Abstract
The ambulatory setting offers potential advantages for elderly patients undergoing elective surgery due to the advancement in both surgical and anesthetic techniques resulting in quicker recovery times, fewer complications, higher patient satisfaction, and reduced costs of care. This review article aims to provide a practical guide to anesthetic management of elderly outpatients. Important considerations in the preoperative evaluation of elderly outpatients with co-existing diseases, as well as the advantages and disadvantages of different anesthetic techniques on a procedural-specific basis, and recommendations regarding the management of common postoperative complications (e.g., pain, postoperative nausea and vomiting [PONV], delirium and cognitive dysfunction, and gastrointestinal dysfunction) are discussed. The role of anesthesiologists as perioperative physicians is important for optimizing surgical outcomes for elderly patients undergoing ambulatory surgery. The implementation of high-quality, evidence-based perioperative care programs for the elderly on an ambulatory basis has assumed increased importance. Optimal management of perioperative pain using opioid-sparing multimodal analgesic techniques and preventing PONV using prophylactic antiemetics are key elements for achieving enhanced recovery after surgery.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Performing a thorough preoperative assessment, careful hemodynamic and respiratory monitoring during the perioperative period, use of short-acting anesthetic drugs which are adjusted based on patient age and co-existing medical conditions, achieving complete neuromuscular recovery at the end of the procedure, and preventative analgesia are important factors in insuring an optimal recovery for elderly outpatients. |
Strategies to minimize the use of opioids in the perioperative period will minimize adverse side effects (e.g., ventilator depression, constipation, urinary retention) and postoperative opioid dependency. Use of effective non-opioid analgesic techniques is an important factor in expanding the use of ambulatory surgery for the elderly surgical population. |
A combination of prophylactic antiemetic drugs should be administered to elderly outpatients with moderate-to-high risk of developing postoperative nausea and vomiting in order to facilitate the recovery process. |
1 Introduction
Population aging is a global public health challenge [1,2,3]. The proportion of the world population aged ≥60 years will increase from 11.0% in 2010 to 21.8% in 2050 while the portion aged ≥80 years (i.e., ‘oldest-old’ or geriatric) will increase from 1.5 to 4.3% (http://esa.un.org/unpd/wpp/unpp/p2k0data.asp). It has been estimated that elderly people require surgery four times more often than the rest of the population, and that this number will increase by 25% by 2020 [4]. Given the economic and social pressure to reduce healthcare expenditures, it is imperative that strategies are developed to meet these growing demands and to ensure higher quality care for elderly surgical patients [5, 6].
Given the recent advances in anesthesia, surgery, and monitoring technology, the ambulatory setting offers many potential advantages for elderly patients undergoing elective surgery [3, 7]. The most common procedures for elderly outpatients can be performed with minimally invasive surgery techniques and are safely manageable in ambulatory settings. However, age is independently associated with a greater rate of unanticipated hospital admission within 30 days of ambulatory surgery [8]. Efficient ambulatory surgery centers rely on anesthesia regimens that facilitate early recovery, provide high-quality analgesia, and have low levels of postoperative nausea and vomiting (PONV) and low rates of unplanned hospital admission [9]. This review article aims to provide a practical guide to anesthetic management of elderly outpatients.
2 Preoperative Assessment
The preoperative assessment should identify potential risk factors of the proposed operative procedure (e.g., diabetes mellitus, cardiovascular disease, pulmonary disorders, hepatic disease, or renal impairment) [10,11,12]. Comprehensive history and physical examination and appropriate laboratory testing are the initial steps to preoperative evaluation. A study by Chung et al. involving 1000 outpatients reported that there was no difference in the incidence of adverse perioperative events between those receiving no preoperative laboratory testing and those undergoing ‘routine’ lab testing [13]. These data suggest that for the majority of older patients with well-controlled (stable) coexisting diseases, routine (screening) lab tests are not necessary. Preoperative diagnostic tests should be performed selectively, and limited to higher risk patients with known comorbidities, and the type of procedure to be performed [14,15,16].
2.1 Diabetes Mellitus Assessment
In the perioperative period, uncontrolled diabetes mellitus can result in electrolyte imbalances, dehydration, and wound infections [17, 18]. An optimal interventional strategy for uncontrolled diabetes mellitus utilizing intravenous (IV) insulin for type I diabetics, and oral hypoglycemic drugs and/or parenteral insulin for type II diabetics are necessary. There are no present guidelines on perioperative glycemic control. A reasonable approach would be to maintain blood glucose at <200 mg/dL intraoperatively and <150 mg/dL postoperatively, but avoid levels <80 mg/dL [18]. Minor ambulatory surgery is not associated with a clinically relevant increase in glucose. The small increase in blood glucose level after surgery is clinically insignificant and may be attributed to the use of dexamethasone for PONV prophylaxis [19].
2.2 Cardiac Risk Assessment
Cardiovascular diseases are common among elderly outpatients, including hypertension, chronic heart failure, arrhythmias, and ischemic heart disease. Elderly patients are more vulnerable to perioperative cardiac adverse events [20]. Therefore, it is critical to evaluate elderly patients with higher risk of cardiac complications according to the American College of Cardiology and American Heart Association (ACC/AHA) algorithm for noncardiac surgery [21]. Preoperative risk assessment should focus on three elements: the surgical risk for cardiac events after the planned procedure, patient functional capacity, and risk indices [22]. Evaluation of cardiac reserve is more important than the resting value of ejection fraction. Functional capacity should be >4 metabolic equivalents (METs), which corresponds to the ability to climb two flights of stairs. There is a clear consensus to continue most chronic medications up to and including the day of surgery (particularly β-blockers and statins) [14, 23]. However, less compelling evidence exists on continuing calcium-channel blockers, angiotensin-converting enzyme inhibitors, and angiotensin-receptor-blocking drugs [23, 24]. A controversial issue in elderly outpatients is perioperative continuation of antithrombotic drugs and/or platelet inhibitors [25], particularly when regional anesthesia is planned. A comprehensive guideline by the European Society of Anesthesiologists on Regional Anesthesia suggests that elderly patients should continue antiplatelet drug therapy if they are undergoing ambulatory procedures [26, 27].
2.3 Respiratory Risk Assessment
Advanced age is an important predictor of postoperative pulmonary complications, including aspiration, pulmonary edema, atelectasis, and pneumonia [17]. Patient-related risk factors include age >60 years, ASA ≥II, chronic obstructive pulmonary disease (COPD), functional dependence, and congestive heart failure, while procedure-related risk factors include prolonged surgery (>3 h), head and neck surgery, thoracic surgery, neurosurgery, abdominal surgery, vascular surgery, emergency surgery, and general anesthesia [17]. In a review, Smetana and Conde further identified pulmonary hypertension and obstructive sleep apnea (OSA) as emerging pulmonary risk factors [28]. Regarding laboratory testing to reduce risk, serum albumin level <35 g/L is a strong predictor of increased postoperative pulmonary complication risk. The guideline of the American College of Physicians recommends that clinicians should employ strategies to reduce postoperative pulmonary complications in elderly patients who are at high risk after clinical risk stratification. Preoperative spirometry and chest radiography may be useful in patients with past history of COPD or asthma, along with risk reduction strategies (e.g., incentive spirometry and deep breathing exercises) [29]. In COPD patients, preoperative tiopropium may reduce postoperative pulmonary complication rates, and preoperative systemic corticosteroids may also be useful in the elderly with chronic pulmonary diseases [17]. Smoking cessation has been shown to decrease risk of perioperative complications, and should be strongly encouraged at least 4 weeks before surgery [30, 31]. Patients with OSA had an increased risk of perioperative events requiring additional anesthetic management [15]. A decision regarding the suitability of elderly obese patients with OSA for ambulatory surgery depends on the invasiveness of the procedure, the choice of anesthesia, the severity of the airway obstruction, the presence of comorbidities, the need for postoperative opioid analgesics, and the level of home care [3].
2.4 Renal Function Assessment
Preventing perioperative acute renal failure is another important consideration in elderly patients, particularly those with preexisting renal insufficiency, diabetes, and long-standing hypertension. Serum creatinine concentration is an insensitive indicator of renal function in the elderly as it is influenced by nonrenal factors such as age, sex, total muscle mass, race, and diet [32]. Strategies used to optimize an elderly patient’s renal function include optimal hydration, careful blood pressure control, electrolyte imbalance correction, drug dosage adjustment, avoidance of nephrotoxic drugs, monitoring blood glucose, and estimating creatinine clearance [17]. Patients with pre-existing renal failure and at risk for postoperative renal impairment should have their urine output monitored carefully throughout the perioperative phase and adequate hydration should be provided to prevent any deterioration [33].
2.5 Functional Status Assessment
Improving functional status may be as important as optimizing their medical treatment, as many elderly patients are frail (up to 30%). Functional level assessment should be practiced throughout the perioperative period using a validated scale that includes an assessment of weakness, weight loss, exhaustion, low physical activity, and slowed walking speed [34]. Identification of risk factors for these geriatric syndromes may promote restoration of compensatory ability and prevent the onset of functional dependence [35]. Functional exercise capacity can be increased through structured training programs, and has been shown to improve outcomes in elderly surgical patients [36, 37].
2.6 Nutritional Risk Assessment
Poor nutritional status is associated with perioperative complications that increase the risk for pneumonia, extended intubation, prolonged wound healing and infection, sepsis, and 30-day mortality [38]. Protein energy malnutrition is the most common nutritional inadequacy found in the geriatric population. Several nutritional status indicators (e.g., serum albumin concentration, mid-arm muscle circumference, usual weight percentage, presence of decubitus ulcers, dysphagia) have proven to be valuable predictors of mortality in geriatric patients [39].
3 Effects of Aging on Anesthetic Drugs
Age-related pharmacokinetic changes in drug distribution, metabolism, and elimination have a significant impact on drug dosing in elderly patients [3, 40] (Table 1). Aging results in an overall loss of neurons in both cerebral cortex and spinal cord, which enhances the sensitivity of the elderly to centrally active anesthetic drugs (e.g., opioid analgesics, volatile agents, benzodiazepines, and sedative hypnotics [IV anesthetics] [41,42,43]) and the local anesthetics used in neuraxial and peripheral nerve blocks (PNBs). An age-related increase in adipose tissue leads to an expansion of the ‘lipid (deep) reservoir’ (i.e., total volume of distribution) for centrally active anesthetic drugs, contributing to prolonged elimination half-life values and an increased duration of action of these drugs in the elderly. However, the reduction in total body water decreases the central volume of distribution for water-soluble drugs, resulting in higher average and peak plasma drug concentrations and an enhanced peak effect [44]. Older patients with poor nutrition can have a 20% or more decrease in albumin levels. Because many anesthetic drugs are highly bound to albumin (e.g., propofol, diazepam), even modest decreases in albumin levels can increase free-drug concentrations, contributing to increased sensitivity to these drugs in the elderly. Liver mass decreases by 20–40% during the typical human lifespan, with a concomitant decline in hepatic blood flow [45, 46]. The kidneys lose approximately 10% of parenchymal thickness per decade of life [47], accompanied by a 10% decline in renal blood flow per decade, contributing to a 30–50% decrease in creatinine clearance between the ages of 20 and 90 years [44]. Impaired hepatic and renal functions in elderly patients affects the metabolism and excretion of many different anesthetic, analgesic, and muscle relaxant drugs [48].
4 Anesthesia Management
Elderly patients may have a higher risk of complications for general anesthesia compared with local anesthesia [49]. However, in a case-control study, age per se was not an independent risk factor of serious morbidity or mortality under general anesthesia in the ambulatory setting [50]. The use of general anesthesia can be dictated by patient preference, surgeon preference, type of surgery, or by the patient’s preexisting comorbidities. With neuraxial anesthesia, age-related cardiovascular alterations, sympathetic block, and consequent decrease in peripheral vascular resistance may produce hypotension and bradycardia, with potentially dangerous consequences in the case of reduced cardiovascular reserve. The choice of the anesthetic technique appears to play a minor role in recovery from anesthesia or in the occurrence of minor postoperative complications or home discharge, except for the use of total intravenous anesthesia to reduce the risk for PONV [51].
4.1 General Anesthesia
Reduced levels of neurotransmitters, neuronal density, and innervation of skeletal muscles are predictable consequences of aging and cause a reduction in the anesthetic requirement [22]. Reducing the dosage and careful titration of anesthetic drugs is essential for elderly outpatients (Table 2). Both reduced cardiac index (which causes increased induction time) and reduced baroreflex response (which causes reduced compensatory tachycardia) may allow overdosing of IV anesthetics. Discrepancies between end-expiratory and plasma concentration, which are due to reduced alveolar exchanges, increase the risk for overdosing inhaled anesthetics. Elderly patients have up to a 2-fold increase in sensitivity to the ventilatory-depressant effects of opioid analgesics compared with younger patients, and thus require lower doses [52]. Benzodiazepines also exert a more potent and prolonged sedative, amnestic, and respiratory-depressant effect in older patients [53]. The α-2 agonist dexmedetomidine can be used as an alternative to opioid analgesics for maintenance of spontaneous ventilation in the fragile elderly patient [54]. However, residual sedation and hypotension after discontinuation of a dexmedetomidine infusion can be problematic in the ambulatory setting [55]. Psychomimetic ketamine-related reactions appear less common in elderly patients, particularly in combination with benzodiazepine or propofol [56]. Despite a continuing controversy regarding its potential to increase PONV and risk of postoperative myocardial infarction, use of nitrous oxide as an adjuvant to the volatile and IV anesthetics can be beneficial for elderly outpatients because of its rapid elimination and anesthetic- and analgesic-sparing effects [57]. The dose of neuromuscular blocking drugs should be modestly reduced in the elderly because of reduced rates of elimination.
In the ambulatory setting, anesthetics should ideally be fast-acting with quick offset time and minimal postoperative side effects. The old adage to “start low and go slow” applies when administering anesthetic medications to elderly patients in the ambulatory setting. Elderly outpatients should have minimal, if any, sedative premedication in the ambulatory setting to avoid prolonging emergence from anesthesia. Short-acting benzodiazepines (a dose of 0.5–1 mg of midazolam IV) and propofol (20 mg) are preferred for premedication if necessary to reduce anxiety and facilitate patient positioning [58]. An empathic patient-centered intervention can reduce preoperative anxiety and increase surgical recovery, wound healing, and patient satisfaction [59]. Since elderly patients have a less-compliant vasculature system and a higher incidence of chronic hypertension, they are more prone to develop hypotension after induction of anesthesia. General anesthesia induction with titrated IV doses of propofol will minimize acute cardiorespiratory depression. A small dose of a potent opioid (e.g., fentanyl 0.5 μg/kg IV) may be useful prior to the procedures to minimize acute autonomic responses and movements in response to painful stimuli. Less soluble volatile anesthetics such as desflurane or sevoflurane allow for ideal anesthetic depth and can facilitate a quicker postoperative recovery process. Total intravenous anesthesia with propofol alone or in conjunction with short-acting opioids (e.g., remifentanil) is gaining popularity. The ester-based muscle relaxants (e.g., cisatracurium) have a more predictable duration of effect in the ambulatory setting than do the steroidal-based muscle relaxants (rocuronium). However, use of steroidal muscle relaxants allows for the use of the new cyclodextrin reversal drug sugammadex when the standard anticholinesterase reversal drugs fail to adequately reverse the residual neuromuscular blockade [48, 60]. Supraglottic airway devices are associated with minimal hemodynamic changes during general anesthesia [61]. A laryngeal mask airway should be considered for qualifying patients as an alternative to tracheal intubation. If tracheal intubation is necessary, neuromuscular blocking agents should be either short or intermediate-acting. Use of the Bispectral Index (BIS) monitor can facilitates early recovery after general anesthesia [62]. A preliminary study suggested that avoiding prolonged periods of ‘deep’ hypnosis (i.e., low BIS values) may be associated with decreased morbidity and mortality in the elderly population [63].
Target-controlled infusion (TCI) is an anesthetic technique which is designed to improve the perioperative titration of IV drugs for anesthesia and sedation [64]. TCI systems implement pharmacokinetic principles for administration of anesthetic drugs with the expectation that such administration would produce more predictable and stable blood concentrations of the drug in the patient during surgery (or diagnostic) procedures [65]. On the basis of the pharmacokinetic description of an anesthetic drug, TCI can calculate an approximate time course of drug concentration during and after turning off the infusion, thereby providing guidance on IV drug administration during and/or after the procedure. The effect of age in decreasing the anesthetic requirement for propofol should be taken into account when the propofol concentration is targeted using a TCI system [52, 66]. Target concentrations of propofol for anesthesia and/or sedation should be reduced in elderly patients. This is important to avoid perioperative hemodynamic instability. However, the decrease in the overall dose requirement of propofol is relatively modest in most patients [67]. A systematic review revealed that there is no clinically significant difference in terms of quality of anesthesia or adverse events between TCI and a less costly, manually controlled infusion of propofol [68].
4.2 Neuraxial Anesthesia
Spinal anesthetics can result in a complete, dense, and widespread sensory block, allowing for optimal surgical conditions. Epidural anesthesia may provide more precise control of the duration of neuraxial blockade in the ambulatory setting. The term unilateral spinal anesthesia is a one-sided block with minimal sensory and motor block on the non-operative side [69]. The cardiovascular stability following unilateral spinal anesthesia is one of the most important benefits for elderly outpatients with heart disease. Enk et al. concluded the importance of “low-dose, low-volume and low-flow” in the induction of unilateral spinal anesthesia [70]. Maintaining the lateral decubitus position for 10 min produced a unilateral motor block in 87–100% of the patients when a low dose of hyperbaric bupivacaine (3–4 mg) was used [69]. Using a unilateral technique, hyperbaric ropivacaine 7.5 mg and levobupivacaine 5 mg provided adequate spinal anesthesia for knee arthroscopy, with home-readiness after 197 min [71].
Clinicians need to carefully consider both the type and dose of local anesthetics used to prevent unwanted prolonged anesthetic effects that may delay discharge. The prolonged recovery associated with the traditional doses of spinal anesthetics is problematic when used for short-stay surgery procedures (e.g., hernia repair, prostate biopsy, and knee arthroscopy) [72]. When lidocaine is administered, the 50-mg dose should not be exceeded [73]. For single-lower-extremity procedures, hyperbaric bupivacaine 5 mg produces a reliable unilateral block with a reasonable time to home-readiness and a low incidence of side effects [74]. Compared with traditional dosage, utilization of mini-dose intrathecal lidocaine (10–30 mg), bupivacaine (3.5–7 mg), or ropivacaine (5–10 mg) with a potent opioid (e.g., fentanyl 10–25 μg) [22, 69], has been shown to provide optimal surgical conditions while still facilitating faster recovery of motor and sensory function. Unfortunately, even with this technique, the time to discharge home is unpredictable and remains longer than with general anesthetic techniques.
Short-acting local anesthetics (e.g., articaine, chloroprocaine, and prilocaine) are potentially beneficial for day-case surgery in the elderly population due to the shorter duration of the motor block, more predictable discharge times, and less frequent residual transient neurologic symptoms (TNS) [75,76,77]. In a preliminary study, Casati et al. reported that 2-chloroprocaine (50 mg) provided excellent anesthesia with a faster recovery than lidocaine (50 mg) after knee arthroscopy [75]. Short-acting intrathecal drugs should only be used for procedures lasting <1 h (e.g. knee arthroscopy). Use of shorter acting neuraxial local anesthetic agents may provide superior recovery profiles in the ambulatory setting [78]. Studies involving ultrashort-acting local anesthetics or use of small doses of conventional local anesthetics combined with adjuvants like clonidine can achieve recovery times after ambulatory surgery that are similar to those found with general anesthesia [77, 79]. However, spinal and epidural anesthetic techniques can result in perioperative hypotension, postoperative urinary retention, nausea and vomiting, dizziness, and delayed ambulation time, particularly in elderly male outpatients [80]. When IV sedation is used to supplement neuraxial anesthetic techniques, the risks of respiratory depression and hemodynamic instability may be higher than with general anesthesia [81].
4.3 Local Anesthetics
Local anesthetics are commonly administered by different routes and at differing sites to manage acute surgical pain (Table 3). Advances in PNB techniques, such as ultrasound guidance, multistimulation, and ambulatory continuous perineural catheters, have contributed to a renewed interest in their use [82, 83]. PNBs significantly reduce opioid requirements postoperatively, facilitate the recovery process, and enable earlier ambulation and discharge home (i.e., ‘fast-track’ recovery) after orthopedic ambulatory surgical procedures in the elderly [3, 84, 85]. A study by Saporito et al. revealed that the use of continuous regional analgesia with PNBs has an overall positive impact on overall healthcare costs by decreasing the incidence of unplanned ambulatory visits and unscheduled readmissions, without compromising on the quality of analgesia, patient satisfaction, or safety [86]. Another study also found that PNBs reduce total perioperative costs and reduce length of stay after ambulatory foot surgery [5]. Although additional preparation time may be required prior to the start of surgery when ‘major’ PNBs are performed, improvements in methods of neural localization (e.g., ultrasound techniques) have made this method more reliable [87]. PNBs may also be used as adjuncts to general anesthesia. They improve perioperative analgesia, thereby reducing unanticipated admission as a result of pain or side effects of opioid analgesics [88].
Peripheral regional anesthesia (e.g., femoral and sciatic nerve blocks) provides adequate pain control for total knee arthroplasty. However, these techniques require careful titration of the local anesthetic to avoid impairment of motor function and delays in the rehabilitation process [89]. As a result of ongoing concerns regarding exacerbations in motor weakness and impairment of rehabilitation, distal nerve block techniques [e.g., adductor canal block (ACB) and tibial nerve block in the popliteal fossa] have gained increasing popularity [90]. For this reason, use of the ACB may facilitate the recovery process compared with use of a femoral nerve block for total knee arthroplasty in the elderly population.
Periarticular infiltration and local infiltration analgesia (LIA) provide satisfactory pain relief, and allow virtually immediate mobilization and earlier discharge from hospital following knee and hip surgery [91, 92]. If used as the primary anesthetic, patients can bypass the post-anesthesia care unit (PACU) and thereby reduce recovery costs [93, 94]. However, some studies have questioned the analgesic efficacy of local infiltration after more painful surgical procedures [95]. The location, volume, and timing of local anesthetic administration, as well as the accuracy and consistency of the pumps, are key factors in determining efficacy of pain relief following local infiltration [84].
The use of continuous local anesthetic infusion techniques (e.g., perineural blocks, wound infiltration) have become increasingly popular due to their ability to control moderate-to-severe pain after major ambulatory surgery procedures [96,97,98]. A study revealed that perineural analgesia (interscalene or popliteal catheters) improved pain control, reduced opioid-related side effects, and significantly increased patients’ ability to perform activities of daily living at home [98]. In contrast with epidural analgesia, continuous PNB techniques provide pain relief superior to systemic opioid analgesia with a lower incidence of side effects and reduced risk of long-term opioid dependency [99]. These beneficial effects of PNBs can now be extended to the postdischarge period in the patients’ home with the availability of perineural catheters and disposable pumps. However, the clear benefits of these approaches for managing pain after surgery must be balanced against the cost of the equipment and the resources need to safely manage these systems outside the hospital environment.
Liposomal bupivacaine is a novel formulation of bupivacaine. This formulation aims to sustain safe therapeutic levels of bupivacaine for up to 72 h after administration [100]. Local infiltration with liposomal bupivacaine reduced postoperative pain and decreased opioid consumption and opioid-related adverse events after superficial surgery procedures (e.g., hernia repairs and hemorrhoidectomy) [101]. A recent study found that liposomal bupivacaine via subcostal transversus abdominis plane infiltration provided superior analgesia up to 72 h after injection when compared with non-liposomal bupivacaine [102]. Therefore, liposomal bupivacaine is an effective, safe, and long-acting local anesthetic that can be used for both local infiltrations at the surgical site and for prolonged perineural blocks. SABER-Bupivacaine is another extended-release formulation which reduced acute postoperative pain and the need for supplemental opioids after inguinal hernia surgery [74, 103].
Despite the well documented advantages of local anesthetic techniques, the use of perineural catheters in the ambulatory setting is limited by concerns over staffing, equipment needs, and potential for complications. Local anesthetics can be used alone or in association with other adjunctive drugs to decrease latency, and to increase their analgesic effect (e.g., clonidine) [104]. Several investigative groups have documented prolongations of PNBs with the addition of dexamethasone (~8 mg) to mepivacaine (332 vs 228 min), ropivacaine (22.2 vs 11.8 h), and bupivacaine (22.4 vs 14.8 h) [105, 106]. A study by Abdallah et al. revealed that the effectiveness of IV dexamethasone in prolonging the duration of analgesia was similar to perineural dexamethasone when administered as part of a single-injection supraclavicular block with a long-acting local anesthetic [107]. PNBs with local anesthetics minimize the endocrine-metabolic responses to surgical trauma and facilitate the recovery process [108].
5 Monitored Anesthesia Care
For minor procedures in elderly patients, monitored anesthesia care (MAC) is an excellent alternative to general and regional anesthesia. Guidelines for MAC in elderly patients have been published by Ekstein et al. [109]. Standard monitoring practices for MAC include pulse oximetry, blood pressure, ECG, heart rate, sedation, and respiratory rate or capnography. Clinical signs of adequate spontaneous ventilation (e.g., maintenance of a patent airway and respiratory rate >10 breaths per minute, hemoglobin oxygen saturation values >90%) must be continuously monitored because of the risk of ventilatory depression in the elderly. Often, patients undergoing procedures with MAC are able to bypass the PACU, resulting in reduced costs, early discharge home, and excellent patient acceptance.
Anesthetic drugs (e.g., midazolam, etomidate, propofol, dexmedetomidine) are frequently used for sedation, either by intermittent bolus or by continuous IV infusion. Propofol produces a rapid and controllable reduction of consciousness with a predictably rapid recovery and is the ‘drug of choice’ for MAC sedation in elderly outpatients [110]. Small doses of midazolam provide anxiolysis and anterograde amnesia but may delay discharge. Midazolam combined with an opioid analgesic for brief diagnostic procedures can produce prolonged sedation in elderly patients [111]. When used in combination with propofol, dexmedetomidine can increase the risk for cardiovascular depression. Since the leading cause of serious patient injury associated with MAC is over-sedation and subsequent respiratory depression, the careful evaluation of the level of sedation is critically important [112]. Overall, MAC has shorter recovery time than general anesthesia or regional anesthesia and is more cost-beneficial for superficial ambulatory operations.
6 Postoperative Side Effects and Complications
6.1 Postoperative Pain Management
Despite expansion of the armamentarium of analgesic drugs and techniques used for pain management, many elderly outpatients continue to experience unacceptable levels of pain after surgery [3, 113,114,115]. Unrelieved postoperative pain may result in clinical and psychological changes that increase morbidity and mortality and hamper the rehabilitation process, and decrease patient satisfaction with their surgical experience [116]. Extensive use of opioids is associated with a variety of perioperative side effects, including cardio-respiratory depression, drowsiness and sedation, hallucinations, PONV, pruritus, urinary retention, ileus, and constipation, which can contribute to a delayed hospital discharge and resumption of normal activities of daily living [3, 113,114,115]. More importantly, in a retrospective study involving over 390,000 outpatients aged >66 years who had undergone minor ambulatory surgery procedures (e.g., cataract surgery, varicose vein stripping, hernia repair), Alam et al. [117] reported that opioid-naive patients receiving an opioid analgesic medication within 7 days after undergoing a minor surgical procedure were 44% more likely to continue using opioids 1 year after the operation. The adaptation of multimodal (or balanced) analgesic techniques as the standard approach for the prevention of pain in the ambulatory setting is one of the keys to improving the recovery process following increasingly complex day-case surgery procedures in elderly patients while reducing the long-term risk of chronic opioid dependency [116, 118, 119].
The expanding number of elderly surgical outpatients requires a perioperative analgesic regimen that is highly effective, has minimal side effects, is intrinsically safe, and can be safely managed away from the hospital or surgical center [120, 121]. A multimodal analgesic regimen should be adjusted to meet the needs of elderly patients by taking into consideration their pre-existing medical conditions, types of surgery, and previous experiences with analgesic drugs for treating postoperative pain [122]. The non-opioid analgesic therapies will likely assume an increasingly important role in facilitating the recovery process and improving the satisfaction for elderly ambulatory surgery patients. Combining long-acting local anesthesia and more potent non-opioid analgesics [e.g., parenteral nonsteroidal anti-inflammatory drugs (NSAIDs)] has become the standard analgesic approach for the management of elderly patients undergoing ambulatory surgery (Table 4) [123].
The use of multiple chronic medications is common among elderly patients undergoing ambulatory surgery procedures [124]. Drug selection and dosage must be adjusted to make pain management as safe as possible in elderly outpatients. Opioid analgesics should be used primarily as ‘rescue’ analgesic medication for the short-term treatment of moderate-to-severe acute pain which failed to respond to a ‘preventative’ analgesic regimen involving the use of one or more non-opioid analgesic drugs. Acetaminophen is a commonly used non-opioid analgesic with a well established safety and tolerability profile in the elderly, and dosage adjustments were not recommended despite a reduced clearance rate [125]. The efficacy of NSAIDs in preventing postoperative pain and reducing the requirement for opioids is also well documented [126,127,128]. However, the potential occurrence of adverse effects (e.g., gastrointestinal bleeding and thrombotic events) is a consideration in elderly patients. For procedures at low risk for hemorrhage, traditional nonselective NSAIDs are a more cost-effective alternative. Because the side effects of these agents are dose and time dependent, NSAIDs and acetaminophen should only be used in the early postdischarge period (2–5 days). Cyclooxygenase-2 (COX-2) inhibitors are useful adjuncts to opioids, but long-term use may increase the risk of cardiovascular events. A single dose of glucocorticoid steroid can reduce pain following ambulatory surgery without increasing postoperative bleeding risk in elderly outpatients. Among nontraditional drugs, ketamine has been used to reduce early postoperative pain [114]. Gabapentanoids can reduce pain, but pregabalin causes sedation and dizziness. Dexmedetomidine infusion 0.2–0.8 mg/kg/min decreased perioperative propofol, remifentanil and fentanyl use, postoperative antiemetic requirements, and reduced the PACU stay, but did not facilitate late recovery [129]. White and colleagues have also reported beneficial effects on postoperative pain management when an infusion of esmolol is used to control autonomic responses during ambulatory surgery [130].
6.2 Postoperative Nausea and Vomiting
PONV and post-discharge nausea and vomiting (PDNV) are common and distressing complications following surgery and anesthesia, and may result in dehydration, electrolyte imbalance, wound dehiscence, pulmonary aspiration, and delayed hospital discharge [131,132,133]. A wide variety of antiemetic drugs and non-pharmacologic techniques (e.g., acupressure, acupuncture, and transcutaneous electrical stimulation) are available for the treatment and prevention of PONV. 5-HT3 receptor antagonists are recommended as the first-line regimen for PONV prophylaxis. Dexamethasone, a corticosteroid, has been shown to be effective administered at a dose of 4–12 mg IV [134, 135]. NK-1 receptor antagonists with a long elimination half-life were effective for the prophylaxis and treatment of PONV [136]. The NK-1 receptor antagonist aprepitant appears to be more effective in decreasing the incidence of PONV as compared with ondansetron [137]; however, it is very costly. Droperidol remains the most cost-effective antiemetic therapy despite concerning extrapyramidal side effects and the potential for prolonging the electrocardiographic QT interval when large doses (>1.25 mg) of the drug are administered [138]. Metoclopramide is probably the most commonly used antiemetic for treatment of PONV, in particular when the 5-HT3 compounds and/or droperidol prophylaxis has failed. Prophylactic doses and timing for the administration of antiemetics are summarized in Table 5. Apfel et al. reported that droperidol, dexamethasone, and ondansetron possess similar antiemetic efficacy when administered for antiemetic prophylaxis [139].
A combination antiemetic therapy using drugs that act at different neuroreceptor sites has been recommended for the ‘at-risk’ patients [133, 140]. Previous clinical studies have demonstrated that the use of a combination of prophylactic antiemetic drugs can reduce the incidence of PONV and PDNV while improving patient satisfaction with their quality of recovery and facilitating the recovery process compared with the use of a single antiemetic drug modality [131]. A meta-analysis suggested that combining dexamethasone with a 5-HT3 receptor antagonist provided greater antiemetic efficacy, and this combination therapy was recommended as the ‘optimal’ choice for prophylaxis against PONV [141]. The combination of dexamethasone with either granisetron, or haloperidol was also more effective than single-drug therapy [142]. The impact of PDNV requires that the prophylactic treatment of this complication would ideally extend well beyond the time of discharge from the hospital [143, 144]. New research is centered on different antiemetics, administered at various time points, to evaluate the effects on reducing PDNV. A study demonstrated patients who received the combination of ondansetron 4 mg IV and ondansetron oral disintegrating tablet 8 mg immediately before discharge had less severe nausea and fewer vomiting episodes compared with ondansetron 4 mg IV alone (3 vs 23%) [145].
Patient, anesthetic, and surgical factors all contribute to the incidence of emetic symptoms in the postoperative period (Table 6) [146]. In addition to using a combination of antiemetics with different mechanisms of action, the multifactorial etiology of PONV might be better addressed by the adoption of a multimodal approach to reduce the baseline risk for PONV. Several effective strategies are recommended to reduce the baseline risk for PONV, such as (1) local and regional anesthesia (e.g., local infiltration and/or peripheral nerve blocks); (2) propofol induction and maintenance; (3) minimization of perioperative opioids; (4) minimized use of volatile anesthetics; (5) avoidance of nitrous oxide and reversal drugs; and (6) insuring adequate intraoperative hydration [147, 148].
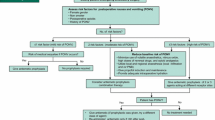
Pharmacological management of PONV and PDNV should be tailored to the patients’ risk level using scoring systems developed by Apfel et al. [139, 140, 149]. These investigators identified four primary predictors for developing PONV: female sex, nonsmoking status, history of PONV or motion sickness, and use of postoperative opioids [150]; and for PDNV: female gender, history of PONV after previous operation, age <50 years, opioid administration and nausea prior to discharge home [139]. PONV prophylaxis is rarely warranted in low-risk patients (<1 factor). However, moderate-risk patients benefit from a single or often multiple antiemetic interventions. ‘Multimodal’ therapy (e.g., triple antiemetic prophylaxis) should be routinely used for all high-risk patients (Table 7) [147]. Optimal management of perioperative pain using opioid-sparing multimodal analgesic techniques and preventing PONV and PDNV using prophylactic antiemetics are both key elements for facilitating both early and late recovery after ambulatory surgery [151].
6.3 Postoperative Delirium and Cognitive Dysfunction
Elderly patients undergoing surgical intervention often have postoperative delirium (POD) and cognitive dysfunction (POCD). POCD is associated with prolonged hospitalization, delayed recovery, and an increased risk of disability and mortality. Age has been increasingly reported as a significant and independent risk factor for POCD [152]. In a large-scale study involving patients aged >60 years, Canet et al. found a lower incidence of POCD 1 week after minor surgery in an ambulatory (vs inpatient) setting. However, no significant difference for the incidence of POCD was found at the later assessment [153]. POD is an acute temporary change in orientation and cognition, whereas POCD is a more subtle and persistent impairment in intellectual/cognitive performance. POD occurs in 5–15% of elderly patients undergoing noncardiac surgery and POCD occurs in 10–13% at 3 months, and can have significant socioeconomic and medical implications [154].
Healthcare professionals caring for surgical patients should perform an assessment of delirium risk factors, including age >65 years, chronic cognitive decline or dementia, poor vision or hearing, severe illness (e.g., ICU admission), and presence of infection [155]. Healthcare providers should evaluate elderly outpatients who develop POD and POCD for possible precipitating conditions (e.g., uncontrolled pain, hypoxia, pneumonia, infections, electrolyte abnormalities, urinary retention, fecal impaction, hypoglycemia). Although elderly patients may have a higher incidence of transient (early) cognitive dysfunction after general anesthesia in comparison with local/regional techniques, there appears to be no causative relationship between general anesthesia and long-term POCD [156]. Several nonpharmacologic and pharmacologic interventions (e.g., reorientation, environmental modifications, antipsychotic agents) are commonly used when delirium occurs and are associated with varying levels of success [157].
7 Enhanced Recovery After Surgery (ERAS)
ERAS programs are multimodal care pathways that aim to decrease intraoperative blood loss, decrease postoperative complications, and reduce recovery times. Studies have suggested that adoption of ERAS decreases postoperative complications by 50%, reduces length of stay by 30%, and decreases readmission rates, thereby lowering overall health costs [158]. Enhanced recovery pathways vary amongst institutions but include key elements such as hemodynamic optimization, early ambulation, and standardized multimodal pain and emesis control regimens. Fast-tracking can be facilitated with the use of short- and ultrashort-acting anesthetic drugs, together with their optimal titration using new monitoring technologies (e.g., BIS monitoring) [159,160,161]. Fasting from solid food for only 6 h and perioperative liquid-carbohydrate loading up to 2 h prior to surgery appeared to be safe and reduced recovery times. Appropriate use of IV fluids to prevent adverse effects associated with decreased physiologic reserve is important when caring for elderly surgical patients [162]. In general, the administration of IV fluids should take into account the combined effects of aging, anesthetics, and analgesics on physiologic responses [163]. Deep vein thrombosis prophylaxis, antibiotic prophylaxis, and thermoregulation were also found to help reduce postsurgical complications. Chewing gum, prokinetic agents, oral laxatives, and an early resumption to normal diet leads to a faster return to normal bowel function.
8 Keys to Future Expansion of Ambulatory Surgery for the Elderly Population
Technological advancements in surgery, improvements in anesthesia monitoring, and pain control will lead to the future expansion of ambulatory surgery for the elderly [164]. New anesthetic drugs (e.g., non-opioid analgesics, sugammadex, depo-bupivacaine) have the potential to reduce morbidity and facilitate a faster recovery for elderly patients [165]. The use of mobile health systems and home telemedicine can lead to improved follow-up care and avoidance of unnecessary emergency room visits [166]. The ASA is encouraging anesthesiologists to include the ‘surgical home’ care program as part of perioperative care [167]. However, these technological advances must be made available at a reasonable cost to the patient and the healthcare system.
The surgical approach (e.g., open vs minimal access) may influence outcomes, complications, and recovery rates. Minimally invasive surgery requires smaller incisions, reduces analgesic requirements, and decreases blood loss [168]. As such, laparoscopy may decrease postoperative complications, pain, and discharge time compared with open surgery [169]. There are clearly economic, societal, and patient benefits in treating elderly surgical patients on an ambulatory basis. However, the economic benefits related to avoiding hospitalization after surgery must be balanced against the additional costs associated with unplanned hospital admissions to treat postoperative complications, as well as the increased need for postoperative medical and social support in extended care facilities and the home environment. Age per se and preexisting medical conditions should not preclude an elderly patient from undergoing ambulatory surgery [170]. However, poor baseline functional status in elderly patients will increase their risk of an adverse outcome after ambulatory surgery. There is general agreement that elderly patients scheduled for surgery in an ambulatory unit should be reasonably fit and any underlying medical conditions should be under stable control.
9 Conclusion
As ambulatory surgery continues to expand in our aging society, implementing evidence-based perioperative care programs for the elderly will assume increased importance. A synergistic and proactive approach is required by the entire healthcare team to facilitate resumption of normal activities after ambulatory surgery in the elderly population. Increasing use of PNBs for anesthesia and perioperative analgesia has the potential to transform anesthesia services for the elderly in the ambulatory setting [171]. Increased use of opioid-sparing multimodal approaches to pain management and adequate treatment of anesthesia and surgery-related side effects is essential to ensure higher levels of patient satisfaction and a faster recovery after ambulatory surgery [172]. Given the recent advances in surgical techniques (e.g., minimally invasive surgery), anesthetic pharmacology, regional anesthesia, and postoperative pain management, the ambulatory setting can offer many potential advantages for the elderly surgical patient.
References
Strom C, Rasmussen LS, Steinmetz J. Practical management of anaesthesia in the elderly. Drugs Aging. 2016;33(11):765–77.
Lutz W, Sanderson W, Scherbov S. The coming acceleration of global population ageing. Nature. 2008;451(7179):716–9.
White PF, White LM, Monk T, Jakobsson J, Raeder J, Mulroy MF, et al. Perioperative care for the older outpatient undergoing ambulatory surgery. Anesth Analg. 2012;114(6):1190–215.
Naughton C, Feneck RO. The impact of age on 6-month survival in patients with cardiovascular risk factors undergoing elective non-cardiac surgery. Int J Clin Pract. 2007;61(5):768–76.
Saporito A, Anselmi L, Borgeat A, Aguirre JA. Can the choice of the local anesthetic have an impact on ambulatory surgery perioperative costs? Chloroprocaine for popliteal block in outpatient foot surgery. J Clin Anesth. 2016;32:119–26.
Aurini L, White PF. Anesthesia for the elderly outpatient. Curr Opin Anaesthesiol. 2014;27(6):563–75.
Jani SR, Shapiro FE, Gabriel RA, Kordylewski H, Dutton RP, Urman RD. A comparison between office and other ambulatory practices: analysis from the National Anesthesia Clinical Outcomes Registry. J Healthc Risk Manag. 2016;35(4):38–47.
De Oliveira GS Jr, Holl JL, Lindquist LA, Hackett NJ, Kim JY, McCarthy RJ. Older adults and unanticipated hospital admission within 30 days of ambulatory surgery: an analysis of 53,667 ambulatory surgical procedures. J Am Geriatr Soc. 2015;63(8):1679–85.
O’Donnell BD, Iohom G. Regional anesthesia techniques for ambulatory orthopedic surgery. Curr Opin Anaesthesiol. 2008;21(6):723–8.
Shnaider I, Chung F. Outcomes in day surgery. Curr Opin Anaesthesiol. 2006;19(6):622–9.
McGory ML, Kao KK, Shekelle PG, Rubenstein LZ, Leonardi MJ, Parikh JA, et al. Developing quality indicators for elderly surgical patients. Ann Surg. 2009;250(2):338–47.
Jakobsson J. Ambulatory anaesthesia: there is room for further improvements of safety and quality of care—is the way forward further simple but evidence-based risk scores? Curr Opin Anaesthesiol. 2010;23(6):679–81.
Chung F, Yuan H, Yin L, Vairavanathan S, Wong DT. Elimination of preoperative testing in ambulatory surgery. Anesth Analg. 2009;108(2):467–75.
Smith I, Jackson I. Beta-blockers, calcium channel blockers, angiotensin converting enzyme inhibitors and angiotensin receptor blockers: should they be stopped or not before ambulatory anaesthesia? Curr Opin Anaesthesiol. 2010;23(6):687–90.
Stierer TL, Wright C, George A, Thompson RE, Wu CL, Collop N. Risk assessment of obstructive sleep apnea in a population of patients undergoing ambulatory surgery. J Clin Sleep Med. 2010;6(5):467–72.
Kertai MD, Palanca BJ, Pal N, Burnside BA, Zhang L, Sadiq F, et al. Bispectral index monitoring, duration of bispectral index below 45, patient risk factors, and intermediate-term mortality after noncardiac surgery in the B-Unaware Trial. Anesthesiology. 2011;114(3):545–56.
Dewan SK, Zheng SB, Xia SJ. Preoperative geriatric assessment: comprehensive, multidisciplinary and proactive. Eur J Intern Med. 2012;23(6):487–94.
Kohl BA, Schwartz S. Surgery in the patient with endocrine dysfunction. Med Clin N Am. 2009;93(5):1031–47.
Polderman JA, Van Velzen L, Wasmoeth LG, Eshuis JH, Houweling PL, Hollmann MW, et al. Hyperglycemia and ambulatory surgery. Minerva Anestesiol. 2015;81(9):951–9.
Chow WB, Rosenthal RA, Merkow RP, Ko CY, Esnaola NF. Optimal preoperative assessment of the geriatric surgical patient: a best practices guideline from the American College of Surgeons National Surgical Quality Improvement Program and the American Geriatrics Society. J Am Coll Surg. 2012;215(4):453–66.
Fleischmann KE, Beckman JA, Buller CE, Calkins H, Fleisher LA, Freeman WK, et al. 2009 ACCF/AHA focused update on perioperative beta blockade. J Am Coll Cardiol. 2009;54(22):2102–28.
Bettelli G. Anaesthesia for the elderly outpatient: preoperative assessment and evaluation, anaesthetic technique and postoperative pain management. Curr Opin Anaesthesiol. 2010;23(6):726–31.
Poldermans D, Bax JJ, Boersma E, De Hert S, Eeckhout E, Fowkes G, et al. Guidelines for pre-operative cardiac risk assessment and perioperative cardiac management in non-cardiac surgery: the Task Force for Preoperative Cardiac Risk Assessment and Perioperative Cardiac Management in Non-cardiac Surgery of the European Society of Cardiology (ESC) and endorsed by the European Society of Anaesthesiology (ESA). Eur J Anaesthesiol. 2010;27(2):92–137.
Auron M, Harte B, Kumar A, Michota F. Renin–angiotensin system antagonists in the perioperative setting: clinical consequences and recommendations for practice. Postgrad Med J. 1029;2011(87):472–81.
Siguret V, Gouin-Thibault I, Gaussem P, Pautas E. Optimizing the use of anticoagulants (heparins and oral anticoagulants) in the elderly. Drugs Aging. 2013;30(9):687–99.
Servin FS. Is it time to re-evaluate the routines about stopping/keeping platelet inhibitors in conjunction to ambulatory surgery? Curr Opin Anaesthesiol. 2010;23(6):691–6.
Hall R, Mazer CD. Antiplatelet drugs: a review of their pharmacology and management in the perioperative period. Anesth Analg. 2011;112(2):292–318.
Smetana GW, Conde MV. Preoperative pulmonary update. Clin Geriatr Med. 2008;24(4):607–24, vii.
Qaseem A, Snow V, Fitterman N, Hornbake ER, Lawrence VA, Smetana GW, et al. Risk assessment for and strategies to reduce perioperative pulmonary complications for patients undergoing noncardiothoracic surgery: a guideline from the American College of Physicians. Ann Intern Med. 2006;144(8):575–80.
Smetana GW. Postoperative pulmonary complications: an update on risk assessment and reduction. Cleve Clin J Med. 2009;76(Suppl 4):S60–5.
Myers K, Hajek P, Hinds C, McRobbie H. Stopping smoking shortly before surgery and postoperative complications: a systematic review and meta-analysis. Arch Intern Med. 2011;171(11):983–9.
National Kidney Foundation. K/DOQI clinical practice guidelines for chronic kidney disease: evaluation, classification, and stratification. Am J Kidney Dis. 2002;39(2 Suppl 1):S1–266.
De Hert S, Imberger G, Carlisle J, Diemunsch P, Fritsch G, Moppett I, et al. Preoperative evaluation of the adult patient undergoing non-cardiac surgery: guidelines from the European Society of Anaesthesiology. Eur J Anaesthesiol. 2011;28(10):684–722.
Makary MA, Segev DL, Pronovost PJ, Syin D, Bandeen-Roche K, Patel P, et al. Frailty as a predictor of surgical outcomes in older patients. J Am Coll Surg. 2010;210(6):901–8.
Abelha FJ, Botelho M, Fernandes V, Barros H. Determinants of postoperative acute kidney injury. Crit Care. 2009;13(3):R79.
Fahlman MM, Topp R, McNevin N, Morgan AL, Boardley DJ. Structured exercise in older adults with limited functional ability. J Gerontol Nurs. 2007;33(6):32–9.
Carli F, Zavorsky GS. Optimizing functional exercise capacity in the elderly surgical population. Curr Opin Clin Nutr Metab Care. 2005;8(1):23–32.
Katlic MR. Consider surgery for elderly patients. Cmaj. 2010;182(13):1403–4.
Dudrick SJ. Nutrition management of geriatric surgical patients. Surg Clin N Am. 2011;91(4):877–96.
Rivera R, Antognini JF. Perioperative drug therapy in elderly patients. Anesthesiology. 2009;110(5):1176–81.
Tang J, Eckenhoff MF, Eckenhoff RG. Anesthesia and the old brain. Anesth Analg. 2010;110(2):421–6.
Greene NH, Attix DK, Weldon BC, Smith PJ, McDonagh DL, Monk TG. Measures of executive function and depression identify patients at risk for postoperative delirium. Anesthesiology. 2009;110(4):788–95.
Smith PJ, Attix DK, Weldon BC, Greene NH, Monk TG. Executive function and depression as independent risk factors for postoperative delirium. Anesthesiology. 2009;110(4):781–7.
Aymanns C, Keller F, Maus S, Hartmann B, Czock D. Review on pharmacokinetics and pharmacodynamics and the aging kidney. Clin J Am Soc Nephrol. 2010;5(2):314–27.
Schmucker DL. Age-related changes in liver structure and function: implications for disease ? Exp Gerontol. 2005;40(8–9):650–9.
Jankovic R, Bogicevic A, Stosic B, Pavlovic A, Petrovic A, Markovic D, et al. Preoperative preparation of geriatric patients. Acta Chir Iugosl. 2011;58(2):169–75.
Gourtsoyiannis N, Prassopoulos P, Cavouras D, Pantelidis N. The thickness of the renal parenchyma decreases with age: a CT study of 360 patients. AJR Am J Roentgenol. 1990;155(3):541–4.
Cope TM, Hunter JM. Selecting neuromuscular-blocking drugs for elderly patients. Drugs Aging. 2003;20(2):125–40.
Fischer B. Benefits, risks, and best practice in regional anesthesia: do we have the evidence we need? Reg Anesth Pain Med. 2010;35(6):545–8.
Arbous MS, Meursing AE, van Kleef JW, de Lange JJ, Spoormans HH, Touw P, et al. Impact of anesthesia management characteristics on severe morbidity and mortality. Anesthesiology. 2005;102(2):257–68 (quiz 491–492).
Pasetto LM, Lise M, Monfardini S. Preoperative assessment of elderly cancer patients. Crit Rev Oncol Hematol. 2007;64(1):10–8.
Minto CF, Schnider TW, Egan TD, Youngs E, Lemmens HJ, Gambus PL, et al. Influence of age and gender on the pharmacokinetics and pharmacodynamics of remifentanil. I. Model development. Anesthesiology. 1997;86(1):10–23.
Jacobs JR, Reves JG, Marty J, White WD, Bai SA, Smith LR. Aging increases pharmacodynamic sensitivity to the hypnotic effects of midazolam. Anesth Analg. 1995;80(1):143–8.
Kunisawa T, Hanada S, Kurosawa A, Suzuki A, Takahata O, Iwasaki H. Dexmedetomidine was safely used for sedation during spinal anesthesia in a very elderly patient. J Anesth. 2010;24(6):938–41.
Makary L, Vornik V, Finn R, Lenkovsky F, McClelland AL, Thurmon J, et al. Prolonged recovery associated with dexmedetomidine when used as a sole sedative agent in office-based oral and maxillofacial surgery procedures. J Oral Maxillofac Surg. 2010;68(2):386–91.
White PF, Vasconez LO, Mathes SA, Way WL, Wender LA. Comparison of midazolam and diazepam for sedation during plastic surgery. Plast Reconstr Surg. 1988;81(5):703–12.
Tang J, Chen L, White PF, Wender RH, Naruse R, Kariger R, et al. Use of propofol for office-based anesthesia: effect of nitrous oxide on recovery profile. J Clin Anesth. 1999;11(3):226–30.
Elvir Lazo OL, White PF, Tang J, Yumul R, Cao X, Yumul F, et al. Propofol versus midazolam for premedication: a placebocontrolled, randomized doubleblinded study. Minerva Anestesiol. 2016;82(11):1170–9.
Pereira L, Figueiredo-Braga M, Carvalho IP. Preoperative anxiety in ambulatory surgery: the impact of an empathic patient-centered approach on psychological and clinical outcomes. Patient Educ Couns. 2016;99(5):733–8.
Lenz A, Hill G, White PF. Emergency use of sugammadex after failure of standard reversal drugs. Anesth Analg. 2007;104(3):585–6.
Zaballos M, Ginel MD, Portas M, Barrio M, Lopez AM. Awake insertion of a Laryngeal Mask Airway-Proseal as alternative to awake fiberoptic intubation in management of anticipated difficult airway in ambulatory surgery. Braz J Anesthesiol. 2016;66(5):539–42.
Song D, Joshi GP, White PF. Titration of volatile anesthetics using bispectral index facilitates recovery after ambulatory anesthesia. Anesthesiology. 1997;87(4):842–8.
Monk TG, Saini V, Weldon BC, Sigl JC. Anesthetic management and one-year mortality after noncardiac surgery. Anesth Analg. 2005;100(1):4–10.
Schnider TW, Minto CF, Struys MM, Absalom AR. The safety of target-controlled infusions. Anesth Analg. 2016;122(1):79–85.
White M, Kenny GN, Schraag S. Use of target controlled infusion to derive age and gender covariates for propofol clearance. Clin Pharmacokinet. 2008;47(2):119–27.
Schnider TW, Minto CF, Shafer SL, Gambus PL, Andresen C, Goodale DB, et al. The influence of age on propofol pharmacodynamics. Anesthesiology. 1999;90(6):1502–16.
Ouattara A, Boccara G, Lemaire S, Kockler U, Landi M, Vaissier E, et al. Target-controlled infusion of propofol and remifentanil in cardiac anaesthesia: influence of age on predicted effect-site concentrations. Br J Anaesth. 2003;90(5):617–22.
Leslie K, Clavisi O, Hargrove J. Target-controlled infusion versus manually-controlled infusion of propofol for general anaesthesia or sedation in adults. Cochrane Database Syst Rev. 2008;3:Cd006059.
Korhonen AM. Use of spinal anaesthesia in day surgery. Curr Opin Anaesthesiol. 2006;19(6):612–6.
Enk D, Prien T, Van Aken H, Mertes N, Meyer J, Brussel T. Success rate of unilateral spinal anesthesia is dependent on injection flow. Reg Anesth Pain Med. 2001;26(5):420–7.
Cappelleri G, Aldegheri G, Danelli G, Marchetti C, Nuzzi M, Iannandrea G, et al. Spinal anesthesia with hyperbaric levobupivacaine and ropivacaine for outpatient knee arthroscopy: a prospective, randomized, double-blind study. Anesth Analg. 2005;101(1):77–82.
Pavlin DJ, Rapp SE, Polissar NL, Malmgren JA, Koerschgen M, Keyes H. Factors affecting discharge time in adult outpatients. Anesth Analg. 1998;87(4):816–26.
Daszkiewicz A, Janik A, Sliwczynska M, Karpe J, Misiolek H. A comparison of selective and conventional spinal anaesthesia for ambulatory surgery. Anaesthesiol Intensive Ther. 2016;48(4):220–7.
Tan M, Law LS, Gan TJ. Optimizing pain management to facilitate enhanced recovery after surgery pathways. Can J Anaesth. 2015;62(2):203–18.
Casati A, Fanelli G, Danelli G, Berti M, Ghisi D, Brivio M, et al. Spinal anesthesia with lidocaine or preservative-free 2-chlorprocaine for outpatient knee arthroscopy: a prospective, randomized, double-blind comparison. Anesth Analg. 2007;104(4):959–64.
Forster JG. Short-acting spinal anesthesia in the ambulatory setting. Curr Opin Anaesthesiol. 2014;27(6):597–604.
Sell A, Tein T, Pitkanen M. Spinal 2-chloroprocaine: effective dose for ambulatory surgery. Acta Anaesthesiol Scand. 2008;52(5):695–9.
Camponovo C. Spinal 1% 2-chloroprocaine versus general anesthesia for ultra-short outpatient procedures: a retrospective analysis. Acta Biomed. 2014;85(3):265–8.
Nishikawa K, Yoshida S, Shimodate Y, Igarashi M, Namiki A. A comparison of spinal anesthesia with small-dose lidocaine and general anesthesia with fentanyl and propofol for ambulatory prostate biopsy procedures in elderly patients. J Clin Anesth. 2007;19(1):25–9.
Auroy Y, Benhamou D, Bargues L, Ecoffey C, Falissard B, Mercier FJ, et al. Major complications of regional anesthesia in France: the SOS Regional Anesthesia Hotline Service. Anesthesiology. 2002;97(5):1274–80.
Vila H Jr, Soto R, Cantor AB, Mackey D. Comparative outcomes analysis of procedures performed in physician offices and ambulatory surgery centers. Arch Surg. 2003;138(9):991–5.
Fredrickson MJ, Leightley P, Wong A, Chaddock M, Abeysekera A, Frampton C. An analysis of 1505 consecutive patients receiving continuous interscalene analgesia at home: a multicentre prospective safety study. Anaesthesia. 2016;71(4):373–9.
Marhofer P, Anderl W, Heuberer P, Fritz M, Kimberger O, Marhofer D, et al. A retrospective analysis of 509 consecutive interscalene catheter insertions for ambulatory surgery. Anaesthesia. 2015;70(1):41–6.
White PF. The changing role of non-opioid analgesic techniques in the management of postoperative pain. Anesth Analg. 2005;101(5 Suppl):S5–22.
Maalouf DB, Dorman SM, Sebeo J, Goytizolo EA, Gordon MA, Yadeau JT, et al. Prospective, randomized double-blind study: does decreasing interscalene nerve block volume for surgical anesthesia in ambulatory shoulder surgery offer same-day patient recovery advantages? Reg Anesth Pain Med. 2016;41(4):438–44.
Saporito A, Calciolari S, Ortiz LG, Anselmi L, Borgeat A, Aguirre J. A cost analysis of orthopedic foot surgery: can outpatient continuous regional analgesia provide the same standard of care for postoperative pain control at home without shifting costs? Eur J Health Econ. 2016;17(8):951–61.
Messeha MM. Real-time ultrasound-guided comparison of adductor canal block and psoas compartment block combined with sciatic nerve block in laparoscopic knee surgeries. Anesth Essays Res. 2016;10(2):305–11.
Rawal N, Hylander J, Nydahl PA, Olofsson I, Gupta A. Survey of postoperative analgesia following ambulatory surgery. Acta Anaesthesiol Scand. 1997;41(8):1017–22.
Charous MT, Madison SJ, Suresh PJ, Sandhu NS, Loland VJ, Mariano ER, et al. Continuous femoral nerve blocks: varying local anesthetic delivery method (bolus versus basal) to minimize quadriceps motor block while maintaining sensory block. Anesthesiology. 2011;115(4):774–81.
Sawhney M, Mehdian H, Kashin B, Ip G, Bent M, Choy J, et al. Pain after unilateral total knee arthroplasty: a prospective randomized controlled trial examining the analgesic effectiveness of a combined adductor canal peripheral nerve block with periarticular infiltration versus adductor canal nerve block alone versus periarticular infiltration alone. Anesth Analg. 2016;122(6):2040–6.
Wall PD, Sprowson AP, Parsons N, Parsons H, Achten J, Balasubramanian S, et al. Protocol for a single-centre randomised controlled trial of multimodal periarticular anaesthetic infiltration versus single-agent femoral nerve blockade as analgesia for total knee arthroplasty: Perioperative Analgesia for Knee Arthroplasty (PAKA). BMJ Open. 2015;5(12):e009898.
Novais EN, Kestel L, Carry PM, Sink E, Strupp K. Local infiltration analgesia compared with epidural and intravenous PCA after surgical hip dislocation for the treatment of femoroacetabular impingement in adolescents. J Pediatr Orthop. 2016. doi:10.1097/BPO.0000000000000725.
Cherprenet AL, Rambourdin-Perraud M, Laforet S, Faure M, Guesmi N, Baud C, et al. Local anaesthetic infiltration at the end of carotid endarterectomy improves post-operative analgesia. Acta Anaesthesiol Scand. 2015;59(1):107–14.
Bhattacharjee DP, Biswas C, Haldar P, Ghosh S, Piplai G, Rudra JS. Efficacy of intraarticular dexamethasone for postoperative analgesia after arthroscopic knee surgery. J Anaesthesiol Clin Pharmacol. 2014;30(3):387–90.
Guo Q, Li R, Wang L, Zhang D, Ma Y. Transversus abdominis plane block versus local anaesthetic wound infiltration for postoperative analgesia: a systematic review and meta-analysis. Int J Clin Exp Med. 2015;8(10):17343–52.
White PF, Issioui T, Skrivanek GD, Early JS, Wakefield C. The use of a continuous popliteal sciatic nerve block after surgery involving the foot and ankle: does it improve the quality of recovery? Anesth Analg. 2003;97(5):1303–9.
Ilfeld BM, Enneking FK. Continuous peripheral nerve blocks at home: a review. Anesth Analg. 2005;100(6):1822–33.
Capdevila X, Dadure C, Bringuier S, Bernard N, Biboulet P, Gaertner E, et al. Effect of patient-controlled perineural analgesia on rehabilitation and pain after ambulatory orthopedic surgery: a multicenter randomized trial. Anesthesiology. 2006;105(3):566–73.
Richman JM, Liu SS, Courpas G, Wong R, Rowlingson AJ, McGready J, et al. Does continuous peripheral nerve block provide superior pain control to opioids? A meta-analysis. Anesth Analg. 2006;102(1):248–57.
Premkumar A, Samady H, Slone H, Hash R, Karas S, Xerogeanes J. Liposomal bupivacaine for pain control after anterior cruciate ligament reconstruction: a prospective, double-blinded, randomized, positive-controlled trial. Am J Sports Med. 2016;44(7):1680–6.
Haas E, Onel E, Miller H, Ragupathi M, White PF. A double-blind, randomized, active-controlled study for post-hemorrhoidectomy pain management with liposome bupivacaine, a novel local analgesic formulation. Am Surg. 2012;78(5):574–81.
Hutchins JL, Kesha R, Blanco F, Dunn T, Hochhalter R. Ultrasound-guided subcostal transversus abdominis plane blocks with liposomal bupivacaine vs. non-liposomal bupivacaine for postoperative pain control after laparoscopic hand-assisted donor nephrectomy: a prospective randomised observer-blinded study. Anaesthesia. 2016;71(8):930–7.
Hadj A, Hadj A, Hadj A, Rosenfeldt F, Nicholson D, Moodie J, et al. Safety and efficacy of extended-release bupivacaine local anaesthetic in open hernia repair: a randomized controlled trial. ANZ J Surg. 2012;82(4):251–7.
Bryskin RB, Londergan B, Wheatley R, Heng R, Lewis M, Barraza M, et al. Transversus abdominis plane block versus caudal epidural for lower abdominal surgery in children: a double-blinded randomized controlled trial. Anesth Analg. 2015;121(2):471–8.
Parrington SJ, O’Donnell D, Chan VW, Brown-Shreves D, Subramanyam R, Qu M, et al. Dexamethasone added to mepivacaine prolongs the duration of analgesia after supraclavicular brachial plexus blockade. Reg Anesth Pain Med. 2010;35(5):422–6.
Cummings KC 3rd, Napierkowski DE, Parra-Sanchez I, Kurz A, Dalton JE, Brems JJ, et al. Effect of dexamethasone on the duration of interscalene nerve blocks with ropivacaine or bupivacaine. Br J Anaesth. 2011;107(3):446–53.
Abdallah FW, Johnson J, Chan V, Murgatroyd H, Ghafari M, Ami N, et al. Intravenous dexamethasone and perineural dexamethasone similarly prolong the duration of analgesia after supraclavicular brachial plexus block: a randomized, triple-arm, double-blind, placebo-controlled trial. Reg Anesth Pain Med. 2015;40(2):125–32.
Buvanendran A, Kroin JS, Berger RA, Hallab NJ, Saha C, Negrescu C, et al. Upregulation of prostaglandin E2 and interleukins in the central nervous system and peripheral tissue during and after surgery in humans. Anesthesiology. 2006;104(3):403–10.
Ekstein M, Gavish D, Ezri T, Weinbroum AA. Monitored anaesthesia care in the elderly: guidelines and recommendations. Drugs Aging. 2008;25(6):477–500.
Arain SR, Ebert TJ. The efficacy, side effects, and recovery characteristics of dexmedetomidine versus propofol when used for intraoperative sedation. Anesth Analg. 2002;95(2):461–6.
Lichtor JL, Alessi R, Lane BS. Sleep tendency as a measure of recovery after drugs used for ambulatory surgery. Anesthesiology. 2002;96(4):878–83.
Balci C, Karabekir HS, Kahraman F, Sivaci RG. Comparison of entropy and bispectral index during propofol and fentanyl sedation in monitored anaesthesia care. J Int Med Res. 2009;37(5):1336–42.
White PF. Pain management for the elderly in the ambulatory setting. Pain Manag. 2015;5(4):233–6.
White PF. Multimodal pain management—the future is now! Curr Opin Investig Drugs. 2007;8(7):517–8.
White PF. Pain management after ambulatory surgery—where is the disconnect? Can J Anaesth. 2008;55(4):201–7.
Falzone E, Hoffmann C, Keita H. Postoperative analgesia in elderly patients. Drugs Aging. 2013;30(2):81–90.
Alam A, Gomes T, Zheng H, Mamdani MM, Juurlink DN, Bell CM. Long-term analgesic use after low-risk surgery: a retrospective cohort study. Arch Intern Med. 2012;172(5):425–30.
Gondusky JS, Choi L, Khalaf N, Patel J, Barnett S, Gorab R. Day of surgery discharge after unicompartmental knee arthroplasty: an effective perioperative pathway. J Arthroplasty. 2014;29(3):516–9.
Cao X, Elvir-Lazo OL, White PF, Yumul R, Tang J. An update on pain management for elderly patients undergoing ambulatory surgery. Curr Opin Anaesthesiol. 2016;29(6):674–82.
White PF. Ambulatory anesthesia advances into the new millennium. Anesth Analg. 2000;90(5):1234–5.
Lamplot JD, Wagner ER, Manning DW. Multimodal pain management in total knee arthroplasty: a prospective randomized controlled trial. J Arthroplasty. 2014;29(2):329–34.
Fanelli G, Berti M, Baciarello M. Updating postoperative pain management: from multimodal to context-sensitive treatment. Minerva Anestesiol. 2008;74(9):489–500.
White PF. What are the advantages of non-opioid analgesic techniques in the management of acute and chronic pain? Expert Opin Pharmacother. 2017;18(4):329–33.
Barnett SR. Polypharmacy and perioperative medications in the elderly. Anesthesiol Clin. 2009;27(3):377–89.
Gupta A, Jakobsson J. Acetaminophen, nonsteroidal anti-inflammatory drugs, and cyclooxygenase-2 selective inhibitors: an update. Plast Reconstr Surg. 2014;134(4 Suppl 2):24s–31s.
Rana MV, Desai R, Tran L, Davis D. Perioperative pain control in the ambulatory setting. Curr Pain Headache Rep. 2016;20(3):18.
White PF, Tang J, Wender RH, Zhao M, Time M, Zaentz A, et al. The effects of oral ibuprofen and celecoxib in preventing pain, improving recovery outcomes and patient satisfaction after ambulatory surgery. Anesth Analg. 2011;112(2):323–9.
Aurini L, Borghi B, White PF, TOGNù A, Rossi B, Fini G, et al. Treatment of chronic cervicobrachial pain with periradicular injection of meloxicam. Minerva Anestesiol. 2016;82(4):411–8.
Keplinger M, Marhofer P, Kettner SC, Marhofer D, Kimberger O, Zeitlinger M. A pharmacodynamic evaluation of dexmedetomidine as an additive drug to ropivacaine for peripheral nerve blockade: a randomised, triple-blind, controlled study in volunteers. Eur J Anaesthesiol. 2015;32(11):790–6.
White PF, Wang B, Tang J, Wender RH, Naruse R, Sloninsky A. The effect of intraoperative use of esmolol and nicardipine on recovery after ambulatory surgery. Anesth Analg. 2003;97(6):1633–8.
Glass PS, White PF. Practice guidelines for the management of postoperative nausea and vomiting: past, present, and future. Anesth Analg. 2007;105(6):1528–9.
Brettner F, Janitza S, Prull K, Weninger E, Mansmann U, Kuchenhoff H, et al. Gender-specific differences in low-dose haloperidol response for prevention of postoperative nausea and vomiting: a register-based cohort study. PLoS One. 2016;11(1):e0146746.
Cao X, White PF, Ma H. An update on the management of postoperative nausea and vomiting. J Anesth. 2017;31(4):617–26.
Wang JJ, Ho ST, Lee SC, Liu YC, Ho CM. The use of dexamethasone for preventing postoperative nausea and vomiting in females undergoing thyroidectomy: a dose-ranging study. Anesth Analg. 2000;91(6):1404–7.
Maitra S, Som A, Baidya DK, Bhattacharjee S. Comparison of ondansetron and dexamethasone for prophylaxis of postoperative nausea and vomiting in patients undergoing laparoscopic surgeries: a meta-analysis of randomized controlled trials. Anesthesiol Res Pract. 2016;2016:7089454.
Liu M, Zhang H, Du BX, Xu FY, Zou Z, Sui B, et al. Neurokinin-1 receptor antagonists in preventing postoperative nausea and vomiting: a systematic review and meta-analysis. Medicine (Baltimore). 2015;94(19):e762.
Milnes V, Gonzalez A, Amos V. Aprepitant: a new modality for the prevention of postoperative nausea and vomiting: an evidence-based review. J Perianesth Nurs. 2015;30(5):406–17.
White PF. Droperidol: a cost-effective antiemetic for over thirty years. Anesth Analg. 2002;95(4):789–90.
Apfel CC, Korttila K, Abdalla M, Kerger H, Turan A, Vedder I, et al. A factorial trial of six interventions for the prevention of postoperative nausea and vomiting. N Engl J Med. 2004;350(24):2441–51.
Tang J, Chen X, White PF, Wender RH, Ma H, Sloninsky A, et al. Antiemetic prophylaxis for office-based surgery: are the 5-HT3 receptor antagonists beneficial? Anesthesiology. 2003;98(2):293–8.
Henzi I, Walder B, Tramer MR. Dexamethasone for the prevention of postoperative nausea and vomiting: a quantitative systematic review. Anesth Analg. 2000;90(1):186–94.
Chu CC, Shieh JP, Tzeng JI, Chen JY, Lee Y, Ho ST, et al. The prophylactic effect of haloperidol plus dexamethasone on postoperative nausea and vomiting in patients undergoing laparoscopically assisted vaginal hysterectomy. Anesth Analg. 2008;106(5):1402–6.
Kolodzie K, Apfel CC. Nausea and vomiting after office-based anesthesia. Curr Opin Anaesthesiol. 2009;22(4):532–8.
Chinnappa V, Chung F. Post-discharge nausea and vomiting: an overlooked aspect of ambulatory anesthesia? Can J Anaesth. 2008;55(9):565–71.
Gan TJ, Franiak R, Reeves J. Ondansetron orally disintegrating tablet versus placebo for the prevention of postdischarge nausea and vomiting after ambulatory surgery. Anesth Analg. 2002;94(5):1199–200.
White PF, Sacan O, Nuangchamnong N, Sun T, Eng MR. The relationship between patient risk factors and early versus late postoperative emetic symptoms. Anesth Analg. 2008;107(2):459–63.
Gan TJ, Diemunsch P, Habib AS, Kovac A, Kranke P, Meyer TA, et al. Consensus guidelines for the management of postoperative nausea and vomiting. Anesth Analg. 2014;118(1):85–113.
Sansonnens J, Taffe P, Burnand B. Higher occurrence of nausea and vomiting after total hip arthroplasty using general versus spinal anesthesia: an observational study. BMC Anesthesiol. 2016;16(1):44.
Apfel CC, Philip BK, Cakmakkaya OS, Shilling A, Shi YY, Leslie JB, et al. Who is at risk for postdischarge nausea and vomiting after ambulatory surgery? Anesthesiology. 2012;117(3):475–86.
Apfel CC, Laara E, Koivuranta M, Greim CA, Roewer N. A simplified risk score for predicting postoperative nausea and vomiting: conclusions from cross-validations between two centers. Anesthesiology. 1999;91(3):693–700.
White PF. Multimodal therapy facilitates a fast-track recovery after surgery—are potential drug-drug interactions of clinical significance in the perioperative period? J Clin Anesth. 2017;39:98–9.
Moller JT, Cluitmans P, Rasmussen LS, Houx P, Rasmussen H, Canet J, et al. Long-term postoperative cognitive dysfunction in the elderly ISPOCD1 study. ISPOCD investigators. International Study of Post-Operative Cognitive Dysfunction. Lancet. 1998;351(9106):857–61.
Canet J, Raeder J, Rasmussen LS, Enlund M, Kuipers HM, Hanning CD, et al. Cognitive dysfunction after minor surgery in the elderly. Acta Anaesthesiol Scand. 2003;47(10):1204–10.
Rasmussen LS, Johnson T, Kuipers HM, Kristensen D, Siersma VD, Vila P, et al. Does anaesthesia cause postoperative cognitive dysfunction? A randomised study of regional versus general anaesthesia in 438 elderly patients. Acta Anaesthesiol Scand. 2003;47(3):260–6.
Mohanty S, Rosenthal RA, Russell MM, Neuman MD, Ko CY, Esnaola NF. Optimal perioperative management of the geriatric patient: a best practices guideline from the American College of Surgeons NSQIP and the American Geriatrics Society. J Am Coll Surg. 2016;222(5):930–47.
Rasmussen LS, Steinmetz J. Ambulatory anaesthesia and cognitive dysfunction. Curr Opin Anaesthesiol. 2015;28(6):631–5.
NICE. Delirium: diagnosis, prevention and management. NICE clinical guideline 103 2010; NICE guideline. Available at: http://www.nice.org.uk/CG103.
Lemanu DP, Singh PP, Stowers MD, Hill AG. A systematic review to assess cost effectiveness of enhanced recovery after surgery programmes in colorectal surgery. Colorectal Dis. 2014;16(5):338–46.
White PF, Song D. Bispectral index monitoring and fast tracking after ambulatory surgery: an unexpected finding? Anesthesiology. 2004;100(1):194–5.
White PF. Fast-tracking in ambulatory anesthesia: a new concept? Not! Can J Anaesth. 2002;49(7):759.
Fredman B, Sheffer O, Zohar E, Paruta I, Richter S, Jedeikin R, et al. Fast-track eligibility of geriatric patients undergoing short urologic surgery procedures. Anesth Analg. 2002;94(3):560–4.
Kim G, Kim MH, Lee SM, Choi SJ, Shin YH, Jeong HJ. Effect of pre-warmed intravenous fluids on perioperative hypothermia and shivering after ambulatory surgery under monitored anesthesia care. J Anesth. 2014;28(6):880–5.
Lobo DN. Fluid overload and surgical outcome: another piece in the jigsaw. Ann Surg. 2009;249(2):186–8.
White PF. Facilitating recovery from anesthesia: assessing the costs and benefits of anesthetic drugs. Anesth Analg. 2010;110(2):273–5.
McDonagh DL, Benedict PE, Kovac AL, Drover DR, Brister NW, Morte JB, et al. Efficacy, safety, and pharmacokinetics of sugammadex for the reversal of rocuronium-induced neuromuscular blockade in elderly patients. Anesthesiology. 2011;114(2):318–29.
Lozada MJ, Nguyen JT, Abouleish A, Prough D, Przkora R. Patient preference for the pre-anesthesia evaluation: telephone versus in-office assessment. J Clin Anesth. 2016;31:145–8.
Holt NF. Trends in healthcare and the role of the anesthesiologist in the perioperative surgical home—the US perspective. Curr Opin Anaesthesiol. 2014;27(3):371–6.
Nix J, Smith A, Kurpad R, Nielsen ME, Wallen EM, Pruthi RS. Prospective randomized controlled trial of robotic versus open radical cystectomy for bladder cancer: perioperative and pathologic results. Eur Urol. 2010;57(2):196–201.
Bochner BH, Dalbagni G, Sjoberg DD, Silberstein J, Keren Paz GE, Donat SM, et al. Comparing open radical cystectomy and robot-assisted laparoscopic radical cystectomy: a randomized clinical trial. Eur Urol. 2015;67(6):1042–50.
White PF, Kehlet H, Neal JM, Schricker T, Carr DB, Carli F. The role of the anesthesiologist in fast-track surgery: from multimodal analgesia to perioperative medical care. Anesth Analg. 2007;104(6):1380–96.
White PF. Choice of peripheral nerve block for inguinal herniorrhaphy: is better the enemy of good? Anesth Analg. 2006;102(4):1073–5.
White PF, Kehlet H. Improving postoperative pain management: what are the unresolved issues? Anesthesiology. 2010;112(1):220–5.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
XZC, PFW, and HM declare that there is no conflict of interest.
Funding
No funding was received for this article.
Rights and permissions
About this article
Cite this article
Cao, X., White, P.F. & Ma, H. Perioperative Care of Elderly Surgical Outpatients. Drugs Aging 34, 673–689 (2017). https://doi.org/10.1007/s40266-017-0485-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40266-017-0485-3