al. 2014). A substantial number of QTLs has been reported for various drought related traits, pertaining to roots, shoot, physiology, phenology, partitioning and yield (Table 1). Schneider et al. (1997) reported strong genotype by environment interaction that affected the QTL expression, and as such their potential for use in marker-assisted selection for drought tolerance was inconclusive. Selection based on relative changes in yield under stress and non-stress conditions suffers from confounding effects that increases the complexity of breeding. Therefore, it is imperative to identify less complex underlying traits related to the drought that will improve efficiency of selection for drought tolerance and separate these traits into major components which may help further understanding of the genetic basis.
Why roots: harnessing the variation for resource acquisition
Despite the substantial experimental support for importance of root traits in drought tolerance across all crops (Hund et al. 2009; Vadez et al. 2011; Zaman-Allah et al. 2011; Bishopp and Lynch 2015; Sofi et al. 2017; Khalid 2017), comparatively, less studies has been done in legumes on root traits in breeding for drought-tolerant varieties. In fact, most of the stress breeding programs in grain legumes have concentrated on above-ground traits, on account of ease of measurement and have primarily ignored root traits, because of the difficulties associated with root recovery and evaluating root traits in situ (Marshall et al. 2016). However, during last three decades, a renewed interest in identifying and evaluating root system traits related to improved adaptation to water-limited environments has been witnessed (Palta et al. 2011). The potential target traits that can be used as effective surrogates include increased root biomass distribution at depth to improve deep water capture (Manske and Vlek 2002; Teran et al. 2019), rooting depth and faster root elongation rates to extract water from deeper soil strata (Hurd 1974; O'Brien 1979), reduced vessel diameter of xylem to conserve soil water (Richards and Passioura 1989), seminal root angle for extracting water from full soil depth (Manschadi et al. 2006) and optimal ontogenic shift (dry matter partitioning to root and shoot that improves water capture across the soil strata (Reynolds et al. 2007). Substantial phenotypic plasticity of root traits under diverse physical and chemical properties of soil, and lack of reliable, high-throughput and cost-effective screening techniques render root studies highly challenging. As a result, information on genetic variability of root traits in most legumes including common bean is still far from adequate.
Root architecture refers to the spatial configuration of root systems that determines plant anchorage, ability to absorb water and nutrients, and inter- and intra-plant communication and competition. It is proposed that modification of root system architecture can be a potential alternative approach to increase the crop yield, leading to a second green revolution (Den Herder et al. 2010). Root system of plants confers upon them morphological, structural, and physiological plasticity to change in environment (Root plasticity). Therefore, integrating root architectural and physiological phenes will help in breeding drought-tolerant genotypes (Osmont et al. 2007; Kashiwagi et al. 2006; Lynch 2011). Breeding for better root traits in crops requires not only efficient and improved methods of screening but also in-depth knowledge of specific functions of roots namely water extraction and nutrients absorption from the soil. An extensive root system confers drought avoidance as the enhanced root length and biomass density per se could increase the water extraction from soil resulting in increased yield under drought stress (Turner et al. 2001; Kashiwagi et al. 2006). Common bean is invariably grown under rainfed condition with deficit soil moisture especially in the post-rainy season (Kumar and Abbo 2001). The rainfed environment in India is characterized with varying intensities and distribution of rainfall during crop season from almost dry spell to > 400 mm precipitation (Birthal et al. 2014; Purushothaman et al. 2016). As such, terminal drought of varying intensities is a major constraint to grain legumes productivity in India.
Large number of studies has shown that roots play an important role under drought stress, yet many studies have indicated that shoot traits are the most important under drought stress (Price et al. 2002). This dichotomy of opinion arises because roots are involved in water uptake, and the absorbed water is lost as water vapor through transpiration by shoots. A genotype possessing limited transpiration can be classified as a water saver, while, a genotype having deep proliferative root can increase water absorption by enhanced soil exploration, and here roots play an important role. Thus, the availability of water at reproductive stages depends on the amount of water used at vegetative stage. Efficient water use at an earlier stage allows more water to be available at later growth stages (conservative pattern of water use). In fact, drought-tolerant chickpea genotypes were reported to have a lower water uptake and stomatal conductance at the vegetative stage, making available sufficient soil moisture during the reproductivephase (Zaman-Allah et al. 2011). However, after the flowering stage, the drought-tolerant chickpea genotypes were able to extract more water than drought sensitive genotypes resulting in higher yield. Similarly, in soybean, simulation modeling study has shown that an increased growth rate of the root system, lead to the faster depletion of soil moisture, making the crop more vulnerable to even modest dry periods later in the season, leading to an adverse effect on soybean yield and it is not a beneficial trait in soybean for yield improvement (Sinclair et al. 2010a, b). However, water conservation trait namely early stomatal closure under soil drying and reduced maximum transpiration rate, which has been observed as a slow wilting phenotype have resulted in increased grain yield (Sinclair et al. 2010a, b).
It can be safely premised that crops which depend on stored soil moisture at later stages of growth should expresses water conservative shoot trait at the early vegetative stage and deep and branched root system at later growth stages. This temporal variation might be looked at the perspective of carbohydrate allocation to plant parts under drought stress. Earlier research related the rooting depth differences with higher water uptake and grain yield (Vadez et al. 2008). However, as a whole plant response of root system, association studies with grain yield revealed a positive correlation (Bishopp and Lynch 2015) or negative or neutral correlation (Schoppach et al. 2013; Zaman-Allah et al. 2011). Thus, it indicates that for improving drought tolerance in crops, the structural and functional characteristics of root system along with shoot traits, i.e., a whole plant perspective should be considered. Therefore, drought tolerance is an interplay between shoot and root trait. Consequently, both root and shoot traits should be considered together at the whole plant level during breeding drought-tolerant genotypes.
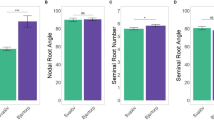
Common bean root system comprises of the taproot, adventitious root, and a variable number of basal roots, from which the rest of the root system develops by lateral branching. The common bean taproot may reach a depth of about 1–1.5 m, and possesses extensive network of lateral roots mainly concentrated in top 30 cm. The depth of tap root ranges from about 7 cm at emergence to about 30–40 cm at the flowering and can reach upto100–115 cm at maturity (Beebe et al. 2013), even thought rooting depths of upto 200 cm have been reported in green house experiments (Khalid 2017). Absorption of water occurs mainly at top 50–70 cm of soil depth. In areas of recurrent drought, the indeterminate genotypes with the profuse branching of shoot and roots are better adapted to drought stress. The intermittent precipitation will not recharge the deeper soil strata, and hence, the genotypes with lateral and adventitious roots are involved in water absorption. The variation in root diameter and lateral root density among common bean genotypes under the well-watered condition is shown in Fig. 2a, b. Under high evapo-transpiration (5–6 mm day−1) conditions, water can be depleted soon, and deep soil taproot is highly useful for water absorption. Sponchiado et al. (1989) and White et al. (1990) have reported that drought adaptation in common bean is associated with greater root length density and deeper soil moisture extraction, and deep rooting has positive association with seed yield, crop growth, cooler canopy as well as greater soil water extraction. In grafting experiments where shoots and roots of resistant and susceptible genotypes were interchanged, expression of drought resistance was positively correlated with the root system of the resistance genotype (White and Castillo 1992). In BAT477, a drought tolerant genotype (Beebe 2012) reported a dimorphic root system with deep roots for water extraction and shallow roots for nutrient absorption (top soil foraging), a model which can be used for breeding drought tolerance as well as nutrient use efficiency. However, the same author proposed that deeper roots alone are not sufficient to assure drought tolerance in common bean and it has to be combined with water-conserving shoot traits. A number of potential target root traits are presented in Fig. 1.
Integration of root traits with shoot traits for improving drought tolerance
Understanding the relationship between root traits with the above-ground traits will contribute to improved productivity under drought stress. Root architectural traits in different soil profiles are essential for yield improvement under stress on account of their high heritabilities under drought stress (Asfaw et al 2012; Riaz et al. 2013; Khalid 2017), and positive asssociation with yield traits (Riaz et al. 2013; Polania et al. 2016; Khalid 2017). However, extensive use of roots as the target traits for developing climate resilience suffers from the difficulties associated with studying roots, especially under field conditions. In comparison, the above ground shoot traits can be easily and reliably measured and quantified; however, the above ground shoot traits have to be linked with hidden half of the plant with the perspective of improving drought tolerance. In the following sections we discuss some of potential above ground traits that have been shown to be correlated with improved drought tolerance as well as better grain yield under stress. A number of potential target shoot traits are presented in Fig. 2.
Canopy temperature depression
Canopy temperature is one of the many physiological traits that may help to identify drought-tolerant cultivars. Canopy temperature depression (CTD) is the difference between air temperature and plant canopy temperature (Tuberosa 2012). Under water stress, there is an observed decrease in stomatal conductance, when available soil moisture is insufficient to meet the evaporative demands, resulting in increased canopy temperature (Urban et al. 2007). Canopy architectural traits (phyllotaxy, angle, area etc.) can also influence canopy temperature, usually mediated by the angle of leaves to the light source as well as the degree of mutual-shading in the canopy (Zheng et al. 2008). Canopy temperature has thus been used to elucidate whole plant-based information on the water status of the crop under stress (Mahan et al. 2011). Blum (2015) has proposed ideotypes of crop plants based on Canopy Temperature Depression for use in plant breeding as per the agro-ecological zones and drought types. As per the proposed model, plants are classified as isohydric (“water saving”) and anisohydric (“water spending”) plant model. The water saver plant ideotypes possess a distinct advantage in the stress conditions, whereas the water spender ideotype can have relatively better performance under moderate drought stress. Polania et al (2016) proposed that the water spenders can be better suited for areas that experience frequent intermittent drought stress with soils that can store greater amount of available water in deeper soil strata. However, water savers can be more conveniently grown in semiarid to dry areas with frequent terminal droughts. The water savers or isohydric genotypes invariably possess a shallower root system with thin roots resulting in intermediate root growth and penetration ability. They are early maturing and have high water use efficiency, limited transpiration, leaf area and canopy biomass development, reduced sink strength but possess better remobilization of photosynthates to pod and seed. In contrast, the water spenders or anisohydric genotypes are characterized by a relatively vigorous and deep root system with thicker roots, those results in faster root growth rate and penetration ability. Such genotypes are early maturing and have highly effective water use, moderate transpiration and faster leaf area and canopy biomass development, moderate sink strength and superior photosynthate remobilization to pod and seed.
Genotypes with cooler canopy temperature (higher CTD) under drought stress have been reported to extract greater amount of available soil moisture to effect evaporative cooling and therefore avoid excessive dehydration, both under greenhouse and field conditions (Ludlow and Muchow 1990; Reynolds et al. 2009). CTD can be related directly to the genetic potential of root’s capacity to explore soil moisture under high evaporative demand (Hammer et al. 2009; Pinto and Reynolds 2015). Canopy temperature depression can, therefore, be used as effective proxy trait for the genotypic potential of root development and biomass partitioning under drought stress (Bhandari 2016). In common bean, genotypes with higher CTD (cooler canopy) have been reported to enhanced ability to extract water by virtue of deeper roots and also produce 30% more yield, largely driven by an increase of upto 40% in root dry weight at 60–120 cm (Lopes and Reynolds 2010). Canopy temperature depression has been shown to be correlated with yield under drought stress (Blum 1989; Rashid et al. 1999; Pinto and Reynolds 2015; Purushothaman et al. 2017; Table 2) and hot irrigated conditions (Amani et al. 1996; Pinto and Reynolds 2015). Under irrigated conditions also, canopy temperature depression is an indication of genotypic yield potential under drought and could effectively be used as a proxy trait to assess genotypic response to drought (Mohammadi et al. 2012). Blum (1989) used canopy temperatures in wheat genotypes under water stress to characterize their yield stability under varying moisture conditions. In terms of yield based indices such as drought susceptibility index (DSI), a positive correlation was found between DSI and CT in stress situations. Genotypes with higher CT at midday tend to suffer relatively greater yield reductions under drought stress. Our studies in common bean and cowpea revealed that CTD can be a reliable indicator of crop performance under both irrigated and water stress conditions. Under irrigated conditions yield changed linearly with CTD, while, under drought stress, both negative and positive values of CTD could be identified, and interestingly in both classes, high yielding genotypes were identified. The water savers probably have the ability to sense water deficit in early phases of growth, theeby triggering conservative water use that could be used in later stages of growth (Khalid 2017). However, the reduced water use is invariably achieved by plant traits and environmental responses, and could also reduce yield potential especially under optimum conditions (Blum 2005).
Leaf relative water content
Leaf water content (RWC) that determines the leaf water status can reliably reflect the balance between the amount of water in the leaf tissue and transpiration rate (Lugojan and Ciulca 2011). It provides a reliable basis for establishing a relationship between leaf water status and plant metabolism under drought stress. RWC can be easily measured and can robustly indicate water status for comparison of tissue responses, by normalizing water content by expressing it relative to the hydrated (fully turgid) state (Lawlor and Cornic 2002). In fact leaf relative water content (RWC) has been reported as a relatively better indicator of water status than water potential (Sinclair and Ludlow 1985). Under drought stress, RWC can be efficiently used in the identification of tolerance of plants to stress by inducing osmotic adjustment due to the accumulation of osmoprotectants (Ritchie et al. 1990). Compared to susceptible genotypes, the tolerant genotype are expected to have higher RWC that helps them maintain better protoplast hydration for a longer duration under drought conditions as compared susceptible genotype, which is critical for optimum physiological functions and growth processes. Variation in drought response among genotypes may be associated with dehydration avoidance through lower stomatal conductance leading to higher transpiration efficiency (Khan et al. 2007). RWC is a rapid method of identification of tolerant genotypes, especially when large germplasm sets are initially screened for characterizing drought response for quick and efficient narrowing of working material to a manageable number of candidate lines for use in a more focused way. Relative water content is a semi-high throughput method for identification of drought tolerant lines (Knepper and Mou 2015). Studies on legumes such as common bean, faba bean and cowpea have shown that maintenance of a relatively high RWC during mild drought is indicative of drought tolerance (Khan et al. 2007; Rosales-Serna et al. 2004; Gull et al. 2019). Omae et al. (2005) reported that genotypic differences in RWC of bean correlated with grain yield under drought conditions. Similarly, in common bean the RWC was positively correlated with leaf dry weight under irrigated (r2 = 0.24***) and drought (r2 = 0.20***) stress conditions, indicating differences in leaf water status exist among bean cultivars, which is linked to the drought tolerance mechanisms (Abenavoli et al. 2016). RWC has been used as an integrative indicator of internal plant water status under drought conditions to identify drought-resistant cultivars in different crops such as of barley (Matin et al. 1989) and common bean (França et al. 2000; Choudhury et al. 2011).
Stay green trait
Delayed senescence under drought stress is one of the most critical traits targeted for yield improvement in many crops. This trait is an indirect estimate of photosynthetic potential since it is associated with chlorophyll concentration and senescence phenomenon (Thomas and Howarth 2000). The stay-green trait, a phenomenon where quantitative trait loci (QTL) for delayed leaf senescence, biomass and grain yield are co-located has been described in detail in wheat, sorghum, and maize (Christopher et al. 2016; Borrell et al. 2000; Wang et al. 2012). Crops with the stay-green phenotype generally retain green leaf area for a long period after flowering compared to senescent genotypes. An example of stay green genotype of the common bean. Stay green prolongs the duration of carbon assimilation during the grain filling period, thereby producing greater yields; especially under terminal drought. It is also associated with relatively smaller size of canopy at flowering, reduced water use before flowering, greater soil moisture extraction after flowering and enhanced uptake of nitrogen during grain filling, and greater leaf nitrogen content (Borrell et al. 2000). Plant exhibiting stay green phenotype is characterized by maintenance of functional green leaf area under drought, which will have associated with a continued photosynthetic rate under drought and quick recovery after drought stress (Thomas and Howarth 2000). The stay-green character at the post-flowering stage can be easily assessed visually using standard scoring scales for retention of the green-leaf area. Expression of stay green trait under favorable conditions has proved to be beneficial in corn, as evidenced by significant improvement of grain yield (Duvick 2005). In maize, multiple QTLs for stay-green and grain yield have been co-localised (Zheng et al. 2009). While in maize, stay green (SG) was found to be associated with higher nitrogen use (Blum 2006), in sorghum, SG has been found to be associated with more favorable water status as well as better root architectural features (Mace et al. 2012). Asfaw et al. (2012) have reported that in common bean expression of stay green trait have associated with remobilization of stored carbohydrate to the reproductive parts for a more extendedperiod leading to increased grain yield.
Stay-green has been used as an effective trait for improving crop adaptation to drought in crops such as wheat and sorghum (Borrell et al. 2014; Christopher et al. 2014; Gregersen et al. 2013; Jordan et al. 2012; Lopes and Reynolds 2012). There are certain important considerations from a breeding standpoint for effective utilization of this trait. Stay green trait has been classified as functional stay-green (which is characterized by prolonged duration of photosynthesis and accumulation of assimilates in grains), or non-functional stay-green (which is characterized by a disruption in the chlorophyll recycling process or remobilization of nitrogen from leaf to grain and, in most cases, is not associated with any yield benefit (Christopher et al. 2016). Only functional stay-green is of interest for plant breeders. In plants, functional stay-green is accomplished by varying dynamics of leaf-greenness in some different ways (Thomas and Howarth 2000). Plants may be exhibit stay-green by way of maintaining greener canopy around anthesis before the onset of senescence, delayed onset of senescence process, or slow pace of senescence (Thomas and Howarth 2000; Harris et al. 2007; Christopher et al. 2014).
Photosystem ii efficiency
Photosynthesis is by far the most important biological reactions on this planet. It is the primary biochemical process for accumulation of dry matter and significantly determines patterns of plant growth and development, which are strongly influenced by the environment (McCree 1986). In common bean, drought stress at its initial stages reduces photosynthesis mainly due to closure of stomata (Amede and Schubert 2003; Miyashita et al. 2005). However, as the stress progresses, non-stomatal inhibition of photosynthesis may become more critical (Lawlor and Cornic 2002; Medrano et al. 2002). Drought stress significantly implicates component processes of photosynthesis such as the photochemical chlorophyll fluorescence quenching, photosystem II quantum yield and electron transport rate, mainly by disrupting the thylakoid electron transport system, carbon reduction and the CO2 capture via stomatal control, coupled with an increase in carbohydrates accumulation, oxidative destruction of lipids and disturbing water balance (Allen and Ort 2001). The basic principle of chlorophyll fluorescence analysis is pretty straightforward. Light energy captured by chlorophyll molecules is channelized through either of the three routes: (i) used to drive photochemical processes in photosynthesis, (ii) lost as heat, or (iii) re-emitted as light-chlorophyll fluorescence. These three processes are essentially competitive, such that any increase in the efficiency of one will negatively implicate the efficiency and yield of the other two. The photochemical processes are linked to fixation of CO2 as they supply ATP, and NADPH (reduced form of nicotinamide adenine dinucleotide phosphate), and are also regulated by other electron sinks including photorespiration, Mehler reaction, and nitrogen reduction (Noctor et al. 2002). Drought stress invariably damages oxygen-evolving complex of photosystem (PS) II as well as reaction centers of PS II (Subrahmanyam et al. 2006). In experimental procedures, the difference between chlorophyll fluorescence values recorded with closed and open reaction centers is known as variable fluorescence (Fv = Fm − Fo), which is a measure of absorbed light energy that would be used in photosynthesis if all reaction centers are in the open state. The relative values of variable and maximum fluorescence are used to measure the quantum efficiency of the photochemical reaction (also referred to as the PS II yield) and measured as Fv/Fm. Another ratio Fo/Fm is a measure of the damage caused to thylakoid membranes, and PSII yield and thylakoid membrane damage are used as an indicativeparameter for assessment of crop health under drought stress (Maxwell and Johnson 2000). In common bean, photosynthesis and other traits related to photosynthesis undergo significant reduction in susceptible cultivars as compared to tolerant ones (Subrahmanyam et al. 2006).
Carbon isotope discrimination
One of the classical examples of uptake or assimilation of a particular isotope in preference to another isotope of the same element by living organisms is the preferential fixation of the 12C isotope (which is lighter but more abundant) compared to 13C (which is heavier but less abundant). The relative proportion of 12C and 13C isotopes is different in photosynthetic plants and the atmosphere, and is also implicated by stressful conditions. Carbon isotope discrimination (CID) was defined as the ratio of 13C/12C in atmosphere and the photosynthetic plant product (Farquhar et al. 1982). In plants such discriminations is achieved manly due to the fact that carbon dioxide containing 13C diffuses more slowly than CO2 containing 12C and the ability of Rubisco to discriminate between the two isotopes. The 12C/13C isotopic discrimination is more pronounced in C3 plants such as common bean resulting in differential isotopic signatures in C3 and C4 plants and the 12C/13C ratios can provide an insight about the physiological state and atmospheric conditions, especially the information about the stomatal conductance. Most of the studies have reported inverse relationship between carbon isotope discrimination (CID) and water use efficiency (WUE), implying thereby that greater 13C discrimination is associated with lower values of WUE (indicating more water use and transpiration). Selection for low 13C discrimination values has been suggested as an effective proxy trait to improve WUE in C3 crops (Khan et al. 2007; Easlon et al. 2014). CID is advantageous as it is integrative of overall crop development, is high throughput, and relatively less costly with fairly high heritability (Easlon et al. 2014). In common bean, CID has been found to be associated grain yield as well as root length density under moderate water stress in non-arid environments (Polania et al. 2016). Increased water use is associated with increased accumulation of carbon and plant growth. Even though increased dry matter partitioned to pod and seed contributes to higher grain yield under water stress (Rao 2014), yet the recent study of Smith et al. (2019) in the leaf tissue did not reveal any impact of drought through δ13C.
Membrane stability index
The membrane electrolyte leakage has been used as a “predictive” selection criterion of water stress tolerance as a whole plant response (Bajji et al. 2001). Changes in the electrical impedance and electrolyte leakage provide a measure of membrane stability of drought tolerance in plants under drought stress conditions (Bajji et al. 2001). The leakage defines the membrane’s ability to take up and retain solutes and, therefore, is a reflection of changes in both membrane potential and membrane permeability induced by stressful environments (Agarie et al. 1995). In fact, the cellular membranes are one of the initial targets of many stresses, and it is a generally accepted premise that the tolerant genotypes stably maintain structural and functional integrity of cellular membranes under stress conditions (Bajji et al. 2001). The degree of damage to cellular membranes induced by drought can be indirectly estimated through electrolyte leakage measurements from the cells with fair degree of reliability. A lower value of MSI (membrane stability index) indicates the amount of lipid peroxidation occurring under higher oxidative stress owing to the loss of cell turgidity under drought conditions (Moussa and Abdel-Aziz 2008). The consequent reduction in cell membrane stability invariably leads to higher permeability of the membrane which increases leakage of electrolyte from the cell (Baroowa and Gogoi 2012). Overproduction of ROS (reactive oxygen species) can increase the photo-inhibition and induce damage to the biological membrane system through electrolyte leakage (Meng et al. 2014). Hence, peroxidation of membrane lipid triggered by ROS can be considered as one of the prime causes of injury and reduced stability of cell membrane under drought. Diaminobenzidine tetrahydrochloride (DAB) assay has been suggested as an effective qualitative assessment of plant response to biotic and abiotic stress and measures the intensity of oxidative burst under stress. In the staining process, the evolved hydrogen peroxide oxidizes the DAB and produces a dark brown precipitate. Genetic variability for DAB staining under drought stress in common bean. The initial oxidative burst through evolution of reactive oxygen species (ROS) including hydrogen peroxide either through NADPH oxidases or peroxidases is an early response to stress (Bindschedler et al. 2006) that may exist singly or in combination in different plant species,. The qualitative evolution of such ROS components can be differentially tracked in different parts of the plant under stress to assess the most vulnerable part under stress.
Limited transpiration rate as a response to drought
Stomata act as primary regulators of internal plant water status and carbon assimilation. They exert their control on both uptake of CO2 and water by controlling transpiration rate; and therefore, have a major role in photosynthesis as well as transpiration efficiency. Stomatal pores account for only 5% of the leaf area but account for 70% water loss by plants (Hetherington and Woodward 2003). Under conditions of high VPD (vapor pressure deficit) in irrigated conditions, limited transpiration rate has been used as an effective selection criterion to identify water conservative genotypes (especially those genotypes that exhibit conservative pre-anthesis water use), thereby allowing sustained post-flowering physiological activity during reproductive development (Sinclair et al. 2017). Based on this premise, maintenance of optimal stomatal control over transpiration rate is critical for overcoming photosynthetic inhibition under drought stress (Bota et al. 2004). There is significant genetic variation for size and density of stomata in legumes such as common bean (Comstock and Ehleringer 1993), soybean (Buttery et al. 1993) and faba bean (Peksen et al. 2006). Smaller size and higher density of stomata have been associated with improved drought tolerance in Arabidopsis (Masle et al. 2005) possibly through increased transpiration efficiency which is a measure of biomass produced per unit of water transpired by a plant (Condon et al. 2004). Similarly, in Arabidopsis mutants having reduced stomatal density and increased stomata size, improved drought tolerance was observed largely driven by reduced transpiration and higher accumulation of biomass (Pillitteri and Torii 2012). Transpiration efficiency can be an effective proxy trait for deeper root system as deeper roots enhance moisture uptake (Uga et al. 2013). Genotypes that extract moisture from deeper soil strata (60–120 cm), invariably, maintain a cooler canopy temperature on account of higher stomatal conductance (Pask and Reynolds 2013). Therefore, a focussed strategy that seeks simultaneous improvements to root architectural traits (root depth and density at deeper soil strata) for enhanced exploration of water from deeper soil layers and the transpiration efficiency (limited transpiration) to conserve above ground water, can be adopted to improve drought tolerance. The possible traits for a desirable ideotype include deeper roots as well as higher transpiration efficiency (Isohydric) with stringent stomatal regulation on plant water status that maintains a relatively constant water potential in leaves regardless of soil moisture status. Limited transpiration can, however, have a negative effect on yield under certain environmental conditions especially under high VPD, where assimilation of carbon is reduced (Sinclair et al. 1984). A major area of investigation would be to elucidate whether water conservation in early growth stages is sufficiently rewarded later by improved water availability for sustained dry matter growth during the seed-fill period. Limited-transpiration trait most likely improves the preferential partitioning of water use from vegetative to reproductive parts, which, in effect will increase seed yield (Sinclair and Rufty 2012; Vadez et al. 2014). In maize, limited transpiration has been effectively used to discriminate drought-susceptible and tolerant hybrids on the basis of their differential capacity for restricted water use during the pre-flowering stage, and consequential improvement in the plant water status at flowering and increased post-flowering water use (Cooper et al. 2014).
Nitrogen fixation under water stress
Like most other legumes, common bean derives part of their nitrogen requirement through the process of biological nitrogen fixation. Water stress severely limits the process efficiency of biological nitrogen fixation (BNF) of legumes plants on account of greater sensitivity of the process of symbiotic N2 fixation to moisture deficit (Purcell 2009; Mhadhbi et al. 2011). Therefore, the crops that are dependent on nitrogen fixation, will experience nitrogen deficiency under drought stress on account of reduced N2 fixation. In fact, the increased sensitivity of nitrogen fixation to drought is a major constraint on N2 accumulation and yield of legumes grown under moisture stress (Serraj et al. 1999). The reduced N2 fixation activity in common bean can be severely implicating nitrogen nutrition because the species is not known as an efficient nitrogen fixer even under well-watered conditions (Diouf et al. 2008). The negative implications of water stress on rhizobia include morphological changes (Shoushtari and Pepper 1985) as well as reduction in infection and nodulation capacity and number and size of nodules (Williams et al. 1994). In common bean, drought stress causes an increased accumulation of abscisic acid (ABA) in leaves with a concomitant reduction in indole acetic acid (IAA) and gibberellic acid (GA3) as well as zeatin content in leaves (Figueiredo et al. 2008). However, the effect on nodulation and N2 fixation varies with the developmental stage of the plants. During vegetative growth, water stress has been reported to be more limiting to nodulation and nitrogen fixation as compared to the reproductive stage (Cabriales and Castellanos 1993).
The persistence of rhizobial populations in desert soils and the effective nodulation of native legume (Jenkins et al. 1987) substantiate the assumption that rhizobia can exist in soils with limiting moisture levels. Thus, it can be reasonably assumed that selection of rhizobial strains for response to moisture stress tolerance within the range of their legume host can be effectively exercised. This is especially important in view of the experimental evidence that association of legumes can significantly increase drought tolerance in common bean (Aydi et al. 2012; Sofi et al. 2017). The increased drought tolerance conferred by rhizobial inoculation in legumes is invariably due to disaccharide trehalose that plays a role in osmo-regulation when rhizobia are growing under osmotic stress (Saurez et al. 2008). Trehalose accumulates to higher levels in cells of R. leguminosarum and under water stress acts as a preferred source of carbon among many carbon sources such as mannitol, sucrose, or lactose, but the slow growing strains accumulate trehalose only when cultured with mannitol as the carbon source. In some cases, concentration of glycine betaine has been reported to increases in tolerant strains of R. meliloti ( Smith et al. 1988). de Silva et al. (1996) found a major role of ureide level and the degree of drought tolerance in soybean.
From a breeding point of view, any strategy to harness BNF in relation to drought stress, the effective approach should include selection for differential genotypic response to rhizobacterial inoculation, selection for competitive rhizobacterial strains, and identification of adaptive crop-microbial associations as well as genetic modification of rhizobacteria (Sofi et al. 2018a, b). Of the two component partners in legume-rhizobium association, the microbial partner is far more sensitive than the host plant. This is obvious from increased sensitivity of symbiotic association environmental stress as compared to the uninfected legume host (Swaine et al. 2007). There is substantial evidence of existence of adequate genetic variability among common bean genotypes for nitrogen fixation ability (Hardarson et al. 1993) and in amount of total nitrogen fixed (Vadez et al. 1999) under irrigated conditions. Substantial variability has also been reported in common bean genotypes in the soil water threshold content at which nitrogen fixation begins to decline (Devi et al. 2010) as well as rooting depth and root biomass under stress with rhizobial inoculation (Sofi et al. 2017).
Reproductive success under water stress
Drought stress leads to abortion of reproductive organs that significantly limits seed yield. The failure of fertilization and subsequent arrest of ovary tissue growth causes abortion of ovary and grain, a trait selected for by evolutionary processes and largely driven by limitations of sucrose availability to ovarian tissue (Boyer and McLaughlin 2007; Ruan et al. 2012). The casual phenomenon of such abortion of reproductive parts has been well documented in crops like maize, safflower and pea. The cessation of growth is more severe in the youngest reproductive organs due to the effect of drought on the respective timing of the development of ovary cohorts and the signaling pathways between youngest and oldest reproductive organs (Turc and Tardieu 2018). The youngest reproductive organs in the developmental sequence are relatively more vulnerable to abortion allow older organs finish their development under limited resources. Water stress drives increased abortion of reproductive organs through a signaling network that originates from the first fertilized ovaries as well as arresting the expansive growth of all ovaries at a precise stage. In crops like maize such developmental mechanisms have been reported account for 90% of water stress driven abortion and are invariably irreversible after a certain stage: typically 3 days after silk emergence (Turc and Tardieu (2018). In common bean such mechanism has also been reported to be operative under water stress and genotypic differences have been found in terms of the level of sucrose available for metabolism in the seeds under drought stress conditions (Gebeyehu et al. 2010). Such a process results in decrease in pod number and seed number in beans while as relatively less effect on 100-seed weight is recorded (Khalid 2017).Studies on sensitivity of flower development, anther dehiscence, pollen tube growth and post-fertilization stages such as pod and seed set revealed higher sensitivity of male parts whereas functionality of female whorl was relatively unaffected. Stress reduced pollen viability, penetration of pollen tube into the stigma that results in reduced pod and seed set in the susceptible genotypes. In common bean, the combined effect of lower pollen viability and impaired female performance in a large proportion of the flowers results in significant decline in the pod and seed set under water stress (Gross and Kigel 1994).
Conclusions
Abiotic stress such as drought and high temperature pose dual challenges for a plant regarding enhanced resource acquisition as well as efficient resource mobilization and remobilization and as such plants need to make adaptive changes in root architectural traits to increase water and nutrient acquisition whereas the above ground trait efficiencies should ensure optimum resource allocation towards enhanced productivity through physiological adaptations. Putting both below and above ground adaptive mechanisms into an integrated system is imperative for identifying effective proxy traits that could be linked to higher productivity under stress. In fact, the future improvements in crop yields are expected to come largely by harnessing improved root architecture, resource acquisition as well as nitrogen fixation (Herder et al. 2010). Even though rooting depth, root length density, root hairs, root branching and root biomass have been related to improved drought tolerance, there have been reports where the root functionality attributes such as root hydraulics have been implicated more than just a profuse root system, which may not necessarily result in increased water acquisition because, more than just the architectural phenes, roots need to have the optimum hydraulic characteristics (Vadez et al. 2014). In terms of increasing yield under stress, a vigorous root can be essentially useful under water-limited conditions in rainfed environments, whereas, in the areas where crops grow on the stored moisture in soil, a vigorous root system may increase the risk of pre-anthesis soil water depletion, thereby leaving inadequate moisture during grain filling. Recently, traits like root aerenchyma tissue that reduces the metabolic cost of resource acquisition have received attention for improving drought tolerance (Zhu et al. 2010). In a recent review, Lynch (2018) has outlined the evolutionary changes in root system in terms of changes in relatively higher investments favoured by natural selection for crop grown under multitude of stresses and intense competition, variable resource availability which are no longer useful in high-input farming systems. The input intensive agriculture has promoted relatively parsimonious root systems that optimize water capture through progressive reduction in root branching, fewer axial roots, lateral root density and a root anatomy that decreases the root also comprises carbon cost. The “parsimonious root ideotype” as proposed by Lynch (2018) also encompasses certain anatomical features such as reduced cortical cell files, greater relative composition of aerenchyma cells as we’ll as large cortical cells. Ye et al (2018) has provided a detailed account of such beneficial root traits and outlined the need of harnessing natural genetic variations in available germplasm. A large number of loci (QTLs) associated with root system architecture and the beneficial root traits that can accelerate the genetic improvement of yield under water deficit have been identified and already introgressed successfully in high yielding cultivars (Varshney, 2016). From a breeding standpoint it is highly relevant to link traits to productivity rather than just survival as in the case of “non-functional stay green” (Christopher et al. 2016) or “lazy pod syndrome” (Beebe et al. 2008), wherein, the plants fail to harness the adaptive advantages into economic yield.
Breeding success for developing drought resilient genotypes will primarily require (i) designing an appropriate plant with theoretically improved adaptation, (ii) identifying genetic resources possessing appropriate complementary allelic variation, (iii) standardizing and implementing phenotyping protocols and experimental procedures to maximize resolution of physiological trait expression, and (iv) genetic dissection of identified traits (Reynolds and Langridge 2016). Plant breeders need to pick the generalized trait correlations as well as specific combinations that can help identify physiologically efficient as well as high yielding genotypes. The above statement is especially true in case of CTD, where high yielding genotypes, under stress, can be associated with both positive and negative values of CTD (Khalid 2017).
Sufficient evidence in the form of experimental datasets are available, that reinforce the need to integrate both above and below ground phenes to have a well-defined set of traits that can be utilized in developing drought resilient varieties (Sofi et al. 2018a, b). Fortunately for plant breeders, there is a substantial variation for potential root and shoot traits in cultivated varieties and germplasm resources available with national and international breeding programmes as well as desirable correlations with yield under stress. We have discussed here a set of traits with experimental data in common bean to support our premise that traits related to root architecture and above-ground physiology in isolation cannot be as fruitful. Designing an improved plant type for root architecture and physiological traits is a unique aspect of physiological breeding, in the sense that such traits are invariably genetically simple and can have handled through traditional plant breeding methods. However, developments in phenomics and genomics are increasing access to more complex traits (Mir et al. 2012; Araus et al. 2014). However, a fair amount of pragmatic skepticism still exists among plant breeders (Reynolds and Langridge 2016). There is a need to understand the fact that plant breeders can use the traits related to below and above ground adaptive traits, as they express high heritability under drought stress, given the fact that yield per se suffers from low heritability under stress (Cabello et al. 2014).
Intensive research and breeding efforts have allowed the selection of climate-resilient varieties with improved yields while crop physiologists have essentially focused on physiological and molecular aspects underlying stress tolerance. Drought research has progressed rapidly, as evidenced by a substantial wealth of information on mechanistic aspects as well as applied research products in form of improved germplasm, genes and QTLs. From identification of tolerant lines and understanding the role of effective surrogate traits, we have now entered an era, where research efforts are focusing on uncovering and manipulating genes/QTLs and mechanisms involved in cellular and whole-plant responses related to crop acclimation to drought scenarios (Tricker et al. 2018; Varshney et al. 2018). Driven by advances in genomics and advances in precise phenomics, a large number of loci governing turgor management and hormonal regulation, plant architecture and developmental biology that are implicated in drought have been mapped and cloned (Varshney et al. 2018). The interactions between roots, shoots, the rhizosphere microbiome and reproductive traits and how these collectively drive crop response under the limited water availability have also received much attention (Tardieu et al. 2018).
References
Abenavoli, M. R., Leone, M., Sunseri, F., Bacchi, M., & Sorgona, A. (2016). Root phenotyping for drought tolerance in bean landraces from Calabria (Italy). Journal of Agronomy and Crop Science, 202(1), 1–12.
Agarie, S., Hanaoka, N., Kubota, F., Agata, W., & Kaufman, P. B. (1995). Measurement of cell membrane stability evaluated by electrolyte leakage as a drought and heat tolerance test in rice (Oryza sativa L.). Journal of the Faculty of Agriculture, Kyushu University, 40(1), 233–240.
Allen, D. J., & Ort, D. R. (2001). Impact of chilling temperatures on photosynthesis in warm climate plants. Trends in Plant Science, 6, 36–42.
Amani, I., Fischer, R. A., & Reynolds, M. P. (1996). Canopy temperature depression association with yield of irrigated spring wheat cultivars in a hot climate. Journal of Agronomy and Crop Science, 176(2), 119–129.
Amede, T., and Schubert, S. (2003). Mechanisms of drought resistance in grain: II Stomatal regulation and root growth. SINET: Ethiopian Journal of Science, 26(2), 137–144.
Amongi, W., Nkalubo, S. T., Ochwo-Ssemakula, M., Gibson, P. T., & Edema, R. (2014). Development of intermittent drought stress tolerant common bean genotypes in Uganda. African Crop Science Journal, 22(4), 303–315.
Araus, J. L., Li, J., Parry, M. A., & Wang, J. (2014). Phenotyping and other breeding approaches for a New Green Revolution. Journal of Integrative Plant Biology, 56(5), 422–424.
Asadi, M., Bahrami, S., & Ansari Samani, R. (2010). The effect of Stachys lavandulifolia Vahl. and Mespilus germanica L. leaves hydroalcoholic extracts on Leishmania Major (MRHO/IR/75/ER) in vitro. Jundishapur Journal of Natural Pharmaceutical Products, 5(1), 39–43.
Asfaw, A., Blair, M. W., & Struik, P. C. (2012). Multienvironment quantitative trait loci analysis for photosynthate acquisition, accumulation, and remobilization traits in common bean under drought stress. G3: Genes Genomes Genetics, 2(5), 579–595.
Aydi, S. S., Aydi, S., & Abdelly, C. (2012). Inoculation with the native Rhizobium gallicum 8a3 improves osmotic stress tolerance in common bean drought-sensitive cultivar. Acta Agriculturae Scandinavica, Section B-Soil and Plant Science, 62(2), 179–187.
Bajji, M., Lutts, S., & Kinet, J. M. (2001). Water deficit effects on solute contribution to osmotic adjustment as a function of leaf ageing in three durum wheat (Triticum durum Desf.) cultivars performing differently in arid conditions. Plant Science, 160(4), 669–681.
Baroowa, B., & Gogoi, N. (2012). Effect of induced drought on different growth and biochemical attributes of black gram (Vigna mungo L.) and green gram (Vigna radiata L.). Journal of Environmental Research and Development, 6(3A), 584–593.
Beebe, S. E. (2012). Common bean breeding in the tropics. Plant Breeding Reviews, 36, 357–426.
Beebe, S. E., Rao, I. M., Blair, M. W., & Acosta-Gallegos, J. A. (2013). Phenotyping common beans for adaptation to drought. Frontiers in Plant Physiology, 4, 1.
Beebe, S. E., Rao, I. M., Cajiao, C., & Grajales, M. (2008). Selection for drought resistance in common bean also improves yield in phosphorus limited and favorable environments. Crop Science, 48(2), 582–592.
Bhandari, M. (2016). Use of infrared thermal imaging for estimating canopy temperature in wheat and maize (Doctoral dissertation), 96 pp.
Bindschedler, L. V., Dewdney, J., Blee, K. A., Stone, J. M., Asai, T., Plotnikov, J., et al. (2006). Peroxidase-dependent apoplastic oxidative burst in Arabidopsis required for pathogen resistance. The Plant Journal, 47(6), 851–863.
Birthal, P. S., Khan, M. T., Negi, D. S., & Agarwal, S. (2014). Impact of climate change on yields of major food crops in india: implications for food security. Agricultural Economics Research Review., 27, 145–155.
Bishopp, A., & Lynch, J. P. (2015). The hidden half of crop yields. Nature Plant, 1, 8.
Blair, M. W., González, L. F., Kimani, P. M., & Butare, L. (2010). Genetic diversity, inter-gene pool introgression and nutritional quality of common beans (Phaseolus vulgaris L.) from Central Africa. Theoretical and Applied Genetics, 121(2), 237–248.
Blum, A. (1989). Osmotic adjustment and growth of barley genotypes under drought stress. Crop Science, 29(1), 230–233.
Blum, A. (2005). Drought resistance, water-use efficiency, and yield potential—are they compatible, dissonant, or mutually exclusive? Australian Journal of Agricultural Research, 56(11), 1159–1168.
Blum, A. (2006). Drought adaptation in cereal crops: a prologue. In J. M. Ribaut (Ed.), Drought adaptation in cereals (pp. 3–15). Binghamton: The Haworth Press Inc.
Blum, A. (2015). Stress, strain, signaling, and adaptation–not just a matter of definition. Journal of Experimental Botany, 67(3), 562–565.
Borrell, A. K., Hammer, G. L., & Henzell, R. G. (2000). Does maintaining green leaf area in sorghum improve yield under drought? II Dry matter production and yield. Crop Science, 40, 1037–1048.
Borrell, A. K., Oosterom, E. J., Mullet, J. E., George-Jaeggli, B., Jordan, D. R., Klein, P. E., & Hammer, G. L. (2014). Stay-green alleles individually enhance grain yield in sorghum under drought by modifying canopy development and water uptake patterns. New Phytologist, 203(3), 817–830.
Bota, J., Medrano, H., & Flexas, J. (2004). Is photosynthesis limited by decreased Rubisco activity and RuBP content under progressive water stress? New Phytologist, 162(3), 671–681.
Broughton, W. J., Hernández, G., Blair, M., Beebe, S., Gepts, P., & Vanderleyden, J. (2003). Beans (Phaseolus spp.): Model food legumes. Plant and Soil, 252, 55–128.
Buttery, B. R., Tan, C. S., Buzzell, R. I., Gaynor, J. D., & MacTavish, D. C. (1993). Stomatal numbers of soybean and response to water stress. Plant and Soil, 149(2), 283–288.
Cabello, R., Monneveux, P., Bonierbale, M., & Khan, M. A. (2014). Heritability of yield components under irrigated and drought conditions in andigenum potatoes. American Journal of Potato Research, 91(5), 492–499.
Cabriales, J. J., & Castellanos, J. Z. (1993). Effects of water stress on N2 fixation and grain yield of Phaseolus vulgaris L. In F. A. Bliss & G. Hardarson (Eds.), Enhancement of biological nitrogen fixation of common bean in Latin America (pp. 151–155). Dordrecht: Springer.
Chaves, M. M., Maroco, J. P., & Pereira, J. S. (2003). Understanding plant responses to drought - from genes to the whole plant. Functional Plant Biology, 30, 239–264.
Christopher, J. T., Christopher, M. J., Borrell, A. K., Fletcher, S., & Chenu, K. (2016). Stay-green traits to improve wheat adaptation in well-watered and water-limited environments. Journal of Experimental Botany, 67(17), 5159–5172.
Christopher, J. T., Veyradier, M., Borrell, A. K., Harvey, G., Fletcher, S., & Chenu, K. (2014). Phenotyping novel stay-green traits to capture genetic variation in senescence dynamics. Functional Plant Biology, 41(11), 1035–1048.
Comstock, J., & Ehleringer, J. (1993). Stomatal response to humidity in common bean (Phaseolus vulgaris): Implications for maximum transpiration rate, water-use efficiency and productivity. Functional Plant Biology, 20(6), 669–691.
Condon, A. G., Richards, R. A., Rebetzke, G. J., & Farquhar, G. D. (2004). Breeding for high water-use efficiency. Journal of Experimental Botany, 55(407), 2447–2460.
Cooper, M., Gho, C., Leafgren, R., Tang, T., & Messina, C. (2014). Breeding drought-tolerant maize hybrids for the US Corn-Belt: Discovery to product. Journal of Experimental Botany, 65, 6191–6204. https://doi.org/10.1093/jxb/eru064.
Dai, A. (2013). Increasing drought under global warming in observations and models. Nature Climate Change, 3, 52–58.
de Silva, M., Purcell, L. C., & King, C. A. (1996). Soybean petiole ureide response to water deficits and decreased transpiration. Crop Science, 36, 611–616.
Den Herder, G., Van Isterdael, G., Beeckman, T., & De Smet, I. (2010). The roots of a new green revolution. Trends in Plant Science, 15, 600–607.
Devi, M. J., Sinclair, T. R., & Vadez, V. (2010). Genotypic variation in peanut for transpiration response to vapor pressure deficit. Crop Science, 50, 191–196.
Diouf, A., Diop, T. A., & Gueye, M. (2008). Nodulation in situ of common bean (Phaseolus vulgaris L.) and field outcome of an elite symbiotic association in Senegal. Research Journal of Agriculture and Biological Sciences, 4(6), 810–818.
Duvick, D. N. (2005). The contribution of breeding to yield advances in maize (Zea mays L.). Advances in Agronomy, 86, 83–145.
Easlon, H. M., Nemali, K. S., Richards, J. H., Hanson, D. T., Juenger, T. E., & McKay, J. K. (2014). The physiological basis for genetic variation in water use efficiency and carbon isotope composition in Arabidopsis thaliana. Photosynthesis Research, 119, 119–129.
Fang, X., Turner, N. C., Yan, G., Li, F., & Siddique, K. H. (2010). Flower numbers, pod production, pollen viability, and pistil function are reduced and flower and pod abortion increased in chickpea (Cicer arietinum L.) under terminal drought. Journal of Experimental Botany, 61(2), 335–345.
FAO–Food and Agriculture Organization of the United Nations. (2016). Food and agricultural commodities production. http://faostat.fao.org/site/339/default.aspx.
Farquhar, G. D., O’Leary, M. H., & Berry, J. A. (1982). On the relationship between carbon isotope discrimination and the intercellular carbon dioxide concentration in leaves. Functional Plant Biology, 9(2), 121–137.
Figueiredo, M. V., Burity, H. A., Martínez, C. R., & Chanway, C. P. (2008). Alleviation of drought stress in the common bean (Phaseolus vulgaris L.) by co-inoculation with Paenibacillus polymyxa and Rhizobium tropici. Applied Soil Ecology, 40(1), 182–188.
França, M. G. C., Thi, A. T. P., Pimentel, C., Rossiello, R. O. P., Zuily-Fodil, Y., & Laffray, D. (2000). Differences in growth and water relations among Phaseolus vulgaris cultivars in response to induced drought stress. Environmental and Experimental Botany, 43(3), 227–237.
Ganeshan, S., Gaur, P. M., & Chibbar, R. N. (2012). Pulse crops: biotechnological strategies to enhance abiotic stress tolerance. In N. Tuteja, S. S. Gill, & R. Tuteja (Eds.), improving crop productivity in sustainable agriculture (pp. 382–403). Singapore: Wiley-Blackwell.
Gebeyehu, S., Wiese, H., & Schubert, S. (2010). Effects of drought stress on seed sink strength and leaf protein patterns of common bean genotypes. African Crop Science Journal, 18(2), 75–88.
Gregersen, P. L., Culetic, A., Boschian, L., & Krupinska, K. (2013). Plant senescence and crop productivity. Plant Molecular Biology, 82(6), 603–622.
Gross, Y., & Kigel, J. (1994). Differential sensitivity to high temperature of stages in the reproductive development of common bean (Phaseolus vulgaris L.). Field Crops Research, 36(3), 201–212.
Gull, M., Sofi, P. A., & Asmat, A. (2019). Physiological and biochemical response of cowpea (Vigna unguiculata L.) landraces of Kashmir valley under water stress. Electronic Journal of Plant Breeding, 10(4), 1461–1470.
Hammer, G. L., Dong, Z., McLean, G., Doherty, A., Messina, C., Schussler, J., et al. (2009). Can changes in canopy and/or root system architecture explain historical maize yield trends in the US corn belt? Crop Science, 49(1), 299–312.
Hardarson, G., Bliss, F. A., Cigales-Rivero, M. R., Henson, R. A., Kipe-Nolt, J. A., Longeri, L., et al. (1993). Genotypic variation in biological nitrogen fixation by common bean. Plant and Soil, 152, 59–70.
Harris, K., Subudhi, P. K., Borrell, A., Jordan, D., Rosenow, D., Nguyen, H., et al. (2007). Sorghum stay-green QTL individually reduce post-flowering drought-induced leaf senescence. Journal of Experimental Botany, 58(2), 327–338.
Hatfield, J. L., Boote, K. J., Kimball, B. A., Ziska, L. H., Izaurralde, R. C., Ort, D., et al. (2011). Climate impacts on agriculture: implications for crop production. Agronomy Journal, 103, 351–370.
Hetherington, A. M., & Woodward, F. I. (2003). The role of stomata in sensing and driving environmental change. Nature, 424(6951), 901.
Hund, A., Ruta, N., & Liedgens, M. (2009). Rooting depth and water use efficiency of tropical maize inbred lines, differing in drought tolerance. Plant and Soil, 318(1–2), 311–325.
Hurd, E. A. (1974). Phenotype and drought tolerance in wheat. Agricultural Meteorology, 14(1–2), 39–55.
Jenkins, M. B., Virginia, R. A., & Jarrel, W. M. (1987). Ecology of fast-growing and slow-growing mesquite-nodulating rhizobia in Chihuahua and Sonoran desert ecosystems. Soil Science Society of America Journal, 53, 543–549.
Jordan, D. R., Hunt, C. H., Cruickshank, A. W., Borrell, A. K., & Henzell, R. G. (2012). The relationship between the stay-green trait and grain yield in elite sorghum hybrids grown in a range of environments. Crop Science, 52(3), 1153–1161.
Kashiwagi, J., Krishnamurthy, L., Crouch, J. H., & Serraj, R. (2006). Variability of root length density and its contributions to seed yield in chickpea (Cicer arietinum L.) under terminal drought stress. Field Crops Research, 95, 171–181.
Katungi, E., Farrow, A., Mutuoki, T., Gebeyehu, S., Karanja, D., Alamayehu, F., Buruchara, R. (2010). Improving common bean productivity: An analysis of socioeconomic factors in Ethiopia and Eastern Kenya. In: Baseline report tropical legumes II. Centro Internacional de Agricultura Tropical-CIAT, Cali, Colombia.
Khalid, R. (2017). Root architecture and biomass partitioning in relation to drought stress in common bean. M.Sc thesis submitted to SKUAST-K (173 pp).
Khan, H. U. R., Link, W., Hocking, T. J., & Stoddard, F. L. (2007). Evaluation of physiological traits for improving drought tolerance in faba bean (Vicia faba L.). Plant and Soil, 292(1), 205–217.
Knepper, C., & Mou, B. (2015). Semi-High Throughput screening for potential drought-tolerance in lettuce (Lactuca sativa) germplasm collections. Journal of Visualized Experiments : JoVE, 98, 52492.
Kulkarni, A., Gadgil, S., & Patwardhan, S. (2016). Monsoon variability, the 2015 Marathwada drought and rainfed agriculture. Current Science, 111, 1182–1193.
Kumar, J., & Abbo, S. (2001). Genetics of flowering time in chickpea and its bearing on productivity in semiarid environments. Advances in Agronomy, 72, 107–138.
Lawlor, D. W., & Cornic, G. (2002). Photosynthetic carbon assimilation and associated metabolism in relation to water deficits in higher plants. Plant, Cell and Environment, 25(2), 275–294.
Lioi, L., & Piergiovanni, A. R. (2013). European common bean. In M. Singh, H. D. Upadhyaya, & I. S. Bisht (Eds.), Genetic and genomic resources of grain legume improvement (pp. 11–40). Oxford: Elsevier.
Lopes, M. S., & Reynolds, M. P. (2010). Partitioning of assimilates to deeper roots is associated with cooler canopies and increased yield under drought in wheat. Functional Plant Biology, 37(2), 147–156.
Ludlow, M. M., & Muchow, R. C. (1990). A critical evaluation of traits for improving crop yields in water-limited environments1. In N. C. Brady (Ed.), Advances in agronomy (Vol. 43, pp. 107–153). New York: Academic Press.
Lugojan, C., & Ciulca, S. (2011). Evaluation of relative water content in winter wheat. Journal of Horticulture, Forestry and Biotechnology, 15(2), 173–177.
Lynch, J. P. (2011). Root phenes for enhanced soil exploration and phosphorus acquisition: Tools for future crops. Plant Physiology, 156, 1041–1049.
Lynch, J. P. (2018). Rightsizing root phenotypes for drought resistance. Journal of Experimental Botany, 69(13), 3279–3292.
Mace, E. S., Singh, V., Van Oosterom, E. J., Hammer, G. L., Hunt, C. H., & Jordan, D. R. (2012). QTL for nodal root angle in sorghum (Sorghum bicolor L. Moench) co-locate with QTL for traits associated with drought adaptation. Theoretical and Applied Genetics, 124, 97–109.
Mahan, F., Isazadeh, A., & Khanli, L. M. (2011). Using an active fuzzy ECA rule-based negotiation agent in e-commerce. International Journal of Electronic Commerce Studies, 2(2), 127–148.
Makunde, G. S., Beebe, S., Blair, M. W., Chirwa, R., & Lungu, D. (2007). Inheritance of drought tolerance traits in Andean x Andean and Andean x Mesoamerican F2 populations. Annual Report-Bean Improvement Cooperative, 50, 159.
Mallya, G., Mishra, V., Niyogi, D., Tripathi, S., & Govindaraju, R. S. (2016). Trends and variability of droughts over the Indian monsoon region. Weather Climate Extremes, 12, 43–68.
Manschadi, A. M., Christopher, J., & Hammer, G. L. (2006). The role of root architectural traits in adaptation of wheat to water-limited environments. Functional Plant Biology, 33(9), 823–837.
Manske, G. G., & Vlek, P. L. (2002). Root architecture–wheat as a model plant. Plant roots: The Hidden Half, 3, 249–259.
Marshall, A. H., Collins, R. P., Humphreys, M. W., & Scullion, J. (2016). A new emphasis on root traits for perennial grass and legume varieties with environmental and ecological benefits. Food and energy security, 5(1), 26–39.
Masle, J., Gilmore, S. R., & Farquhar, G. D. (2005). The ERECTA gene regulates plant transpiration efficiency in Arabidopsis. Nature, 436(7052), 866–870.
Matin, M. A., Brown, J. H., & Ferguson, H. (1989). Leaf water potential, relative water-content, and diffusive resistance as screening techniques for drought resistance in barley. Agronomy Journal, 81, 100–105.
Maxwell, K., & Johnson, G. N. (2000). Chlorophyll fluorescence—a practical guide. Journal of Experimental Botany, 51(345), 659–668.
McCree, K. J. (1986). Whole-plant carbon balance during osmotic adjustment to drought and salinity stress. Functional Plant Biology, 13(1), 33–43.
Medrano, H., Escalona, J. M., Bota, J., Gulías, J., & Flexas, J. (2002). Regulation of photosynthesis of C3 plants in response to progressive drought: Stomatal conductance as a reference parameter. Annals of Botany, 89(7), 895–905.
Meng, X., Yin, B., Feng, H. L., Zhang, S., Liang, X. Q., & Meng, Q. W. (2014). Overexpression of R2R3-MYB gene leads to accumulation of anthocyanin and enhanced resistance to chilling and oxidative stress. Biologia Plantarum, 58(1), 121–130.
Mhadhbi, H., Chihaoui, S., Mhamdi, R., Mnasri, B., Jebara, M., & Mhamdi, R. (2011). A highly osmotolerant rhizobial strain confers a better tolerance of nitrogen fixation and enhances protective activities to nodules of Phaseolus vulgaris under drought stress. African Journal of Biotechnology, 10, 4555–4563.
Mir, R. R., Zaman-Allah, M., Sreenivasulu, N., Trethowan, R., & Varshney, R. K. (2012). Integrated genomics, physiology and breeding approaches for improving drought tolerance in crops. Theoretical and Applied Genetics, 125(4), 625–645.
Mishra, A., & Liu, S. C. (2014). Changes in precipitation pattern and risk of drought over India in the context of global warming. Journal of Geophysics and Research on Atmosphere., 119, 7833–7841.
Miyashita, K., Tanakamaru, S., Maitani, T., & Kimura, K. (2005). Recovery responses of photosynthesis, transpiration, and stomatal conductance in kidney bean following drought stress. Environmental and experimental botany, 53(2), 205–214.
Mohammadi, R., Armion, M., Kahrizi, D., & Amri, A. (2012). Efficiency of screening techniques for evaluating durum wheat genotypes under mild drought conditions. International Journal of Plant Production, 4(1), 11–24.
Monneveux, P., Sanchez, C., & Tiessen, A. (2008). Future progress in drought tolerance in maize needs new secondary traits and cross combinations. Journal of Agricultural Science, 146(3), 287–300.
Moussa, H. R., & Abdel-Aziz, S. M. (2008). Comparative response of drought tolerant and drought sensitive maize genotypes to water stress. Australian Journal of Crop Science, 1(1), 31–36.
Noctor, G., Veljovic-Jovanovic, S. O. N. J. A., Driscoll, S., Novitskaya, L., & Foyer, C. H. (2002). Drought and oxidative load in the leaves of C3 plants: A predominant role for photorespiration? Annals of Botany, 89(7), 841–850.
O’Brien, L. (1979). Genetic variability of root growth in wheat (Triticum aestivum L.). Australian Journal of Agricultural Research, 30(4), 587–595.
Omae, H., Kumar, A., Kashiwaba, K., & Shono, M. (2005). Influence of level and duration of high temperature treatments on plant water status in snap bean (Phaseolus vulgaris L.). Japanese Journal of Tropical Agriculture, 49(3), 238–242.
Osmont, K. S., Sibout, R., & Hardtke, C. S. (2007). Hidden branches: Developments in root system architecture. Annual Review of Plant Biology, 58, 93–113.
Palta, J. A., Chen, X., Milroy, S. P., Rebetzke, G. J., Dreccer, M. F., & Watt, M. (2011). Large root systems: are they useful in adapting wheat to dry environments? Functional Plant Biology, 38(5), 347–354.
Pask, A. J. D., & Reynolds, M. P. (2013). Breeding for yield potential has increased deep soil water extraction capacity in irrigated wheat. Crop Science, 53(5), 2090–2104.
Peksen, E., Peksen, A., & Artik, C. (2006). Comparison of leaf and stomatal characteristics in faba bean (Vicia faba L.). Journal of Biological Sciences, 6, 360–364.
Pillitteri, L. J., & Torii, K. U. (2012). Mechanisms of stomatal development. Annual Review of Plant Biology, 63, 591–614.
Pinto, R. S., & Reynolds, M. P. (2015). Common genetic basis for canopy temperature depression under heat and drought stress associated with optimized root distribution in bread wheat. Theoretical and Applied Genetics, 128(4), 575–585.
Polania, J. A., Poschenrieder, C., Beebe, S., & Rao, I. M. (2016). Effective use of water and increased dry matter partitioned to grain contribute to yield of common bean improved for drought resistance. Frontiers in Plant Science, 7, 660.
Porch, T. G., & Jahn, M. (2001). Effects of high-temperature stress on microsporogenesis in heat-sensitive and heat-tolerant genotypes of Phaseolus vulgaris. Plant, Cell and Environment, 24(7), 723–731.
Prakash, S. (2018). Capabilities of satellite-derived datasets to detect consecutive Indian monsoon droughts of 2014 and 2015. Current Science (00113891), 114(11), 2362–2368.
Price, A. H., Cairns, J. E., Horton, P., Jones, H. G., & Griffiths, H. (2002). Linking drought-resistance mechanisms to drought avoidance in upland rice using a QTL approach: Progress and new opportunities to integrate stomatal and mesophyll responses. Journal of Experimental Botany, 53(371), 989–1004.
Purcell, L. C. (2009). Physiological responses of N2 fixation to drought and selecting genotypes for improved N2 fixation. In H. B. Krishna & D. W. Emerich (Eds.), Nitrogen fixation in crop production, Agronomy Monograph (Vol. 52, pp. 211–238). Madison: ASA CSSA SSSA.
Purushothaman, R., Krishnamurthy, L., Upadhyaya, H. D., Vadez, V., & Varshney, R. K. (2017). Shoot traits and their relevance in terminal drought tolerance of chickpea (Cicer arietinum L.). Field Crop Research, 197, 10–27.
Rao, I. M. (2014). Advances in improving adaptation of common bean and Brachiaria forage grasses to abiotic stress in the tropics. In M. Pessarakli (Ed.), Handbook of plant and crop physiology (pp. 847–889). New York: CRC Press, Taylor and Francis.
Rashid, A., Stark, J. C., Tanveer, A., & Mustafa, T. (1999). Use of canopy temperature measurements as a screening tool for drought tolerance in spring wheat. Journal of Agronomy and Crop Science, 182(4), 231–238.
Reynolds, M., & Langridge, P. (2016). Physiological breeding. Current Opinion in Plant Biology, 31, 162–171.
Reynolds, M., Manes, Y., Izanloo, A., & Langridge, P. (2009). Phenotyping approaches for physiological breeding and gene discovery in wheat. Annals of Applied Biology, 155(3), 309–320.
Reynolds, M. P., Pierre, C. S., Saad, A. S., Vargas, M., & Condon, A. G. (2007). Evaluating potential genetic gains in wheat associated with stress-adaptive trait expression in elite genetic resources under drought and heat stress. Crop Science, 47, S-172.
Riaz, A. T. I. F., Younis, A., Taj, A. R., Karim, A., Tariq, U., Munir, S., & Riaz, S. I. T. W. A. T. (2013). Effect of drought stress on growth and flowering of marigold (Tagetes erecta L.). Pakistan Journal of Botany, 45(S1), 123–131.
Richards, R. A., & Passioura, J. B. (1989). A breeding program to reduce the diameter of the major xylem vessel in the seminal roots of wheat and its effect on grain yield in rain-fed environments. Australian Journal of Agricultural Research, 40(5), 943–950.
Ritchie, S. W., Nguyan, H. T., & Holaday, A. S. (1990). Leaf Water content and gas exchange parameters of two wheat genotypes differing in drought resistance. Crop Science, 30, 105–111.
Rosales-Serna, R., Kohashi-Shibata, J., Acosta-Gallegos, J. A., Trejo-López, C., Ortiz-Cereceres, J., & Kelly, J. D. (2004). Biomass distribution, maturity acceleration and yield in drought-stressed common bean cultivars. Field Crops Research, 85(2–3), 203–211.
Ruan, Y.-L., Patrick, J. W., Bouzayen, M., Osorio, S., & Fernie, A. R. (2012). Molecular regulation of seed and fruit set. Trends in Plant Science, 17, 656–665.
Rubiales, D., & Mikic, A. (2015). Introduction: legumes in sustainable agriculture. Critical Reviews in Plant Science, 34, 2–3.
Saurez, R., Wong, A., Ramírez, M., Barraza, A., Orozco, M. D. C., Cevallos, M. A., et al. (2008). Improvement of drought tolerance and grain yield in common bean by overexpressing trehalose-6-phosphate synthase in rhizobia. Molecular Plant-Microbe Interactions, 21(7), 958–966.
Schneider, K. A., Brothers, M. E., & Kelly, J. D. (1997). Marker-assisted selection to improve drought resistance in common bean. Crop Science, 37(1), 51–60.
Schoppach, R. M., Wauthelet, D., Jeanguenin, L., & Sadok, W. (2013). Conservative water use under high evaporative demand associated with smaller root metaxylem and limited trans-membrane water transport in wheat. Functional Plant Biology, 41, 257–269.
Serraj, R., Buhariwalla, H. K., Sharma, K. K., Gaur, P. M., & Crouch, J. H. (2004a). Crop improvement of drought resistance in pulses: A holistic approach. Indian Journal of Pulse Research, 17, 1–13.
Serraj, R., Krishnamurthy, L., Kashiwagi, J., Kumar, J., Chandra, S., & Crouch, J. H. (2004b). Variation in root traits of chickpea (Cicer arietinum L.) grown under terminal drought. Field Crops Research, 88(2–3), 115–127.
Serraj, R., Sinclair, T. R., & Purcell, L. C. (1999). Symbiotic N2 fixation response to drought. Journal of Experimental Botany, 50(331), 143–155.
Shoushtari, N. H., & Pepper, I. L. (1985). Mesquite rhizobia isolated from the Sonoran desert: Competitiveness and survival in soil. Soil Biology and Biochemistry, 17, 803–806.
Sinclair, T. R., Devi, J., Shekoofa, A., Choudhary, S., Sadok, W., Vadez, V., & Rufty, T. (2017). Limited-transpiration response to high vapor pressure deficit in crop species. Plant Science, 260, 109–118.
Sinclair, T. R., & Ludlow, M. M. (1985). Who taught plants thermodynamics? The unfulfilled potential of plant water potential. Functional Plant Biology, 12(3), 213–217.
Sinclair, T. R., Messina, C. D., Beatty, A., & Samples, M. (2010a). Assessment across the United States of the benefits of altered soybean drought traits. Agronomy Journal, 102(2), 475–482.
Sinclair, T. R., Messina, C. D., Beatty, A., & Samples, M. (2010b). Assessment across the United States of the benefits of altered soybean drought traits. Agronomy Journal, 102, 475–482.
Sinclair, T. R., & Rufty, T. W. (2012). Nitrogen and water resources commonly limit crop yield increases, not necessarily plant genetics. Global Food Security, 1(2), 94–98.
Sinclair, T. R., Tanner, C. B., & Bennett, J. M. (1984). Water-use efficiency in crop production. BioScience, 34(1), 36–40.
Singh, S. P. (1995). Selection for water-stress tolerance in interracial populations of common bean. Crop Science, 35(1), 118–124.
Singh, S. P. (2007). Drought resistance in the race Durango dry bean landraces and cultivars. Agronomy journal, 99(5), 1219–1225.
Smith, L. T., Pocard, J. A., Bernard, T., & Le Rudulier, D. (1988). Osmotic control of glycine betaine biosynthesis and degradation in Rhizobium meliloti. Journal Bacteriology, 70, 3142–3149.
Smith, M. R., Veneklaas, E., Polania, J., Rao, I. M., Beebe, S. E., & Merchant, A. (2019). Field drought conditions impact yield but not nutritional quality of the seed in common bean (Phaseolus vulgaris L.). PLoS ONE, 14(6), e0217099.
Sofi, P. A., Baba, Z. A., Hamid, B., & Meena, R. S. (2018a). Harnessing soil Rhizobacteria for improving drought resilience in legumes. In R. S. Meena, A. Das, G. S. Yadav, R. Lal (Eds.), Legumes for soil health and sustainable management (pp. 235–275). Singapore: Springer.
Sofi, P. A., Djanaguiraman, M., Siddique, K. H. M., & Prasad, P. V. V. (2018b). Reproductive fitness in common bean (Phaseolus vulgaris L.) under drought stress is associated with root length and volume. Indian Journal of Plant Physiology, 23(4), 796–809.
Sofi, P. A., Saba, I., & Amin, Z. (2017). Root architecture and rhizobial inoculation in relation to drought stress response in common bean (Phaseolus vulgaris L.). Journal of Applied and Natural Science, 9(1), 502–507.
Sponchiado, B. N., White, J. W., Castillo, J. A., & Jones, P. G. (1989). Root growth of four common bean cultivars in relation to drought tolerance in environments with contrasting soil types. Experimental Agriculture, 25, 249–257.
Subrahmanyam, D., Subash, N., Haris, A., & Sikka, A. K. (2006). Influence of water stress on leaf photosynthetic characteristics in wheat cultivars differing in their susceptibility to drought. Photosynthetica, 44(1), 125.
Swaine, E. K., Swaine, M. D., & Killham, K. (2007). Effects of drought on isolates of Bradyrhizobium elkanii cultured from Albizia adianthifolia seedlings of different provenances. Agroforestry Systems, 69(2), 135–145.
Tardieu, F., Simonneau, T., & Muller, B. (2018). The physiological basis of drought tolerance in crop plants: a scenario-dependent probabilistic approach. Annual Review of Plant Biology, 69, 733–759.
Teran, J. C., Konzen, E. R., Medina, V., Palkovic, A., Ariani, A., Tsai, S. M., et al. (2019). Root and shoot variation in relation to potential intermittent drought adaptation of Mesoamerican wild common bean (Phaseolus vulgaris L.). Annals of Botany, 124, 917–932. https://doi.org/10.1093/aob/mcy221.
Thomas, H., & Howarth, C. J. (2000). Five ways to stay green. Journal of Experimental Botany, 51(supp_1), 329–337.
Tricker, P. J., ElHabti, A., Schmidt, J., & Fleury, D. (2018). The physiological and genetic basis of combined drought and heat tolerance in wheat. Journal of Experimental Botany, 69(13), 3195–3210.
Tuberosa, R. (2012). Phenotyping for drought tolerance of crops in the genomics era. Frontiers in Physiology, 3, 347.
Turc, O., & Tardieu, F. (2018). Drought affects abortion of reproductive organs by exacerbating developmentally driven processes via expansive growth and hydraulics. Journal of Experimental Botany, 69(13), 3245–3254.
Turner, N. C., Wright, G. C., & Siddique, K. H. M. (2001). Adaptation of grain legumes (pulses) to water-limited environments. Advances in Agronomy, 71, 193–231.
Uga, Y., Sugimoto, K., Ogawa, S., Rane, J., Ishitani, M., Hra, N., & Inoue, H. (2013). Control of root system architecture by DEEPER ROOTING 1 increases rice yield under drought conditions. Nature Genetics, 45(9), 1097.
Urban, O., Janous, D., Acosta, M., Czerny, R., Marková, I., Navratil, M., et al. (2007). Ecophysiological controls over the net ecosystem exchange of mountain spruce stand. Comparison of the response in direct vs. diffuse solar radiation. Global Change Biology, 13, 157–168.
Vadez, V., Deshpande, S. P., Kholova, J., Hammer, G. L., Borrell, A. K., Talwar, H. S., & Hash, C. T. (2011). Stay-green quantitative trait loci’s effects on water extraction, transpiration efficiency and seed yield depend on recipient parent background. Functional Plant Biology, 38(7), 553–566.
Vadez, V., Kholová, J., Medina, S., Kakkera, A., & Anderberg, H. (2014). Transpiration efficiency: New insights into an old story. Journal of Experimental Botany, 65, 6141–6153. https://doi.org/10.1093/jxb/eru040.
Vadez, V., Lasso, J. H., Beck, D. P., & Drevon, J. J. (1999). Variability of N2-fixation in common bean (Phaseolus vulgaris L.) under P deficiency is related to P use efficiency. Euphytica, 106(3), 231–242.
Vadez, V., Rao, S., Kholova, J., Krishnamurthy, L., Kashiwagi, J., Ratnakumar, P., et al. (2008). Root research for drought tolerance in legumes: Quo vadis? Journal of Food Legumes, 21, 77–85.
van Schoonhoven, A., & Voysest, O. (1989). Common beans in Latin America and their constraints (pp. 33–57). Colombia: Bean Production problems in tropics. Centro Internacional de Agrícultura Tropical. Cali.
Varshney, R. K. (2016). Exciting journey of 10 years from genomes to fields and markets: Some success stories of genomics-assisted breeding in chickpea, pigeonpea and groundnut. Plant Science, 242, 98–107.
Varshney, R. K., Tuberosa, R., & Tardieu, F. (2018). Progress in understanding drought tolerance: From alleles to cropping systems. Journal of Experimental Botany, 69(13), 3175–3179.
Vaz Patto, M. C., Amarowicz, R., Aryee, A. N., Boye, J. I., Chung, H. J., Martín-Cabrejas, M. A., et al. (2015). Achievements and challenges in improving the nutritional quality of food legumes. Critical Reviews in Plant Sciences, 34, 105–143.
Vyas, S. P. (2014). Impact and strategies for yield improvement of arid legumes under drought. International Journal of Applied Life Science Engineering, 1, 12–19.
Wang, A.-Y., Li, Y., & Zhang, C.-Q. (2012). QTL mapping for stay-green in maize Zea mays. Canadian Journal of Plant Sciences, 92, 249–256.
White, J., & Singh, S. P. (1991). Physiology of yield potential and stress potential. In: Common bean: Research for crop improvement. In: CABI (pp. 287–383).
White, J. W., & Castillo, J. A. (1992). Evaluation of diverse shoot genotypes on selected root genotypes of common bean under soil water deficits. Crop Science, 32, 762–765.
White, J. W., Castillo, J. A., & Ehleringer, J. R. (1990). Associations between productivity, root growth and carbon isotope discrimination in Phaseolus vulgaris under water deficit. Australian Journal of Plant Physiology, 17, 189–198.
Williams, J., Bulman, M., Huttly, A., Phillips, A., & Neill, S. (1994). Characterization of a cDNA from Arabidopsis thaliana encoding a potential thiol protease whose expression is induced independently by wilting and abscisic acid. Plant Molecular Biology, 25, 259–270.
Wortmann, C.S., R.A. Kirkby, C.A.E. and, and D.J. Allen. 1998. Atlas of common bean (Phaseolus vulgaris L.) production in Africa (Vol. 297). Centro Internacional de Agricultura Tropical, CIAT Publication, Cali.
Ye, H., Roorkiwal, M., Valliyodan, B., Zhou, L., Chen, P., Varshney, R. K., & Nguyen, H. T. (2018). Genetic diversity of root system architecture in response to drought stress in grain legumes. Journal of Experimental Botany, 69(13), 3267–3277.
Zaman-Allah, M., Jenkinson, D. M., & Vadez, V. (2011). A conservative pattern of water use, rather than deep or profuse rooting, is critical for the terminal drought tolerance of chickpea. Journal of Experimental Botany, 62, 4239–4252.
Zheng, H. J., Wu, A. Z., Zheng, C. C., Wang, Y. F., Cai, R., Shen, X. F., & Dong, S. T. (2009). QTL mapping of maize (Zea mays) stay-green traits and their relationship to yield. Plant Breeding, 128(1), 54–62.
Zhu, J., Brown, K. M., & Lynch, J. P. (2010). Root cortical aerenchyma improves the drought tolerance of maize (Zea mays L.). Plant Cell and Environment, 33(5), 740–749.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sofi, P.A., Rehman, K., Gull, M. et al. Integrating root architecture and physiological approaches for improving drought tolerance in common bean (Phaseolus vulgaris L.). Plant Physiol. Rep. 26, 4–22 (2021). https://doi.org/10.1007/s40502-021-00570-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40502-021-00570-8