Abstract
Osteoarthritis is a chronic condition that causes pain and loss of joint function. While currently recommended intra-articular therapies (e.g. corticosteroids and hyaluronic acid) can alleviate some of the symptoms of osteoarthritis, they are currently unable to alter the course of disease progression, and the duration of symptom relief is generally short-term. New therapies are under evaluation for treatment of osteoarthritis; these aim to reverse the pathological structural changes associated with the disease and provide longer-lasting pain relief. This article summarises recent guideline recommendations for intra-articular therapies and highlights some of the new therapies currently in clinical evaluation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
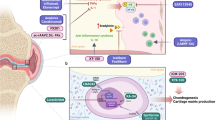
Osteoarthritis is a condition involving inflammation and structural changes in the joint
Osteoarthritis is one of the most common chronic health conditions, and its incidence is on the rise [1]. It is common in older patients, with symptomatic osteoarthritis occurring in 10% of men and 18% of women aged ≥ 60 years [1]. Osteoarthritis is a chronic condition that progresses over many years [2]. It commonly occurs in one or more joints of the knee, hip, hand or foot [1]. The symptoms of osteoarthritis include pain, swelling and a loss of joint function [3]. Osteoarthritis can be caused by pathological changes in the bone, cartilage or synovium [2]. Typically, loss of bone is common in early osteoarthritis and reduced bone reabsorption is seen in late-stage disease. Loss of cartilage is also associated with osteoarthritis and can lead to cartilage softening, fibrillation, and fissuring of superficial layers. Inflammation of the joint synovium, with associated increase infiltration of immune cells and release of inflammatory cytokines, is also common in patients with this disease [2].
Treatments currently used in the clinic focus on symptom management; none of the currently approved treatments for osteoarthritis are effective in delaying or reversing structural damage to the joint [2]. Most guidelines strongly recommend exercise and educational approaches for patients with osteoarthritis [4,5,6]. In addition to these treatments, pharmacological therapies are often required. Nonsteroidal anti-inflammatory drugs (NSAIDs) are strongly recommended, with topical preferred over oral administration due to the risk of adverse events (AEs). If adequate pain relief is not achieved with NSAIDs, or if patients have pre-existing comorbidities which preclude their use, intra-articular therapies involving corticosteroids or hyaluronic acid are often recommended [4, 6].
Ideally, therapy for osteoarthritis involves not only pain relief but also prevention of further joint damage [3]. A number of intra-articular therapies currently in clinical evaluation aim to repair damage to the joint by correcting pathological changes in bone and cartilage remodelling. These approaches will be of more benefit to some patients than others, and tools that can stratify patients with osteoarthritis into subgroups to identify likely responders are sorely needed [7]. Additionally, new intra-articular therapies designed for longer-term pain relief are also being evaluated [3].
This article briefly summarises the intra-articular therapies currently used in the clinic and the recommendations for their use according to recent guidelines, including from the American College of Rheumatology (ACR) [4]; the European Society for Clinical and Economic Aspects of Osteoporosis, Osteoarthritis and Musculoskeletal Diseases (ESCEO) [5]; and the Osteoarthritis Research Society International (OARSI) [6], and highlights the new intra-articular therapies currently being evaluated in clinical trials for treatment of osteoarthritis as reviewed by Assi et al. [3].
Consider intra-articular therapies for treatment of osteoarthritis ...
Intra-articular corticosteroids and hyaluronic acid have a long history of use in the clinic for treatment of osteoarthritis [8, 9]. The advantage of intra-articular therapies is that they directly deliver drugs to the joint [3]; this can be quite effective given that osteoarthritis is typically a localised disease. Furthermore, this has the effect of limiting off-target systemic effects of treatment [3]. There is a small but significant risk of infection with intra-articular injection, particularly with corticosteroids [10], as well as technical difficulties in accessing some joints (e.g. hip and small joints of the hand) [3] and requires a skilled practitioner to administer. For injections into the hip, the ACR recommends use of ultrasound to guide the injection [4]. In the clinic, approximately 38% of patient with knee osteoarthritis receive intra-articular corticosteroids and approximately 13% receive intra-articular hyaluronic acid [11].
There is also recent interest in platelet-rich plasma as an intra-articular therapy for osteoarthritis and there is some evidence to support its efficacy [12]; however, the ACR [4] and the OARSI [6] guidelines strongly recommend against its use in knee and hip osteoarthritis due to inconsistencies in its formulation and because the evidence to support its efficacy come from low quality trials. There is no recommendation on the use of intra-articular platelet-rich plasma for hand osteoarthritis due to lack of data [4].
…including corticosteroids…
Guideline recommendations for intra-articular corticosteroids in knee, hip and hand osteoarthritis are mixed, but generally favours its use in these indications (Table 1). Most studies of intra-articular corticosteroids have been in the knee [3]. The evidence supports the use of intra-articular corticosteroids for short-term pain relief in knee and hip osteoarthritis but not in hand osteoarthritis (Table 1). The effect size and duration of effect of intra-articular corticosteroids in knee osteoarthritis varied between trials [10, 13, 14]; differences in the formulation (water soluble, insoluble, slow-release) and dose of corticosteroids may account for some of these variations [10, 14]. The class of corticosteroid used is not thought to have a large impact on efficacy [3]. There have been reports of rapid destructive osteoarthritis following intra-articular corticosteroid injection but there is insufficient evidence that this is linked to use of intra-articular corticosteroids and may instead be due to underlying disease [10, 13].
…and hyaluronic acid
Hyaluronic acid is the main component of the synovial fluid and cartilage matrix; it provides lubrication to joints and protects cartilage from mechanical degradation [15]. Guideline recommendations for use of hyaluronic acid in knee osteoarthritis is mixed, while limited guidance is available for other joints (Table 1). The evidence generally supports the use of intra-articular hyaluronic acid for reducing pain in patients with osteoarthritis (Table 1) [15,16,17]. There is heterogeneity between study outcomes [17] and some controversy as to whether the effect size is clinically relevant [4, 18]. Variability in study protocols including frequency of administrations and type of hyaluronic acid used (e.g. differences in molecular weight, animal or recombinant in origin, whether or not it is conjugated to other active drugs) may also contribute to the heterogeneity in the efficacy of intra-articular hyaluronic acid [16].
A number of new intra-articular therapies are being investigated in osteoarthritis
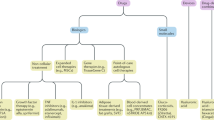
New intra-articular therapies for treatment of osteoarthritis are currently undergoing clinical evaluation (Table 2). A better understanding of the pathology behind osteoarthritis and advances in new methods of delivery of therapeutic substances are driving the development of new classes of treatments for the disease [2, 3]. Identification of key proinflammatory cytokines that are involved in pain and destruction of joint tissue has aided in the development of many immunomodulatory therapies; additionally, the signalling involved in bone and cartilage remodelling have also been targeted [3]. Slow-release formulations are being evaluated to develop therapeutic options with longer durations of effect. Similarly, gene therapies allow for release of therapeutic signalling molecules for longer durations of time [3]. The evidence for efficacy of these new intra-articular therapies currently under investigation is summarised in Table 2.
The primary endpoint in most studies was statistically significant benefit but whether improvements were clinically significant was not assessed (Table 2). Identifying intra-articular therapies with significant clinical responses can be challenging given the large effect size and interpatient variability of placebo responses with this mode of drug administration [21]. Notably, studies that involve multiple injections and with a longer duration (≈ 12 weeks or more) are more prone to placebo responses [21].
Short-term pain relief and improved function may be achieved with anti-inflammatory and immunomodulatory therapies
Intra-articular therapies targeting inflammation, include new slow-release formulations of corticosteroids, NSAID-hyaluronate conjugates, as well as drugs that target specific immunological pathways which can reduce inflammation but may also modify structural aspects of disease (Table 2). In clinical trials, slow-release corticosteroids (EP-104IAR, TLC599) reduced pain in the short-term, as did the intra-articular NSAID-hyaluronate conjugate (Table 2).
A number of new therapies modulate specific immune pathways [interleukin (IL)-10, transforming growth factor (TGF)-β1, IL-1, tumour necrosis factor (TNF)] and can reduce inflammation but may also improve joint structure [2, 3]. Gene therapy to promote expression of IL-10 was efficacious in reducing pain and improving function (Table 2). Cell and gene therapy with TissueGene-C, involving intra-articular injection of TGF-β1-expressing cells, also successfully reduced pain and improved function, and these improvements were observed up to 12 months after administration (Table 2). Although TissueGene-C was briefly approved in South Korea, this approval has since been withdrawn due a discrepancy between the cell type that was provided and what was written in the product manufacturing data [3]. The evidence supporting use of anti-TNF and anti-IL-1 therapies are still limited (Table 2).
Structure modifying therapies may have long-term effects on disease
Intra-articular structure-modifying drugs aim to slow decline or reverse joint damage in patients with osteoarthritis and involves drugs that reduces loss or promotes growth of cartilage as well as drugs that reduce pathological bone changes associated with osteoarthritis [2]. In clinical trials, cartilage thickness was improved with sprifermin and LNA043, and pathological bone remodelling was reduced with TPX-100 (Table 2). Functional improvements and pain relief in a subset of patients receiving lorecivivint were also reported (Table 2). Although structural improvements are observed with many of these treatments it is not currently clear if these changes will translate into symptomatic improvements (Table 2). In the trials where pain is reduced (e.g. with lorecivivint), this was only seen in long-term follow-up (Table 2).
Long-term pain relief may be achieved with therapies targeting pain receptors
Therapies aiming to reduce pain have largely centred around the transient receptor potential vanilloid-1 (TRPV-1) channel, which is expressed on pain receptors in joints and transmit nociceptive signals to the brain [3]. CNTX-4975 and MTX-071 bind to TRPV-1 and prolongs channel opening and results in desensitisation or loss of these neurons [3]. This leads to reduced pain in patients with osteoarthritis in clinical trials (Table 2). Notably, the pain relief with MTX-071 appears to be long-lasting in some patients (Table 2).
Take home messages
-
Osteoarthritis can involve pathological changes to the bone, cartilage and/or synovium of joints.
-
Recommend exercise and educational therapies to all patients with osteoarthritis.
-
Consider intra-articular corticosteroids and hyaluronic acid in patients where NSAIDs are not appropriate or have failed to achieve adequate benefit.
-
Be aware of the new intra-articular therapies for treatment of osteoarthritis currently under investigation, including immunomodulatory therapies, structure modifying therapies and therapies that directly target pain receptors.
References
Allen KD, Thoma LM, Golightly YM. Epidemiology of osteoarthritis. Osteoarthr Cartil. 2022;30(2):184–95.
Oo WM, Hunter DJ. Repurposed and investigational disease-modifying drugs in osteoarthritis (DMOADs). Ther Adv Musculoskelet Dis. 2022;14:1–29.
Assi R, Quintiens J, Monteagudo S, et al. Innovation in targeted intra-articular therapies for osteoarthritis. Drugs. 2023;83(8):649–63.
Kolasinski SL, Neogi T, Hochberg MC, et al. 2019 American college of rheumatology/arthritis foundation guideline for the management of osteoarthritis of the hand, hip, and knee. Arthritis Rheumatol. 2020;72(2):220–33.
Bruyere O, Honvo G, Veronese N, et al. An updated algorithm recommendation for the management of knee osteoarthritis from the european society for clinical and economic aspects of osteoporosis, osteoarthritis and musculoskeletal diseases (ESCEO). Semin Arthritis Rheum. 2019;49(3):337–50.
Bannuru RR, Osani MC, Vaysbrot EE, et al. OARSI guidelines for the non-surgical management of knee, hip, and polyarticular osteoarthritis. Osteoarthr Cartil. 2019;27(11):1578–89.
Mobasheri A, Kapoor M, Ali SA, et al. The future of deep phenotyping in osteoarthritis: how can high throughput omics technologies advance our understanding of the cellular and molecular taxonomy of the disease? Osteoarthr Cartil Open. 2021;3(4): 100144.
Dieppe PA, Sathapatayavongs B, Jones HE, et al. Intra-articular steroids in osteoarthritis. Rheumatol Rehabil. 1980;19(4):212–7.
Rydell N, Balazs EA. Effect of intra-articular injection of hyaluronic acid on the clinical symptoms of osteoarthritis and on granulation tissue formation. Clin Orthop Relat Res. 1971;80:25–32.
Choueiri M, Chevalier X, Eymard F. Intraarticular corticosteroids for hip osteoarthritis: a review. Cartilage. 2021;13(1):122S-131S.
Bedard NA, DeMik DE, Glass NA, et al. Impact of clinical practice guidelines on use of intra-articular hyaluronic acid and corticosteroid injections for knee osteoarthritis. J Bone Joint Surg Am. 2018;100(10):827–34.
Gato-Calvo L, Magalhaes J, Ruiz-Romero C, et al. Platelet-rich plasma in osteoarthritis treatment: review of current evidence. Ther Adv Chronic Dis. 2019;10:2040622319825567.
Arroll B, Goodyear-Smith F. Corticosteroid injections for osteoarthritis of the knee: meta-analysis. BMJ. 2004;328(7444):869.
Saltychev M, Mattie R, McCormick Z, et al. The magnitude and duration of the effect of intra-articular corticosteroid injections on pain severity in knee osteoarthritis: a systematic review and meta-analysis. Am J Phys Med Rehabil. 2020;99(7):617–25.
Altman R, Hackel J, Niazi F, et al. Efficacy and safety of repeated courses of hyaluronic acid injections for knee osteoarthritis: a systematic review. Semin Arthritis Rheum. 2018;48(2):168–75.
Chavda S, Rabbani SA, Wadhwa T. Role and effectiveness of intra-articular injection of hyaluronic acid in the treatment of knee osteoarthritis: a systematic review. Cureus. 2022;14(4):e24503.
Rutjes AW, Juni P, da Costa BR, et al. Viscosupplementation for osteoarthritis of the knee: a systematic review and meta-analysis. Ann Intern Med. 2012;157(3):180–91.
Altman RD, Ike RW, Hamburger MI, et al. Missing the mark? American college of rheumatology 2019 guidelines for intraarticular hyaluronic acid injection and osteoarthritis knee pain. J Rheumatol. 2022;49(8):958–60.
Estee MM, Cicuttini FM, Page MJ, et al. Efficacy of corticosteroids for hand osteoarthritis - a systematic review and meta-analysis of randomized controlled trials. BMC Musculoskelet Disord. 2022;23(1):665.
Miller LE, Fredericson M, Altman RD. Hyaluronic acid injections or oral nonsteroidal anti-inflammatory drugs for knee osteoarthritis: systematic review and meta-analysis of randomized trials. Orthop J Sports Med. 2020;8(1):2325967119897909.
Yu SP, van Middelkoop M, Deveza LA, et al. Predictors of placebo response to local (intra-articular) therapy in osteoarthritis—an individual participant data meta-analysis. Arthritis Care Res. 2023. https://doi.org/10.1002/acr.25212.
Hunter DJ, Chang CC, Wei JC, et al. TLC599 in patients with osteoarthritis of the knee: a phase IIa, randomized, placebo-controlled, dose-finding study. Arthritis Res Ther. 2022;24(1):52.
Malone A, Price J, Price N, et al. Safety and pharmacokinetics of EP-104IAR (sustained-release fluticasone propionate) in knee osteoarthritis: a randomized, double-blind, placebo-controlled phase 1 trial. Osteoarthr Cartil Open. 2021;3(4): 100213.
Nishida Y, Kano K, Nobuoka Y, et al. Efficacy and safety of diclofenac-hyaluronate conjugate (diclofenac etalhyaluronate) for knee osteoarthritis: a randomized phase III trial in Japan. Arthritis Rheumatol. 2021;73(9):1646–55.
Kim MK, Ha CW, In Y, et al. A multicenter, double-blind, phase III clinical trial to evaluate the efficacy and safety of a cell and gene therapy in knee osteoarthritis patients. Hum Gene Ther Clin Dev. 2018;29(1):48–59.
Thewlis D, Collins S, Huston M, et al. Functional improvements in knee kinematics during walking gait over six months following treatment with XT-150: pooled results from two clinical trials [abstract no. 60]. Osteoarthr Cartil. 2023;31(1):S121–2.
Hochberg MC, Guermazi A, Guehring H, et al. Effect of intra-articular sprifermin vs placebo on femorotibial joint cartilage thickness in patients with osteoarthritis: the FORWARD randomized clinical trial. JAMA. 2019;322(14):1360–70.
Yazici Y, McAlindon TE, Gibofsky A, et al. Lorecivivint, a novel intraarticular CDC-like kinase 2 and dual-specificity tyrosine phosphorylation-regulated kinase 1A inhibitor and wnt pathway modulator for the treatment of knee osteoarthritis: a phase II randomized trial. Arthritis Rheumatol. 2020;72(10):1694–706.
Yazici Y, McAlindon TE, Gibofsky A, et al. A phase 2b randomized trial of lorecivivint, a novel intra-articular CLK2/DYRK1A inhibitor and wnt pathway modulator for knee osteoarthritis. Osteoarthr Cartil. 2021;29(5):654–66.
Saxer F, Laurent D, Scotti C, et al. Cartilage regeneration after treatment with LNA043, a novel disease-modifying osteoarthritis drug candidate: results from an imaging-based proof-of-concept trial in patients with focal articular cartilage lesions in the knee [abstract no. V-272]. Osteoarthr Cartil. 2022;30(1):206–7.
McGuire D, Bowes M, Brett A, et al. Study TPX-100-5: intra-articular TPX-100 significantly delays pathological bone shape change and stabilizes cartilage in moderate to severe bilateral knee OA. Arthritis Res Ther. 2021;23(1):242.
Stevens RM, Ervin J, Nezzer J, et al. Randomized, double-blind, placebo-controlled trial of intraarticular trans-capsaicin for pain associated with osteoarthritis of the knee. Arthritis Rheumatol. 2019;71(9):1524–33.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The preparation of this review was not supported by any external funding.
Authorship and conflict of interest
S. Fung is a salaried employee of Adis International Ltd/Springer Nature and declares no relevant conflicts of interest. All authors contributed to this article and are responsible for its content.
Ethics approval, Consent to participate, Consent for publication, Availability of data and material, Code availability
Not applicable.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fung, S. Towards intra-articular therapies for more than just symptom management in osteoarthritis. Drugs Ther Perspect 39, 382–387 (2023). https://doi.org/10.1007/s40267-023-01029-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40267-023-01029-6