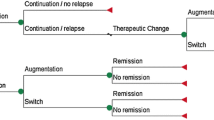
Abstract
Background
Various treatment approaches are available for depression. Given the scarcity of healthcare resources, it is important to optimise treatment availability in an efficient manner. Economic evaluations can inform the optimal allocation of healthcare resources. However, there is currently no review synthesising what is known about the cost effectiveness of treatments for depression in low- and middle-income countries (LMICs).
Methods
This review identified articles from six database searches: APA PsycINFO, CINAHL Complete, Cochrane Library, EconLit, Embase and MEDLINE Complete. Trial- and model-based economic evaluations published between 1 January 2000 and 3 December 2022 were included. The quality of health economic studies (QHES) instrument was used to assess the quality of the included papers.
Results
This review comprised 22 articles, with most studies (N = 17) focusing exclusively on the adult population. Even though evidence regarding the cost effectiveness of antidepressants for treating various forms of depression was inconsistent; an atypical antipsychotic (aripiprazole) was frequently reported to be cost effective for treatment-resistant depression. Task shifting (aka task sharing) to lay health workers or non-specialist health care providers appeared to be a cost-effective approach for treating depression in LMICs.
Conclusions
Overall, this review found mixed evidence on the cost effectiveness of depression treatment choices among LMICs, with some indication that task sharing with lay health workers may be cost effective. Future research will be needed to fill the gaps around the cost effectiveness of depression treatments in younger people and beyond healthcare facilities.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
There is mixed evidence regarding the cost effectiveness of depression treatments in low- and middle-income countries (LMICs). |
The review found that task shifting (aka task sharing) to lay health workers or non-specialist health care providers appears to be cost effective for treating depression in LMICs settings. |
Cost-effectiveness evidence is particularly scarce in younger populations and community-based settings. Further studies are required to fill these gaps. |
1 Background
Major depressive disorder (MDD) is a mental illness marked by somatic and cognitive symptoms that limit one's capacity to function and is characterised by depressed mood and the loss of interest in daily activities [1, 2]. According to the Global Burden of Disease Study 2019, MDD accounted for 1.5% of global disability-adjusted life-years (DALYs) in 2019; and contributed to 29.6% of DALYs within the subset of all mental disorders [3]. Moreover, the global DALYs burden for MDD increased by 28% during the COVID-19 pandemic, which disproportionately impacted females and younger age groups [4]. While the global pandemic has caused a significant increase in depression prevalence [4, 5], a rising trend in prevalence rates was already observed prior to the pandemic, particularly in Sub-Saharan Africa and Oceania [6].
In addition to health burden, depression is associated with a significant economic burden. In high-income country settings, indirect costs (absenteeism or presenteeism) and direct costs (medical costs, prescription drug costs) accounted for over 61% and 35% of the total cost for MDDs, respectively, with the remainder of costs attributable to suicide [7]. Similarly, findings from low- and middle-income countries (LMICs) reported that indirect (including the costs of suicide) and direct costs of depression account for 84% and 16% of the total cost of depression [8]. The economic burden of MDD is compounded by the lack of public health insurance or reimbursement schemes covering care and treatment for severe mental diseases (for example, globally, 27% of countries lack such coverage), forcing service users to pay out of pocket, which accounted for 40–43% of mental health costs in African and South-East Asian regions [9, 10].
Despite the significant increase in the disease burden of MDD over the last two decades [11], numerous treatment options are available that can be selected based on disease severity and patient characteristics [12, 13]. Depression treatments can be broadly categorised into two types: (1) pharmacological treatment, including the most commonly used antidepressants, such as tricyclic antidepressants (TCAs) (e.g., amitriptyline, desipramine, imipramine, nortriptyline), selective serotonin reuptake inhibitors (SSRIs) (e.g., citalopram, fluvoxamine, fluoxetine, sertraline, paroxetine) and serotonin and norepinephrine reuptake inhibitors (SNRIs) (e.g., duloxetine, venlafaxine, milnacipran) [14]; and (2) non-pharmacological treatments consisting of psychological interventions such as cognitive behavioural therapy (CBT) and interpersonal therapy (IPT), as well as complementary and alternative medicine, dietary intervention and physical exercise [15]. Despite the availability of various treatments for depression, there are disparities in treatment rates, with 48.3% seeking care in higher-income countries and 16.8% in lower-income countries [16]. Improving treatment availability and making the right choice from the available options is important for decision makers. Economic evaluations can, in turn, assist with such healthcare decision making [17].
To our knowledge, there is no review synthesising what is known about the cost effectiveness of pharmacological and non-pharmacological treatment options for depression in LMICs. This review aims to provide a comprehensive summary of the cost effectiveness of treatment options for depression in LMICs that is useful for decision makers and other researchers (e.g., to inform model-based economic evaluations). It addresses the following questions concerning the cost-effectiveness evidence for depression treatments in LMICs:
-
(i)
Which interventions provide the best value for money?
-
(ii)
How are interventions delivered?
-
(iii)
In which setting are they delivered (e.g., primary care, the hospital or community-based care)?
2 Methods
This systematic review aligns with the latest Preferred Reporting Items for a Systematic Review and Meta-Analyses (PRISMA) guidelines [18] and has been registered on PROSPERO (CRD42021288015).
2.1 Search Strategy
The PICO (Population, Interventions, Comparison and Outcomes) framework was used to construct the search strategy for electronic databases. The search was built around four blocks, with terms related to depression (block 1), intervention (block 2), cost effectiveness (block 3) and LMICs (block 4). The search included keywords and subject headings, which were subject to database-specific truncation, wildcard and/or proximity operators. The keywords and terms in each category were combined with the database’s subject headings using the Boolean operator ‘OR’; after that, the Boolean operator ‘AND’ was used to combine each block into the main search (see Electronic Supplementary Material [ESM]). Initial searches were conducted in six electronic databases (APA PsycInfo; CINAHL Complete; Cochrane Library; EconLit; Embase; and MEDLINE Complete) on 16 August 2021 and updated on 3 December 2022. The Global Health Cost-Effectiveness Analysis (GH CEA) Registry [19] was also searched, and a targeted Google search was performed. A search was undertaken on the World Bank and World Health Organization websites to find grey literature. Finally, a reference list search of included studies was conducted and a forward citation search in Scopus identified studies that cited the included studies.
2.2 Eligibility
The inclusion criteria comprised economic evaluation studies (e.g., cost-effectiveness analysis [CEA], cost-utility analysis [CUA] and cost-benefit analysis [CBA]) and return-on-investment studies associated with the treatment of depression in LMICs [20]. Studies involving patients with comorbidities were excluded but were eligible if the study’s primary goal was to determine the cost effectiveness of depression treatment. Trial- and model-based studies published between 1 January 2000 and 3 December 2022 were searched; literature older than 20 years was excluded to identify the most recent evidence. Primary research, conference abstracts, books, book chapters, dissertations and reviews were included in the search to check for any eligible primary studies; while editorials, commentaries and articles with ‘no full text’ or ‘full text written in a language other than English’ were excluded. Partial economic evaluations that just reported cost estimates or treatment outcomes (but not both) were excluded. Studies that looked for pharmacological and non-pharmacological therapies were incorporated based on the eligibility criteria regardless of participant age.
2.3 Study Selection
Study selections were made in accordance with PRISMA guidelines. EndNote software [21] was used to de-duplicate the total records retrieved from the six databases, which were then exported to the Covidence platform for screening [22]. Two authors (YBB and NL) completed title/abstract screening for the unique articles, while a third author (LE) resolved disputes between the first two authors.
2.4 Data Extraction and Analysis
Data extraction was performed by the study's primary author (YBB) and double-checked by another co-author (LE), with any discrepancies resolved through discussion. Data were collected on the following study characteristics: author; year; country; target population; intervention type; intervention delivery setting and intervention providers. Details of the study methods concerning the economic evaluation type, analytical approach, perspective, time horizon, discount rate, cost items, currency, reference year and health outcomes were also extracted. For model-based evaluations, the model type (e.g., decision tree, Markov model or microsimulation) was described as part of the analytical approach, while the design of the clinical trial and the sample size were described for trial-based evaluations. The study's main findings, including the difference in costs and health outcomes for intervention versus comparator, were extracted. The incremental cost-effectiveness ratio was summarised for the base-case analysis, uncertainty analysis and authors’ conclusions. Costs included in the respective study were categorised based on the Second Panel on Cost-Effectiveness in Health and Medicine's recommendations [23], which differentiate between (i) formal healthcare sector costs (medical costs), including future related and unrelated medical costs paid by third-party payers or out-of-pocket by patients; (ii) informal healthcare sector costs, such as patient time and unpaid caregiver time and (iii) non-healthcare sector costs, such as productivity losses or cost of social services. Health outcomes such as quality-adjusted life-year (QALY) gains, DALYs averted, remission and depression-free days were also extracted. To aid the comparison of various depression management techniques, incremental cost-effectiveness ratios (ICERs), benefit-to-cost ratios and average cost-effectiveness ratios were converted to 2021 United States dollars (2021 US$) using the CCEMG-EPPI-Centre online tool [24]. A value judgement for cost effectiveness was based primarily on the author's conclusion. Furthermore, this judgement was discussed based on comparing the ICER value with the willingness-to-pay (WTP) criterion. The WHO's cost-effectiveness recommendation [25], which uses thresholds of 1–3 GDP, is no longer recommended [26]. Therefore, we used alternative recommendations such as GDP-based thresholds of 0.5–1.5 GDP and opportunity cost thresholds of 0.5 GDP [26,27,28].
2.5 Quality Assessment
The quality of the included papers was assessed by two reviewers (YBB & NL) using the quality of health economic studies (QHES) instrument. This tool includes assessments of the study objectives; perspectives; variable estimates; data sources; costs; outcome measures; model structure and description of analysis. It consists of 16 yes/no questions, each with a distinct weighting based on the health economist expert review [29]. A recent study has modified the QHES tool to improve the grading system and increase its flexibility in evaluating studies with different characteristics [30]. This review used the modified version of the QHES tool with further amendments (e.g., ‘none’ as a scoring option has been added to all questions) to improve scoring.
3 Results
3.1 Study Selection
A total of 9962 records were retrieved from the six databases and 5953 unique articles after de-duplication were exported to the Covidence platform for screening. Irrelevant articles were removed in the title/abstract screening, leaving 179 articles for full-text screening and 14 papers that were deemed suitable for inclusion. The reference lists and a forward citation search of included studies yielded eight additional publications for review. The total of 22 included studies covered a range of interventions, economic evaluation methodologies and geographic locations (Fig. 1).
3.2 Study Characteristics
In-scope studies encompassed a total of 19 country-specific studies from India (32%) [31,32,33,34,35,36], Pakistan [37,38,39], Thailand [40,41,42], Uganda [43, 44], Brazil [45], Colombia [46], Ethiopia [47], Nigeria [48] and Turkey [49]; and three regional studies involving countries from Sub-Saharan Africa [50, 51] and Asia [50, 52]. Most studies (N = 17) focused exclusively on the adult population, while five studies involve populations of all ages (children, adolescents or elderly populations) [37, 47, 48, 51]. A number of studies examined specific sub-populations, such as women with perinatal depression [32, 36, 39], or conditions like post-stroke depression [33].
Of the 22 studies, seven studies examined pharmacological treatments only, whereas the remaining studies assessed treatment approaches that incorporated non-pharmacological interventions alone (N = 3) or in combination with pharmacological interventions (N = 12). Pharmacological treatments such as selective serotonin reuptake inhibitors (SSRIs), selective serotonin and norepinephrine reuptake inhibitors (SNRIs) and tricyclic antidepressants (TCAs) were compared with one another [45, 46, 49, 52], or with non-pharmacological treatments, either alone or in combination with pharmacological treatments [35, 42, 47, 48]. In addition, studies examined the role of task shifting in mental health treatment provision from clinicians to lay health workers [31, 32, 39, 43]. Healthcare facilities with clinicians were the leading service delivery locations (N = 19); however, three studies evaluated treatment provision in community settings, such as rural villages [39, 44] and participant residences [32] with trained lay health workers, targeting an adult population aged 18 years and above (Table 1).
3.3 Details of the Study Methods
CUA was the most common type of economic evaluation framework (N = 11) in which health outcomes were measured as QALY gains (N = 6) [31, 35, 36, 41, 44, 49] or DALYs averted (N = 5) [42, 43, 47, 48, 51]. This was followed by CEA studies (N = 10) which examined a range of outcomes such as improved symptom scores [32,33,34, 38, 39, 52] or remission [40, 45, 46]. Only a single study, comparing conventional depression treatment (e.g., antidepressants) to a herbal remedy (hypericum perforatum), used CBA [37]. Model-based economic evaluations (N = 10) incorporated data from various sources (e.g., RCT, survey, systematic review). Among trial-based economic evaluations (N = 11), the data source for nine studies were RCTs [31, 32, 34,35,36, 38, 39, 43, 52] as well as comparative follow-up (observational) [33] and survey-based [37] studies.
Out of 22 studies included in this review, 11 involved model-based economic evaluations utilising a decision tree (N = 4) [41, 45, 46], Markov cohort model (N = 5) [44, 47, 48, 50, 51] and microsimulation (N=2) [42, 49]. Most studies included in this review adopted a 1-year (N = 10) or 6-month (N = 4) analytic time horizon. There were a few studies that included a time horizon longer than 1 year (N = 3) [42, 47, 51] or a lifetime time horizon (N = 2) [44, 49]. Some studies used shorter time horizon such as 6 weeks [41], 8 weeks [33, 52] or 3 months [35]. Half of the economic analyses (N = 11) only considered medical costs (i.e., medications, healthcare staff, laboratory investigations and hospitalisations) and employed a healthcare sector/provider perspective. For pharmacological treatments, with the exception of two studies that only considered medication costs [33, 52], most studies also accounted for the costs of healthcare staff, laboratory tests, hospitalisations and even productivity impacts, as seen in one study [40]. On the other hand, many studies of non-pharmacological treatments considered beyond medical costs and incorporated the costs of patient/caregiver time and productivity impacts [31, 32, 34,35,36,37,38,39] and/or used a partial societal perspective (Table 2).
3.4 Cost-Effectiveness Results
Cost-effectiveness results are reported in Table 3 by treatment type (i.e., pharmacological, non-pharmacological or a combination of both) and study design (i.e., model-based studies versus trial-based economic evaluations) to facilitate meaningful comparisons.
3.4.1 Pharmacological Treatments
Model-based economic evaluations In CEA studies that compared pharmacological interventions, the cost effectiveness of antidepressants (i.e., SSRIs, SNRIs and TCAs) varied depending on the setting. For instance, amitriptyline (TCA) [46] in the Colombian setting and escitalopram (SSRI) [40] in the Thailand setting were the dominant treatment options compared with fluoxetine (SSRI) and venlafaxine (SNRI). Both studies used a decision tree with a time horizon of 6 months, with the Thai study adopting a societal perspective and the Colombian study adopting a payer perspective. In addition, there were differences in how they measured costs, with the Thai study additionally including productivity costs (e.g., work absenteeism). A CEA study in the Brazilian setting indicated that SNRIs were the dominant treatment option over TCAs and SSRIs for MDD treatment [45]. Several studies examined the cost effectiveness of atypical antipsychotics for the management of treatment-resistant MDD. A CUA comparing aripiprazole (an atypical antipsychotic) versus placebo as an adjunctive therapy for Thai MDD patients with inadequate response to antidepressants concluded that aripiprazole was not cost effective, with a relatively high ICER value (US$333/QALY) despite positive QALY gains [41]. However, a Turkish study comparing aripiprazole with two other atypical antipsychotics (olanzapine and quetiapine) [49] found that aripiprazole was a dominant (i.e., cost saving) adjunctive treatment in treatment-resistant depression.
Trial-based economic evaluations A CEA conducted alongside a clinical trial in a Chinese setting including a sample of 437 patients with recurrent MDD concluded that vortioxetine (serotonin modulator and stimulator) was a cost-effective alternative to venlafaxine [52] based on the threshold of one times GDP per capita. By contrast, a CEA study from an Indian setting revealed that venlafaxine was dominant over fluoxetine when managing post-stroke depression [33]; however, this study was based on evidence from a prospective comparative study with a small sample size (n = 30).
3.4.2 Non-Pharmacological Treatments
Model-based economic evaluations In a CUA based on microsimulation analysis, cognitive behavioural therapy (CBT) was the dominant option over fluoxetine for episodic and maintenance treatment of depressive disorder in the hospital setting in Thailand [42].
Trial-based economic evaluations There was mixed evidence about the cost effectiveness of non-pharmacological treatment versus pharmacological treatment for depression. According to one CBA in Pakistan [37], conventional antidepressants (fluoxetine) demonstrated better value in terms of benefit-to-cost ratio compared with a herbal remedy (hypericum perforatum); however, the authors concluded that herbal treatments are cost effective compared with conventional treatment for depression by providing justification related to methodological limitations [37]. According to a CEA analysis based on RCT data from 450 people in India, antidepressants (fluoxetine) were found to be more cost effective than psychological treatment and, in turn, psychological treatment was no better than placebo [34] from both the patient and health care perspective.
3.4.3 Combination Treatments
Model-based economic evaluations A regional study comparing various forms of psychosocial intervention with antidepressants found that combining intensive psychosocial intervention with an antidepressant is associated with lower ICER values than a psychosocial intervention alone in Asian (US$334/healthy life-year [HLY]) and African (US$93/HLY) contexts [50]. Findings from studies that employed a CUA framework have shown that combining treatments is not always cost effective and that there are situations where monotherapy might be more cost effective than a combination of treatments. For example, a regional study in an African and Asian context showed that SSRIs were more cost effective than a combination of psychosocial treatments and SSRIs, with a lower ICER value (Africa: ICER values of US$1149/DALY and US$2282/DALY for SSRIs and combination treatments, respectively; Asia: ICER values of US$1012/DALY and US$2032/DALY for SSRIs and combination treatments, respectively) [51]. A study in Ethiopia found that SSRIs had a lower ICER value (US$457/DALY) compared with a combination of TCAs with psychotherapy, followed by a combination of SSRIs with psychotherapy (US$1026/DALY) [47]. Compared with drug treatment alone, a combination of TCAs, psychotherapy and proactive case management was a cost-effective treatment option in the primary health care setting of Nigeria [48]. One economic analysis from Uganda [44] compared Group Interpersonal Therapy (Group IPT) versus Group IPT with a booster involving additional sessions of Group IPT delivered monthly for 2 years. It was found that Group IPT with a booster was dominant over Group IPT alone and had a lower ICER (US$1428/QALY) compared with placebo. According to the authors, implementing Group IPT with a booster depends on the decision makers’ WTP.
Trial-based economic evaluations Studies on treatment approaches, such as task shifting to lay health workers, were identified. Compared with usual care, a stepped-care approach with collaborative care involving trained lay health workers who provide psychosocial intervention for mild depression was cost effective, resulting in increased QALY gains, reduced psychiatric symptom scores and fewer lost work days [31]. The task-shifting approach was also associated with lower cost, thus making the intervention dominant compared with usual care. A CUA study on HIV patients with MDD [43] found that group support psychotherapy was a cost effective (under the threshold of WHO's recommendation) approach compared with group HIV education to managing depression among HIV patients in the Ugandan setting using trained lay health workers. In CUA studies [35, 36] from India, incorporating a healthy activity program into usual care was found to be cost effective compared with usual care alone, under a WTP threshold of US$60 per unit of improvement on Patient Health Questionnaire-9 (PHQ-9) score from both a health system and a societal perspective. Similarly, a CEA that compared a healthy thinking program with usual care in Pakistan [39] and India [32] appeared to be a cost-effective approach to the treatment of depression during pregnancy. Another study from Pakistan [38] found that adding a problem management plus (PM+) scheme (which includes stress management, behavioural activation and social support by trained lay health workers under supervision) to usual care was a cost-effective approach. This RCT-based analysis has revealed that the PM+ programme has a higher ICER value for international supervisors (US$2019 per case treated) than local supervisors (US$402 per case treated). However, in both cases, under a WTP threshold of US$67 per one-point improvement in depression and anxiety score or US$57 per one-point increase in functioning score, the PM+ with enhanced usual care was a cost-effective treatment programme for managing depression.
3.5 Quality Assessment
The quality score of included papers ranged from 45.5 to 98.0 using the QHES checklist, with a mean score of 83.6 (SD = 15.6) and, except for two studies, all the included studies scored > 50. According to the relative weight of the criteria, studies in the review scored relatively lower quality regarding the perspectives (most studies mentioned the perspective of the studies but did not state the reason for the selected perspectives), uncertainty handling, cost and outcome measurement criteria, model or study method transparency, direction and magnitude of bias. Studies scored higher quality in terms of their stated objectives, health outcome measurement scales, variable estimations, data extraction and incremental analysis. The quality score of the studies was reported based on the type of interventions and the study design. Model-based studies that examined the cost effectiveness of pharmacological treatment scored better, with a mean (SD) of 93.7 (2.7), compared with studies evaluating the cost effectiveness of non-pharmacological or combination treatments, which had a mean (SD) of 81.1 (11.4). The mean (SD) scores were reported from trial-based economic analysis for all intervention types, including pharmacological [66 (27.6)] and non-pharmacological [83.5 (17.6)] (Tables 4 and 5).
4 Discussion
This study synthesised evidence from studies conducted across a wide range of LMICs throughout Asia, Latin America, Sub-Saharan Africa and the Middle East. Most (17 out of 21) of the economic studies on depression treatment in LMICs focused exclusively on the adult population; with the remaining (4 out of 21) studies analysing populations with all ages [37, 47, 48, 51]. More evidence on cost effectiveness of interventions targeting children and adolescents is needed given that depression represents a significant health burden in this age range [53]. Healthcare settings (e.g., primary care and hospital) were the dominant service delivery setting, with most services being delivered by clinicians (e.g., psychiatrists and physicians). A minority of studies examined services delivered in community settings (e.g., rural villages or participant homes) by trained lay health workers [32, 39, 44]. This review shows that interventions involving lay health workers were cost effective in LMICs, which could be due to better health outcomes with lower costs (dominant) [31, 32, 36] or higher costs but with low ICERs (cost effective) [35, 39, 43]. The more patient-centred nature of the services may account for the superior health outcomes of services provided by lay health workers [36].
Despite the inconsistencies in cost-effectiveness evidence of treatment options, engaging lay health workers and using a community setting for service delivery appears to be a cost-effective approach for depression treatment in LMICs. Therefore, task shifting toward lay health workers and involvement of non-healthcare settings in service delivery should be promoted in LMICs to improve mental health care services [54]. Task shifting is a process whereby specific tasks are moved to health workers with less training and fewer qualification to make more efficient use of existing human resources. It involves delegating some delineated tasks to newly created cadres of health workers who receive specific, competency-based training [55]. For instance, the Friendship Bench program in Zimbabwe has demonstrated improvements in mental health for rural women with depression using a brief psychological intervention delivered by village health workers [56]. One challenge to scale-up and implementation of task shifting is the lack of cost-effectiveness evidence to support decision making [57]. However, this review demonstrated the cost effectiveness of task shifting in different scenarios, including the involvement of lay health workers in stepped care programmes [31], thinking healthy programmes for pregnant women with depression [32], problem management plus for community members with a high level of distress [38], and healthy activity programmes for depressed patients [35] in general and depressed patients with specific conditions like HIV/AIDS [43].
Studies that examined the cost effectiveness of atypical antipsychotics (e.g., aripiprazole, olanzapine and quetiapine) for managing treatment-resistant MDD were included. A study comparing aripiprazole with other atypical antipsychotics [49] found that aripiprazole was the dominant adjunctive treatment in treatment-resistant depression in the Turkish setting. By contrast, a CUA that compared aripiprazole with placebo for Thai MDD patients concluded that aripiprazole was not cost effective due to its high ICER value (US$267/remission; US$333/QALY) [41]. However, the estimated WTP threshold in Thailand ranges between Baht59,000 (US$5600) and Baht285,000 (US$27,052), or 0.4–2 times the GDP per capita [58]. Therefore, the ICER value associated with aripiprazole is less than the estimated WTP or below the WHO's cost-effectiveness recommendation [25] of 1–3 GDP or alternative suggestions such as GDP-based criteria of 0.5–1.5 GDP and opportunity cost thresholds of 0.5 GDP [26,27,28]. As a result, aripiprazole may be a cost-effective option for treatment-resistant depression in Thailand.
Cost-effectiveness studies of drug treatment for depression revealed mixed evidence [41, 45, 46, 49, 52]. This variation could be explained by differences in study design, types of models, time horizon or data source. Furthermore, the difference in data inputs could explain the inconsistency of findings across studies, in which some studies included only medical costs [44,45,46,47,48, 52]; while other studies incorporated productivity costs or caregiver/family costs [31, 32, 34,35,36,37, 39, 40, 43]. Evidence synthesis is constrained by variation across methods (e.g., measurement of resource use or valuation of health benefits) alongside differences in study design (e.g., different study perspectives or time horizons). The heterogeneity of evidence also seen in this review is a common challenge in synthesising cost-effectiveness evidence, making it even more difficult for decision makers to identify evidence to support their decisions [59].
Due to the well-known side effects of antidepressants, such as decreased libido, addiction, withdrawal symptoms and others [60], non-pharmacological treatments such as psychotherapy are recognised as a preferred option in the long run for depression treatment with no adverse effects [61]. Unfortunately, this review identified only a handful of cost-effectiveness studies that compared non-pharmacological with pharmacological treatments. The evidence on the cost effectiveness of non-pharmacological treatment is not well established in LMICs. Like pharmacological treatments, the existing evidence on the cost effectiveness of non-pharmacological treatment of depression is inconsistent. For example, studies from three settings reported conflicting results [34, 37, 42]. In India, antidepressants were more cost effective than psychological treatment [34], while in Thailand, CBT was reported as the preferred treatment option over fluoxetine [42]. Despite the inconsistency in findings, in a setting with limited resources, the use and scale-up of non-pharmacological interventions, such as brief psychological interventions, can improve health outcomes and be cost saving [35]. This may necessitate changes to the overall health system, including provider training, but it leads to more effective use of the limited resources available [62].
The cost-effectiveness evidence on the combination of non-pharmacological treatment with pharmacological interventions were also reported. Studies from African and Asian settings found that monotherapy use of SSRIs was more cost effective than combination treatments [51]. In Ethiopia, a country in Sub-Saharan Africa, SSRIs were a cost-effective alternative to a combination of SSRIs with psychotherapy [47]. However, a study from Nigeria revealed combination treatment to be a cost-effective treatment option compared with either pharmacological or non-pharmacological treatment alone [48]. All the included studies, except for two, scored >50 for quality score. Model-based studies that assessed cost effectiveness of pharmacological treatments scored better than studies that assessed the cost effectiveness of non-pharmacological or combination treatments. This could be attributed to the availability of quality data for pharmacological treatments. Because of the diversity of evidence, decision makers must carefully select relevant evidence for their setting. This type of review aids in presenting these diverse data to researchers and decision makers in a comprehensive manner, making it easier to utilise the existing evidence.
The absence of an established threshold for country-specific WTP in low- and middle-income countries and the lack of evidence supporting the recommended threshold values, such as thresholds of 1–3 GDP, pose difficulties [26, 28]. As a result, some studies assumed a higher threshold value (US$2000) [51], while others assumed a lower value (US$8.60) [31]. Therefore, it is important to highlight the need for a country-specific threshold for WTP to improve the process of establishing cost-effectiveness evidence as well as to facilitate the application of cost-effectiveness studies.
Of the studies reviewed, 59% (N = 13) were from middle-income countries and 32% (N = 7) were from low-income countries, with the remaining 9% (N = 2) covering both. All of the studies that examined the cost effectiveness of pharmacological interventions (five model-based and two trial-based) were conducted in middle-income countries. In contrast, studies from low-income countries frequently looked at non-pharmacological treatments that were delivered using trained lay providers and have found that these interventions were cost effective [37,38,39, 43, 44]. This review also highlights a lack of evidence in pharmacological treatments in LMICs, where most of the evidence is limited to amitriptyline and fluoxetine [45,46,47,48, 51]. Other pharmacological treatments, such as escitalopram and mirtazapine, which have been recognised as effective and cost-effective antidepressants in high-income countries, lack adequate evidence in LMICs [63]. Studies included in the review relied on international data to estimate the effectiveness of interventions [46, 48]. This implies that the model-based economic evaluations of pharmacological treatments could be restricted by the lack of local effectiveness data. The delivery of psychotherapy such as CBT is largely limited to healthcare facilities or home visits [32, 39]. The use and cost effectiveness of delivering it online, which is a cost-effective method in high-income nations [64], has not been explored well, possibly due to its rarity in LMICs [65].
This review has several limitations. The variability across studies was the main challenge in synthesising the evidence and drawing consistent conclusions. This includes variations in the outcome measures that generate DALYs, QALYs or depression symptom scores; variations in data sources; choice of parameter inputs for model-based studies and methodological approaches for trial-based studies. Standard procedures, such as using QALYs or DALYs as an outcome measure instead of different natural units, may help reduce such heterogeneity and aid in improving evidence synthesis for healthcare decision making in LMICs [66]. The lack of country-specific threshold values to determine cost effectiveness in various settings constrains evidence synthesis.
5 Conclusion
This review provides a comprehensive overview of the cost-effectiveness evidence of depression treatment in LMICs, including pharmacological and non-pharmacological treatments. It highlights several evidence gaps for intervention choices that require further research. Overall, findings showed inconsistency in the evidence of the cost effectiveness of pharmacological, non-pharmacological or combination treatments. However, the evidence consistently depicted that task shifting to lay health care workers or non-specialist health care providers may be considered a cost-effective approach in treating depression in LMICs settings. The evidence is scarce in the younger population and community-based settings and further studies are required to fill these gaps. Despite its drawbacks, this review provides helpful information to decision makers in LMICs, including indicating treatment options or delivery approaches to be considered for depression treatment in LMICs. It also identifies evidence gaps that researchers should investigate further (e.g., depression treatment for younger populations and service delivery in non-healthcare settings) and an area that decision makers should pay attention to (e.g., task shifting to lay health workers).
References
American Psychiatric Association. What is depression? 2020. https://www.psychiatry.org/patients-families/depression/what-is-depression. Accessed 10 Jan 2022.
World Health Organization. Depression and other common mental disorders: global health estimates. World Health Organization; 2017.
Institute for Health Metrics and Evaluation. Major depressive disorder — Level 4 cause. 2019. https://www.healthdata.org/results/gbd_summaries/2019/major-depressive-disorder-level-4-cause. Accessed 29 Jul 2022.
Santomauro DF, Herrera AMM, Shadid J, Zheng P, Ashbaugh C, Pigott DM, et al. Global prevalence and burden of depressive and anxiety disorders in 204 countries and territories in 2020 due to the COVID-19 pandemic. The Lancet. 2021;398(10312):1700–12.
Wu T, Jia X, Shi H, Niu J, Yin X, Xie J, et al. Prevalence of mental health problems during the COVID-19 pandemic: a systematic review and meta-analysis. J Affect Disord. 2021;281:91–8.
Liu Q, He H, Yang J, Feng X, Zhao F, Lyu J. Changes in the global burden of depression from 1990 to 2017: Findings from the Global Burden of Disease study. J Psychiatr Res. 2020;126:134–40.
Greenberg PE, Fournier A-A, Sisitsky T, Simes M, Berman R, Koenigsberg SH, et al. The economic burden of adults with major depressive disorder in the United States (2010 and 2018). Pharmacoeconomics. 2021;39(6):653–65.
Hu T-w, He Y, Zhang M, Chen N. Economic costs of depression in China. Soc Psychiatry Psychiatric Epidemiol. 2007;42(2):110–6.
The Lancet Global H. Mental health matters. Lancet Glob Health. 2020;8(11):e1352.
World Health Organization. Mental health atlas. 2018. https://apps.who.int/iris/bitstream/handle/10665/272735/9789241514019-eng.pdf?ua=1. Accessed 12 Dec 2021.
Vos T, Lim SS, Abbafati C, Abbas KM, Abbasi M, Abbasifard M, et al. Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. The Lancet. 2020;396(10258):1204–22.
Melinda S, Lawrence R, Jeanne S. Depression Treatment. 2021. https://www.helpguide.org/articles/depression/depression-treatment.htm. Accessed 12 Dec 2021.
Pacher P, Kecskemeti V. Trends in the development of new antidepressants. Is there a light at the end of the tunnel? Curr Med Chem. 2004;11(7):925–43.
Fasipe OJ. Neuropharmacological classification of antidepressant agents based on their mechanisms of action. Arch Med Health Sci. 2018;6(1):81.
Gartlehner G, Gaynes BN, Amick HR, Asher GN, Morgan LC, Coker-Schwimmer E, et al. Comparative benefits and harms of antidepressant, psychological, complementary, and exercise treatments for major depression: an evidence report for a clinical practice guideline from the American College of Physicians. Ann Intern Med. 2016;164(5):331–41.
Mekonen T, Chan GC, Connor JP, Hides L, Leung J. Estimating the global treatment rates for depression: a systematic review and meta-analysis. J Affect Disord. 2021;295:1234–42.
Knapp M. Health economics as a tool for decision-making in mental health care. Mental health economics. Springer; 2017. p. 179–90.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, The PRISMA, et al. statement: an updated guideline for reporting systematic reviews. BMJ. 2020;2021:372.
Center for the Evaluation of Value and Risk in Health (CEVR) Tufts Medical Center. Global health Cost-effectiveness analysis registry (GHCEA). https://cevr.tuftsmedicalcenter.org/databases/cea-registry. Accessed 29 Jul 2021.
The World Bank. World Bank Country and Lending Groups. 2021. https://datahelpdesk.worldbank.org/knowledgebase/articles/906519-world-bank-country-and-lending-groups. Accessed 11 Aug 2021.
Endnote. Version 20 [Computer program]. Philadelphia, PA: Clarivate; 2020. https://www.monash.edu/esolutions/software/endnote-install. Accessed 01 Oct 2022.
Veritas Health Innovation Ltd Covidence systematic review software. Melbourne, Australia2021. www.covidence.org. Accessed 6 Feb 2023.
Sanders GD, Neumann PJ, Basu A, Brock DW, Feeny D, Krahn M, et al. Recommendations for conduct, methodological practices, and reporting of cost-effectiveness analyses: second panel on cost-effectiveness in health and medicine. JAMA. 2016;316(10):1093–103.
CCEMG & EPPI-Centre. CCEMG - EPPI-Centre Cost Converter. 2019. https://eppi.ioe.ac.uk/costconversion/default.aspx. Accessed 04 Dec 2021.
Tan-Torres Edejer T, WHO G, Baltussen R, Adam T, Hutubessy R, Acharya A, et al. Making choices in health: WHO guide to cost-effectiveness analysis. 2003. https://apps.who.int/iris/bitstream/handle/10665/42699/9241546018.pdf;jsessionid=AD4B4C0774DDEEDE1C82F41AB720F68F?sequence=1. Accessed 28 Feb 2023.
Kazibwe J, Gheorghe A, Wilson D, Ruiz F, Chalkidou K, Chi Y-L. The use of cost-effectiveness thresholds for evaluating health interventions in low-and middle-income countries from 2015 to 2020: a review. Value Health. 2021;25:385–9.
Iino H, Hashiguchi M, Hori S. Estimating the range of incremental cost-effectiveness thresholds for healthcare based on willingness to pay and GDP per capita: a systematic review. PLoS ONE. 2022;17(4): e0266934.
Woods B, Revill P, Sculpher M, Claxton K. Country-level cost-effectiveness thresholds: initial estimates and the need for further research. Value Health. 2016;19(8):929–35.
Ofman JJ, Sullivan SD, Neumann PJ, Chiou C-F, Henning JM, Wade SW, et al. Examining the value and quality of health economic analyses: implications of utilizing the QHES. J Manag Care Pharm. 2003;9(1):53–61.
Roberts SLE, Healey A, Sevdalis N. Use of health economic evaluation in the implementation and improvement science fields—a systematic literature review. Implement Sci. 2019;14(1):1–13.
Buttorff C, Hock RS, Weiss HA, Naik S, Araya R, Kirkwood BR, et al. Economic evaluation of a task-shifting intervention for common mental disorders in India. Bull World Health Organ. 2012;90:813–21.
Fuhr DC, Weobong B, Lazarus A, Vanobberghen F, Weiss HA, Singla DR, et al. Delivering the Thinking Healthy Programme for perinatal depression through peers: an individually randomised controlled trial in India. The Lancet Psychiatry. 2019;6(2):115–27.
Parvathy J, Philip T, Abhijith K, Sreeni S, Panicker N, Nambiar V. Fluoxetine vs venlafaxine: economic evaluation in post stroke depression. Int J Pharm Sci Rev Res. 2016;41(1):234–6.
Patel V, Chisholm D, Rabe-Hesketh S, Dias-Saxena F, Andrew G, Mann A. Efficacy and cost-effectiveness of drug and psychological treatments for common mental disorders in general health care in Goa, India: a randomised, controlled trial. The Lancet. 2003;361(9351):33–9.
Patel V, Weobong B, Weiss HA, Anand A, Bhat B, Katti B, et al. The Healthy Activity Program (HAP), a lay counsellor-delivered brief psychological treatment for severe depression, in primary care in India: a randomised controlled trial. The Lancet. 2017;389(10065):176–85.
Weobong B, Weiss HA, McDaid D, Singla DR, Hollon SD, Nadkarni A, et al. Sustained effectiveness and cost-effectiveness of the Healthy Activity Programme, a brief psychological treatment for depression delivered by lay counsellors in primary care: 12-month follow-up of a randomised controlled trial. PLoS Med. 2017;14(9): e1002385.
Ahmad S, Azhar I, Masroor D, Ahmed N. Preference of Herbal Therapies: Cost of Illness and Cost Benefit Analysis for Major Diseases in the City of Karachi, Pakistan. Indian J Pharm Sci. 2019;81(5):885–91.
Hamdani SU, Rahman A, Wang D, Chen T, van Ommeren M, Chisholm D, et al. Cost-effectiveness of WHO problem management plus for adults with mood and anxiety disorders in a post-conflict area of Pakistan: randomised controlled trial. Br J Psychiatry. 2020;217(5):623–9.
Sikander S, Ahmad I, Atif N, Zaidi A, Vanobberghen F, Weiss HA, et al. Delivering the Thinking Healthy Programme for perinatal depression through volunteer peers: a cluster randomised controlled trial in Pakistan. The Lancet Psychiatry. 2019;6(2):128–39.
Kongsakon R, Bunchapattanasakda C. The treatment of major depressive disorders (MDD) in Thailand using escitalopram compared to fluoxetine and venlafaxine: a pharmacoeconomic evaluation. J Med Assoc Thail Chotmaihet Thangphaet. 2008;91(7):1117–28.
Leelahanaj T. The cost-effectiveness of aripiprazole as adjunctive therapy in major depressive disorder: Thai economic model. J Med Assoc Thail Chotmaihet Thangphaet. 2010;93:S43-50.
Prukkanone B, Vos T, Bertram M, Lim S. Cost-effectiveness analysis for antidepressants and cognitive behavioral therapy for major depression in Thailand. Value Health. 2012;15(1):S3–8.
Nakimuli-Mpungu E, Musisi S, Wamala K, Okello J, Ndyanabangi S, Birungi J, et al. Effectiveness and cost-effectiveness of group support psychotherapy delivered by trained lay health workers for depression treatment among people with HIV in Uganda: a cluster-randomised trial. Lancet Glob Health. 2020;8(3):e387–98.
Siskind D, Baingana F, Kim J. Cost-effectiveness of group psychotherapy for depression in Uganda. J Ment Health Policy Econ. 2008;11(3):127–33.
Machado M, Iskedjian M, Ruiz I, Einarson TR. The economic impact of introducing serotonin-noradrenaline reuptake inhibitors into the Brazilian national drug formulary-Cost-effectiveness and budget-impact analyses. Pharmacoeconomics. 2007;25(1):67-76.
Machado M, Lopera MM, Diaz-Rojas J, Jaramillo LE, Einarson TR. Pharmacoeconomics of antidepressants in moderate-to-severe depressive disorder in Colombia. Rev Panam Salud Publica. 2008;24:233–9.
Strand KB, Chisholm D, Fekadu A, Johansson KA. Scaling-up essential neuropsychiatric services in Ethiopia: a cost-effectiveness analysis. Health Policy Plan. 2016;31(4):504–13.
Gureje O, Chisholm D, Kola L, Lasebikan V, Saxena S. Cost-effectiveness of an essential mental health intervention package in Nigeria. World Psychiatry. 2007;6(1):42.
Saylan M, Treur M, Postema R, Dilbaz N, Savas H, Heeg B, et al. Cost-effectiveness analysis of aripiprazole augmentation treatment of patients with major depressive disorder compared to olanzapine and quetiapine augmentation in Turkey: a microsimulation approach. Value Health Reg Issues. 2013;2(2):171–80.
Bertram MY, Chisholm D, Watts R, Waqanivalu T, Prasad V, Varghese C. Cost-effectiveness of population level and individual level interventions to combat non-communicable disease in Eastern Sub-Saharan Africa and South East Asia: a WHO-CHOICE analysis. Int J Health Policy Manag. 2021;10(Special Issue on WHO-CHOICE Update):724–33.
Chisholm D, Saxena S. Cost effectiveness of strategies to combat neuropsychiatric conditions in sub-Saharan Africa and South East Asia: mathematical modelling study. BMJ. 2012;344:e609.
Wang G, Zhao K, Reynaud-Mougin C, Loft H, Ren H, Eriksen H-LF, et al. Successfully treated patients with vortioxetine versus venlafaxine: a simplified cost-effectiveness analysis based on a head-to-head study in Asian patients with major depressive disorder. Curr Med Res Opin. 2020;36(5):875–82.
Grover S, Raju VV, Sharma A, Shah R. Depression in children and adolescents: a review of Indian studies. Indian J Psychol Med. 2019;41(3):216–27.
Hoeft TJ, Fortney JC, Patel V, Unützer J. Task-sharing approaches to improve mental health care in rural and other low-resource settings: a systematic review. J Rural Health. 2018;34(1):48–62.
World Health Organization. Task shifting: rational redistribution of tasks among health workforce teams: global recommendations and guidelines. 2007. https://apps.who.int/iris/bitstream/handle/10665/43821/9789?sequence=1. Accessed 28 Feb 2023.
Fernando S, Brown T, Datta K, Chidhanguro D, Tavengwa NV, Chandna J, et al. The Friendship Bench as a brief psychological intervention with peer support in rural Zimbabwean women: a mixed methods pilot evaluation. Glob Ment Health. 2021;8:e31
Javadi D, Feldhaus I, Mancuso A, Ghaffar A. Applying systems thinking to task shifting for mental health using lay providers: a review of the evidence. Glob Ment Health. 2017;4:e14
Thavorncharoensap M, Teerawattananon Y, Natanant S, Kulpeng W, Yothasamut J, Werayingyong P. Estimating the willingness to pay for a quality-adjusted life year in Thailand: does the context of health gain matter? ClinicoEcon Outcomes Res CEOR. 2013;5:29.
Shields GE, Elvidge J. Challenges in synthesising cost-effectiveness estimates. Syst Rev. 2020;9(1):1–7.
Read J, Williams J. Adverse effects of antidepressants reported by a large international cohort: emotional blunting, suicidality, and withdrawal effects. Curr Drug Saf. 2018;13(3):176–86.
Guess P. The emperor’s new drugs: exploding the antidepressant myth. Ethical Human Psychol Psychiatry. 2011;13(1):83.
Fuhr DC, Acarturk C, Sijbrandij M, Brown FL, Jordans MJ, Woodward A, et al. Planning the scale up of brief psychological interventions using theory of change. BMC Health Serv Res. 2020;20(1):1–9.
Khoo AL, Zhou HJ, Teng M, Lin L, Zhao YJ, Soh LB, et al. Network meta-analysis and cost-effectiveness analysis of new generation antidepressants. CNS Drugs. 2015;29:695–712.
Ahern E, Kinsella S, Semkovska M. Clinical efficacy and economic evaluation of online cognitive behavioral therapy for major depressive disorder: a systematic review and meta-analysis. Expert Rev Pharmacoecon Outcomes Res. 2018;18(1):25–41.
Naeem F, Gobbi M, Ayub M, Kingdon D. Psychologists experience of cognitive behaviour therapy in a developing country: a qualitative study from Pakistan. Int J Ment Heal Syst. 2010;4(1):1–9.
Pitt C, Vassall A, Teerawattananon Y, Griffiths UK, Guinness L, Walker D, et al. Foreword: health economic evaluations in low-and middle-income countries: methodological issues and challenges for priority setting. Health Econ. 2016;25(Suppl Suppl 1):1–5.
Acknowledgements
We appreciate the work that each co-author put into the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions.
Conflict of Interest
Yared Belete Belay, Lidia Engel, Yong Yi Lee, Ngoc Le and Cathrine Mihalopoulos declare that they have no conflicts of interest.
Availability of Data and Materials
The data supporting the findings of this study are available within the article or its Electronic Supplementary Material.
Ethics Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Code availability
Not applicable.
Authors’ Contributions
From the topic conception until the preparation of the final draft, all authors made equal contributions. Regarding their respective roles, YBB, CM, LE and YL contributed to developing the search strategy, analysing the data and writing the manuscript. YBB, NL and LE contributed to the study screening, article selection and quality assessment. YBB and LE did data extraction, and all authors supported with the review, editing and manuscript submission.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Belay, Y.B., Engel, L., Lee, Y.Y. et al. Cost Effectiveness of Pharmacological and Non-pharmacological Treatments for Depression in Low- and Middle-Income Countries: A Systematic Literature Review. PharmacoEconomics 41, 651–673 (2023). https://doi.org/10.1007/s40273-023-01257-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40273-023-01257-8