Abstract
Over the past decade, functional imaging by means of 18F-fluorodeoxyglucose positron emission tomography (FDG-PET/CT) has improved tumor staging and treatment planning leading to somewhat higher survival rates, in particular in NSCLC patients. This review focuses on the recent insight gained and at current challenges encountered while pursuing improved outcome in patients suffering from NSCLC or SCLC.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Non-small cell (NSCLC) and small cell lung cancer (SCLC) are tumor entities characterized by both a high incidence and a high mortality rate [1]. Risk factors for both include tobacco smoking and exposure to chemicals. SCLC accounts for approximately 15% of all lung cancers and is an aggressive tumor entity characterized by a rapid doubling time and early dissemination [2]. Since there is no effective screening method so far and symptoms are vague, about two-thirds of newly diagnosed patients present with locally advanced disease or even distant metastases, the latter in particular being the case in SCLC [3, 4]. Treatment of early-stage NSCLC consists of surgical tumor resection and lymphadenectomy in medically fit patients or (stereotactic) radiotherapy in frail patients or those refusing surgery [5]. Treatment of irresectable locally advanced NSCLC and limited disease SCLC consists of a combination of radiotherapy and platinum-based chemotherapy, preferably given concurrently, but also sequentially in patients with impaired medical condition [6,7,8].
PET imaging for staging in lung cancer
Typically, primary lung tumors are diagnosed on a planar X-ray acquired for diagnosis of pulmonary complaints, i.e., suspicion of pneumonia, work-up prior to cardiac procedures or rheumatologic disease. Thereafter, CT is the method of choice to depict the extent of the primary tumor and intrathoracic lymph node metastases in clinical routine [9]. However, staging on the basis of additional functional imaging by means of FDG-PET/CT has been shown to outperform that of anatomical imaging, and thus has found its way into numerous guidelines as well as clinical practice. This is true for detecting the primary tumor, particularly in the presence of atelectasis, for selecting the affected lymph nodes to be treated and for diagnosing distant metastasis.
The value of FDG-PET/CT for staging of locally advanced NSCLC has recently been summarized by Grootjans et al. [10]. A thorough review of the literature on FDG-PET/CT in staging of SCLC is thus far lacking and therefore given here.
Carter et al. [9] found an upstaging from limited to extensive disease in 19% of SCLC patients and a downstaging from extensive to limited SCLC in 8% of patients using FDG-PET in combination with conventional imaging methods. In addition, in a proof-of-principle study by Saima et al. [11], the rate of agreement between CT and FDG-PET/CT was calculated using Cohen’s kappa. They found a strong (κ = 0.82) correlation for determination of primary tumor, a fair (κ = 0.24) value for lymph nodes and only poor (κ = 0.12) agreement rates for metastases for both imaging techniques. In their population of 23 patients, 47% were upstaged with visceral and bone metastases not detected by means of only CT. Furthermore, PET imaging is more sensitive and specific for detection of distant metastases, except for brain metastases, compared to CT or magnetic resonance imaging (MRI) [9, 12, 13]. Overall, these findings support the important role of FDG-PET/CT for improvement of initial SCLC staging with corresponding prognostic implications.
In SCLC, additional functional imaging techniques, e.g., a 99mTc-labeled vasopressin conjugate as a potential radiopharmaceutical for imaging of oncogene receptors overexpressed, have only been reported in single case studies or in preclinical investigations, but were thus far not translated into clinical routine [14].
PET imaging for radiation treatment planning
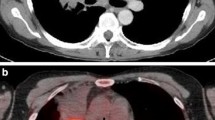
Since primary radiochemotherapy is the mainstay of treatment for patients with locally advanced NSCLC or limited disease SCLC and appropriate performance status, functional imaging could provide important information, especially on target volumes definition for irradiation [6, 8, 15, 16] (Fig. 1).
a Passive scattered proton beam therapy plan (in transverse, sagittal and frontal direction) for irradiation of a cT4N2M0 non-small cell carcinoma patient. b depicts the respective functional and anatomical imaging information gathered by 18Fluorodeoxyglucose-PET-CT at initial diagnosis and taken into account for radiation treatment planning
The added value of FDG-PET for delineating the primary tumor over CT alone has been elegantly shown by Steenbakkers et al. [17]. The standard deviation of interobserver variation of the gross tumor volume (GTV) contoured by 11 experts in thoracic radiotherapy significantly decreased when offering the participants an FDG-PET/CT as opposed to a CT scan only (both without intravenous contrast agent). Moreover, a good correlation between the macroscopic tumor extension determined in the resection specimen and the size of the tumor on pre-treatment has been reported [18, 19]. Using FDG-PET/CT, atelectasis surrounding the tumor as well as tumor infiltration of the mediastinum can be better depicted largely affecting the precision of defining the target volume, and decreasing the irradiated volume as well as the dose to tumor-surrounding radiation-sensitive organs at risk.
In the era of two- and three-dimensional radiotherapy, being based on planar X-ray imaging or computed tomography, respectively, increasing the radiation dose from 50 to 66 Gy in NSCLC was merely hampered by the large, elective mediastinal volume irradiated. That is why in the early 2000s, there were increasing efforts to identify the affected lymph nodes and subsequently irradiate these selectively and to a higher dose. De Ruysscher et al. [20] and Belderbos et al. [21] pioneered this approach in locally advanced NSCLC patients both using a FDG-PET/CT-based selective nodal irradiation (SNI) using a 3D radiation technique. In the first study, 44 locally advanced NSCLC patients were irradiated to doses of 61.2–64.8 Gy (1.8 Gy fractions b.i.d.) and after a median follow-up period an isolated nodal recurrence rate of 2.3% was reported [20]. The same incidence of regional recurrences was reported by Belderbos et al. [21] in a cohort of 88 NSCLC patients treated with escalating radiation doses. Hypothesizing that the incidental dose to the non-selected regional lymph nodes may at least in part be responsible for these favorable results, we recently retrospectively analyzed the regional recurrence rate of locally advanced NSCLC patients having undergone SNI in the era of intensity-modulated radiation therapy (IMRT) [22]. Based on treatment plans and outcome data of 183 patients, the isolated nodal recurrence rate was 1.6% and the combined locoregional recurrence rate 2.2%. Thus, the concept of SNI is still valid in the era of high-conformal radiotherapy. The guideline proposed by Senan et al. [23] is still applicable nowadays, stating that FDG-PET positive lymph nodes should be included in the GTV as should PET-negative lymph nodes with either a large necrotic core on CT or with pathological conformation on cytology.
In contrast to NSCLC, where SNI has been accepted as gold standard, there is no final consensus on nodal irradiation volumes in SCLC, even though most institutions nowadays adhere to the selective concept. In a small prospective study on omission of elective nodal irradiation (ENI) in patients with SCLC, noteworthy based on CT scans only, an unexpectedly high rate of isolated nodal failures (11%) occurred [24]. In contrast, another prospective phase II study on CT-based omission of ENI in 38 SCLC patients revealed no isolated nodal recurrence [25]. These results are supported by further retrospective data on CT-based SNI in SCLC [26]. Conversely, radiation treatment planning based on FDG-PET/CT scans reported by van Loon et al. [27] and Shirvani et al. [28] revealed considerably lower rates of isolated nodal failures in the order of 2–3%. These data seem to support the use of SNI also in limited-stage SCLC when incorporating FDG-PET/CT thus resulting in reduction of radiation dose to organs at risk and potentially increase dose to the target [29, 30].
Regarding this, the results of the recently published CONVERT trial are also of major interest [31]. This multicenter randomized phase III superiority trial compared the currently used twice-daily irradiation (45 Gy in 1.5 Gy fractions b.i.d. [8]) to a once-daily radiotherapy with a higher total dose in line with locally advanced NSCLC (66 Gy in 2 Gy fractions), both with concurrent chemotherapy. Interestingly, the survival outcomes did not differ significantly between both regimes and toxicity was both lower than expected and similar in both groups. In this study, FDG-PET/CT was non-mandatory for staging and radiation treatment planning including the definition of the target volume for mandatory SNI. Since the results are somewhat unexpected, the authors discussed that potentially the prolonged overall treatment time may be the cause and that dose escalation to a biologically defined sub-volume may be of benefit, again underlining possible advantages of specific functional imaging techniques in SCLC.
Prognostic value of PET imaging before, during and after treatment
In addition to treatment planning and staging accuracy, functional imaging, especially different FDG-PET parameters, may also have prognostic value in patients with lung cancer. In locally advanced NSCLC patients, repeat FDG-PET/CT imaging has been conducted for two reasons—first to predict treatment outcome by comparing pre- to per-treatment functional imaging, and second to correlate the high uptake volume prior to treatment with that of the recurrent disease.
A recent review has summarized the role of FDG-PET/CT for early response evaluation in NSCLC [32]. From this it can be appreciated, that results differ widely, some of which are given here. Van Elmpt et al. [33] performed FDG-PET/CT scans before and in the second week of radiochemotherapy in 34 consecutive locally advanced NSCLC patients. They found that CT-based tumor volume did not correlate with overall survival (OS), whereas a decrease in mean standardized uptake value (SUVmean) of 20 ± 21% was found in those patients surviving 2 years as opposed to an increase by 2 ± 22% in non-survivors. In a prospective imaging study, 28 locally advanced NSCLC patients underwent FDG-PET/CT before treatment, at the end of the second week of treatment, and 2 weeks and 3 months after completion of radiochemotherapy [34]. These FDG-PET/CT scans were evaluated regarding maximum standardized uptake value (SUVmax), metabolic tumor volume (MTV), and total lesion glycolysis (TLG). The authors found pre-treatment TLG to be a prognostic factor for worse progression-free survival (PFS). Moreover, a decrease in TLG of more than 38% between pre- and per-treatment imaging was associated with a significantly longer PFS. Grootjans et al. [35] evaluated the use of TLG derived from different automatic segmentations algorithms for early response monitoring (i.e., prior to and in second week of treatment) in 27 of these NSCLC patients undergoing radiochemotherapy. The authors reported pre-treatment TLG of the tumor to be predictive for PFS and OS, and the addition of the TLG of the metastatic lymph nodes to improve assessment. Moreover, the difference between pre- and per-treatment TLG of the summed primary tumor and lymph nodes was again statistically significantly associated with PFS and OS. Bearing these results in mind, patient selection for treatment intensification may be a next step. However, it would be appealing to define the target for dose escalation to spare the tumor-surrounding organs at risk.
To find a putative target volume for dose escalation, Aerts et al. [36] assessed 22 locally advanced stage NSCLC patients with persistent FDG-PET/CT after radio(chemo)therapy taken from a total cohort of 55 patients. The authors found that the high FDG uptake volume (standardized uptake value 50%; SUV50%) largely corresponds with that prior to treatment. For all patients, the hotspot of the residual volume (SUV90%) was completely inside the GTV, and largely overlapped with the pre-radio(chemo)therapy SUV50% volume. This finding was validated in an independent patient cohort [37] and led to the design of a prospective multicenter phase II clinical trial, called the PET-boost trial. In this trial, the radiation dose in locally advanced NSCLC patients was increased to either the entire GTV or the SUV50% volume keeping within the tolerance doses for radiation-sensitive organs at risk [38]. The study was finished in October 2017 after completion of patient accrual and results are eagerly being awaited.
In SCLC, several studies on the prognostic value of FDG-PET/CT imaging have been published during the last years and reporting conflicting results. Starting with the SUVmax, as the most frequently used PET parameter, an association between high pre-treatment SUVmax and worse OS or PFS was found in some investigations [39,40,41] whereas other authors could not confirm these correlations [42, 43]. One possible explanation could be the lacking correlation between SUVmax of the primary tumor and disease stage [9]. In contrast, volume-based PET parameters seem to have a more homogeneous association with patient outcome. In particular, the MTV has been shown to provide significant prognostic information on OS and PFS in patients with SCLC [41, 43, 44].
Nevertheless, most of these studies have been conducted retrospectively in heterogeneous patient cohorts limiting the studies’ validity. Some authors even propagate the use of these parameters only in sub-groups, e.g., patients with limited-stage SCLC. Lee et al. [45] evaluated tumor metabolic activities using FDG-PET/CT and their relationship with markers of biological behavior (e.g., lactate dehydrogenase, glucose transporter 1) in SCLC patients. In multivariate analyses, pre-treatment PET parameters in combination with some biological markers remain significant. These results suggest better prognostic values by combining functional imaging with additional biomarkers.
There is only limited data on the significance of FDG-PET/CT for validity of early response or post-therapeutic restaging in SCLC patients. One study on response to first-line chemotherapy in extensive disease SCLC patients suggested that greater MTV and TLG may correlate with poor response [46]. The authors concluded that both PET parameters may be used for therapy decisions in patients who are not suitable for first-line chemotherapy [46]. An earlier investigation on the same topic revealed early metabolic response in CT and FDG after start of chemotherapy as significant prognostic factor for survival in patients with SCLC [47]. One final aim out of these data could be patient selection according to predicted outcome for individualization of treatment.
The use of FDG-PET for post-therapeutic restaging in SCLC patients is currently not a clinical routine. In some studies, comparing this functional imaging modality to CT, 20–57% of the patients were found to have more tumor mass and 14–38% less disease burden than expected using CT alone [9]. Despite this heterogeneity, FDG-PET is suitable for evaluation of residual and/or recurrent disease and may thus be offered to symptomatic patients in good general condition amenable to first-line treatment if residual or recurrent disease is confirmed.
Current developments
During the past few years, radiomics, i.e., automated analyses of large amounts of imaging features, has been shown to provide new potential for personalized medicine [48,49,50]. With the help of such unique information about imaging-based tumor characteristics, in particular non-small cell, lung carcinoma, individualized, multidisciplinary strategies for improved patient outcome seem possible [51]. However, up till now no prospective clinical studies on this approach can be found on www.clinicaltrials.gov thus far, a prerequisite for this experimental approach being introduced in clinical practice.
Except for FDG-PET, other PET tracers depicting tumor characteristics, such as hypoxia and tumor cell proliferation have been investigated in NSCLC. HX4, a hypoxia-related PET tracer, was found to represent overlapping, but also distinct tumor subvolumes in NSCLC patients undergoing both HX4- and FDG-PET imaging [52]. So, depending on the findings of the above-mentioned PET-boost study, HX4 may be a different target for dose escalation. Moreover, the value of repeat 18fluorothymidine (FLT-)PET for outcome prediction in NSCLC patients undergoing radio(chemo)therapy was assessed in 37 locally advanced patients [53]. Paradoxically, stable FLT-PET readings in the second week of treatment were associated with longer overall survival and progression-free survival, as opposed to the hypothesis that patients with decreased FLT-PET uptake would do better, as found in other solid tumors [54, 55]. Thus, the use of this tracer for scientific purposes has been abandoned.
With the introduction of combined MR photon-based linear accelerators in the field of radiation oncology, the value of MRI for identification of affected lymph nodes in the hilum and mediastinum has been assessed in a recent meta-analysis [56]. In the per-patient and per-nodal analysis, this study confirmed that in particular functional MR imaging may augment selective nodal irradiation. Thus far, however, this approach has not been tested in the context of a clinical trial.
Conclusion
In summary, the introduction of FDG-PET/CT in staging, radiation treatment planning and response evaluation has immensely altered the management and target volume definition in lung cancer patients. Currently, studies incorporating metabolic tumor information in defining the boost target volume are being conducted and results are eagerly awaited.
References
Fruh M, De Ruysscher D, Popat S, Crino L, Peters S, Felip E (2013) Small-cell lung cancer (SCLC): ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol 24(Suppl 6):vi99–vi105. https://doi.org/10.1093/annonc/mdt178
Siegel RL, Miller KD, Jemal A (2016) Cancer statistics 2016. CA Cancer J Clin 66(1):7–30. https://doi.org/10.3322/caac.21332
Cuffe S, Moua T, Summerfield R, Roberts H, Jett J, Shepherd FA (2011) Characteristics and outcomes of small cell lung cancer patients diagnosed during two lung cancer computed tomographic screening programs in heavy smokers. J Thorac Oncol 6(4):818–822. https://doi.org/10.1097/JTO.0b013e31820c2f2e
Jett JR, Schild SE, Kesler KA, Kalemkerian GP (2013) Treatment of small cell lung cancer: diagnosis and management of lung cancer: American college of chest physicians evidence-based clinical practice guidelines. Chest 143(5 Suppl):e400S–e419S. https://doi.org/10.1378/chest.12-2363
Palma D, Visser O, Lagerwaard FJ, Belderbos J, Slotman B, Senan S (2011) Treatment of stage I NSCLC in elderly patients: a population-based matched-pair comparison of stereotactic radiotherapy versus surgery. Radiother Oncol 101(2):240–244. https://doi.org/10.1016/j.radonc.2011.06.029
Auperin A, Le Pechoux C, Rolland E, Curran WJ, Furuse K, Fournel P, Belderbos J, Clamon G, Ulutin HC, Paulus R, Yamanaka T, Bozonnat MC, Uitterhoeve A, Wang X, Stewart L, Arriagada R, Burdett S, Pignon JP (2010) Meta-analysis of concomitant versus sequential radiochemotherapy in locally advanced non-small-cell lung cancer. J Clin Oncol 28(13):2181–2190. https://doi.org/10.1200/JCO.2009.26.2543
Faivre-Finn C, Snee M, Ashcroft L, Appel W, Barlesi F, Bhatnagar A, Bezjak A, Cardenal F, Fournel P, Harden S, Le Pechoux C, McMenemin R, Mohammed N, O’Brien M, Pantarotto J, Surmont V, Van Meerbeeck JP, Woll PJ, Lorigan P, Blackhall F, Team CS (2017) Concurrent once-daily versus twice-daily chemoradiotherapy in patients with limited-stage small-cell lung cancer (CONVERT): an open-label, phase 3, randomised, superiority trial. Lancet Oncol 18(8):1116–1125. https://doi.org/10.1016/S1470-2045(17)30318-2
Turrisi AT 3rd, Kim K, Blum R, Sause WT, Livingston RB, Komaki R, Wagner H, Aisner S, Johnson DH (1999) Twice-daily compared with once-daily thoracic radiotherapy in limited small-cell lung cancer treated concurrently with cisplatin and etoposide. New Engl J Med 340(4):265–271. https://doi.org/10.1056/nejm199901283400403
Carter BW, Glisson BS, Truong MT, Erasmus JJ (2014) Small cell lung carcinoma: staging, imaging, and treatment considerations. Radiographics 34(6):1707–1721. https://doi.org/10.1148/rg.346140178
Grootjans W, de Geus-Oei LF, Troost EG, Visser EP, Oyen WJ, Bussink J (2015) PET in the management of locally advanced and metastatic NSCLC. Nat Rev Clin Oncol 12(7):395–407. https://doi.org/10.1038/nrclinonc.2015.75
Saima R, Humayun B, Khalid NI (2017) Triage of limited versus extensive disease on (18)F-FDG PET/CT scan in small cell lung cancer. Asia Ocean J Nucl Med Biol 5(2):109–113. https://doi.org/10.22038/aojnmb.2017.8751
Mitchell MD, Aggarwal C, Tsou AY, Torigian DA, Treadwell JR (2016) Imaging for the pretreatment staging of small cell lung cancer: a systematic review. Acad Radiol 23(8):1047–1056. https://doi.org/10.1016/j.acra.2016.03.017
Treadwell JR, Mitchell MD, Tsou A, Torigian D, Aggarwal C, Schoelles KM (2016) Imaging for the Pretreatment Staging of Small Cell Lung Cancer. Comparative Effectiveness Review No. 174. Agency for Healthcare Research and Quality, Rockville, MD
Gniazdowska E, Kozminski P, Bankowski K, Ochman P (2014) 99mTc-labeled vasopressin peptide as a radiopharmaceutical for small-cell lung cancer (SCLC) diagnosis. J Med Chem 57(14):5986–5994. https://doi.org/10.1021/jm500272r
De Ruysscher D, Nestle U, Jeraj R, Macmanus M (2012) PET scans in radiotherapy planning of lung cancer. Lung Cancer 75(2):141–145. https://doi.org/10.1016/j.lungcan.2011.07.018
Pignon JP, Arriagada R, Ihde DC, Johnson DH, Perry MC, Souhami RL, Brodin O, Joss RA, Kies MS, Lebeau B et al (1992) A meta-analysis of thoracic radiotherapy for small-cell lung cancer. New Engl J Med 327(23):1618–1624. https://doi.org/10.1056/nejm199212033272302
Steenbakkers RJ, Duppen JC, Fitton I, Deurloo KE, Zijp LJ, Comans EF, Uitterhoeve AL, Rodrigus PT, Kramer GW, Bussink J, De Jaeger K, Belderbos JS, Nowak PJ, van Herk M, Rasch CR (2006) Reduction of observer variation using matched CT-PET for lung cancer delineation: a three-dimensional analysis. Int J Radiat Oncol Biol Phys 64(2):435–448. https://doi.org/10.1016/j.ijrobp.2005.06.034
van Baardwijk A, Bosmans G, Boersma L, Buijsen J, Wanders S, Hochstenbag M, van Suylen RJ, Dekker A, Dehing-Oberije C, Houben R, Bentzen SM, van Kroonenburgh M, Lambin P, De Ruysscher D (2007) PET-CT-based auto-contouring in non-small-cell lung cancer correlates with pathology and reduces interobserver variability in the delineation of the primary tumor and involved nodal volumes. Int J Radiat Oncol Biol Phys 68(3):771–778. https://doi.org/10.1016/j.ijrobp.2006.12.067
Stroom J, Blaauwgeers H, van Baardwijk A, Boersma L, Lebesque J, Theuws J, van Suylen RJ, Klomp H, Liesker K, van Pel R, Siedschlag C, Gilhuijs K (2007) Feasibility of pathology-correlated lung imaging for accurate target definition of lung tumors. Int J Radiat Oncol Biol Phys 69(1):267–275. https://doi.org/10.1016/j.ijrobp.2007.04.065
De Ruysscher D, Wanders S, van Haren E, Hochstenbag M, Geeraedts W, Utama I, Simons J, Dohmen J, Rhami A, Buell U, Thimister P, Snoep G, Boersma L, Verschueren T, van Baardwijk A, Minken A, Bentzen SM, Lambin P (2005) Selective mediastinal node irradiation based on FDG-PET scan data in patients with non-small-cell lung cancer: a prospective clinical study. Int J Radiat Oncol Biol Phys 62(4):988–994. https://doi.org/10.1016/j.ijrobp.2004.12.019
Belderbos JS, Heemsbergen WD, De Jaeger K, Baas P, Lebesque JV (2006) Final results of a phase I/II dose escalation trial in non-small-cell lung cancer using three-dimensional conformal radiotherapy. Int J Radiat Oncol Biol Phys 66(1):126–134. https://doi.org/10.1016/j.ijrobp.2006.04.034 (S0360-3016(06)00731-0 [pii])
Martinussen HM, Reymen B, Wanders R, Troost EG, Dingemans AC, Ollers M, Houben R, De Ruysscher D, Lambin P, van Baardwijk A (2016) Is selective nodal irradiation in non-small cell lung cancer still safe when using IMRT? Results of a prospective cohort study. Radiother Oncol 121(2):322–327. https://doi.org/10.1016/j.radonc.2016.10.001
Senan S, Chapet O, Lagerwaard FJ, Ten Haken RK (2004) Defining target volumes for non-small cell lung carcinoma. Semin Radiat Oncol 14(4):308–314. https://doi.org/10.1016/j.semradonc.2004.07.004
De Ruysscher D, Bremer RH, Koppe F, Wanders S, van Haren E, Hochstenbag M, Geeraedts W, Pitz C, Simons J, ten Velde G, Dohmen J, Snoep G, Boersma L, Verschueren T, van Baardwijk A, Dehing C, Pijls M, Minken A, Lambin P (2006) Omission of elective node irradiation on basis of CT-scans in patients with limited disease small cell lung cancer: a phase II trial. Radiother Oncol 80(3):307–312. https://doi.org/10.1016/j.radonc.2006.07.029
Colaco R, Sheikh H, Lorigan P, Blackhall F, Hulse P, Califano R, Ashcroft L, Taylor P, Thatcher N, Faivre-Finn C (2012) Omitting elective nodal irradiation during thoracic irradiation in limited-stage small cell lung cancer-evidence from a phase II trial. Lung Cancer 76(1):72–77. https://doi.org/10.1016/j.lungcan.2011.09.015
Watkins JM, Wahlquist AE, Zauls AJ, Shirai K, Garrett-Mayer E, Aguero EG, Silvestri GA, Sherman CA, Sharma AK (2010) Involved-field radiotherapy with concurrent chemotherapy for limited-stage small-cell lung cancer: disease control, patterns of failure and survival. J Med Imaging Radiat Oncol 54(5):483–489. https://doi.org/10.1111/j.1754-9485.2010.02201.x
van Loon J, De Ruysscher D, Wanders R, Boersma L, Simons J, Oellers M, Dingemans AM, Hochstenbag M, Bootsma G, Geraedts W, Pitz C, Teule J, Rhami A, Thimister W, Snoep G, Dehing-Oberije C, Lambin P (2010) Selective nodal irradiation on basis of (18)FDG-PET scans in limited-disease small-cell lung cancer: a prospective study. Int J Radiat Oncol Biol Phys 77(2):329–336. https://doi.org/10.1016/j.ijrobp.2009.04.075
Shirvani SM, Komaki R, Heymach JV, Fossella FV, Chang JY (2012) Positron emission tomography/computed tomography-guided intensity-modulated radiotherapy for limited-stage small-cell lung cancer. Int J Radiat Oncol Biol Phys 82(1):e91–e97. https://doi.org/10.1016/j.ijrobp.2010.12.072
Han D, Qin Q, Hao S, Huang W, Wei Y, Zhang Z, Wang Z, Li B (2014) Feasibility and efficacy of simultaneous integrated boost intensity-modulated radiation therapy in patients with limited-disease small cell lung cancer. Radiat Oncol 9:280. https://doi.org/10.1186/s13014-014-0280-9
Bütof R, Gumina C, Valentini C, Sommerer A, Appold S, Zips D, Löck S, Baumann M, Troost E (2017) Sites of recurrent disease and prognostic factors in SCLC patients treated with radiochemotherapy. Clin Transl Radiat Oncol 7:36–42
Faivre-Finn C, Snee M, Ashcroft L, Appel W, Barlesi F, Bhatnagar A, Bezjak A, Cardenal F, Fournel P, Harden S, Le Pechoux C, McMenemin R, Mohammed N, O’Brien M, Pantarotto J, Surmont V, Van Meerbeeck JP, Woll PJ, Lorigan P, Blackhall F (2017) Concurrent once-daily versus twice-daily chemoradiotherapy in patients with limited-stage small-cell lung cancer (CONVERT): an open-label, phase 3, randomised, superiority trial. Lancet Oncol 18(8):1116–1125. https://doi.org/10.1016/s1470-2045(17)30318-2
Cremonesi M, Gilardi L, Ferrari ME, Piperno G, Travaini LL, Timmerman R, Botta F, Baroni G, Grana CM, Ronchi S, Ciardo D, Jereczek-Fossa BA, Garibaldi C, Orecchia R (2017) Role of interim (18)F-FDG-PET/CT for the early prediction of clinical outcomes of non-small cell lung cancer (NSCLC) during radiotherapy or chemo-radiotherapy. A systematic review. Eur J Nucl Med Mol Imaging 44(11):1915–1927. https://doi.org/10.1007/s00259-017-3762-9
van Elmpt W, Ollers M, Dingemans AM, Lambin P, De Ruysscher D (2012) Response assessment using 18F-FDG PET early in the course of radiotherapy correlates with survival in advanced-stage non-small cell lung cancer. J Nucl Med 53(10):1514–1520. https://doi.org/10.2967/jnumed.111.102566
Usmanij EA, de Geus-Oei LF, Troost EG, Peters-Bax L, van der Heijden EH, Kaanders JH, Oyen WJ, Schuurbiers OC, Bussink J (2013) 18F-FDG PET early response evaluation of locally advanced non-small cell lung cancer treated with concomitant chemoradiotherapy. J Nucl Med 54(9):1528–1534. https://doi.org/10.2967/jnumed.112.116921
Grootjans W, Usmanij EA, Oyen WJ, van der Heijden EH, Visser EP, Visvikis D, Hatt M, Bussink J, de Geus-Oei LF (2016) Performance of automatic image segmentation algorithms for calculating total lesion glycolysis for early response monitoring in non-small cell lung cancer patients during concomitant chemoradiotherapy. Radiother Oncol 119(3):473–479. https://doi.org/10.1016/j.radonc.2016.04.039
Aerts HJ, van Baardwijk AA, Petit SF, Offermann C, Loon J, Houben R, Dingemans AM, Wanders R, Boersma L, Borger J, Bootsma G, Geraedts W, Pitz C, Simons J, Wouters BG, Oellers M, Lambin P, Bosmans G, Dekker AL, De Ruysscher D (2009) Identification of residual metabolic-active areas within individual NSCLC tumours using a pre-radiotherapy (18)Fluorodeoxyglucose-PET-CT scan. Radiother Oncol 91(3):386–392. https://doi.org/10.1016/j.radonc.2009.03.006
Aerts HJ, Bussink J, Oyen WJ, van Elmpt W, Folgering AM, Emans D, Velders M, Lambin P, De Ruysscher D (2012) Identification of residual metabolic-active areas within NSCLC tumours using a pre-radiotherapy FDG-PET-CT scan: a prospective validation. Lung Cancer 75(1):73–76. https://doi.org/10.1016/j.lungcan.2011.06.003
van Elmpt W, De Ruysscher D, van der Salm A, Lakeman A, van der Stoep J, Emans D, Damen E, Öllers M, Sonke J-J, Belderbos J (2012) The PET-boost randomised phase II dose-escalation trial in non-small cell lung cancer. Radiother Oncol 104(1):67–71
Go SI, Song HN, Kang JH, Kang MH, Kim MJ, Jung J, Chung SI, Choi BH, Hwang IG, Kim SH, Ling H, Lee GW (2014) The clinical impact of the sum of the maximum standardized uptake value on the pretreatment with F-FDG-PET/CT in small-cell lung cancer. Oncology 86(1):1–9. https://doi.org/10.1159/000357136
Pandit N, Gonen M, Krug L, Larson SM (2003) Prognostic value of [18F]FDG-PET imaging in small cell lung cancer. Eur J Nucl Med Mol Imaging 30(1):78–84. https://doi.org/10.1007/s00259-002-0937-8
Park SB, Choi JY, Moon SH, Yoo J, Kim H, Ahn YC, Ahn MJ, Park K, Kim BT (2014) Prognostic value of volumetric metabolic parameters measured by [18F]fluorodeoxyglucose-positron emission tomography/computed tomography in patients with small cell lung cancer. Cancer Imaging 14:2. https://doi.org/10.1186/1470-7330-14-2
Chong S, Lee KS (2007) Spectrum of findings and usefulness of integrated PET/CT in patients with known or suspected neuroendocrine tumors of the lung. Cancer Imaging 7:195–201. https://doi.org/10.1102/1470-7330.2007.0028
Kim SJ, Chang S (2015) Limited prognostic value of SUVmax measured by F-18 FDG PET/CT in newly diagnosed small cell lung cancer patients. Oncol Res Treat 38(11):577–585. https://doi.org/10.1159/000441289
Nobashi T, Koyasu S, Nakamoto Y, Kubo T, Ishimori T, Kim YH, Yoshizawa A, Togashi K (2016) Prognostic value of fluorine-18 fludeoxyglucose positron emission tomography parameters differs according to primary tumour location in small-cell lung cancer. Br J Radiol 89(1059):20150618. https://doi.org/10.1259/bjr.20150618
Lee J, Kim JO, Jung CK, Kim YS, Yoo Ie R, Choi WH, Jeon EK, Hong SH, Chun SH, Kim SJ, Kim YK, Kang JH (2014) Metabolic activity on [18f]-fluorodeoxyglucose-positron emission tomography/computed tomography and glucose transporter-1 expression might predict clinical outcomes in patients with limited disease small-cell lung cancer who receive concurrent chemoradiation. Clin Lung Cancer 15(2):e13–e21. https://doi.org/10.1016/j.cllc.2013.09.005
Yu X, Zhu Y, Wang J, Song X, Zhu L, Men X, Li X, Dai D, Xu W (2017) Pretreatment metabolic parameters measured by 18F-FDG-PET to predict the outcome of first-line chemotherapy in extensive-stage small-cell lung cancer. Nucl Med Commun 38(2):193–200. https://doi.org/10.1097/mnm.0000000000000637
van Loon J, Offermann C, Ollers M, van Elmpt W, Vegt E, Rahmy A, Dingemans AM, Lambin P, De Ruysscher D (2011) Early CT and FDG-metabolic tumour volume changes show a significant correlation with survival in stage I–III small cell lung cancer: a hypothesis generating study. Radiother Oncol 99(2):172–175. https://doi.org/10.1016/j.radonc.2011.03.014
Aerts HJ, Velazquez ER, Leijenaar RT, Parmar C, Grossmann P, Cavalho S, Bussink J, Monshouwer R, Haibe-Kains B, Rietveld D, Hoebers F, Rietbergen MM, Leemans CR, Dekker A, Quackenbush J, Gillies RJ, Lambin P (2014) Decoding tumour phenotype by noninvasive imaging using a quantitative radiomics approach. Nat Commun 5:4006. https://doi.org/10.1038/ncomms5006
Carvalho S, Leijenaar RT, Velazquez ER, Oberije C, Parmar C, van Elmpt W, Reymen B, Troost EG, Oellers M, Dekker A, Gillies R, Aerts HJ, Lambin P (2013) Prognostic value of metabolic metrics extracted from baseline positron emission tomography images in non-small cell lung cancer. Acta Oncol 52(7):1398–1404. https://doi.org/10.3109/0284186X.2013.812795
de Jong EE, van Elmpt W, Leijenaar RT, Hoekstra OS, Groen HJ, Smit EF, Boellaard R, van der Noort V, Troost EG, Lambin P, Dingemans AC (2017) [18F]FDG PET/CT-based response assessment of stage IV non-small cell lung cancer treated with paclitaxel-carboplatin-bevacizumab with or without nitroglycerin patches. Eur J Nucl Med Mol Imaging 44(1):8–16. https://doi.org/10.1007/s00259-016-3498-y
Lee G, Lee HY, Park H, Schiebler ML, van Beek EJR, Ohno Y, Seo JB, Leung A (2017) Radiomics and its emerging role in lung cancer research, imaging biomarkers and clinical management: state of the art. Eur J Radiol 86:297–307. https://doi.org/10.1016/j.ejrad.2016.09.005
Zegers CM, van Elmpt W, Reymen B, Even AJ, Troost EG, Ollers MC, Hoebers FJ, Houben RM, Eriksson J, Windhorst AD, Mottaghy FM, De Ruysscher D, Lambin P (2014) In vivo quantification of hypoxic and metabolic status of NSCLC tumors using [18F] HX4 and [18F] FDG-PET/CT imaging. Clin Cancer Res 20(24):6389–6397. https://doi.org/10.1158/1078-0432.CCR-14-1524
Everitt S, Ball D, Hicks RJ, Callahan J, Plumridge N, Trinh J, Herschtal A, Kron T, Mac Manus M (2017) Prospective study of serial imaging comparing fluorodeoxyglucose positron emission tomography (PET) and fluorothymidine PET during radical chemoradiation for non-small cell lung cancer: reduction of detectable proliferation associated with worse survival. Int J Radiat Oncol Biol Phys 99(4):947–955. https://doi.org/10.1016/j.ijrobp.2017.07.035
Troost EG, Bussink J, Hoffmann AL, Boerman OC, Oyen WJ, Kaanders JH (2010) 18F-FLT PET/CT for early response monitoring and dose escalation in oropharyngeal tumors. J Nucl Med 51(6):866–874. https://doi.org/10.2967/jnumed.109.069310 (jnumed.109.069310 [pii])
Hoeben BA, Troost EG, Span PN, van Herpen CM, Bussink J, Oyen WJ, Kaanders JH (2013) 18F-FLT PET during radiotherapy or chemoradiotherapy in head and neck squamous cell carcinoma is an early predictor of outcome. J Nucl Med 54(4):532–540. https://doi.org/10.2967/jnumed.112.105999
Peerlings J, Troost EGC, Nelemans PJ, Lambin P, Beets-Tan RG, Hoffmann AL (2016) Meta-analysis on the use of MRI for nodal staging in non-small cell lung cancer. Radiology 281(1):151631
Acknowledgements
This work was in part funded by German Cancer Consortium (DKTK), Partner Site Dresden, and German Cancer Research Center (DKFZ), Heidelberg, Germany; and National Center for Tumor Diseases (NCT), Partner Site Dresden, Germany: German Cancer Research Center (DKFZ), Heidelberg, Germany; Faculty of Medicine and University Hospital Carl Gustav Carus, Technische Universität Dresden, Dresden, Germany, and Helmholtz Association / Helmholtz-Zentrum Dresden – Rossendorf (HZDR), Dresden, Germany.
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author(s) declare that they have no competing interests.
Ethical approval
This article does not contain any studies with human or animal subjects performed by any of the authors. Therefore, no ethical approval is required.
Informed consent
This article does not contain any studies with human or animal subjects performed by any of the authors. Therefore, no informed consent is required.
Additional information
The members of the National Center for Tumor Diseases are listed at the end of the article.
Rights and permissions
About this article
Cite this article
Bütof, R., Troost, E.G.C. The role of functional imaging in lung cancer. Clin Transl Imaging 6, 441–447 (2018). https://doi.org/10.1007/s40336-018-0300-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40336-018-0300-0