Abstract
Most running studies have considered level running (LR), yet the regulation of locomotor behaviour during uphill (UR) and downhill (DR) running is fundamental to increase our understanding of human locomotion. The purpose of this article was to review the existing literature regarding biomechanical, neuromuscular and physiological adaptations during graded running. Relative to LR, UR is characterized by a higher step frequency, increased internal mechanical work, shorter swing/aerial phase duration, and greater duty factor, while DR is characterized by increased aerial time, reduced step frequency and decreased duty factor. Grade also modifies foot strike patterns, with a progressive adoption of a mid- to fore-foot strike pattern during UR, and rear-foot strike patterns during DR. In UR, lower limb muscles perform a higher net mechanical work compared to LR and DR to increase the body’s potential energy. In DR, energy dissipation is generally prevalent compared to energy generation. The increased demands for work as running incline increases are met by an increase in power output at all joints, particularly the hip. This implies that UR requires greater muscular activity compared to LR and DR. Energy cost of running (C r) linearly increases with positive slope but C r of DR decreases until a minimum slope is reached at −20 %, after which C r increases again. The effects of slope on biomechanics, muscle contraction patterns and physiological responses have important implications for injury prevention and success of athletes engaged in graded running competitions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Grade-specific biomechanical adaptations occur in uphill and downhill running. These include changes in foot strike pattern and ground reaction forces, joint kinematics and kinetics, and impact shock. |
The observed adaptations directly impact neuromuscular activation, as demonstrated by changes in lower limb electromyographic activity with grade, these changes being specific to the considered muscles. |
Energy cost of running is also affected by slope, which linearly increases as slope increases and linearly decreases as slope decreases until a minimum value is observed at −20 % grade. |
1 Introduction
Running performance derives from a combination of anatomical, physiological and behavioural characteristics that are uniquely evolved in humans [1, 2]. Accordingly, the study of human running has always been of great interest for exercise physiologists and biomechanists in order to increase our understanding of the physiological and biomechanical bases of running capabilities in humans. However, the vast majority of studies have considered only level running (LR). This is not surprising because until recently, the major running events have been road races such as 10 km, half-marathon and marathon, which are usually run on flat courses [3]. This, however, is not always the case. South Africa, for instance, has held the world’s most famous ultra-marathon race since 1921 [3], the Comrades Marathon, which consists of a ~90-km long course that varies each year between the ‘up’ run [87 km, 1167 m of positive elevation change (D+)] starting from Durban, and the ‘down’ run [89 km, 1810 m of negative elevation change (D−)] starting from Pietermaritzburg. More importantly, ultra-trail running has become increasingly popular [4]. Indeed, in the last 40 years, there has been an exponential growth of participation in ultra-trail races (Fig. 1), likely due to greater appeal of these competitions compared to road and track events [4]. There are countless races worldwide over a broad range of distances and variations in slope. In Europe, Ultra-Trail du Mont Blanc® (UTMB) and Tor des Geants® (TdG) are famous mountain ultra-marathon races, characterized not only by an extreme distance/duration (166 and 330 km, respectively) but also a high elevation (±9500 and ±24000 m, respectively) [5–8]. The total number of participants is now close to 10000 over five different distances for the UTMB and 800 for the TdG, with 6500 and 5000 runners refused in the 2015 edition, respectively. In North America, the 161 km Western States Endurance Run® (with 5500 m of D+ and 7000 m of D−) has increased exponentially in the number of finishers from 1977 to 2008 [4].
Number of 161-km ultra-trail races in North America from 1977 through 2008 (dashed line) and number of >100-km ultra-trail races in France (solid line). Based on data from Hoffman et al. [4] and from personal data
Ultra-trail and shorter trail running races typically involve running over (extremely) long distances on rough terrain with large positive/negative elevation changes throughout [5–8]. Thus, the study of the physiological and biomechanical changes associated with graded running is important because the ability to appropriately regulate locomotor behaviour in response to changes in grade is fundamental to increase our understanding of the control of human locomotion [9–15]. Running speed was reported to decrease by 0.1 to 0.3 km·h−1 for every 1 % change in gradient [16, 17], leading to important biomechanical, neuromuscular and physiological adaptations. Events such as ultra-trail running are likely to be at the extremes of human tolerance [18], and understanding these adaptations is of paramount importance for health, training and competition reasons.
The purpose of this review is to provide a comprehensive overview of the literature related to the biomechanical, neuromuscular and physiological responses to graded running. In this review, manuscripts were acquired by searching the electronic databases of MEDLINE, PubMed, ScienceDirect, SPORTDiscus and Web of Science using the following keywords in various combinations: ‘level’, ‘uphill’, ‘downhill’, ‘incline’, ‘decline’, ‘grade’, ‘gradient’, ‘slope’, ‘hilly’, ‘running’, ‘physiology’, ‘biomechanics’. Due to the narrow focus on graded running, a relatively limited number of articles were retrieved, and consequently, no limit to the search period was applied. Electronic database searching was supplemented by examining the bibliographies of relevant articles. In the literature, the terms ‘slope’ and ‘gradient’ are interchangeably used, and are employed in this paper to indicate running with variations in slope unless specified otherwise. The important effects of graded running on fatigue and tissue damage warrant their own critical evaluation, and are therefore not considered in the current review.
2 Biomechanical Adaptations
2.1 Spatiotemporal Parameters and Centre of Mass Kinematics
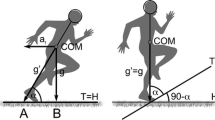
Several studies have systematically examined the influence of uphill running (UR) on kinematic variables [10, 12, 14, 19–23] (Fig. 2) and observed differences when compared to level running (LR). In UR, higher gradients have been reported to be associated with a higher step frequency [10, 12, 19, 24] and consequently increased internal mechanical work [12]. On the other hand, Gottschall and Kram [10] did not observe any variation in contact and aerial times at a given running speed (10.8 km·h−1) between LR, UR and downhill running (DR) (±5.2, 10.5 and 15.8 %). Thus, no changes in duty factor, step frequency, and stride length were observed. Similarly, Telhan et al. [24] reported no changes in step frequency or length when comparing –7 % DR, LR, and 7 % UR at 11 km·h−1. UR is characterized by a shorter swing/aerial phase duration and a greater proportion of the stride cycle spent in stance, i.e. duty factor (e.g. ~33 vs. ~29 % for UR and LR, respectively) [23].
Contact time was found to be constant in DR at 10 km·h−1, while aerial time increased at –8 and –5 % grade compared to LR and UR (2, 5 and 8 %) at the same speed, resulting in a lower step frequency in DR [19, 25]. Similarly, a greater stride length was reported during DR at 12 km·h−1 and –17.4 % compared to 17.4 % UR at a similar running speed [26].
Changes in external mechanical energy both during UR and DR are concomitant with changes in running kinematics (Fig. 2; Table 1). Lower vertical displacement of the centre of mass (CoM) during the stance phase and greater vertical displacement during the aerial phase were illustrated in DR compared to UR (e.g. ~–20 and +44 % at 12 km·h−1 and ±17.4 % grade, respectively) [26]. In this study [26], the vertical displacement during the braking (downward displacement) and propulsive (upward displacement) phases was not separated. Lussiana et al. [25] specified that running at 10 km·h−1 and –8 and –5 % grades induced greater downward displacement of the CoM during the stance phase compared to running at shallow or positive grades.
The use of laboratory settings to simulate UR and DR implies significant technical challenges, limiting the subject’s ability to regulate running speed [17]. Accordingly, Townshed et al. [17] investigated speed regulation and kinematics parameters during a 9525-m overground UR and DR. They illustrated that, when compared with LR, stride length was 20.5 % shorter and 16.2 % longer during UR and DR, respectively, suggesting that running speed during overground graded running is predominantly regulated by stride length.
2.2 Foot strike and Ground Reaction Forces
Grade has been shown to modify foot strike pattern during running. Gottschall and Kram [10] and Lussiana et al. [19] reported that runners progressively adopted a mid-foot or forefoot strike pattern as slope changed from LR to UR. Lussiana et al. [19] also reported a less pronounced heel strike angle during running on inclined versus declined gradients for a constant running speed. During downhill trail running conditions, it was observed that high-level trail runners (based on their skill in DR) adopted a midfoot strike pattern, whereas medium-level trail runners adopted a rearfoot strike pattern, irrespective of the technical profile of the run (e.g. steep or shallow slope, with or without rocks) [27]. In real trail running, however, runners may not adopt a single foot strike pattern because of the need to adapt to uneven surfaces, as was observed in a high-level trail runner [28]. It has been recently observed that the foot strike pattern adopted during a 6.5-km downhill trail run (with 1264-m of negative elevation change and run as fast as possible) differently influences the components of tibial shock [29]. Although a forefoot strike reduces impact severity and impact frequency content along the tibial transverse axis, a rearfoot strike decreases them in the tibial axial direction.
Gottschall and Kram [10] investigated the ground reaction forces in running at 10.8 km·h−1 at grades from –15.8 to +15.8 %. Normal (i.e. perpendicular to running surface) impact force peaks decreased with slope, but normal active force peaks remained the same. On the contrary, the parallel propulsive force peaks and impulses increased with grade [10]. That is, the parallel propulsive impulse was 57 % lower in DR and 68 % higher in UR at –15.8 and +15.8 % slope, respectively, when compared to LR. These changes likely reflect the need for developing greater propulsive forces to generate the required upward acceleration imposed by grade [22]. Recently, Padulo et al. [22] investigated ground reaction forces during running at 15 km·h−1 on a 50-m asphalt road with different grades. These authors observed increases in forces and impulses of 12 and 14.7 %, respectively, as the slope increased from 0 to 7 %.
The impact force at foot contact is determined by the effective lower extremity mass, landing velocity and passive shoe stiffness [30, 31]. Effective mass changes as a function of joint angle, therefore if kinematics do not drastically change across gradients, for a given shoe, only the speed at which the foot strikes the ground would influence the impact force. As such, one would expect higher impact forces in DR than in LR or UR. Indeed, a greater impact peak force was observed during –7 % DR compared to LR or 7 % UR at 11 km·h−1 [24]. Similarly, Gottschall and Kram [10] reported greater normal impact forces at –15.8, –10.5 and –5.2 % DR than in LR for a similar running speed (Fig. 3a, b), as well as higher loading rates at the –15.8 and –10.5 % gradients compared to LR. Regarding the antero-posterior component of force, greater braking peak and impulse were observed by Gottschall and Kram [10] in the three tested DR conditions compared to LR. For instance, during –15.8 % DR, the braking impulse increased by 108 % compared to LR [10].
Ground reaction forces (GRFs) expressed as percentage of body weight (BW) in different gradient conditions during treadmill running at 10.8 km·h−1: a normal; b parallel. Adapted from Gottschall and Kram [10], with permission. c Typical signals of tibial accelerations for the vertical (solid line) and transversal (dashed line) components measured along the tibial anteromedial aspect in various conditions of slope during a trail running race. From authors’ personal data
2.3 Mechanics and Joint Kinetics
Knowledge of the changes in joint mechanics and kinetics during UR and DR running may be important to understand the adaptive mechanisms induced by sloped running, and can provide additional information on the risk factors for lower limb injuries. In the following sections, we discuss how graded running may impact joint mechanics and kinetics. A summary of the studies investigating the effect of UR and DR on the main kinematics and kinetics parameters at ankle, knee and hip joints is presented in Table 2.
2.3.1 Mechanical Energy Fluctuations of the CoM
During LR, runners’ CoM behaviour oscillates cyclically during each stride [32], and both the potential and kinetic energy fluctuations are in-phase [15, 33]. External work, defined as the sum of potential, and horizontal and vertical kinetic work associated with the displacement of the CoM, was measured at different grades (−15 to 15 %) and speeds (7.9 to 11.8 km·h−1) [12]. It was found that negative external work (i.e. the work done to decelerate the body’s CoM with respect to the environment [9]) linearly increased as the slope decreased [12]. For instance, negative external work was ~25 % of the total external work during UR at 15 % grade, but was ~75 % during DR at –15 % grade. Therefore, more net mechanical energy generation is required in UR, i.e. energy generation is greater than absorption. Minetti et al. [12] measured internal and external mechanical work during UR at different speeds. They reported that both positive external mechanical work (i.e. the work done to move the body’s CoM with respect to the environment [9]) and internal mechanical work (i.e. the work done to move the lower limbs with respect to the CoM [9]) per unit of distance travelled increase linearly with slope. Thus, UR requires a greater net mechanical work with each step to increase the body’s potential energy [13], and the muscles must perform greater net positive work both to raise the body’s CoM to its height at toe-off and to give it sufficient kinetic energy to reach its highest point during the aerial phase [14].
In DR, albeit positive external work is necessary at shallow slopes (−5 %), energy dissipation, rather than energy generation, is generally observed [14, 15]. Snyder et al. [15] measured the mechanical energy fluctuations of the CoM during LR, UR and DR at 10.8 km·h−1. They observed that some positive mechanical energy is still required at shallow grades (−5.6 and –10.5 %), but almost all mechanical energy is negative at –15.6 % grade. Accordingly, at 12 km·h−1, both the total negative and positive work of the joints were 38 % greater and 265 % lower in DR (−17.4 %) than in UR (+17.4 %), respectively [26].
2.3.2 Ankle Joint Kinematics and Kinetics
Combined with video analysis, ground reaction forces can provide useful information about the joint torques of the support leg during running. Data on the effects of grade on ankle kinematics are rather heterogeneous, but there is basically no major or consistent change between LR, UR, and DR both in terms of ankle kinematics and negative work. Ankle positive work does not change at shallow slopes but increases in UR at steep slopes compared to DR.
Over 30 min of DR (−7 %) and LR (5 % faster than the individual anaerobic threshold: 12.7 ± 0.7 km·h−1), ankle range of motion during the braking phase (dorsiflexion) only increased after 15 min of DR [34]. Comparing UR (30 % grade) and LR at 16.0 km·h−1, Swanson and Caldwell [23] observed that the ankle was more dorsiflexed at foot strike and exhibited a lower dorsiflexion range of motion in UR. However, the plantar flexion range of motion was greater during the propulsive phase in UR. Ankle angular velocity was also considerably lower during the stance phase in UR than in LR. During the swing phase, ankle range of motion was greater in UR than in LR, with angular velocity being similar between the two conditions. Although Telhan et al. [24] observed no changes in ankle kinematics between LR, UR and DR (±7 %) at 11 km·h−1, Buczek and Cavanagh [35] showed that the instant of maximum dorsiflexion, ankle peak power absorption, ankle negative work and the relative duration of the ankle negative work period over the stance phase increased by 10, 36, 52 and 34 %, respectively, during –8.3 % DR compared to LR at a given speed (15.9 km·h−1). Furthermore, the power generation at the ankle joint was decreased by 49 % in DR (−17.4 % grade) compared to UR (+17.4 %) at 12 km·h−1 and no difference in ankle negative work was observed between DR and UR [26]. Roberts and Belliveau [13] observed no variation in the net work done at the ankle joint in UR compared to LR at 10.8 to 12.6 km·h−1 and 0, 10 and 21.2 % grades.
2.3.3 Knee Joint Kinematics and Kinetics
Knee flexion at contact increases in UR compared to LR and DR [23, 24]. During the braking phase, knee power absorption increases in DR as a result of a greater knee range of motion; while during the propulsion phase, knee power generation increases at steep UR gradients but is constant across grades at shallow slopes [26].
DR was associated with substantially more knee extension at initial contact than LR [34, 35] and UR [19] at a given speed [range: 10–16 km·h−1]. DR was also associated with greater knee range of motion in the sagittal plane during the braking phase [34, 35], as well as a delayed time to peak knee flexion [35] at a constant running speed (~13 or 16 km·h−1) compared to LR. These kinematic changes lead to greater negative work (+21 %) and relative duration of the negative work at the knee over the stance phase (+7 %) compared to LR [35], as well as increased power absorption at the knee compared to LR and UR [24]. DeVita et al. [26] observed that 54 % more power was developed at the knee joint during DR at 12 km·h−1 and –17.4 % compared to UR at +17.4 % and the same speed, which may be attributed to greater energy absorption at the knee joint during the braking phase of running. When comparing UR (30 % grade) and LR at 16.0 km·h−1, it was observed that the knee was more flexed at foot strike in UR than in LR (59.7° vs. 21.0°, respectively) [23]. These authors also reported a lower knee range of motion during the braking phase (flexion), but a greater knee range of motion during the propulsion phase (extension) in UR. When comparing UR and LR at 10.8 and 12.6 km·h−1 at 0, 10 and 21.2 % grades, Roberts and Belliveau [13] observed no effect of slope on the net work done at the knee joint.
2.3.4 Hip Joint Kinematics and Kinetics
In DR, the hip range of motion increases, inducing greater hip power absorption compared to LR [24, 26, 34]. Conversely in UR, hip power generation increases compared to DR [13].
More specifically, greater hip range of motion was reported during the braking phase at –7 % DR compared to LR at 12.7 ± 0.7 km·h−1 [34]. Swanson and Caldwell [23] observed that the hip was more flexed at foot strike in UR (30 % grade) than in LR at the same speed (16 km·h−1). Although in LR the hip flexed during the braking phase and then extended during the propulsion phase, the authors stated that in UR the hip was extending rapidly at foot strike, more slowly during the braking phase, and rapidly again during the propulsion phase. The authors also reported a greater hip range of motion during the propulsion phase (extension) in UR than in LR. Swanson and Caldwell [23] investigated the kinetics of the lower limb muscles during LR and UR at 16.2 km·h−1 and 30 % grade. They observed that average hip power during the swing phase was ~200 % higher during UR. Roberts and Belliveau [13] extended this work by measuring joint kinematics during running between 10.8 and 12.6 km·h−1 at 0, 10 and 21.2 % grades. They found that the net work done at the hip increased with running incline (~+140 % at 21.2 % grade). Using a musculoskeletal model, Yokozawa et al. [36] confirmed these previous findings, illustrating that the estimated muscle torque of the hip extensors and flexors were greater during UR at different speeds and 9.1 % grade.
Hip power during the braking phase was also found to be higher at –7 % DR compared to LR at the same running speed (11 km·h−1) [24]. Hip joint power was lower at –17.4 % DR compared to 17.4 % UR, for a similar speed [26]. It is worth mentioning that DeVita et al. [26] noticed that the ground reaction force vector was directed farther from the hip joint centre in UR than in DR, lengthening the lever arm. The authors proposed that the shorter moment arm at the hip reduced the work demand on the hip flexor and/or extensor muscles.
2.3.5 Summary
In summary, the overall joint work on the lower limbs seems to be 28 % higher in UR than in DR [26]. However, it also seems that graded running does not dramatically affect the distribution of negative work between the joints. Both during DR and UR, the knee joint performed the highest negative work (DR: 63 %, UR: 58 %), followed by the ankle (DR: 23 %, UR: 30 %) and the hip (DR: 15 %, UR: 12 %) [26]. This distribution changes when positive work is considered. Hip seems to be the most stressed joint (DR: 48 %, UR: 55 %), followed by the ankle (DR and UR: 32 %) and knee (DR: 20 %, UR: 13 %) [26]. Thus, there are differences in joint stabilization between UR and DR that may directly influence, for example, the risk of developing graded running-injuries.
2.4 Impact Shock Attenuation in Downhill
As mentioned in Sect. 2.2, initial foot contact kinetics change as a function of slope. Impact accelerations are also largely affected by DR. DeVita et al. [26] postulated that the greater impact force in DR would likely cause larger accelerations of musculo-skeletal tissues, requiring greater energy dissipation by muscles as well as the heel pad, bones and spinal discs. In laboratory experiments (i.e. standardized running speed) and in real practice (i.e. running speed increases as the gradient decreases), negative correlations were observed between slope and axial, transverse (i.e. along the axis of the anteromedial tibial aspect) and resultant peak tibial accelerations [28, 37, 38] (Fig. 3c), as well as with high-frequency vibration content, i.e. median frequency, along these three acceleration components [28]. Hamill et al. [38] reported that tibial shock increased by 30 % during DR on a –8.7 % slope compared to LR at the same speed. Chu and Caldwell [37] observed an average of 23 % and 48 % increase in peak tibial and head acceleration, respectively, at –12 % DR compared to LR (15 km·h−1). Increases of 51 and 125 % in impact-related frequencies (i.e. power spectral densities within the 12–20 Hz bandwidth) were also observed at the tibia and head, respectively [39]. According to these findings, DR induces a decrease in shock attenuation [37]. However, Mizrahi et al. [34] observed similar peak tibial acceleration but larger peak sacral acceleration in –7 % DR compared to LR at 12.7 ± 0.7 km·h−1. These authors also reported a lower amplitude within the impact frequency range at the tibia during DR compared to LR, but no differences at the sacrum were observed. Interestingly, Chu and Caldwell [37] found a bimodal response in peak shock attenuation in DR, i.e. half of the subjects illustrated increased shock attenuation during DR compared to LR while the other half illustrated decreased shock attenuation. Analysing these two subgroups, the authors found several differences in their respective kinematic adaptations to DR: (i) those with reduced shock attenuation in DR displayed a 5° greater dorsiflexion and 4.3° lower hip flexion at heel strike than those with increased shock attenuation; (ii) at mid-stance, the subgroup with reduced shock attenuation exhibited greater dorsiflexion, knee flexion, and lower hip flexion compared to the subgroup with increased shock attenuation; (iii) the subgroup with reduced shock attenuation increased the stance and stride duration compared to the subgroup with increased shock attenuation. Therefore, the results of Chu and Caldwell [37] suggest that shock attenuation can be increased during DR by adopting a less pronounced heel strike and a forward leaning trunk. In this sense, it has been recently observed during a downhill trail run that the more anterior the foot strike pattern, the greater the axial and resultant impact-related vibrations (i.e. 12–20 Hz) between tibia and sacrum. Since knee flexion at initial contact increases when forefoot striking (e.g. Shih et al. [40]), one could assume that the improved shock attenuation with anterior foot strike patterns could be related to a greater knee flexion at initial contact. Indeed, Gottschall and Kram [10] proposed that impact forces can be moderated by increasing knee flexion at initial contact and reducing stride length during DR. This is in line with previous observations that increased knee flexion improves shock attenuation during various dynamic and static tasks [41–44].
3 Neuromuscular Adaptations
Table 3 shows differences in the electromyographic (EMG) activity of different muscle groups investigated as a function of running grade. Abe et al. [45] compared vastus lateralis activity among 0 and ±5 % grades at 11.9 km·h−1 and observed lower activity during the concentric phase (i.e. propulsion) of DR compared to UR but not LR. These authors did not observe any differences between slopes in the intensity and duration of vastus lateralis activity during the eccentric phase (i.e. braking). One would expect more vastus lateralis activity during the eccentric phase in DR. Indeed, as previously mentioned, the absolute and relative negative works as well as the percentage of stance time in negative work were significantly higher for knee extensor muscles in DR than in LR [35]. A possible explanation for the lack of significant difference in the study of Abe et al. [45] could be the rather low gradient examined (±5 %) which may have induced a minimal change in the power absorption performed by the knee extensors, suggesting also that a minimal gradient of –7 % is necessary to significantly increase knee power absorption [24, 35]. The ratio between vastus lateralis muscle activity during the eccentric phase to activity during the concentric phase was significantly greater during DR compared to UR [45]. Mizrahi et al. [34] investigated the consequences of 30-min of DR (slope: –7 %) compared to LR at a speed slightly higher than anaerobic threshold (12.7 ± 0.7 km·h−1). They found no difference in EMG activity of the rectus femoris muscle between LR and DR in the first 15 min of running. While the effects of fatigue are beyond the scope of the present review, it can be noted that differences between LR and DR running appeared after 15 min [34].
The slope-related changes in UR biomechanics as discussed above would be expected to require higher activation patterns of lower limb muscles, i.e. greater motor unit recruitment in UR. However, the effects of slope on EMG activity are not the same for all lower limb muscles. This is likely due to the varying roles of different lower limb muscles in producing force at various phases of the gait cycle [46] and to the fact that slope alters joint mechanics non-uniformly [26]. EMG studies have provided considerable information on the timing of individual muscle activity throughout the gait cycle of UR. Globally, a greater activation compared to LR is usually found in the iliopsoas, gluteus maximus, adductor muscles, hamstrings and vastii muscles, tibialis anterior, and gastrocnemius (Table 3). More specifically, the hip flexors have been found to generate more energy and higher moments during the swing phase [13, 23]. During the braking/absorption phase (i.e. from foot strike to mid-stance), higher activation has been measured for the gluteus maximus, vastii muscles, gastrocnemius and soleus whereas during the propulsion phase (i.e. from mid-stance to toe-off), higher activation has been measured for the gluteus maximus, hamstrings and vastii muscles, gastrocnemius and soleus [22, 23, 47]. Collectively, these studies illustrated that greater EMG activity in the lower limb muscles (e.g. up to 83 and 100 % for gluteus maximus and vastus lateralis, respectively) exists in UR at a given speed. This greater EMG activity is likely associated with a greater force production [48] primarily for concentric muscle contractions during the second phase of stance [12].
Exercise-induced contrast shifts in magnetic resonance images before and after (time between termination of exercise and completion of the post-exercise image being 11–12 min) high-intensity running at ~115 % of the peak oxygen uptake (VO2peak) were examined by Sloniger et al. [49]. These authors showed that the EMG activity of the lower limb muscle was 6 % greater during UR at 10 % grade compared to LR. Using the same technique, a subsequent study demonstrated that, compared to LR, UR required a greater activation of the vastus group (+23 %) and soleus (+14 %) paralleled by less activation of the rectus femoris (−29 %), gracilis (−18 %) and semitendinosus (−17 %) [50].
It is important to note that most of the aforementioned studies assessed EMG activity/muscle activation at a given absolute speed, limiting their applicability to real world settings where speed is naturally reduced during UR. The behaviour of muscle activity when the exercise intensity (or the energy expenditure) is kept nearly constant is currently unknown.
4 Physiological Consequences of Biomechanical and Neuromuscular Changes
4.1 Energy Cost of Running
In LR, the energy cost of running (C r), defined as the amount of energy spent to transport the subject’s body a given distance [51], does not change with speed when expressed as oxygen uptake [52]. However, when C r is expressed in terms of caloric unit cost it seems to be more sensitive to changes in speed, even when normalized per distance travelled [53, 54]. Changes in the kinetic and potential energy in one stride are almost in-phase [9], implying that the energy storage accomplished by a muscle-tendon unit and passive muscle elasticity (the so-called stretch-shortening cycle [55, 56]) contribute to one of the energy-saving mechanisms during running [45, 57–59]. This feature is recognized as one of the major determinants of C r [9]. Indeed, in running, the storage and release of elastic energy contributes to accelerate the body upwards during the propulsive phase and reduce energy production needed during the concentric phase, since the advantage of elastic energy is how much muscle work it can replace [14, 15, 60]. During LR, it has been estimated that the elastic energy stored in the Achilles’ tendon and the foot arch aponeurosis accounted for approximately 43 % of the total positive mechanical energy at each step [15]. However, during UR and DR, Snyder et al. [14] hypothesized that the use of elastic energy may be compromised due to a mismatch between the possibility to store the elastic energy during landing and to use that elastic energy during take-off. Indeed, at 10 km·h−1 the maximum possible elastic energy use was 20.4 and 11.7 % lower when UR (+5.2 %) and DR (−5.2 %) were compared to LR [14], reflecting more a decrease in the maximum possible elastic energy storage and return rather than a change in the anatomically estimated elastic energy storage [15]. Yet, even if energy released from the stretch-shortening cycle is low and the ability of the muscle tendon units to store elastic energy during landing and to release that energy during take-off is reduced [12], the main explanation for the higher C r in UR is the increased net mechanical energy generation required to overcome the potential energy associated with slope. Thus, greater muscle activity (see above) is required to generate a relatively high amount of positive (concentric) work during the push-off phase in order to both raise the CoM and offset the diminished maximum possible elastic energy storage and return [14]. This ultimately results in a C r increase. In the last decade, several studies have examined the effects of increased gradient on C r. Despite some methodological differences in the way C r was expressed [i.e. oxygen cost (ml·kg−1·m−1) or caloric cost (J·kg−1·m−1)], all studies report a linear increase in C r with each increment in the slope gradient (see an example in Fig. 4) [12, 14, 19, 22, 45, 61, 62].
Metabolic energy cost of running (C r) as a function of grade. Asterisk indicates significantly different from level gradient (P < 0.0001). Based on data from Minetti et al. [61]
When measured on a treadmill at different moderate slopes, a reduction of C r, as well as heart rate, ventilation or total EMG, has been consistently observed in DR compared to LR and UR [45, 63–66], with the effects of grade not being significantly different between males and females [65]. For moderate slopes, the following equation has been proposed [64]:
where v is the running velocity in m min−1. This would suggest that C r (when expressed as oxygen uptake) is 22 % lower at –5 % DR compared to LR. However, when considering steeper slopes (from –45 to +45 %), the following fifth-order polynomial regression has been proposed [61]:
where i is the gradient in %. According to this equation, the decrease in C r is no longer linear after –20 % grade, where the relationship inverts and C r increases with further decreases in downhill slope (Fig. 4) [61]. Note that this optimal slope was found to be lower (−10 %) by the same authors in a previous paper [12] yet C r seems to plateau between –10 and –20 %. This is in line with the downhill slope at which mechanical energy must no longer be generated (−16 %) according to Snyder et al. [15]. In other words, this optimal negative grade can be explained by the fact that on steeper downhill grades, mechanical energy dissipation must occur, whereas on less steep downhill grades, though more mechanical energy is dissipated than generated, some positive mechanical energy must be generated [15]. Interestingly, the slope allowing for the best C r (−20 %) is steeper than the slope at which the best energy cost of walking is observed [61]. Gottschall and Kram [10] argued that at progressively steeper declines, the parallel propulsive impulse decreased exponentially, while the parallel braking impulse increased linearly. This different change in the propulsive and braking impulses could explain the increase in the metabolic cost below –20 % [61] due to higher concentric muscle contractions at steeper versus shallow declines [10].
Vertical speed directly measured during UR is very close to that predicted by Minetti et al. [61] whereas the predicted speed for DR overestimates the measured speed [61]. Minetti et al. [61] identified methodological issues that hampered accurate reproduction of outdoor conditions in the laboratory (e.g. differences between the rough terrain and smooth treadmill surface). We are not aware of any study measuring C r in UR or DR in the field at a constant speed. The key point is that C r measured during running does not reflect the reality in the field. In particular, assuming that C r is independent of speed in DR is probably wrong. Minetti et al. [61] also introduced the notion of vertical cost of running (C r-vert), defined as the energy expenditure to run a distance that corresponds to a vertical displacement of 1 m. Contrary to classic C r, C r-vert was found to be stable below –20 % and above +20 %. Recently, Giovannelli et al. [67] extended this knowledge, observing that at a fixed vertical speed of 1.26 km·h−1 there is a range of angles for which C r-vert is minimized (between 37.2 % and 70 %), with a minimum value at 50.9 %.
4.2 Other Physiological Specificities of Graded Running
It is known that hilly races (even though not characterized by a net change in elevation) are not as fast as level ones. Staab et al. [68] suggested that although running pace changed inversely with percentage grade on hilly courses, subjects were not able to maintain a constant energy expenditure during the race. Indeed, the increase in DR pace was inadequate to maintain a level VO2 [68], i.e. the change from LR to UR and from LR to DR resulted in a 40 % increase and a 27 % decrease in VO2, respectively. This indicates that the greater metabolic demands of UR are not compensated for by the lower metabolic demands of DR [69]. In an attempt to investigate speed regulation during overground running, Townshend et al. [17] showed that while natural pace was reduced in UR and increased in DR compared to LR, these pace changes were not enough to keep VO2 stable: VO2 was found to be 100 % of ventilatory thresholds in UR, 89 % in LR and 79 % in DR. Interestingly, these authors also reported that the velocity in LR was systematically influenced by the preceding slope, i.e. UR or DR [17]. It has also been reported that in simulated competition conditions, lactate increased in UR compared to LR even though running pace decreased [68]. In line with this finding, for a given blood lactate concentration, which also corresponded to the same VO2 but obviously different velocities (i.e. ~8.5, 11 and 13.5 km h−1 for UR, LR and DR, respectively), Kolkhorst et al. [70] reported that rating of perceived exertion tended to be higher in DR than LR and UR, and LR tended to be higher than UR. Graded running is associated with modified breathing patterns, i.e. the locomotor-respiratory coupling [71]. It is worth reporting that increasing or decreasing the stride frequency away from preferred values alters the metabolic cost similarly during LR, UR and DR [14] and this is true when the stride frequency is manipulated or the optimal stride frequency applied [14].
Costill et al. [72] demonstrated that glycogen depletion (as assessed by muscle biopsy) was higher in the vastus lateralis, gastrocnemius and soleus muscle fibres after 2-h of UR at 10 % grade compared to 2-h of LR at the same relative intensity (i.e., ~75 % VO2max). Given that glycogen utilization by human skeletal muscle varies as a function of both work done and intensity [73], this finding confirms that a higher percentage of muscle mass is recruited during UR in these muscles. Furthermore, the greater muscle activity observed during UR (see Sect. 3) seems also to be responsible for a higher peak oxygen deficit [49].
A few studies systematically examined the influence of slope on maximal accumulated oxygen deficit, an indicator of the anaerobic capacity [74]. Olesen [62] determined this during treadmill running at 1, 10, 15 and 20 % grade. Compared with running at 1 % grade, maximal accumulated oxygen deficit increased by 37 % at 10.5 % grade and ~80 % at 15 % grade, without any further increase at 20 % grade. Walker et al. [75] and Sloniger et al. [49] reported similar findings, with a 26 and 21 % increase in the maximal accumulated oxygen deficit as treadmill grade was increased from 0 to 10 %, respectively. Together, these findings may indicate that the maximal anaerobic energy production is greater during UR due to increased skeletal muscle mass activation in the lower limbs [49, 62, 75], even if differences in running efficiency as well as testing and calculations procedures (i.e. a linear relationship between work rate and energy demand) cannot be ruled out [76].
In summary, graded running induces specific adaptations related to modified physiological strain proportional to the slope gradients from –20 to +45 %, and natural reductions in pace may not necessarily allow for a reduction in total strain. Furthermore, given the higher muscle activation and the increase in the anaerobic energy production that leads to lower C r, runners exert a higher physiological strain during UR compared to LR. Conversely, due to the mechanical consequences discussed in Sect. 2, DR may result in more mechanical stress, particularly in non-familiarised runners, leading to muscle damage and lower limb injuries.
5 Conclusion and Future Directions
The present review shows that several grade-specific differences exist between LR, UR and DR regarding biomechanics, neuromuscular adaptations and physiological responses. Higher step frequency and increased internal mechanical work, shorter swing/aerial phase duration, and greater duty factor are the main kinematic features of UR. Compared to LR, DR is characterized by a similar contact time and a tendency toward higher aerial time and lower step frequency. A progressive adoption of a mid- to fore-foot and rear-foot strike pattern has been observed during UR and DR, respectively. During UR, lower limb muscles perform more net mechanical work compared to LR and DR to increase the body’s potential energy. The increased demand for work during UR is met by an increase in power output at all joints, particularly at the hip which induces greater muscular activity compared to LR and DR, and in turn a linear increase in the energy cost of running. In DR, energy dissipation is higher than energy generation and this decreases the energy cost during DR until a minimum is reached at –20 % and increases again at steeper negative slopes. Thus, the metabolic cost associated with various types of muscle contractions remains a valid explanation for the high and low cost of UR and DR, respectively, from –20 to +45 % grades.
DR increases tibial shock and impact force, which have been associated with overuse injuries. Additionally, the muscle activity required for the increased power and eccentric energy absorption during DR would place additional stress on musculoskeletal tissues. However, since in graded (e.g. trail) running, the locomotion pattern changes more often than in LR, overuse injuries related to repetitive movement may be attenuated in graded running compared to LR.
In conclusion, the present review represents a useful synthesis of all research describing the relevant biomechanical and (neuro)physiological changes associated with graded running. However, as we have highlighted throughout this review, important gaps in our biomechanical and physiological understanding of graded running still exist. In particular, controlled training studies or well-designed interventional experiments are needed to investigate the effect of manipulating both running speed and positive/negative slope at the same relative intensity on muscle activity.
References
Bramble DM, Lieberman DE. Endurance running and the evolution of Homo. Nature. 2004;432:345–52.
Lieberman DE, Bramble DM. The evolution of marathon running : capabilities in humans. Sports Med. 2007;37:288–90.
Burfoot A. The history of the marathon : 1976-present. Sports Med. 2007;37:284–7.
Hoffman MD, Ong JC, Wang G. Historical analysis of participation in 161 km ultramarathons in North America. Int J Hist Sport. 2010;27:1877–91.
Millet GY, Tomazin K, Verges S, et al. Neuromuscular consequences of an extreme mountain ultra-marathon. PLoS One. 2011;6:e17059.
Saugy J, Place N, Millet GY, et al. Alterations of neuromuscular function after the world’s most challenging mountain ultra-marathon. PLoS One. 2013;8:e65596.
Vernillo G, Rinaldo N, Giorgi A, et al. Changes in lung function during an extreme mountain ultramarathon. Scand J Med Sci Sports. 2015;25:e374–80.
Vernillo G, Savoldelli A, Zignoli A, et al. Influence of the world’s most challenging mountain ultra-marathon on energy cost and running mechanics. Eur J Appl Physiol. 2014;114:929–39.
Cavagna GA, Saibene FP, Margaria R. Mechanical work in running. J Appl Physiol. 1964;19:249–56.
Gottschall JS, Kram R. Ground reaction forces during downhill and uphill running. J Biomech. 2005;38:445–52.
Higham TE, Biewener AA. Integration within and between muscles during terrestrial locomotion: effects of incline and speed. J Exp Biol. 2008;211:2303–16.
Minetti AE, Ardigo LP, Saibene F. Mechanical determinants of the minimum energy cost of gradient running in humans. J Exp Biol. 1994;195:211–25.
Roberts TJ, Belliveau RA. Sources of mechanical power for uphill running in humans. J Exp Biol. 2005;208:1963–70.
Snyder KL, Farley CT. Energetically optimal stride frequency in running: the effects of incline and decline. J Exp Biol. 2011;214:2089–95.
Snyder KL, Kram R, Gottschall JS. The role of elastic energy storage and recovery in downhill and uphill running. J Exp Biol. 2012;215:2283–7.
Mastroianni GR, Zupan MF, Chuba DM, et al. Voluntary pacing and energy cost of off-road cycling and running. Appl Ergon. 2000;31:479–85.
Townshend AD, Worringham CJ, Stewart IB. Spontaneous pacing during overground hill running. Med Sci Sports Exerc. 2010;42:160–9.
Millet GP, Millet GY. Ultramarathon is an outstanding model for the study of adaptive responses to extreme load and stress. BMC Med. 2012;10:77.
Lussiana T, Fabre N, Hebert-Losier K, et al. Effect of slope and footwear on running economy and kinematics. Scand J Med Sci Sports. 2013;23:e246–53.
Padulo J, Annino G, Migliaccio GM, et al. Kinematics of running at different slopes and speeds. J Strength Cond Res. 2012;26:1331–9.
Padulo J, Annino G, Smith L, et al. Uphill running at iso-efficiency speed. Int J Sports Med. 2012;33:819–23.
Padulo J, Powell D, Milia R, et al. A paradigm of uphill running. PLoS One. 2013;8:e69006.
Swanson SC, Caldwell GE. An integrated biomechanical analysis of high speed incline and level treadmill running. Med Sci Sports Exerc. 2000;32:1146–55.
Telhan G, Franz JR, Dicharry J, et al. Lower limb joint kinetics during moderately sloped running. J Athl Train. 2010;45:16–21.
Lussiana T, Hébert-Losier K, Mourot L. Effect of minimal shoes and slope on vertical and leg stiffness during running. J Sport Health Sci. 2015;4:195–202.
DeVita P, Janshen L, Rider P, et al. Muscle work is biased toward energy generation over dissipation in non-level running. J Biomech. 2008;41:3354–9.
Horvais N, Giandolini M. Foot strike pattern during downhill trail running. Footwear Sci. 2013;5:S26–7.
Giandolini M, Pavailler S, Samozino P, et al. Foot strike pattern and impact continuous measurement during a trail running race: proof of concept in a world-class athlete. Footwear Sci. 2015;7:127–37.
Giandolini M, Horvais N, Rossi J, et al. Foot strike pattern differently affects the axial and transverse components of shock acceleration and attenuation in downhill trail running. J Biomech. 2016. doi:10.1016/j.jbiomech.2016.04.001.
Gerritsen KG, van den Bogert AJ, Nigg BM. Direct dynamics simulation of the impact phase in heel-toe running. J Biomech. 1995;28:661–8.
Liu W, Nigg BM. A mechanical model to determine the influence of masses and mass distribution on the impact force during running. J Biomech. 2000;33:219–24.
Ardigò LP, Saibene F, Minetti AE. The optimal locomotion on gradients: walking, running or cycling? Eur J Appl Physiol. 2003;90:365–71.
Saibene F, Minetti AE. Biomechanical and physiological aspects of legged locomotion in humans. Eur J Appl Physiol. 2003;88:297–316.
Mizrahi J, Verbitsky O, Isakov E. Fatigue-induced changes in decline running. Clin Biomech (Bristol, Avon). 2001;16:207–12.
Buczek FL, Cavanagh PR. Stance phase knee and ankle kinematics and kinetics during level and downhill running. Med Sci Sports Exerc. 1990;22:669–77.
Yokozawa T, Fujii N, Ae M. Muscle activities of the lower limb during level and uphill running. J Biomech. 2007;40:3467–75.
Chu JJ, Caldwell GE. Stiffness and damping response associated with shock attenuation in downhill running. J Appl Biomech. 2004;20:291–308.
Hamill CL, Clarke TE, Frederick EC, et al. Effects of grade running on kinematics and impact force. Med Sci Sports Exerc. 1984;16:185.
Shorten MR, Winslow DS. Spectral analysis of impact shock during running. Int J Sport Biomech. 1992;8:288–304.
Shih Y, Lin KL, Shiang TY. Is the foot striking pattern more important than barefoot or shod conditions in running? Gait Posture. 2013;38:490–4.
Derrick TR. The effects of knee contact angle on impact forces and accelerations. Med Sci Sports Exerc. 2004;36:832–7.
Derrick TR, Hamill J, Caldwell GE. Energy absorption of impacts during running at various stride lengths. Med Sci Sports Exerc. 1998;30:128–35.
Edwards WB, Derrick TR, Hamill J. Musculoskeletal attenuation of impact shock in response to knee angle manipulation. J Appl Biomech. 2012;28:502–10.
Lafortune MA, Lake MJ, Hennig EM. Differential shock transmission response of the human body to impact severity and lower limb posture. J Biomech. 1996;29:1531–7.
Abe D, Fukuoka Y, Muraki S, et al. Effects of load and gradient on energy cost of running. J Physiol Anthropol. 2011;30:153–60.
McClay IS, Lake MJ, Cavanagh PR. Muscle activity in running. In: Cavanagh PR, editor. Biomechanics of distance running. Champaign: Human Kinetics; 1990. p. 165–86.
Wall-Scheffler CM, Chumanov E, Steudel-Numbers K, et al. Electromyography activity across gait and incline: the impact of muscular activity on human morphology. Am J Phys Anthropol. 2010;143:601–11.
Mero A, Komi PV. Force-, EMG-, and elasticity-velocity relationships at submaximal, maximal and supramaximal running speeds in sprinters. Eur J Appl Physiol Occup Physiol. 1986;55:553–61.
Sloniger MA, Cureton KJ, Prior BM, et al. Anaerobic capacity and muscle activation during horizontal and uphill running. J Appl Physiol. 1985;1997(83):262–9.
Sloniger MA, Cureton KJ, Prior BM, et al. Lower extremity muscle activation during horizontal and uphill running. J Appl Physiol. 1985;1997(83):2073–9.
di Prampero PE, Atchou G, Bruckner JC, et al. The energetics of endurance running. Eur J Appl Physiol Occup Physiol. 1986;55:259–66.
Margaria R, Cerretelli P, Aghemo P, et al. Energy cost of running. J Appl Physiol. 1963;18:367–70.
Fletcher JR, Esau SP, Macintosh BR. Economy of running: beyond the measurement of oxygen uptake. J Appl Physiol. 1985;2009(107):1918–22.
Shaw AJ, Ingham SA, Folland JP. The valid measurement of running economy in runners. Med Sci Sports Exerc. 2014;46:1968–73.
Nicol C, Avela J, Komi PV. The stretch-shortening cycle : a model to study naturally occurring neuromuscular fatigue. Sports Med. 2006;36:977–99.
Komi PV. Stretch-shortening cycle: a powerful model to study normal and fatigued muscle. J Biomech. 2000;33:1197–206.
Cavagna GA. Storage and utilization of elastic energy in skeletal muscle. Exerc Sport Sci Rev. 1977;5:89–129.
Cavagna GA, Heglund NC, Taylor CR. Mechanical work in terrestrial locomotion: two basic mechanisms for minimizing energy expenditure. Am J Physiol. 1977;233:R243–61.
Cavagna GA, Kaneko M. Mechanical work and efficiency in level walking and running. J Physiol. 1977;268:467–81.
Lichtwark GA, Wilson AM. Interactions between the human gastrocnemius muscle and the Achilles tendon during incline, level and decline locomotion. J Exp Biol. 2006;209:4379–88.
Minetti AE, Moia C, Roi GS, et al. Energy cost of walking and running at extreme uphill and downhill slopes. J Appl Physiol. 1985;2002(93):1039–46.
Olesen HL. Accumulated oxygen deficit increases with inclination of uphill running. J Appl Physiol. 1985;1992(73):1130–4.
Dick RW, Cavanagh PR. An explanation of the upward drift in oxygen uptake during prolonged sub-maximal downhill running. Med Sci Sports Exerc. 1987;19:310–7.
Robergs RA, Wagner DR, Skemp KM. Oxygen consumption and energy expenditure of level versus downhill running. J Sports Med Phys Fit. 1997;37:168–74.
Pivarnik JM, Sherman NW. Responses of aerobically fit men and women to uphill/downhill walking and slow jogging. Med Sci Sports Exerc. 1990;22:127–30.
Maciejczyk M, Więcek M, Szymura J, et al. Comparison of physiological and acid-base balance response during uphill, level and downhill running performed at constant velocity. Acta Physiol Hung. 2013;100:347–54.
Giovanelli N, Ortiz AL, Henninger K, et al. Energetics of vertical kilometer foot races; is steeper cheaper? J Appl Physiol (1985). 2016;120:370–5.
Staab JS, Agnew JW, Siconolfi SF. Metabolic and performance responses to uphill and downhill running in distance runners. Med Sci Sports Exerc. 1992;24:124–7.
Gregor RJ, Costill DL. A comparison of the energy expenditure during positive and negative grade running. J Sports Med Phys Fit. 1973;13:248–52.
Kolkhorst FW, Mittelstadt SW, Dolgener FA. Perceived exertion and blood lactate concentration during graded treadmill running. Eur J Appl Physiol Occup Physiol. 1996;72:272–7.
Takano N. Phase relation and breathong pattern during locomotor/respiratory coupling in uphill and downhill running. Jpn J Physiol. 1990;45:47–58.
Costill DL, Jansson E, Gollnick PD, et al. Glycogen utilization in leg muscles of men during level and uphill running. Acta Physiol Scand. 1974;91:475–81.
Saltin B, Karlsson J. Muscle glycogen utilization during work of different intensities. In: Pernow B, Saltin B, editors. Muscle metabolism during exercise. New York: Springer US; 1971. p. 289–99.
Medbø JI, Mohn AC, Tabata I, et al. Anaerobic capacity determined by maximal accumulated O2 deficit. J Appl Physiol. 1985;1988(64):50–60.
Walker GT, Cureton KJ, DuVal HP, et al. Effects of external loading on peak oxygen deficit during treadmill running (Abstract). Med Sci Sports Exerc. 1994;26:S179.
Bangsbo J. Is the O2 deficit an accurate quantitative measure of the anaerobic energy production during intense exercise? J Appl Physiol. 1985;1992(73):1207–9.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No sources of funding were used to assist in the preparation of this article.
Conflict of interest
Gianluca Vernillo, Marlène Giandolini, W. Brent Edwards, Jean-Benoît Morin, Pierre Samozino, Nicolas Horvais and Guillaume Y. Millet declare that they have no conflicts of interest relevant to the content of this review.
Rights and permissions
About this article
Cite this article
Vernillo, G., Giandolini, M., Edwards, W.B. et al. Biomechanics and Physiology of Uphill and Downhill Running. Sports Med 47, 615–629 (2017). https://doi.org/10.1007/s40279-016-0605-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40279-016-0605-y