Abstract
Purpose of Review
Typhoid fever is one of the commonest global health burdens and has various local and systemic complications. The clinical picture may be more complex in concealed, untreated, or partially treated patients, which form a significant proportion of total cases. So, in this study, we aimed to identify recent and unique intestinal and extra-intestinal complications associated with typhoid fever.
Recent Findings
We searched the terms “Typhoid/Enteric fever AND complication” and “Salmonella Typhi AND complication” in online libraries like PubMed, Scopus, and Embase and collected the recent studies. A total of 45,078 studies were identified, and 42 were included in this study. Searches from various databases suggested that among intestinal complications, bleeding and perforation are more prevalent than other complications like acute pancreatitis, hepatic failure, and acalculus cholecystitis. Similarly, neurological complications, including delirium, are more prevalent than cardiac and respiratory system involvement among extra-intestinal features. The concurrent presence of different intestinal and extra-intestinal complexities is common and requires long-term management.
Summary
Typhoid fever has numerous complications, whose prompt identification and timely management are desirable to decrease morbidity and mortality.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
A bacterial infection with Salmonella typhi causes typhoid or enteric fever. The disease is present in most countries around the world, but it is a public health concern in developing countries, particularly those with poor sanitation and overcrowding. Mogasale et al. [1] estimated that in 2010, the number of cases in low- and middle-income countries of Asia, Africa, and South America was 11.9 million with 129,000 deaths. Similarly, a recent study from India estimated the typhoid fever incidence of 360 cases per 100,000 person-years, with a case fatality rate of 0.2% [2•].
Methodology
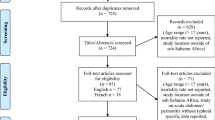
For this study, we searched the studies that discussed/reported complications of typhoid fever, and used the database libraries like PubMed, Scopus, and Embase with Boolean operator. The terms used for search include “Typhoid/Enteric fever AND complication” and “Salmonella Typhi AND complication,” and Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines were followed. Only articles in English language were selected, and we considered observational studies, review articles, and unique case reports. Recent publications were preferred over the older ones, and no articles before the year 2000 were included. Similarly, literature on animals and with inconclusive diagnostic results was also excluded. With this, a total of 45,078 studies were identified, and 42 were included in this report (Fig. 1).
Pathophysiology
Typhoid fever is acquired by the oral route, and the incubation period varies inversely with the dose of S. typhi ingested, ranging from 3 days to 2 months [3]. After ingestion, the bacterium gets established in Peyer’s patches of the small intestine due to resistance to gastric acid, and from there, lymphatic and hematogenous dissemination occurs. Further replication occurs within the reticuloendothelial system. Intestinal hemorrhage and subsequent perforation can be seen due to the hypertrophy and necrosis of Peyer’s patches. Alternatively, chronic intestinal carriage can occur without systemic spread, and carriers can release the bacilli via feces or urine asymptomatically. The clinical features in an untreated patient follow a stereotypical timeline and are summarized in Table 1 [4]. All symptoms may not necessarily be seen in a single patient.
Diagnosis
Diagnosis can be ascertained by blood culture, which carries sensitivity of 50 to 70% [5]. Although the yield of bone marrow culture is more than 90%, it is rarely used clinically due to its invasive nature. Cultures of stool and urine are less sensitive. All other modalities, including serology by enzyme-linked immunosorbent assay (ELISA) and polymerase chain reaction (PCR), are less accurate. The primordial Widal test that detects antibodies against the O and H antigens can be diagnostic in over three-quarters of patients when convalescent serum demonstrates a fourfold rise in titer over the acute phase serum [4].
Treatment
The treatment of typhoid fever has been challenging in the recent past due to emergence of drug resistance. Chloramphenicol, amoxicillin, and trimethoprim-sulfamethoxazole are no longer unfailing [6]. Fluoroquinolone resistance has also emerged in some Asian countries. Azithromycin and the third-generation cephalosporins, ceftriaxone, and cefotaxime, are effective in most of the world. The duration of therapy varies from 10 to 14 days. An extensively drug-resistant strain, which responds only to carbapenems and azithromycin, has been reported to be prevalent in Pakistan since 2016 [7••]. The carriers, who are identified in screening programs, are treated with ciprofloxacin, amoxicillin, or trimethoprim-sulfamethoxazole. Live oral and injectable polysaccharide vaccines are available. They are recommended in endemic regions and in travelers. The efficacy is in the range of 50–60% [8].
Complications
Complications in typhoid fever may be encountered in about 10–15% cases [9]. S. typhi is not an innocuous pathogen that afflicts the intestines only, as is suggested by the term “enteric fever.” The disease has a potential to afflict all the organ systems in the body. Atypical manifestations can lead to diagnostic uncertainty and, thus, a considerable delay in the diagnosis. This review analysis has been performed to highlight the spectrum of complications and discuss their pathogenesis. For the purpose of this study, the complications have been divided into intestinal (Table 2) and extra-intestinal (Table 3) [10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40].
Intestinal Complications
The most common complications of typhoid fever include abdominal organ system, which can be explained by its route of infection acquisition and pathophysiological involvement of small intestine. Due to the highest number of Peyer’s patches in the terminal ileum, perforation of this segment is common in 3–4 weeks of disease and requires surgical intervention in most cases [41]. Further, other abdominal organ involvements have also been documented (Table 2).
The liver involvement is almost universal [42]. In a study, raised alanine transaminase (ALT) and aspartate transaminase (AST) were found in 85% and 75% of the cases, respectively. In the same study, hepatomegaly was seen in 52%, and jaundice was noticeable in 13% of the cases. However, marked elevations of AST and ALT, mimicking acute viral hepatitis, and acute liver failure have also been reported [18, 20].
The reflux of S. typhi containing bile into the pancreatic duct, leading to direct injury, is the postulated mechanism for the development of acute pancreatitis [43]. Also, the direct effect of bacterial toxins and the host immune response are other possibilities. It is not uncommon to have elevated levels of amylase and lipase without any clinical evidence of pancreatitis in patients with typhoid fever. This was confirmed by Das et al. [44], who studied 47 enteric fever patients and found that while 44% of the study subjects had elevated serum enzyme levels, none of them had acute pancreatitis, and this complication did not adversely affect the outcome.
In a recent review including 10,355 typhoid fever cases, it was found that the incidence of intestinal perforation and gastrointestinal bleeding was 1.3% and 3.1%, respectively [45•]. Typhoid fever is a very rare cause of splenic abscess. There can be a single or multiple abscesses, and the management needs to be individualized. Small abscesses can be managed with antibiotics alone, while larger ones can require percutaneous aspiration, pig tail drainage, or splenectomy [46].
Extra-intestinal Complications
Several extra-intestinal complications have been linked to typhoid fever, and almost every organ system involvement has been described. Among these, central nervous system complications are most common (3–35%), followed by pulmonary (1–6%) and cardiovascular system (1–5%) [47]. Other uncommon complexities involve hematological, soft-tissue, joint and bone, and genitourinary system (Table 3). The potential mechanism for extra-intestinal complication includes Salmonella typhi bacteremia.
A spectrum of neurological manifestations is diverse, and many syndromes affecting the central and peripheral nervous systems have been reported. These include aseptic meningitis, metastatic brain abscesses, cerebellar dysfunction, retinitis, myelopathy, neuropathy, pseudo-tumor cerebri, and cranial nerve palsies [14, 32, 35]. In a large observational study, delirium was seen in 42.8%, encephalitis in 25%, polyneuropathy in 8.3%, and extrapyramidal signs in 5.5% of the cases [48]. A type of encephalopathy called “muttering delirium” or “coma vigil,” characterized by picking at clothes or imaginary objects, can occur when typhoid fever is not treated within 2 weeks of onset. It is rapidly reversible with treatment [40].
The involvement of lungs can be seen in the form of pneumonia, empyema, and acute respiratory distress syndrome (ARDS) [30, 37]. A simple cough may be seen in up to 86% of patients [49]. Less commonly, bronchopleural fistulas and pulmonary embolism have also been reported for typhoid fever. These patients usually have previous lung abnormalities or underlying immunocompromised state [47]. Fluoroquinolones, along with ceftriaxone for a period of 2–3 weeks, are recommended for the management of associated pulmonary manifestations.
The cardiac involvement can be seen in up to 4% of all cases in the form of myocarditis or pericarditis [50]. It has been recommended that all patients with Salmonella septicemia be screened for cardiovascular complications [51]. The optimal duration of treatment has not been defined. In their review, Robson et al. [39] found both mitral and aortic valve involvement in native, diseased, and prosthetic valve endocarditis. The treatment duration varied from 4 to 8 weeks.
In their review, Khan et al. [10] found that the renal involvement is very rare and occurs in only 2–3% of patients with typhoid fever. The manifestations can be in the form of cystitis, pyelonephritis, glomerulonephritis, acute tubular necrosis, and acute interstitial nephritis. The pathogenesis is multifactorial and can be a result from direct invasion by the bacilli or from the effect of its toxin [52]. Glomerulonephritis is the result of an autoimmune phenomenon, and diffuse proliferative glomerulonephritis has been seen on renal biopsy [14].
Acute kidney injury (AKI) can also be secondary to sepsis, hypovolemia, rhabdomyolysis, and acute interstitial nephritis (AIN) secondary to the infection or due to drugs used during treatment [10]. AKI occurs in almost all the cases with rhabdomyolysis, and it is managed with prompt administration of intravenous fluids and maintenance of electrolyte balance, particularly hyperkalemia. The pathogenesis of rhabdomyolysis in typhoid fever involves tissue hypoxia, invasion of muscles by the bacteria, and lysosomal enzyme activation. Early institution of hemodialysis is necessary in certain cases, but generally, good recovery of renal function is seen.
Drug-induced AIN requires a high index of suspicion, and the diagnosis should be considered when there is no other apparent cause of AKI and the timing coincides with the initiation of antibiotic therapy. It should be substituted with an alternate antibiotic belonging to a different class. Corticosteroids may be indicated in certain severe cases. The diagnosis can be established by renal biopsy, but it is generally avoided.
Prevention and Vaccines
Low- and middle-income countries have more prevalence of typhoid fever due to poor sanitation and unavailability of safe water. Literature have documented that the transmission risk of S. typhi is almost double in the population exposed to unsafe water [53]. Additionally, overcrowding is also identified as a contributing factor for the communication of this infectious disease, and a study from India found that 14% of the family members of confirmed typhoid children cases shed S. typhi in stool [54]. Thus, proper management and elimination of these modifiable risk factors can reduce the overall global typhoid fever burden [55].
Three major vaccine categories, i.e., Vi-polysaccharide (Vi-PS, parenteral), Ty21a (oral), and typhoid conjugate vaccines (TCVs, parenteral), have been utilized as a control measure for typhoid fever. Vi-PS is a subunit vaccine, which provides 30–70% protection efficacy. A similar efficacy is also provided by Ty21a live vaccine, but has contraindication for pregnant and immunocompromised patients. Both of these vaccines have short-term protection and require booster dose administration at regular intervals. To overcome these burdens, new-generation TCVs are being used, which has an efficacy of > 80% in most clinical trials [55]. These vaccines have the longest protection duration theoretically, and currently, no booster dose is recommended, but studies are being conducted on various TCVs for the requirement of more than one dose [56]. Introduction of TCVs in routine immunization of 9-month to 15-year-old children can reduce the disease burden in a subsequent 10-year time frame [55].
Conclusion
Having a vast spectrum of complications, typhoid fever may have variable presentation. Most of the patients experience minor abdominal symptoms like pain, diarrhea, and anorexia, but grave complications like intestinal bleeding and perforation, acute pancreatitis, and hepatic failure are not uncommon, especially in untreated patients. These patients may also demonstrate concurrent extra-abdominal complications, and nervous system involvement is most common among these. Delirium, cerebellar dysfunction, neuropathy, and myelopathy in the nervous system; cough, pneumonia, and pleuritis in the pulmonary system; and pericarditis and myocarditis in the cardiac system are common manifestations. These complications potentially increase morbidity and mortality and require prolonged antibiotic institution.
Code Availability
Not applicable.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Mogasale V, Maskery B, Ochiai RL, Lee JS, Mogasale VV, Ramani E, et al. Burden of typhoid fever in low-income and middle-income countries: a systematic, literature-based update with risk-factor adjustment. Lancet Glob Health. 2014;2:e570–80. https://doi.org/10.1016/S2214-109X(14)70301-8.
• Cao Y, Karthikeyan AS, Ramanujam K, Raju R, Krishna S, Kumar D, et al. Geographic pattern of typhoid fever in India: a model-based estimate of cohort and surveillance data. J Infect Dis. 2021;224:S475-S483. https://doi.org/10.1093/infdis/jiab187The authors done an extensive work to find out the prevalence of typhoid fever across the country and provided the data of different regions.
Waddington CS, Darton TC, Woodward WE, Angus B, Levine MM, Pollard AJ. Advancing the management and control of typhoid fever: a review of the historical role of human challenge studies. J Infect. 2014;68:405–18. https://doi.org/10.1016/j.jinf.2014.01.006.
Crump JA, Sjölund-Karlsson M, Gordon MA, Parry CM. Epidemiology, clinical presentation, laboratory diagnosis, antimicrobial resistance, and antimicrobial management of invasive Salmonella infections. Clin Microbiol Rev. 2015;28:901–37. https://doi.org/10.1128/CMR.00002-15.
Mogasale V, Ramani E, Mogasale VV, Park J. What proportion of Salmonella typhi cases are detected by blood culture? A systematic literature review. Ann Clin Microbiol Antimicrob. 2016;15:32. https://doi.org/10.1186/s12941-016-0147-z.
Kariuki S, Gordon MA, Feasey N, Parry CM. Antimicrobial resistance and management of invasive Salmonella disease. Vaccine. 2015;33:C21–9. https://doi.org/10.1016/j.vaccine.2015.03.102.
•• Qamar FN, Yousafzai MT, Khalid M, Kazi AM, Lohana H, Karim S, et al. Outbreak investigation of ceftriaxone-resistant Salmonella enterica serotype Typhi and its risk factors among the general population in Hyderabad, Pakistan: a matched case-control study. Lancet Infect Dis. 2018;18:1368–1376. https://doi.org/10.1016/S1473-3099(18)30483-3The authors provided a case-control study on the outbreak of ceftriaxone-resistant typhoid fever and identified contaminated community water source as a causal factor.
Milligan R, Paul M, Richardson M, Neuberger A. Vaccines for preventing typhoid fever. Cochrane Database Syst Rev. 2018;5:CD001261. https://doi.org/10.1002/14651858.CD001261.pub4.
Parry CM, Hien TT, Dougan G, White NJ, Farrar JJ. Typhoid fever. N Engl J Med. 2002;347:1770–82. https://doi.org/10.1056/NEJMra020201.
Khan FY, Al-Ani A, Ali HA. Typhoid rhabdomyolysis with acute renal failure and acute pancreatitis: a case report and review of the literature. Int J Infect Dis. 2009;13:e282–5. https://doi.org/10.1016/j.ijid.2008.11.009.
Huang GC, Chang CM, Ko WC, Huang YL, Chuang YC. Typhoid fever complicated by multiple organ involvement: report of two cases. J Infect. 2005;51:E57–60. https://doi.org/10.1016/j.jinf.2004.08.018.
Ali M, Abdalla H. Salmonella typhi infection complicated by rhabdomyolysis, pancreatitis and polyneuropathy. Arab J Nephrol Transplant. 2011;4:91–3.
Odetunde OI, Ezenwosu OU, Odetunde OA, Azubuike JC. Typhoid glomerulonephritis and intestinal perforation in a Nigerian child. Niger J Clin Pract. 2014;17:655–7. https://doi.org/10.4103/1119-3077.141442.
Meena VK, Kumar N, Nawal R. An unusual presentation of typhoid fever causing aseptic meningitis, acute pancreatitis, acute glomerulonephritis, acute hepatitis. Chin Med J. 2013;126:397–8. https://doi.org/10.3760/cma.j.issn.0366-6999.20121930.
Asano T, Kuwabara K, Takagi A, Hatori T, Hamada H, Imai T, et al. Acute pancreatitis complicating typhoid fever in a 4-year-old girl. Pediatr Int. 2007;49:1004–6. https://doi.org/10.1111/j.1442-200X.2007.02465.x.
Lambotte O, Debord T, Castagné C, Roué R. Unusual presentation of typhoid fever: cutaneous vasculitis, pancreatitis, and splenic abscess. J Infect. 2001;42:161–2. https://doi.org/10.1053/jinf.2000.0783.
Stauffer W, Mantey K, Kamat D. Multiple extraintestinal manifestations of typhoid fever. Infection. 2002;30:113. https://doi.org/10.1007/s15010-002-1121-6.
Joshi N, Bhattacharya M, Yadav S, Rustogi D. Cranial nerve palsies in typhoid fever: report of three cases. Ann Trop Paediatr. 2011;31:255–8. https://doi.org/10.1179/1465328111Y.0000000023.
Julià J, Canet JJ, Lacasa XM, González G, Garau J. Spontaneous spleen rupture during typhoid fever. Int J Infect Dis. 2000;4:108–9. https://doi.org/10.1016/s1201-9712(00)90104-8.
Mitra S, Karthik R, Balaji V, George IA, Kapil A, Abraham OC. Quinolone-resistant Salmonella enteric serovar typhi presenting as acute fulminant hepatitis. J Assoc Physicians India. 2009;57:338–9.
Khan FY, Elouzi EB, Asif M. Acute acalculous cholecystitis complicating typhoid fever in an adult patient: a case report and review of the literature. Travel Med Infect Dis. 2009;7:203–6. https://doi.org/10.1016/j.tmaid.2009.05.006.
Khan FY, El-Hiday AH, Kamel HA. Typhoid osteomyelitis of the lumbar spine. Hong Kong Med J. 2006;12:391–393. https://www.hkmj.org/abstracts/v12n5/391.htm
Khoo HW, Chua YY, Chen JL. Salmonella typhi vertebral osteomyelitis and epidural abscess. Case Rep Orthop. 2016;2016:6798157. https://doi.org/10.1155/2016/6798157.
Albaqali A, Ghuloom A, Al Arrayed A, Al Ajami A, Shome DK, Jamsheer A, et al. Hemolytic uremic syndrome in association with typhoid fever. Am J Kidney Dis. 2003;41:709–13. https://doi.org/10.1053/ajkd.2003.50135.
Parmar RC, Bavdekar SB, Houilgol R, Muranjan MN. Nephritis and cerebellar ataxia: rare presenting features of enteric fever. J Postgrad Med. 2000;46:184–6.
Singh G, Dasgupta M, Gautam V, Behera A, Ray P. Bilateral breast abscesses due to Salmonella enterica serotype typhi. J Glob Infect Dis. 2011;3:402–4. https://doi.org/10.4103/0974-777X.91069.
Non LR, Patel R, Esmaeeli A, Despotovic V. Typhoid fever complicated by hemophagocytic lymphohistiocytosis and rhabdomyolysis. Am J Trop Med Hyg. 2015;93:1068–9. https://doi.org/10.4269/ajtmh.15-0385.
Pandey M, Dhingra B, Sharma S, Chandra J, Pemde H, Singh V. Enteric fever presenting as secondary hemophagocytic lymphohistiocytosis. Indian J Pediatr. 2012;79:1671–2. https://doi.org/10.1007/s12098-012-0728-6.
Pineda MC, Lopinto-Khoury C. Cerebral venous sinus thrombosis secondary to typhoid fever: a case report and brief summary of the literature. Neurologist. 2012;18:202–3. https://doi.org/10.1097/NRL.0b013e31825cf3e1.
Duhil de Bénazé G, Desselas E, Houdouin V, Mariani-Kurkdjian P, Kheniche A, Dauger S, et al. Pneumonia with pleural empyema caused by Salmonella typhi in an immunocompetent child living in a non-endemic country. Paediatr Int Child Health. 2018;38:227–230. https://doi.org/10.1080/20469047.2017.1316938
Prabhushanker M, Topiwalla TT, Ganesan G, Appandaraj S. Bilateral retinitis following typhoid fever. Int J Retina Vitreous. 2017;3:11. https://doi.org/10.1186/s40942-017-0065-z.
Hanel RA, Araújo JC, Antoniuk A, da Silva Ditzel LF, Flenik Martins LT, Linhares MN. Multiple brain abscesses caused by Salmonella typhi: case report. Surg Neurol. 2000;53:86–90. https://doi.org/10.1016/s0090-3019(99)00161-5.
Momen ABI, Twinkle FQ, Rahman A, Quraishi FA. Guillain-Barre syndrome in a case of typhoid fever: an uncommon association. Bangladesh J Med. 2021;32:142–4. https://doi.org/10.3329/bjm.v32i2.53799.
Mishra K, Kaur S, Basu S, Gulati P, Parakh A. Acute transverse myelitis: an unusual complication of typhoid fever. Paediatr Int Child Health. 2012;32:174–6. https://doi.org/10.1179/2046905512Y.0000000011.
Bhatt GC, Dewan V, Dewan T, Yadav TP. Pseudotumour cerebri with multiple cranial nerve palsies in enteric fever. Indian J Pediatr. 2014;81:196–7. https://doi.org/10.1007/s12098-013-0965-3.
Dadia SD, Modi RR, Shirwadkar S, Potdar NA, Shinde CA, Nair AG. Salmonella typhi associated endogenous endophthalmitis: a case report and a review of literature. Ocul Immunol Inflamm. 2018;26:527–32. https://doi.org/10.1080/09273948.2017.1306085.
Dhakad U, Das SK, Srivastva D, Nolkha N. Arthritis and adult respiratory distress syndrome: unusual presentations of typhoid fever. BMJ Case Rep. 2014;2014:bcr2014204266. https://doi.org/10.1136/bcr-2014-204266
Bansal N, Kaistha N, Chander J. Epididymo-orchitis: an unusual manifestation of salmonellosis. J Microbiol Immunol Infect. 2012;45:318–20. https://doi.org/10.1016/j.jmii.2011.09.017.
Robson C, O’Sullivan MVN, Sivagnanam S. Salmonella enteric serovar typhi: an unusual cause of infective endocarditis. Trop Med Infect Dis. 2018;3:35. https://doi.org/10.3390/tropicalmed3010035.
Nair RK, Mehta SR, Kumaravelu S. Typhoid fever presenting as acute psychosis. Med J Armed Forces India. 2003;59:252–3. https://doi.org/10.1016/S0377-1237(03)80023-0.
Vagholkar K, Mirani J, Jain U, Iyengar M, Chavan RK. Abdominal complications of typhoid fever. J Surgery. 2015;10:227–8. https://doi.org/10.7438/1584-9341-10-4-9.
Ali HA, Ahmed MA, Abbdullam U, Zulfa OD, Hina HS. Hepatic dysfunction in typhoid fever. Pat J Med Sci. 2007;12:81–4.
Adhikari S, Shrestha AL, Thapa SR, Ghimire A. A rare cause of acute hepatopancreatitis in a Nepalese teen. Case Rep Pediatr. 2018;2018:8456503. https://doi.org/10.1155/2018/8456503.
Das DK, Shukla S, Murmu MC. Clinical profile of acute pancreatitis with infection due to Salmonella typhi and hepatitis-A in children. Orissa J Paediatr. 2020;21:11.
• Marchello CS, Birkhold M, Crump JA. Complications and mortality of typhoid fever: a global systematic review and meta-analysis. J Infect. 2020;81:902–910. https://doi.org/10.1016/j.jinf.2020.10.030The authors provided meta-analysis on typhoid fever complications and suggested that delay in management correlates with case fatality ratio.
Jha VK, Vidhale T. Multiple splenic abscesses in a case of enteric fever: salvaging spleen through pigtail drainage and antibiotics is a good alternative approach. Indian J Crit Care Med. 2018;22:886–8. https://doi.org/10.4103/ijccm.IJCCM_405_18.
Huang DB, DuPont HL. Problem pathogens: extra-intestinal complications of Salmonella enterica serotype typhi infection. Lancet Infect Dis. 2005;5:341–8. https://doi.org/10.1016/S1473-3099(05)70138-9.
Lakhotia M, Gehlot RS, Jain P, Sharma S, Bhargava A. Neurological manifestations of enteric fever. J Indian Acad Clin Med. 2003;4:196–9.
Su CP, Chen YC, Chang SC. Changing characteristics of typhoid fever in Taiwan. J Microbiol Immunol Infect. 2004;37:109–14.
Esmailpour N, Rasoolinejad M, Abdolbaghi MH. Cardiopulmonary manifestations of typhoid fever: a prospective analysis of 65 cases in Iran. Trop Doct. 2006;36:118–9. https://doi.org/10.1258/004947506776593468.
Baddour LM, Wilson WR, Bayer AS, Fowler VG Jr, Tleyjeh IM, Rybak MJ, et al. Infective endocarditis in adults: diagnosis, antimicrobial therapy, and management of complications: a scientific statement for healthcare professionals from the American Heart Association. Circulation. 2015;132:1435–86. https://doi.org/10.1161/CIR.0000000000000296.
Janssen van Doorn K, Pierard D, Spapen H. Acute renal dysfunction in Salmonella gastroenteritis. J Clin Gastroenterol. 2006;40:910–912. https://doi.org/10.1097/01.mcg.0000212620.20079.34
Khanam F, Ross AG, McMillan NAJ, Qadri F. Toward typhoid fever elimination. Int J Infect Dis. 2022;119:41–3. https://doi.org/10.1016/j.ijid.2022.03.036.
Srinivasan M, Sindhu KN, Giri S, Kumar N, Mohan VR, Grassly NC, et al. Salmonella typhi shedding and household transmission by children with blood culture-confirmed typhoid fever in Vellore. South India J Infect Dis. 2021;224:S593–600. https://doi.org/10.1093/infdis/jiab409.
Kumar S, Ghosh RS, Iyer H, Ray A, Vannice K, MacLennan C, et al. Typhoid in India: an age-old problem with an existing solution. J Infect Dis. 2021;224:S469–74. https://doi.org/10.1093/infdis/jiab441.
Vashishtha VM, Kalra A. The need & the issues related to new-generation typhoid conjugate vaccines in India. Indian J Med Res. 2020;151:22–34. https://doi.org/10.4103/ijmr.IJMR_1890_17.
Acknowledgements
We thank Dr. Ibrahim A Bani for reviewing the manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. The idea for the article was conceived by Saurabh Gaba and Monica Gupta. Material preparation, data collection, and analysis were performed by Saurabh Gaba, Monica Gupta, Gautam Jesrani, and Samiksha Gupta. The first draft of the manuscript was written by Saurabh Gaba, Gautam Jesrani, and Samiksha Gupta, and all authors commented on all versions of the manuscript. Monica Gupta, Ruchi Gaba, and Sarabmeet Singh Lehl drafted and critically revised the work, and all authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
This article does not contain any studies with human or animal subjects performed by any of the authors. No human subjects were involved in this study.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Hot Topics in Tropical Medicine
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gaba, S., Gupta, M., Jesrani, G. et al. Intestinal and Extra-intestinal Complications of Typhoid Fever: an Update. Curr Trop Med Rep 10, 71–78 (2023). https://doi.org/10.1007/s40475-023-00285-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40475-023-00285-w