Abstract
Plants that grow in semiarid environments can sustain a positive equilibrium between water absorption and loss. This equilibrium is reminiscent of structural traits acquired throughout their evolutionary history. Woody species often possess sclerophyllous characteristics when growing in dry environments. Herbs on the other hand commonly shed leaves during unfavorable conditions, such as drought, allowing them to grow and establish themselves rapidly, and therefore are less sclerophyllous. Based on observations of species representing different growth habits, and considering xeric habitats, we raised the hypothesis that woody species will posses sclerophyllous traits and that these traits will vary among growth habits. We evaluated morphoanatomical traits that were previously described in the literature as being adaptations to arid environments, of 16 species, of four different growth habits, that co-occur in the Brazilian “Chaco”. Only vine species showed adaptive convergence to dry environments. Woody species were found to possess sclerophyllous traits, including thicker leaves, well-developed palisade parenchyma and the presence of sclerenchyma. Herbaceous species exhibited mesomorphic traits, including larger leaves and little or no sclerenchyma. We concluded that all the analyzed species possess adaptations to the dry environment, although not all of them are sclerophyllous. As expected, woody species and herbaceous species possess different strategies for adapting to environmental conditions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
The survival of plants in arid environments is aided by their ability to maintain a favorable equilibrium between water absorption and loss during severe droughts (Kassas and Batanouny 1984). Leaves are the main organs controlling this equilibrium and respond effectively to abiotic factors. They also influence other biological processes such as growth and reproduction (Niklas et al. 2007). Leaves aid in the structuring of ecosystem processes and dynamics, and their exposure to changes in environmental conditions has led to the belief that internal and external leaf structures are adaptations to environmental occurrences (Fahn 1986). Leaf anatomy is a decisive aspect of adaptability and the ability of species to adjust when exposed to different environmental conditions (Hanba et al. 2002; Schluter et al. 2003). Foliar features indicate whether plants are adapted to dry environments and/or to seasonal water deficits.
Ecological processes can imply variation in functional traits within or among species and have importance on many levels of investigation. Thus, understanding of morphology and physiology of plants has been sought after in recent years in order to measure their ecological characteristics as well as functional traits (Pérez-Harguindeguy et al. 2013). Leaves are considered a highly plastic organ capable of adapting to environmental factors (Gratani et al. 2006). The main factors that influence variations in leaf structure are: light intensity, water availability and temperature (Esau 1977; Bradshaw 2006).
Woody species from dry environments usually have thicker leaves, cuticles and external periclinal walls of the epidermal cells. Other features include a well-developed sclerenchyma around the vascular units and in other regions of the mesophyll, and well-developed palisade parenchyma (Fahn and Cutler 1992; Fahmy 1997; Elias et al. 2003; Rotondi et al. 2003). Leaf shedding is common among herbaceous species when conditions are unfavorable, such as during droughts. Herbs typically exhibit fast growth and propagate by means of seed dispersal or by underground systems, allowing for fast establishment. Due to these features, herbs may exhibit less sclerophylly compared to woody species (Gentry 1991; Hartley and Jones 1997; Eamus 1999).
Most research on the ecological anatomy of plants from arid environments, especially species from South American biomes, has been mostly focused on specific groups of plants. In the “Cerrado,” which has a rainfall pattern similar to that of the Brazilian “Chaco”, convergence among morphological and anatomical traits has been documented within growth habits (Rossato et al. 2015). Describing the community structure of the seasonal “Chaco” environment can be aided by analyzing the adaptive strategies of co-occurring species of different growth habits. In the case of plants that inhabit environments with pronounced seasonality in water availability, the following questions need to be addressed: (1) Do species have sclerophyllous characteristics? If so, (2) do the leaf characteristics that indicate sclerophylly converge among species of similar growth habits? Our hypothesis is that the anatomical traits studied herein will show divergence among, and convergence within, growth habits. We also hypothesize that woody species will possess more sclerophyllous features than herbaceous species.
2 Materials and methods
Study area and species groups analyzed
– Highlighted as a South American environment with prominent seasonal drought, the “Chaco” has a biogeographical area that comprises savannah rainforests and predominantly sclerophyllous species, and is distributed throughout the central part of the subcontinent (Cabrera and Willink 1980; Pennington et al. 2000). The variety of ecosystems in the “Chaco” provides a high diversity of species and a relatively high rate of endemism when compared to other arid, semiarid and subhumid environments (Hueck 1955; Veloso 1972; Adámoli 1986). Species found in the “Chaco” have easily recognizable structural characteristics, such as leaves with reduced blades and spiny stems.
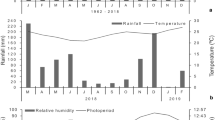
In Brazil, ecosystems with features of the “Chaco” are present only in the municipality of Porto Murtinho in the state of Mato Grosso do Sul, Brazil, where they occur continuously. The “Chaco” in this region possesses approximately 70% of the features of “Chaco sensu stricto” (Prado 1993). It is also the wettest region of the “Chaco” with an average rainfall of 1200 mm/year and is characterized by a hot and dry climate; a predominance of seasonal rains, sometimes concentrated within a short period of 3–4 months; and long periods of droughts (Brasil 1982; Ramella and Spichiger 1989; Pennington et al. 2000). Plant species were collected from the “Chaco–Pantanal” ecotone, which is considered a semiarid region. The highest absolute temperatures in South America are recorded in this biome, with average temperatures ranging between 18 °C (south) and 26 °C (north), with a maximum of 48 °C (Prado 1993; Nunes 2006). The soil is classified as solodic, solonetz and planosol (Brasil 1982; Ramella and Spichiger 1989; Pennington et al. 2000), with great compactness and impermeability.
Sixteen species, selected because they were the most abundant in the study, representing four different growth habits were analyzed (Table 1). Species classification by growth habit followed the classification of Clements (1920), namely trees = woody plants greater than two meters in height, with a well-defined trunk and branches with secondary growth; shrubs = plants between one and three meters in height with tough woody stems, usually with more than one stem without any predominant trunk, with branches rising near or from the base and with secondary growth in its branches; vines = herbaceous plants with long, thin, flexible branches or tendrils, generally supported on upright woody plants; herbs = small plants of up to 40 cm in height with a small aerial branch, suberized slightly or not at all, and with no more than one aerial branch. All climbing species were found growing on trees and shrubs.
Samples were collected during the same time of the year (winter and dry season). Collections were made in two similar communities in a remnant of Tree Steppe Savannah (wooded “Chaco”): on the Anahí farm, 15 km from Porto Murtinho, measuring about 40 ha (21º41′12″S, 57º44′05″W), and on the Dique Beira Rio road near town of Porto Murtinho (21°42′37″S, 57°53′55″W). Both locations are in the southwest region of the state of Mato Grosso do Sul, Brazil. Four specimens of each species were analyzed. Vouchers were sent to the Herbarium CGMS of the Federal University of Mato Grosso do Sul, Brazil.
Morphological analysis
– Thirty fully expanded leaves, collected from the fourth node of each of the four selected specimens of each species, were evaluated. In the case of compound leaves, the median leaflet was evaluated. The Sclerophylly Index (SI, g dm−2) was calculated based on the formula described by Rizzini (1976), who established that sclerophyllous plants have a SI > 0.6 and mesophilic plants a SI < 0.6. Specific leaf mass (SLM, g−1 cm2), or the Sclerophylly Index (Witkowski and Lamont 1991), was calculated as the ratio of leaf area to leaf dry mass. To determine dry mass, leaves were dried in an oven at 70 °C for 72 h and weighed on a precision digital scale. Leaf area was calculated by using ImageJ 1:43 (http://imagej.nih.gov/ij/; Rasband 1997–2015). Dry mass and leaf area were measured without the petiole.
Anatomical analysis
– Segments of four healthy leaves from the fourth node of four different individuals of each species were collected for the anatomical analysis. They were fixed in FAA70 (formaldehyde, acetic acid and 70% ethyl alcohol; Johansen 1940) and stored in 70% ethanol. Leaf blade cross sections were obtained from material embedded in historesin and sectioning with a rotary microtome. The sections were stained with toluidine blue and mounted with Entellan®. Dissociation for analysis of the epidermis was performed by cutting 0.5-cm2 segments, placing them in a 1:1 solution of hydrogen peroxide (30%) and glacial acetic acid and maintaining the solution with segments in an oven at 60 °C for 24 h. Semipermanent slides were prepared from cross sections of leaf blades, with basic fuchsin and astra blue double staining; this dual coloration provides visualization of phenols, polysaccharides and cell wall cellulose (Kraus et al. 1998).
The surface of the leaves was analyzed by dehydrating the fixed leaf samples in an ascending ethanol series up to absolute ethanol. The samples were then submitted to critical point drying using CO2, then placed on stubs and coated with a thin layer of gold prior to analysis. Electron micrographs of the epidermis were obtained using a JEOL JSM-6380LV scanning electron microscope.
Qualitative analysis was based on the following characteristics: leaf (dorsiventral or homogeneous), epidermis of the adaxial and abaxial surface (uni- or pluri-stratified), type of vascular bundle (collateral, bicollateral or amphivasal), occurrence of stomata (hypostomatic, epistomatic or amphistomatic), position of the stomata (below, above or at the same level of the epidermal cells), stomatal crest (present or absent), type of stomata (anomocytic, paracytic or cyclocytic), occurrence of trichomes (abaxial, adaxial or both sides), type of trichomes (tector or glandular), anticlinal cell walls of the epidermis in paradermic view (straight or winding), sclerenchyma (present or absent), bundle sheath (absent, parenchyma, sclerenchyma or both), intercellular spaces of the spongy parenchyma (broad or reduced), phenolic compounds and druses (present or absent).
Quantitative analysis encompassed measuring the thickness of the epidermis (of the abaxial and adaxial surfaces) and of the mesophyll, and total leaf thickness (from one epidermis to the other). Stomata and trichomes per mm2, and length of stomata of the epidermis were calculated. Five sections from different leaves of all the four specimens per species studied were analyzed. Thirty stomata were selected at random for each of the five leaves of all specimens to measure stomata length. The thickness of the photosynthetic tissue of the leaves was calculated by dividing the thickness of the mesophyll by the total thickness of the leaf.
Cross sections were analyzed under a light microscope and photomicrographs captured using a camera. Quantitative analysis was undertaken with ImageJ 1:43 (http://imagej.nih.gov/ij/; Rasband 1997–2015).
Statistical analysis
– Differences in leaf features were tested using one-way ANOVA and considering growth habit as a fixed factor, followed by Tukey’s test for homogeneous groups (α = 0.05). For morphological analyses, 30 leaves of each individual were used, with four individuals being evaluated per species (16 species in total). For anatomical analyses, five leaves of each individual were evaluated. Mean values were obtained from measurements, and differences in means were assessed for leaf features among growth forms and among species.
Since stomatal density, stomatal length and trichome density could not be measured for Capparicordis tweediana, due to difficulties in the dissociation of the material, the species was excluded when analyses required these features. All analyses were performed in the statistical computing environment.
3 Results
Morphology
– Great variation was perceived in leaf morphology and size among species, with evident veins in all species. Trees and herbs had narrower leaves, while shrubs and vines possessed wider leaves (Fig. 1a, b). Vines exhibited the highest values for leaf area (45.06 cm2), dry matter (0.24 g) and specific leaf mass (SLM; 181.38 cm2 g−1), while trees had the lowest SLM (68.38 cm2 g−1). Among species, the highest SLM values occurred in Ruellia erythropus (herb), Aristolochia esperanzae (vine), Jatropha ribifolia (shrub) and Araujia odorata (vine).
According to the Rizzini index, three tree species (Schinopsis balansae, Tabebuia nodosa and Aspidosperma quebracho-blanco), three shrub species (Capparicordis tweediana, Coccoloba paraguariensis and Maytenus ilicifolia) and two herb species (Ruellia erythropus and Stachytarpheta cayennensis) can be classified as sclerophyllous; all other species are considered mesophilic.
Anatomy
– The internal structures of the leaves varied, according to the presence/absence, type, distribution and density of the structures, among the analyzed species (Figs. 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, and 19; Table 2). Regardless of growth habit, all species had uni-stratified epidermis, with the exception of Coccoloba paraguariensis and Aristolochia esperanzae, which featured periclinal division of some cells on the adaxial side (Figs. 7, 13). In most species, the epidermis of the adaxial surface of the leaf was thicker and had bulkier cells when compared to those of the abaxial epidermis (Figs. 3, 4, 7, 8, 11, 13, 14, 16, and 17).
Cross sections of leaves showing the mesophyll, epidermis and veins of the analyzed species. Trees 2 Schinopsis balansae Engl., 3 Prosopis ruscifolia Griseb., 4 Tabebuia nodosa (Mart.) Standl, 5 Aspidosperma quebracho-blanco Schltdl.; Shrubs 6 Capparicordis tweediana (Eichler) Iltis & Carnejo, 7 Coccoloba paraguariensis Lindau, 8 Maytenus ilicifolia (Schrad.) Planch, 9 Jatropha ribifolia Pohl (Baill); Vines 10 Ipomoea ramosissima (Poir) Choisy, 11 Araujia stormiana Morong, 12 Araujia odorata (Hook. & Arn.) Fontella & Goyder, 13 Aristolochia esperanzae Kuntze; Herbs 14 Pterocaulon purpurascens Malme, 15 Ruellia erythropus (Nees) Lindau, 16 Stachytarpheta cayennensis (Reich.) Vahl, 17 Stachytarpheta jamaicensis Vahl. Arrow: crystal. Star: phenolic compounds
The anticlinal wall of the epidermal cells was straight in all tree species and in half of the species representing other growth habits. In the other species, the anticlinal wall of the epidermis is winding or ranges between straight and winding (Table 2).
In tree species, the epidermis of the abaxial surface is thicker than that of the other analyzed habits (Fig. 18). On the other hand, the epidermis of the adaxial surface was thicker in herbs (Fig. 18b). Shrubs had the lowest epidermal thickness for both abaxial and adaxial surfaces (Fig. 18a, b). Although it was not measured separately, it is clear that the woody species had a thicker cuticle on the adaxial epidermis than the herbaceous species. Only seven species exhibited wax deposition on the epidermis, four of them being exclusive to the “Chaco” (Table 2).
Eleven species have amphistomatic leaves, and the stomata are distributed at the same level as the other epidermal cells in eight species; beneath the other epidermal cells in six species; and above the other epidermal cells in only two species (T. nodosa and S. cayennensis). Anomocytic stomata were the most common, with ten species possessing this type of stomata, followed by diacytic stomata in three species; paracytic stomata in two species; and ciclocytic stomata in only S. balansae (Table 2). Stomatal crests were observed in five species.
Variation in stomatal density was not statistically significant among habits (Fig. 19a). Variation in stomatal length allowed the distinction of woody species (trees and shrubs), which had larger stomata, from herb species (herbs and vines), with small stomata (Fig. 19c, d).
The trichome density of the woody species was low, while that of herbaceous species was high (Fig. 19e, f). Only four of the species studied, two trees (Prosopis ruscifolia and Schinopsis balansae) and two shrubs (Maytenus ilicifolia and Jatropha ribifolia), were found to be glabrous. Trichomes occur only on the abaxial side of Aspidosperma quebracho-blanco, but they are distributed on both sides of the leaf in all other species. When growth habits are compared, trichomes occurred on both sides in all vine species and in herbs, whereas trees and shrubs are glabrous or have trichomes only on the abaxial side or on both sides. The highest density of trichomes (Fig. 19f) occurred in herbs and vines, such as Pterocaulon purpurascens, Araujia stormiana, Araujia odorata, Stachytarpheta cayennensis, Aristolochia esperanzae, Ruellia erythropus and Stachytarpheta jamaicensis. In the case of Capparicordis tweediana, a high density of trichomes was evident, but could not be quantified due to difficulties in the dissociation of material. The types of trichomes found were acicular (Fig. 21), acerate (Figs. 21, 23), attenuate (Figs. 22, 23), stellate (Fig. 24), peltate (Figs. 25, 26, 27) and brevicollate (Fig. 22; Payne 1978).
Micromorphology of leaf trichomes of the evaluated species. 20 Abaxial face of Aristolochia esperanzae Kuntze. 21 Abaxial face of Araujia stormiana Morong. 22, 23 Abaxial face of Stachytarpheta cayennensis (Reich.) Vahl. 24 Adaxial face of Capparicordis tweediana (Eichler) Iltis & Carnejo (Mart.). 25 Abaxial face of Coccoloba paraguariensis Lindau. 26 Abaxial face of Tabebuia nodosa Standl. 27 Abaxial face of Ipomoea ramosissima (Poir) Choisy
Leaves were dorsiventral with well-defined palisade and spongy parenchyma in 75% of analyzed species (Figs. 4, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17), and homogeneous in the tree species Prosopis ruscifolia (Fig. 3), Aspidosperma quebracho-blanco (Fig. 5) and Schinopsis balansae (Fig. 2).
Mesophyll thickness and total leaf thickness did not vary among the species with different growth habits. Investment in photosynthetic tissue was high for all habits (> 80%) and did not vary significantly (Fig. 18). However, when species were compared, the vine Aristolochia esperanzae had the lowest photosynthetic investment (72.30%). Comparisons between tree and shrub species revealed little variation in photosynthetic tissues investment among the species, but there was significant variation in this characteristic among vines and herbs (Fig. 18).
Aquifer parenchyma was found in Prosopis ruscifolia, Tabebuia nodosa, Stachytarpheta cayennensis and Stachytarpheta jamaicensis in different tissues of the leaf blade (Fig. 2), but had no relation with growth habit.
Most species (nine) had sclerenchyma in certain areas of the leaf blade. Six out of the seven species that did not have sclerenchyma were vines and herbs (Table 2).
Seven species lacked bundle sheath extensions (Figs. 8, 9, 10, 11, 12, 16, 17). Two species had the bundle sheath composed of sclerenchyma (Figs. 3, 5). The bundle sheath extension was mixed, composed of parenchymal and sclerenchyma cells, in three species (Figs. 2, 4, 7). For trees and shrubs, the vascular bundle was sclerenchymatic or mixed, while for vines and herbs, it varied between absent and parenchymal (Table 2).
Phenolic compounds were detected in various tissues of 11 species, regardless of the growth habit (Figs. 2, 5, 7). Crystals occurred in various tissues of only six species (Figs. 7, 10) and were unrelated to growth habits (Table 2).
The midrib of 11 species had a side bundle. In five of these, the side bundle was closed, in four, it was open, and in two, the bundle was not entirely closed (Table 2), although the latter condition was not associated with growth habit.
4 Discussion
The quantitative and qualitative leaf features of the 16 studied species revealed different adaptive strategies to a dry environment. With the exception of vines, we found no evidence for functional convergence between species and growth habits. This result was probably due to the fact that some species were not exclusive to the “Chaco” and shared functional characteristics with other plant species. Species of the vine habit were the only group that shared similar functional strategies. Species of other growth habits displayed broader morphoanatomical variation when the seasonal drought of the environment is taken into account.
Regardless of taxa and place of occurrence, all species could be separated in two main groups: woody plants (trees and shrubs) with predominantly scleromorphic features and herbs (herbs and vines) with predominantly mesomorphic features. This grouping demonstrates that functional traits were consistent in the current analysis, and that anatomical characteristics may be coupled with morphological and ecophysiological features in investigations of functional diversity. The assessed species, which occur naturally in environments with pronounced seasonality in water availability, possess adaptive convergences for most of the evaluated traits.
Furthermore, the fact that tree species have smaller and narrower leaves (sclerophyllous traits) reflects the drought and high intensity of light in the environment, since these species occur exclusively in the “Chaco”. All vine species have larger leaves and higher values of SLM, which is the main index of high photosynthetic efficiency (Souza et al. 2011). Consequently, the large leaf area of these species may generate the greatest rates of water loss by transpiration, which in turn is compensated for their deciduousness during the drought period. In addition, the species Araujia stormiana and A. odorata possess a strategy of resistance to dry seasons in their underground system. This system has the potential of producing buds and restoring aerial plant parts during the rainy season. As a rule, specialized underground systems contain starch reserves, which, under adverse conditions, may be used by the plant to increase its chance of survival (Paviani and Haridasan 1988; Gentry 1991; Kolb et al. 1998).
Higher SLM values for climbing species contribute to faster growth and establishment during periods with water availability (Hartley and Jones 1997; Eamus 1999) and may facilitate environmental occupation during these periods. Therefore, higher SLM is an adaptive advantage for species that share this habit since it provides speed during establishment and reproduction during favorable periods. Leaf thickness is one of the morphological features that correlate most strongly with SLM (Wilson et al. 1999). Leaves that are exposed to intense luminosity usually possess smaller surfaces, but greater thickness of the leaf blade. This greater thickness is due to increased amounts of photosynthetic tissue and intercellular spaces in the mesophyll (Boeger and Poulson 2006).
The dorsiventral leaf arrangement with well-differentiated palisade and spongy parenchyma possessed by vines and herbs represents a typical characteristic of sclerophyllous plants (Fahn 1986). However, other characteristics involving this group of plants classify them as mesophytic. Further investigation into other features, including ecophysiological traits, is needed to define their ecological position and obtain more conclusive adaptive interpretations.
Of the species studied, those that possessed homogeneous mesophyll were classified as either trees or shrubs that occur exclusively in the “Chaco”. Burrows (2001) described species with an isolateral mesophyll and amphistomatic leaves in a study of 39 sclerophyllous species of the western part of New South Wales, Australia. The aforementioned characteristic was also described by Fahmy (1997) for non-succulent plants in the Egyptian desert and was featured as a common trait in species that naturally inhabit arid environments.
High investment in photosynthetic tissue by all species, except A. esperanzae, is due to high light incidence throughout the entire seasonal period. Consequently, when exposed to high light incidence, amphistomatic leaves are more advantageous than hypostomatic leaves due to higher gas exchange efficiency (Mott et al. 1982). Stomata have a key role in regulating transpiration and the flow of CO2 into the leaf (Pearce et al. 2006). During the dry season in the “Chaco”, there is only a brief period during the day when relative humidity is high. This brief period provides plants an opportunity for gas exchange, and greater stomatal area makes it faster and more efficient (Medri and Lleras 1980).
The greater thickness of the adaxial epidermis observed in herbs may be an important adaptive strategy for greater water retention or thermal insulation against high luminosity. The high investment in cuticle deposition demonstrated by woody species exclusive to the “Chaco” is an important feature for species inhabiting arid environments since it is involved with several important processes, such as limiting the loss of water by evapotranspiration (Pallardy 1981).
The epidermis of woody species possessed cell walls with predominantly straight contours, the opposite of that of herbaceous species. The orientation of anticlinal cell walls is predominantly straight for species of dry environments, and especially those with high light incidence, while they are predominantly described as corrugated for species of humid and/or aquatic environments (Cutter 1978; Medri and Lleras 1980). Straight anticlinal cell walls are a feature common to all species that are exclusive to the “Chaco”, including P. ruscifolia, which also occurs in the “Caatinga” biome, a dry biome similar to the “Chaco” (Prado and Gibbs 1993).
The phenolic compounds found in the mesophyll of most of the studied species are common in species that inhabit dry environments. These substances reduce light transmittance through the epidermal cells and protect photosynthetic tissues against excessive solar radiation (Izaguirre et al. 2007). Thus, phenolic compounds can prevent the harmful effects of irradiation on plants, such as damage from ultraviolet radiation, heat-induced stress on leaves and photoinhibition caused by exposure to high levels of light that damages the photosynthetic system (Jordan et al. 2005).
The features that vary the most among the different growth habits are intercellular spaces of the spongy parenchyma, presence of crystals, presence of aquifer parenchyma, position of stomata in relation to other epidermal cells, presence of a stomatal crest, presence of a bundle sheath extension, types and density of the trichomes, density and length of stomata, and tissue thickness. However, the adaptive value of morphological, anatomical and ecophysiological structures of plant organs in species of different habitats and habits is still unknown. In spite of this, the observed characteristics portray environmental adaptations specific to the dynamics of the particular region of the “Chaco” (Scremin-Dias et al. 2011).
Several of the morphological and anatomical features described for sclerophyllous plants in the present work are adaptive convergences, similar to high stomatal density. However, certain anatomical characteristics are not merely adaptive and may be phylogenetically consistent characteristics of the evaluated groups, such as the traditionally described stomata types, used to characterize botanical families (Metcalfe and Chalk 1988).
Leaf trichomes are common in plants from arid environments (Fahn and Cutler 1992), where the high density of these leaf structures represents an adaptation to low water availability and high temperatures (Sandquist and Ehleringer 1997). Trichomes decrease transpiration rate and reduce leaf temperature by reflecting solar radiation, acting as a barrier to water loss and increasing the depth of the entrapped air layer above the leaf (Sandquist and Ehleringer 1997). Exclusive “Chaco” species are glabrous or feature small amounts of trichomes, when compared to species that also occur in other biomes. However, other features, such as thick cell walls, thick cuticle and efficient cuticle layers, may be restricting water loss of these species and therefore may be functioning as efficient adaptations to the environment (Medri and Lleras 1980; Fahn 1986).
The presence of a bundle sheath extension is common among woody species, in which it is comprised of parenchyma, sclerenchyma or both, differing from that of herbaceous species, in which the sheath, when present, is formed only by parenchyma. A vascular bundle sheath extension connects the vascular bundle of the leaves to the epidermal layers of the adaxial surface and may be an adaptive characteristic for plants belonging to dry environments (Fahn 1986) since they serve to transport water to the vascular bundles, epidermis and photosynthetic tissue (Evert 2006).
Although the analyzed species of different habits share the same habitat, they do not all have the features that characterize sclerophyllous plants. Variation exists among the different functional traits, and especially among the species exclusive to the “Chaco”. In spite of reduced leaf area and larger stomata, trichomes are unusual and, when present, occur only in small quantities. This fact reveals that each species has a different adaptive strategy for dealing with the semiarid environment. The co-occurrence of different growth habits and taxa that exhibit variation in morphological, anatomical and ecophysiological features indicates that evolution of resistance and/or tolerance to seasonal water stress is the result of the interaction of several factors in the life history of a plant.
Kummerow (1938) reported sclerophyllous structures of leaves for species analyzed in the arid environments of the Mediterranean Basin, Southern California, South Africa, South-Western Australia and Central Chile. This demonstrates that the basic anatomical features of leaves of plants in arid environments are very similar. However, other works describe completely different structures for plants growing in the same habitat, such as the work of Evenari (1938), who quantitatively assessed anatomical traits of leaves of arid and semiarid species in an attempt to classify species according to habit, but failed to achieve positive results.
Our results show that the morphological and anatomical patterns observed for the 16 studied species converge only among the species of vine habit. As expected, the analyzed features allowed the separation of species into two groups: woody species (trees and shrubs) with predominantly sclerophyllous features, such as thicker and smaller leaves, well-developed parenchyma and the presence of sclerenchyma, and herbaceous species (vines and herbs), with predominantly mesomorphic features such as larger leaves with little or no sclerenchyma. We thus conclude that, even though these species cohabit the same environment, these two groups present different adaptive strategies for dealing with the dry environment.
References
Adámoli J (1986) Fitogeografia do Pantanal. In: Anais do I Simpósio sobre recursos naturais e sócio-econômicos do Pantanal. Centro de Pesquisa Agropecuária do Pantanal. Embrapa, Brasília
Boeger MRT, Poulson M (2006) Efeitos da radiação ultravioleta-B sobre a morfologia foliar de Arabidopsis thaliana (L.) Heynh. (Brassicaceae). Act Bot Bras 20:329–338. doi:10.1590/S0102-33062006000200008
Bradshaw AD (2006) Unravelling phenotypic plasticity—why should we bother? New Phytol 170:644–648. doi:10.1111/j.1469-8137.2006.01761.x (PMID: 16684227)
Brasil (1982) Projeto RADAMBRASIL. Levantamento de Recursos Naturais. Ministério de Minas e Energia, Rio de Janeiro
Burrows GE (2001) Comparative anatomy of the photosynthetic organs of 39 xeromorphic species from subhumid New South Wales, Australia. Int J Plant Sci 162:411–430. doi:10.1086/319579
Cabrera AL, Willink A (1980) Biogeografía de America Latina. Secretaria General de la Organización de los Estados Americanos, Washington
Clements FC (1920) Plant indicators: the relation of plant communities to process and practice. The Carnegie Institute of Washington, Washington
Cutter EG (1978) Plant anatomy: cells and tissues. William Clowes and Sons, London
Eamus D (1999) Ecophysiological traits of deciduous and evergreen woody species in the seasonally dry tropics. Trends Ecol Evol 14:1–16. doi:10.1016/S0169-5347(98)01532-8
Elias SRM, Assis RM, Stacciarini-Seraphin E, Rezende MH (2003) Anatomia foliar em plantas jovens de Solanum lycocarpum A.St.-Hil. (Solanaceae). Rev Bras Bot 26:169–174. doi:10.1590/S0100-84042003000200004
Esau K (1977) Anatomy of seed plants. Wiley, New York
Evenari M (1938) The physiological anatomy of the transpiratory organs and the conducting systems of certain plants typical of the wilderness of Judaea. Biol J Linn Soc 51:389–407. doi:10.1111/j.1095-8339.1937.tb01913.x
Evert RF (2006) Esau´s plant anatomy. Meristem´s cells, tissues plants of the plant body—their structure, function and development. Wiley, Hoboken
Fahmy GM (1997) Leaf anatomy and its relation to the ecophysiology of some non-succulent desert plants from Egypt. J Arid Environ 36:499–525. doi:10.1006/jare.1996.0217
Fahn A (1986) Structural and functional properties of trichomes of xeromorphic leaves. Ann Bot 57:631–637. doi:10.1093/oxfordjournals.aob.a087146
Fahn A, Cutler D (1992) Xerophytes. Gebruder Borntraeger, Berlin
Gentry AH (1991) The distribution and evolution of climbing plants. In: Putz FE, Mooney HA (eds) The biology of vines. Cambridge University Press, Cambridge, pp 3–53
Gratani L, Covone F, Larcher W (2006) Leaf plasticity in response to light of three evergreen species of the Mediterranean maquis. Trees 20:549–558. doi:10.1007/s00468-006-0070-6
Hanba YT, Kogami H, Terashima L (2002) The effects of growth irradiance on leaf anatomy and photosynthesis in Acer species differing in light demand. Plant Cell Environ 25:1021–1030. doi:10.1007/s00468-006-0070-6
Hartley JJ, Jones GE (1997) Process oriented supplier development: building the capability for change. Int J Purch Mater Manag 33:24–29. doi:10.1111/j.1745-493X.1997.tb00028.x
Hueck K (1955) Bosques chaquenhos e extração de tanino no Brasil. Rev Bras Geogr 17:343–346
Izaguirre MM, Mazza CA, Svatos A, Baldwin IT, Ballaré CL (2007) Solar ultraviolet-B radiation and insect herbivory trigger partially overlapping phenolic responses in Nicotiana attenuata and Nicotiana longiflora. Ann Bot 99:103–109. doi:10.1093/aob/mcl226 (PMID:17210605)
Johansen D (1940) Plant microtechnique. McGraw-Hill Book Company, New York
Jordan GJ, Dillon RA, Weston PH (2005) Solar radiation as a factor in the evolution of scleromorphic leaf anatomy in Proteaceae. Am J Bot 92:789–796. doi:10.3732/ajb.92.5.789
Kassas M, Batanouny KH (1984) Plant ecology. In: Cloudsley-Thompson JL (ed) Sahara desert (key environments). Pergamon Press, Oxford, pp 77–90
Kolb RM, Medri ME, Bianchini E, Pimenta JA, Giloni PC, Correa GT (1998) Anatomia ecológica de Sebastiania commersoniana (Baillon) Smith & Downs (Euphorbiaceae) submetida ao alagamento. Rev Bras Bot 21:305–312. doi:10.1590/S0100-84041998000300010
Kraus JE, Arduin M (1997) Manual básico de métodos em morfologia vegetal. Seropédica, Rio de Janeiro
Kraus JE, de Souza HC, Rezende MH, Castro NM, Vecchi C, Luque R (1998) Astra blue and basic fuchsin double staining of plants materials. Biotech Histochem 73:235–243 (PMID: 9829416)
Kummerow J (1938) Comparative anatomy of sclerophylls of mediterranean climatic areas. In: Mooney HA, diCastri F (eds) Mediterranean type ecosystems. origin and structure. Springer, Berlin, pp 157–167
Medri ME, Lleras E (1980) Aspectos da anatomia ecológica de folhas de Hevea brasiliensis Müell. Arg. Acta Amazon 10:463–493. doi:10.1590/1809-43921980103463
Metcalfe CR, Chalk L (1988) Anatomy of the dicotyledons. Oxford University Press, London
Mott KA, Gibson AC, O’Leary JW (1982) The adaptive significance of amphistomatic leaves. Plant Cell Environ 5:455–460. doi:10.1111/1365-3040.ep11611750
Niklas KJ, Cobb ED, Niinemets U, Reich PB, Sellin A, Shipley B, Wright IJ (2007) “Diminishing returns” in the scaling of functional leaf traits across and within species groups. PNAS 104:8891–8896. doi:10.1073/pnas.0701135104
Nunes GP (2006) Estudo florístico de formações chaquenhas brasileiras e caracterização estrutural de um remanescente de Chaco de Porto Murtinho, MS, Brasil. Dissertation, Universidade Federal de Mato Grosso do Sul
Pallardy SG (1981) Closely related wood plants. In: Kozlowski KK (ed) Water déficits and plant growth. Academic press, New York
Paviani TI, Haridasan M (1988) Tuberosidade em Vochysia thyrsoidea Pohl (Vochysiaceae). Ciência e Cultura 40:998–1003
Payne WW (1978) A glossary of plant hair terminology. Brittonia 30:239–255
Pearce DW, Millard S, Bray DF, Rood SB (2006) Stomatal characteristics of riparian poplar species in a semi-arid environment. Tree Physiol 26:211–218
Pennington RT, Prado DE, Pendry CA (2000) Neotropical seasonally dry forest and quaternary vegetation changes. J Biogeogr 27:261–273. doi:10.1046/j.1365-2699.2000.00397
Pérez-Harguindeguy N, Díaz S, Garnier E, Lavorel S, Poorter H, Jaureguiberry P, Bret-Harte MS, Cornwell WK, Craine JM, Gurvich DE, Urcelay C, Veneklaas EJ, Reich PB, Poorter L, Wright IJ, Ray P, Enrico L, Pausas JG, de Vos AC, Buchmann N, Funes G, Quétier F, Hodgson JG, Thompson K, Morgan HD, ter Steege H, van der Heijden MGA, Sack L, Blonder B, Poschlod P, Vaieretti MV, Conti G, Staver AC, Aquino S, Cornelissen JHC (2013) New handbook for standardized measurement of plant functional traits worldwide. Aust J Bot 61:167–234. doi:10.1071/BT12225
Prado DE (1993) What is the Gran Chaco vegetation in South America? A review. Contribution to the study of the flora and vegetation of the Chaco. Candollea 48:145–172
Prado DE, Gibbs BE (1993) Patterns of species distributions in the dry seasonal forest in the South America. Ann Mo Bot Gard 80:902–917. doi:10.2307/2399937
Ramella L, Spichiger L (1989) Interpretación preliminar del médio físico y de la vegetación del Chaco Boreal. Contribución al estúdio de la flora y de la vegetación del Chaco. Candollea 44:639–680
Rasband WS (1997–2015) ImageJ. U. S. National Institutes of Health, Maryland, http://imagej.nih.gov/ij/
Rizzini CT (1976) Tratado de Fitogeografia do Brasil. Edusp/Hucitec, São Paulo
Rossato DR, Kolb RM, Franco AC (2015) Leaf anatomy is associated with the type of growth form in Neotropical savanna plants. Botany 93:1–12. doi:10.1139/cjb-2015-0001
Rotondi A, Rossi F, Asunis C, Cesaraccio C (2003) Leaf xeromorphic adaptations of some plants of a coastal Mediterranean macchia ecosystem. J Medit Ecol 4:25–35
Sandquist DR, Ehleringer JR (1997) Intraspecific variation of leaf pubescence and drought response in Encelia farinosa associated with contrasting desert environments. New Phytol 135:635–644. doi:10.1046/j.1469-8137.1997.00697.x
Schluter U, Muschak M, Berger D, Altimann N (2003) Photosynthetic performance of an Arabidopsis mutant with elevated stomatal density (sdd1-1) under different light regimes. J Exp Bot 54:867–874
Scremin-Dias E, Lorenz-Lemke AP, Oliveira AKM (2011) The floristic heterogeneity of Pantanal and the occurrence of species with different adaptive strategies to water stress. Braz J Biol 71:275–282. doi:10.1590/S1519-69842011000200006
Souza AP, Pereira JBA, Silva LDB, Guerra JGM, Carvalho DF (2011) Evapotranspiração, coeficientes de cultivo e eficiência do uso da água da cultura do pimentão em diferentes sistemas de cultivo. Acta Sci Agron 33:15–22. doi:10.4025/actasciagron.v33i1.5527
Turner NC (1986) Adaptation to water deficits: a changing perspective. Func Plant Biol 13:175–190. doi:10.1071/PP9860175
Veloso HP (1972) Aspectos fitoecológicos da Bacia do Alto Rio Paraguai. Instituto de Geografia, São Paulo
Wilson PJ, Thompson K, Hodgson JG (1999) Specific leaf area and dry matter content as alternative predictors of plant strategies. New Phyto 143:155–162. doi:10.1046/j.1469-8137.1999.00427.x
Witkowski ETF, Lamont BB (1991) Leaf specific mass confounds leaf density and thickness. Oecologia 88:486–493. doi:10.1007/BF0031
Acknowledgements
The authors thank CAPES for granting a scholarship to the first author and for granting a Research Productivity Scholarship to the second author (Grant 308793/2013-7), and CNPq for Casadinho/Procad research project funding. The authors would like to thank the owners of the Anahi farm, Nelson Cintra and Maria Lucia Cintra, for their help with field collecting.
Author information
Authors and Affiliations
Corresponding author
Additional information
This paper is part of his dissertation in the Programa de Pós-Graduação em Biologia Vegetal, Universidade Federal de Mato Grosso do Sul, Campo Grande, MS, Brazil.
Rights and permissions
About this article
Cite this article
Guerra, A., Scremin-Dias, E. Leaf traits, sclerophylly and growth habits in plant species of a semiarid environment. Braz. J. Bot 41, 131–144 (2018). https://doi.org/10.1007/s40415-017-0416-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40415-017-0416-x