Abstract
Background
Infections frequently occur in patients with dementia and antibiotics are often prescribed, but may also be withheld.
Objectives
The aim of this systematic review is to provide a systematic overview of the prevalence of antibiotic use, and factors associated with prescribing antibiotics in patients with dementia.
Data Sources
A systematic search of MEDLINE, EMBASE, PSYCINFO, CINAHL, and the Cochrane library databases until February 13, 2014 was performed, using both controlled terms and free-text terms.
Results
Thirty-seven articles were included. The point prevalence of antibiotic use in patients with dementia ranged from 3.3 to 16.6 %. The period prevalence ranged from 4.4 to 88 % overall, and from 23.5 to 94 % in variable time frames before death; the median use was 52 % (median period 14 days) and 48 % (median period 22 days), respectively. Most patients with lower respiratory tract infections or urinary tract infections (77–91 %) received antibiotic treatment. Factors associated with antibiotic use related to patients, families, physicians, and the healthcare context. More severe dementia and a poor prognosis were associated with less antibiotic use in various countries. Associations with aspiration and illness severity differed by country.
Conclusions and Implications
Antibiotic use in patients with dementia is substantial, and probably highly associated with the particular healthcare context. Future studies may report antibiotic use by infection type and stage of dementia, and compare cross-nationally.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
The prevalence of antibiotic use for patients with dementia is substantial, varies between countries, and depends on the particular healthcare setting |
More severe dementia and a poor prognosis were related to fewer antibiotics in patients with dementia in various countries |
Future studies investigating antibiotic prescription patterns should report antibiotic use by type of infection, stage of dementia, and goals of antibiotic treatment in multiple settings |
1 Introduction
In 2010, 35.6 million people were estimated to have dementia worldwide and this number is expected to nearly double every 20 years [1]. Dementia patients are susceptible to infections, including respiratory tract infections (RTIs), urinary tract infections (UTIs), and skin and soft tissue infections [2, 3], and decision makers such as physicians, patients, and families are often faced with complex treatment decisions, especially at the end of life.
Treatment decisions about antibiotics, specifically in patients with dementia, imply ethical considerations such as whether to accept potential burden caused by treatment, and weighing best interests against patient and family preferences [4, 5]. Furthermore, patients with dementia may be unable to express symptoms and complaints, and typical symptoms of the infection are often absent [6, 7]. Long-term care environments may involve specific challenges such as the absence of diagnostic resources, which complicates appropriate antibiotic treatment [8, 9]. These challenges in the treatment of infections for patients with dementia may lead to variability in antibiotic use. Eventually this can result in burdensome side effects and the development of antibiotic-resistant microbes, which pose a major health risk, especially in older and institutionalized populations [10, 11].
Studies that report on withholding treatment in advanced stages of dementia often focus on cardiopulmonary resuscitation (CPR), artificial nutrition or hospitalization [12–14], which suggests that antibiotic use is frequently regarded as a routine treatment. In addition, patients, families, and professionals often do not realize that dementia is a life-limiting disease, which may result in the deployment of potentially burdensome medical interventions in dementia including intravenous antibiotics and fluids [15–18]. Antibiotic use in dementia may vary across different countries and settings [19, 20]. However, an overview of the actual proportion of patients with dementia that receive antibiotic treatment worldwide in different settings and for various indications is lacking, as is a mapping of the factors associated with its use.
The objectives of this review are to provide a comprehensive overview of (i) the prevalence of antibiotic use in patients with dementia in general and for specific infections, and of (ii) factors associated with antibiotic treatment or withholding treatment in dementia, in various care settings and countries.
2 Methods
2.1 Literature Search
We performed systematic searches in the bibliographic databases PubMed, EMBASE.com, PsycINFO (via EBSCO) and The Cochrane Library (via Wiley) from inception to February 13, 2014. Search terms included controlled terms from MeSH in PubMed, EMtree in EMBASE.com and thesaurus terms in PsycINFO as well as free-text terms. We used free-text terms only in The Cochrane Library. Search terms expressing ‘dementia’ were used in combination with search terms comprising ‘antibiotics’ (see electronic supplementary material [ESM], online resource A). The references of the identified articles were searched for other relevant publications.
2.2 Inclusion Criteria
Articles were included if they reported about antibiotic use in patients with dementia. We excluded articles if they were (i) not reporting about people with dementia or not referring to a population of which at least 50 % had dementia; (ii) not containing empirical data such as in reviews, editorials, letters, and legal cases; (iii) case reports or n = 1 studies; (iv) not reporting about prevalence of antibiotic use or about factors associated with antibiotic use; (v) written in languages other than English, Dutch, French or German.
2.3 Selection Process
Two reviewers (TM and EPJ) independently screened all potentially relevant titles and abstracts for eligibility. If possibly eligible, the full-text article was retrieved and evaluated. Differences in judgment were resolved through a consensus procedure. Data extraction was performed in duplicate by TM and three extra reviewers (SH, MZ, JTS), independently. We resolved any discrepancies in data extracted by discussion until consensus was reached. Data were extracted using a pilot-tested form that included design of the study, subject characteristics, setting, type of infection, diagnosis of the infection, severity of the dementia, prevalence of antibiotic use, and any factors associated with antibiotic use. When the same data about antibiotic prevalence were published in multiple publications, we used the data from the publication that matched our research question best, or, if indifferent, the first publication that reported on the largest possible appropriate selection of participants. We abstracted any factor that was examined for an association with antibiotic use, regardless of country and setting. We therefore abstracted all factors and subsequently categorized by content.
2.4 Assessment of Methodological Quality and Usefulness
The methodological quality of the included articles was assessed using the Mixed Methods Appraisal Tool (MMAT) [21], which enabled the appraisal of both quantitative and qualitative studies within their methodological domain resulting in comparable quality ratings. The quality ratings range from 0 %, when none of four criteria are met, to 100 % when all criteria are met.
In addition to the MMAT scoring, we developed and applied more specific criteria to rate the usefulness of included articles for the purpose of our review, and rated these as useful, somewhat useful or not useful (for further explanation see ESM, online resource B). The MMAT scores and the usefulness of articles were assessed in duplicate and independently by TM and the three extra reviewers (SH, MZ, JTS) and disagreements were resolved by discussion. Articles that scored ≤25 % on the MMAT were excluded when evaluated as somewhat useful; articles evaluated as not useful were excluded regardless of the MMAT score.
3 Results
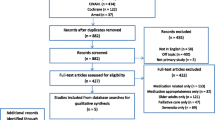
The literature search generated a total of 1,867 references: 892 from PubMed, 843 from EMBASE.com, 114 from PsycINFO, 18 from the Cochrane Library, and an additional 10 references from reference lists. After removing duplicates, 1,556 references remained. After screening titles and abstracts of references retrieved, 49 articles fulfilled the eligibility criteria, and quality and usefulness was adequate for 37 articles (Fig. 1). Five studies were reported in more than one article, and we therefore report data from a total of 34 studies. We found 14 articles reporting about the prevalence of antibiotic use, overall or per infection, 13 articles examining the association of one or more factors with antibiotic use, and 10 articles that reported about both prevalence of antibiotic use and associated factors (Fig. 1).
3.1 Prevalence of Antibiotic Use
Of the 24 articles that reported prevalence of antibiotic use (see ESM, online resource C), most (17) referred to nursing homes, long-term care facilities (LTCFs) or similar settings, and a few referred to a hospital setting (6), or home situation (1). Articles assessed the point prevalence of antibiotic use (3), period prevalence (7), antibiotic use in the last period before death (6) or antibiotic use during a specific infectious episode (8).
In nursing homes in Finland, Italy, and Canada, the point prevalence of antibiotic use ranged between 3.3 and 16.6 % [19, 22, 23]. The point prevalence depended on the setting; in an Italian study, it was 3.3 % in nursing homes, and 15.2 % in community-dwelling patients (Fig. 2a and ESM, online resource C) [19].
The period prevalence of patients who received at least one course of antibiotics in a nursing-home setting was 4.4 % in 3 days in a selection of European countries and Israel [24], and 88 % in a time span of 6 months in the US (Fig. 2b and ESM, online resource C) [25]. The remaining five studies were conducted in hospitals in France, Israel, Canada, and the US, and antibiotic use ranged from 21.8 % during the first 14 days of admission, to 86.2 % during the stay in a hospital with a mean length of stay of 3.6 days. The median period prevalence in these articles was 52 % for a median period of 14 days [26–30].
The six articles that examined period prevalence of antibiotic use until death found percentages that ranged from 23.5 % receiving antibiotics in the last 2 days of life at home in Japan [31], to 94 % during terminal hospitalization in the US [32]. The four remaining studies were conducted in Italian and US nursing homes or hospice agencies and antibiotic use was assessed in the last 6 months, 30 days or 14 days of life, and the last 7 days in hospice care, with a median prevalence of 48 % for a median period of 22 days (Fig. 2c and ESM, online resource C) [28–30, 33–36].
Five articles reported about treatment for patients with lower respiratory infections (LRIs) or pneumonia, mostly diagnosed by clinical criteria (Fig. 2d and ESM, online resource C). Three US articles reported that 85.3–91.1 % of patients received antibiotic therapy [37–39], and two Dutch articles reported percentages of 77 and 79 % [40, 41].
Antibiotic treatment of UTIs was provided in 77.9 % of episodes in US nursing homes [6]. An article that focused on treatment of feverish conditions reported that antibiotics were used in 37.8 % of 172 feverish episodes in 104 patients in the US, of which 93 episodes were RTIs, 67 were UTIs, and 25 were systemic infections; all diagnoses were based on physical examinations [42]. Another article reported that 43.1 % of 102 feverish episodes in 193 patients were treated with antibiotics in Finland [43].
3.2 Factors Associated with Antibiotic Use
Papers reported a total of 57 factors that were associated with antibiotic use and could be categorized into factors that related to (i) the patient’s health status, (ii) persons involved in the decision making such as patients, physicians or families, and (iii) the healthcare context, such as country and setting (Tables 1, 2, 3). For only three articles, the search for factors associated with antibiotic use was the primary goal, and these articles investigated associations of multiple factors with antibiotic use for pneumonia in patients with dementia [37, 41, 44]. Most articles tested one or a few factors that were or were not based on specific hypotheses.
3.2.1 Patient’s Health Status
The majority of factors associated with antibiotic use in the included articles related to the patient’s health status (27/57); four factors were reported in two or more articles, and showed consistent associations with fewer antibiotic treatments (Table 1—highlighted rows). The severity of dementia was studied in four articles and all found that patients with more severe dementia were less likely to receive antibiotics [33, 39, 41, 44]. Furthermore, a poor prognosis was associated with fewer antibiotics in a selection of European nursing homes [24], and in a Dutch study which assessed physicians’ subjective predictions in a survey [45]. The latter reported that three-quarters of the physicians would consider a mortality risk of 75–90 % in spite of treatment sufficiently high to justify withholding antibiotics [45]. Lastly, illness severity and eating dependence, both pre-LRI and at the time of the treatment decision, were associated with fewer antibiotic treatments in two articles [39, 41]. Other factors were only examined in one study.
Eating dependency, drinking insufficiently and being dehydrated, both before a LRI and at the time of the treatment decision, and swallowing difficulty decreased the likelihood of antibiotic treatment in the US and the Netherlands [41, 44]. Patients who had been diagnosed with pneumonia previously [41], and were more ADL (activities of daily living)-dependent [39], were also less likely to receive antibiotics. A high body temperature in the US and the Netherlands [44], and unstable vital signs in the US were positively related to antibiotic treatment for pneumonia [37].
Some articles showed contrasting results. Illness severity at the time of the treatment decision and 2 weeks before the treatment decision related to withholding antibiotics in the Netherlands [41], but in the US, indicators of more severe acute illness were associated with more antibiotic prescriptions [39], or no association was found [44]. In addition, aspiration was associated with withholding antibiotics in the Netherlands [41], but in contrast, patients with suspected aspiration were more frequently treated with antibiotics in the US [37].
3.2.2 Persons Involved in Decision Making
Articles that reported factors associated with persons involved in decision making assessed attitudes of patients, families, and physicians in hypothetical scenarios, or in real-life situations using qualitative designs (Table 2 and ESM, online resource C, table 3). Four studies, conducted in the US and Australia, found that 47 % (71/152) [46], and 73 % (159/218; 38/52) [47, 48] of subjects deciding for themselves were willing to accept antibiotics in general, or specifically for the treatment of pneumonia. Agreeing to antibiotics depended on the severity of the dementia with percentages ranging from 74 % (62/84) choosing antibiotic treatment in the case of early Alzheimer’s disease (AD) to only 25 % (21/84) in the case of severe or late AD in a US study [49].
Three articles assessed attitudes of family members of patients with dementia using a hypothetical scenario, and reported that a majority (range 60–90 %) preferred antibiotic treatment. The highest percentage was found in a US study reporting that 90 % (45/50) of spouses would choose antibiotics for their relative with dementia in case of a life-threatening infection [50]. In the case of critical illness (not further specified), 78 % (40/51) of Chinese family caregivers would agree with antibiotic treatment for their relative with dementia [51]. In a similar scenario, 60 % (30/50) of relatives would agree to oral antibiotics in the UK [52].
Other studies explored attitudes of families and physicians using qualitative designs. A US focus group study pointed out that family members encountered difficulties viewing pneumonia as part of a ‘natural death’ for someone with dementia, and easily agreed to aggressive treatment including intravenous antibiotics. In fact, antibiotics were viewed as a comfort measure, rather than as a technological way of prolonging life [53]. Furthermore, a US survey study found that while spouses were reasonably comfortable to forgo life-sustaining treatments such as CPR and feeding tubes, of all treatments spouses were the most comfortable with the decision to agree to antibiotics and the least comfortable with the decision to forego antibiotics [50].
A qualitative interview study that focused on the role of the physician found that physicians in the Netherlands have more patient contact and therefore know the patients and their relatives better than US physicians, and patient contact helped to start a timely discussion about treatment options for life-threatening infections. Ongoing care discussions before a ‘crisis situation’ occurs were thought to increase the odds of not choosing an aggressive treatment option [54]. Further, physicians preferred not to treat pneumonia when they considered treatment futile [45], and curative treatment was sometimes forgone with an explicit intention to hasten death [55].
3.2.3 Healthcare Context
The articles that described factors regarding the healthcare context indicated that patients living alone [46], and at home, were more likely to receive antibiotics than nursing-home residents or hospital inpatients (Table 3) [19]. In Dutch nursing homes, the percentage of patients who received antibiotics for pneumonia increased with the number of psycho-geriatric beds in the facility [41]. The total number of beds or the number of beds for somatic patients was unrelated to antibiotic use for patients with dementia. Furthermore, antibiotic prescriptions were not related to the religious affiliation of the facility, urbanization level, the level of policy making or the availability of a protocol or policy on treatment or non-treatment [41]. Patients living in the Netherlands were less likely to receive antibiotic treatment than patients residing in the US [20]. Furthermore, patients who had a ‘do not hospitalize’ (DNH) order [37], or an advance directive [41, 56], were less likely to receive antibiotics.
Recommendations of a palliative care team aimed at improving comfort in a hospital did not affect antibiotic use in a US study [26]. In a recent Finnish article, an expert team comprising an infectious disease consultant and a geriatrician visited LTCFs and succeeded in reducing inappropriate use of antibiotics as UTI prophylaxis [23]. The factor time showed no association with antibiotic treatment, as observational studies in the US and the Netherlands found no change in antibiotic prescriptions over time in three and two death cohorts, respectively, between 1985 and 2007 [33, 40].
4 Discussion
To our best knowledge, this is the first review that systematically assesses the prevalence of antibiotic use among patients with dementia, and factors associated with antibiotic prescriptions in this population. Antibiotic use is often substantial (median 48 % within a median period of 14–30 days), but highly variable as the period prevalence ranged from 4.4 to 88 %. However, in attitudinal studies, many patients, families, and physicians prefer to forgo treatment. We found that more severe dementia and a poor prognosis were consistently associated with using less antibiotics in various countries. Associations with aspiration, illness severity, and a number of healthcare contextual factors differed by country.
4.1 Strengths and Limitations
We systematically identified, reviewed, and evaluated the literature concerning antibiotic use in patients with dementia. We considered the quality of the data in two ways, including only studies that met both general methodological criteria and usefulness criteria. The latter were developed because the first were rather unspecific and most studies were not primarily aimed at examining the prevalence of antibiotic use and factors associated with it, but provided data that could be useful to some extent to address the research question of this review. Studies that were somewhat useful were only included when they met the minimum criteria for acceptable methodological quality.
Some limitations should be acknowledged. First, detailed information about the diagnosis of infections and the specific reason to prescribe antibiotics was mostly lacking. Second, we included studies when more than 50 % of subjects had dementia and some populations (4 of the 37 articles had less than 100 % dementia) were therefore not homogeneous. Third, because investigating associations with antibiotic use was often not the primary goal of the studies, only a few factors were examined in multiple articles, leaving little opportunity to compare between countries and settings. Fourth, we are aware of the fact that the findings of studies conducted in the Netherlands in nursing-home settings contributed substantially to the factors we identified as associated with antibiotic use. This indicates the importance of studying the factors associated with antibiotic treatment in countries other than the Netherlands. Last, articles that reported on attitudes of family and patients regarding antibiotics were among the oldest articles included, and often applied a hypothetical scenario or a qualitative approach using focus groups. This leaves room for discussion about what would be chosen in actual practice, and how valuable these results are to represent attitudes towards withholding treatment with antibiotics today. Over the last decades, attitudes regarding providing antibiotics for comfort may have changed and the involvement of patients themselves and family may have gained importance.
4.2 Variability in Antibiotic Use
The prevalence of antibiotic use varied widely between studies. Although the designs of the prevalence studies varied, antibiotic prescription patterns in practice probably also varied widely. Part of this variability is likely caused by little evidence being available on effects to guide practice. Furthermore, the decision about antibiotics may depend on country, setting, and on whether the patient is perceived as approaching the end of life. For example, we found that antibiotics were frequently prescribed in the last 30 days of life in Italy, but, in contrast, the point prevalence of antibiotic use in an earlier period was surprisingly low [19, 34].
4.3 Country and Setting
Studies comparing different countries after pooling individual patient data examined the situation in the US and in the Netherlands. The few studies in other countries did not compare directly with other countries. Antibiotic use in general nursing-home populations varies between European countries; the mean point prevalence of antibiotic use ranged from 1.1 % in Latvia to 15.9 % in Finland [57]. Although this report lacks data about the prevalence in patients with dementia specifically, antibiotic use may be as variable, or perhaps even more variable in these patients.
US nursing-home residents, compared with Dutch residents, were more likely to receive antibiotics for pneumonia. Comparing both countries, the same factor operated in different directions: severity of the infection was associated with fewer antibiotics in the Netherlands, but increased antibiotic use in the US. Differences in training of physicians and differences in the organization of care may explain this. Dutch elderly care physicians follow a 3-year vocational training in elderly care medicine that includes training in advance care planning and decision making in end-of-life care [54, 58]. Further, elderly care physicians in the Netherlands are employed by the nursing home which is their principal site of practice. They therefore meet with their patients regularly, while, in many other countries and settings, care in the nursing home is provided by the general practitioner [58, 59]. Fewer physician contacts potentially result in less certainty about treatment decisions and family preferences, even after adjustment for country [60]. Physicians’ experiences with treatment of patients with dementia, and with a focus on palliative care and withholding treatments, may also play a role.
Patients treated at home were more likely to receive antibiotics for an infection compared with patients in a hospital or nursing-home setting [19, 56]. It was suggested that the use of antibiotics at home may be higher due to urinary catheters, which were five times more common in patients living at home [19]. Furthermore, patients eligible for home care may have specific indications that relate to antibiotic use, such as infections or pressure sores. In many countries, hospitalization of patients with dementia and pneumonia is common and typically involves intravenous antibiotic treatment [61–63]. Treatments such as parenteral antibiotics—which may be the only treatment option for patients with intake problems—may not be available in a home-care or nursing-home setting, depending on the country, which implies that a decision for antibiotics sometimes parallels a decision to hospitalize.
Although few studies assessed cross-national variation in attitudes towards antibiotic treatment for patients with dementia, these attitudes and decision making probably differ. This is supported by variability in treatment decisions between physicians in different countries using hypothetical scenarios regarding chronically ill older patients. For example, 897 physicians from seven countries selected a treatment option from supportive care only to maximum care including admission to the intensive care unit for an 82-year-old man with a gastrointestinal bleeding, and US physicians were among the most aggressive while Australian colleagues were the most conservative [64, 65].
4.4 Decision Making About Antibiotic Treatment
Most family members of patients with dementia would agree to antibiotic treatment for an infection. Many experience emotional difficulties including guilt when deciding to refrain from life-sustaining treatments [17], but being involved in decision making is not always regarded as a burden [66]. To avoid routinely prescribing antibiotics, one of the parties involved should initiate the discussion of withholding curative treatment [67]. Not only whether treatment options are being discussed, but also how they are addressed may make a difference. For example, in a US survey study, a detailed written explanation about pneumonia to inform community-dwelling older people about treatment options surprisingly led to more of the subjects choosing antibiotics in the case of severe dementia and pneumonia [68]. In practice, the physician’s attitude may be important. That is, an attitude which is more passive and deferential to family preferences may result in more aggressive treatment (antibiotics and hospitalization), in contrast to an attitude to treat based on what is perceived by the physician to be in the best interest of the patient. This was observed in a study in which physicians in both the US and the Netherlands indicated that timely discussion of treatment options may decrease the risk of starting inappropriate treatment of pneumonia [54], emphasizing the importance of timely and effective communication between all parties involved in decision making.
4.5 Effects of Antibiotic Treatment: Ethical Considerations
The rationales behind withholding antibiotic treatment for terminally ill patients with dementia or incompetent patients and palliative use of antibiotics are the subject of an ethical debate in the literature [15, 69–74]. As the dementia progresses, the general treatment goal may shift from life prolongation to maintenance of function, and eventually to maximization of comfort [75]. However, the evidence base to guide treatment decisions about use of antibiotics consistent with these goals is small. In advanced dementia, antibiotics may prolong life in only a small minority of patients [76], but hydration status affected survival even more profoundly than antibiotic treatment in nursing-home residents, many of whom had dementia [44]. Antibiotics might also relieve the symptoms of pneumonia in the absence of other proper treatment to relieve symptoms [77], and may be prescribed to provide comfort [41]. However, it remains unclear whether antibiotics actually enhance comfort, and if they do, whether such benefit outweighs the potential burden of antibiotic treatment in severely ill patients with dementia. Furthermore, one may consider that the patient, when cured of the infection, is still exposed to the deterioration of the dementia [78, 79]. When the goal of antibiotic treatment is not to provide comfort, and treatment is not expected to decrease mortality risk [42, 76], some question its usefulness. Others claim that, regardless of the underlying illness and potentially negative consequences, withholding a drug that is effective in a disease treatment is always inappropriate [80].
In decision making about providing antibiotics, prescribers may also consider their decision from a public health point of view and include the emergence of antibiotic resistance into their considerations to start or withhold treatment [69]. However, antibiotic resistance and inappropriate antibiotic use did not emerge as a factor from the studies we included in this review. This is not surprising, considering the novelty of the topic of antibiotic resistance, the limited awareness about it in, for example, the nursing home setting, and difficulty applying general knowledge about resistance in the community in individual cases in clinical practice [11, 69].
4.6 Implications
This review identifies several gaps in knowledge about the prevalence of antibiotic use in patients with dementia, as well as about factors associated with use of antibiotics in this population. The majority of articles focused on overall antibiotic use, or antibiotic use for RTIs, mostly in institutionalized patients (long-term care; hospital). Information about other common infections such as UTIs and skin infections, and about community-dwelling patients, is virtually lacking. Furthermore, few studies focused specifically on patients with advanced or end-stage dementia, and the factors investigated in these studies were not always the same as those assessed in studies that included all stages of dementia. Moreover, we found no studies that examined if factors associated with antibiotic use differed by dementia stage. Future studies should address these gaps, and distinguish types of infections and stages of dementia. We suggest a cross-national study in which a standardized set of factors as identified from our review is examined simultaneously and systematically, to further investigate antibiotic prescription patterns and how these may vary between countries and settings.
Little is known about attitudes and decision making in real practice situations. Qualitative studies using individual interviews or ethnographic designs may assess attitudes among patients, family, healthcare workers, and physicians, and other factors that are important in decision making around antibiotics in practice. Observational studies about antibiotic use in patients with dementia should include the goal of antibiotic treatment, and investigate associations with function, survival, comfort, and quality of life in different settings to contribute to a more evidence-based approach in antibiotic use.
4.7 Conclusion
This review suggests that decision making about starting or withholding antibiotic treatment remains a challenge, involves ethical considerations, and is strongly influenced by the particular healthcare context. Treatment with antibiotics is sometimes withheld, but considerations about this, and perhaps whether use or non-use is considered at all, depend on country, setting, and family and physician preferences. This review provides a basis for further research and an international discussion among stakeholders about the ethical and practical considerations of withholding antibiotic treatment in patients with dementia.
References
Prince M, Bryce R, Albanese E, et al. The global prevalence of dementia: a systematic review and metaanalysis. Alzheimers Dement. 2013;9(1):63–75.
Mitchell SL, Teno JM, Kiely DK, et al. The clinical course of advanced dementia. N Engl J Med. 2009;361(16):1529–38.
Vandervoort A, Van den Block L, van der Steen JT, et al. Nursing home residents dying with dementia in Flanders, Belgium: a nationwide postmortem study on clinical characteristics and quality of dying. J Am Med Dir Assoc. 2013;14(7):485–92.
Hurley AC, Volicer L. Alzheimer Disease: “It’s okay, Mama, if you want to go, it’s okay”. JAMA. 2002;288(18):2324–31.
van der Steen JT, Ooms ME, Ribbe MW, et al. Decisions to treat or not to treat pneumonia in demented psychogeriatric nursing home patients: evaluation of a guideline. Alzheimer Dis Assoc Disord. 2001;15(3):119–28.
D’Agata E, Loeb MB, Mitchell SL. Challenges in assessing nursing home residents with advanced dementia for suspected urinary tract infections. J Am Geriatr Soc. 2013;61(1):62–6.
Scherder E, Herr K, Pickering G, et al. Pain in dementia. Pain. 2009;145(3):276–8.
Loeb M, Simor AE, Landry L, et al. Antibiotic use in Ontario facilities that provide chronic care. J Gen Intern Med. 2001;16(6):376–83.
Zimmer JG, Bentley DW, Valenti WM, et al. Systemic antibiotic use in nursing homes. A quality assessment. J Am Geriatr Soc. 1986;34(10):703–10.
Croft AC, D’Antoni AV, Terzulli SL. Update on the antibacterial resistance crisis. Med Sci Monit. 2007;13(6):RA103–18.
van Buul LW, van der Steen JT, Veenhuizen RB, et al. Antibiotic use and resistance in long term care facilities. J Am Med Dir Assoc. 2012;13(6):568–13.
Jox RJ, Denke E, Hamann J, et al. Surrogate decision making for patients with end-stage dementia. Int J Geriatr Psychiatry. 2012;27(10):1045–52.
Mitchell SL, Teno JM, Intrator O, et al. Decisions to forgo hospitalization in advanced dementia: a nationwide study. J Am Geriatr Soc. 2007;55(3):432–8.
Rurup ML, Onwuteaka-Philipsen BD, Pasman HR, et al. Attitudes of physicians, nurses and relatives towards end-of-life decisions concerning nursing home patients with dementia. Patient Educ Couns. 2006;61(3):372–80.
Congedo M, Causarano RI, Alberti F, et al. Ethical issues in end of life treatments for patients with dementia. Eur J Neurol. 2010;17(6):774–9.
Mitchell SL, Kiely DK, Hamel MB. Dying with advanced dementia in the nursing home. Arch Intern Med. 2004;164(3):321–6.
Sachs GA, Shega JW, Cox-Hayley D. Barriers to excellent end-of-life care for patients with dementia. J Gen Intern Med. 2004;19(10):1057–63.
Morrison RS, Siu AL. Mortality from pneumonia and hip fractures in patients with advanced dementia. JAMA. 2000;284(19):2447–8.
Toscani F, Di GP, Villani D, et al. Treatments and prescriptions in advanced dementia patients residing in long-term care institutions and at home. J Palliat Med. 2013;16(1):31–7.
van der Steen JT, Kruse RL, Ooms ME, et al. Treatment of nursing home residents with dementia and lower respiratory tract infection in the United States and The Netherlands: an ocean apart. J Am Geriatr Soc. 2004;52(5):691–9.
Pace R, Pluye P, Bartlett G, et al. Testing the reliability and efficiency of the pilot Mixed Methods Appraisal Tool (MMAT) for systematic mixed studies review. Int J Nurs Stud. 2012;49(1):47–53.
Daneman N, Gruneir A, Newman A, et al. Antibiotic use in long-term care facilities. J Antimicrob Chemother. 2011;66(12):2856–63.
Rummukainen ML, Jakobsson A, Matsinen M, et al. Reduction in inappropriate prevention of urinary tract infections in long-term care facilities. Am J Infect Control. 2012;40(8):711–4.
Onder G, Liperoti R, Foebel A, et al. Polypharmacy and mortality among nursing home residents with advanced cognitive impairment: results from the SHELTER study. J Am Med Dir Assoc. 2013;14(6):450–12.
Blass DM, Black BS, Phillips H, et al. Medication use in nursing home residents with advanced dementia. Int J Geriatr Psychiatry. 2008;23(5):490–6.
Ahronheim JC, Morrison RS, Morris J, et al. Palliative care in advanced dementia: a randomized controlled trial and descriptive analysis. J Palliat Med. 2000;3(3):265–73.
Catic AG, Berg AI, Moran JA, et al. Preliminary data from an advanced dementia consult service: integrating research, education, and clinical expertise. J Am Geriatr Soc. 2013;61(11):2008–12.
Malyuk RE, Wong C, Buree B, et al. The interplay of infections, function and length of stay (LOS) in newly admitted geriatric psychiatry patients. Arch Gerontol Geriatr. 2012;54(1):251–5.
Nourhashemi F, Gillette S, Cantet C, et al. End-of-life care for persons with advanced Alzheimer disease: design and baseline data from the ALFINE study. J Nutr Health Aging. 2012;16(5):457–61.
Reisfeld S, Paul M, Gottesman BS, et al. The effect of empiric antibiotic therapy on mortality in debilitated patients with dementia. Eur J Clin Microbiol Infect Dis. 2011;30(6):813–8.
Hirakawa Y, Masuda Y, Kuzuya M, et al. End-of-life experience of demented elderly patients at home: findings from DEATH project. Psychogeriatrics. 2006;6(2):60–7.
Ahronheim JC, Morrison RS, Baskin SA, et al. Treatment of the dying in the acute care hospital. Advanced dementia and metastatic cancer. Arch Intern Med. 1996;156(18):2094–100.
Evers MM, Purohit D, Perl D, et al. Palliative and aggressive end-of-life care for patients with dementia. Psychiatr Serv. 2002;53(5):609–13.
Di Giulio P, Toscani F, Villani D, et al. Dying with advanced dementia in long-term care geriatric institutions: a retrospective study. J Palliat Med. 2008;11(7):1023–8.
D’Agata E, Mitchell SL. Patterns of antimicrobial use among nursing home residents with advanced dementia. Arch Intern Med. 2008;168(4):357–62.
Albrecht JS, Gruber-Baldini AL, Fromme EK, et al. Quality of hospice care for individuals with dementia. J Am Geriatr Soc. 2013;61(7):1060–5.
Chen JH, Lamberg JL, Chen YC, et al. Occurrence and treatment of suspected pneumonia in long-term care residents dying with advanced dementia. J Am Geriatr Soc. 2006;54(2):290–5.
Givens JL, Jones RN, Shaffer ML, et al. Survival and comfort after treatment of pneumonia in advanced dementia. Arch Intern Med. 2010;170(13):1102–7.
Mehr DR, van der Steen JT, Kruse RL, et al. Lower respiratory infections in nursing home residents with dementia: a tale of two countries. Gerontologist. 2003;43(2):85–93.
van der Steen JT, Meuleman-Peperkamp I, Ribbe MW. Trends in treatment of pneumonia among Dutch nursing home patients with dementia. J Palliat Med. 2009;12(9):789–95.
van der Steen JT, Ooms ME, Ader HJ, et al. Withholding antibiotic treatment in pneumonia patients with dementia: a quantitative observational study. Arch Intern Med. 2002;162(15):1753–60.
Fabiszewski KJ, Volicer B, Volicer L. Effect of antibiotic treatment on outcome of fevers in institutionalized Alzheimer patients. JAMA. 1990;263(23):3168–72.
Visapaa J. Significant changes in the terminal care of aged patients in the long-term care in Helsinki. Scand J Soc Med. 1998;26(1):53–5.
Szafara KL, Kruse RL, Mehr DR, et al. Mortality following nursing home-acquired lower respiratory infection: LRI severity, antibiotic treatment, and water intake. J Am Med Dir Assoc. 2012;13(4):376–83.
van der Steen JT, Helton MR, Ribbe MW. Prognosis is important in decisionmaking in Dutch nursing home patients with dementia and pneumonia. Int J Geriatr Psychiatry. 2009;24(9):933–6.
Lo B, McLeod GA, Saika G. Patient attitudes to discussing life-sustaining treatment. Arch Intern Med. 1986;146(8):1613–5.
Low JA, Chan DKY, Hung WT, et al. Treatment of recurrent aspiration pneumonia in end-stage dementia: preferences and choices of a group of elderly nursing home residents. Intern Med J. 2003;33(8):345–9.
Reilly RB, Teasdale TA, McCullough LB. Projecting patients’ preferences from living wills: an invalid strategy for management of dementia with life-threatening illness. J Am Geriatr Soc. 1994;42(9):997–1003.
Gjerdingen DK, Neff JA, Wang M, et al. Older persons’ opinions about life-sustaining procedures in the face of dementia. Arch Fam Med. 1999;8(5):421–5.
Mezey M, Kluger M, Maislin G, et al. Life-sustaining treatment decisions by spouses of patients with Alzheimer’s disease. J Am Geriatr Soc. 1996;44(2):144–50.
Kwok T, Twinn S, Yan E. The attitudes of Chinese family caregivers of older people with dementia towards life sustaining treatments. J Adv Nurs. 2007;58(3):256–62.
Potkins D, Bradley S, Shrimanker J, et al. End of life treatment decisions in people with dementia: carers’ views and the factors which influence them. Int J Geriatr Psychiatry. 2000;15(11):1005–8.
Forbes S, Bern-Klug M, Gessert C. End-of-life decision making for nursing home residents with dementia. J Nurs Scholarsh. 2000;32(3):251–8.
Helton MR, van der Steen JT, Daaleman TP, et al. A cross-cultural study of physician treatment decisions for demented nursing home patients who develop pneumonia. Ann Fam Med. 2006;4(3):221–7.
van der Steen JT, Van Der Wal G, Mehr DR, et al. End-of-life decision making in nursing home residents with dementia and pneumonia: Dutch physicians’ intentions regarding hastening death. Alzheimer Dis Assoc Disord. 2005;19(3):148–55.
Parsons C, McCorry N, Murphy K, et al. Assessment of factors that influence physician decision making regarding medication use in patients with dementia at the end of life. Int J Geriatr Psychiatry. 2013.
ESAC. Report on point prevalence survey of antimicrobial prescription in European nursing homes, 2009. 2010.
Koopmans RT, Lavrijsen JC, Hoek JF, et al. Dutch elderly care physician: a new generation of nursing home physician specialists. J Am Geriatr Soc. 2010;58(9):1807–9.
Hoek JF, Ribbe MW, Hertogh CM, et al. The role of the specialist physician in nursing homes: the Netherlands’ experience. Int J Geriatr Psychiatry. 2003;18(3):244–9.
Helton MR, Cohen LW, Zimmerman S, et al. The importance of physician presence in nursing homes for residents with dementia and pneumonia. J Am Med Dir Assoc. 2011;12(1):68–73.
Li Q, Zheng NT, Temkin-Greener H. Quality of end-of-life care of long-term nursing home residents with and without Dementia. J Am Geriatr Soc. 2013;61(7):1066–73.
Givens JL, Selby K, Goldfeld KS, et al. Hospital transfers of nursing home residents with advanced dementia. J Am Geriatr Soc. 2012;60(5):905–9.
Lamberg JL, Person CJ, Kiely DK, et al. Decisions to hospitalize nursing home residents dying with advanced dementia. J Am Geriatr Soc. 2005;53(8):1396–401.
Alemayehu E, Molloy DW, Guyatt GH, et al. Variability in physicians’ decisions on caring for chronically ill elderly patients: an international study. CMAJ. 1991;144(9):1133–8.
Molloy DW, Guyatt GH, Alemayehu E, et al. Factors affecting physicians’ decisions on caring for an incompetent elderly patient: an international study. CMAJ. 1991;145(8):947–52.
Jacobs VE, van Zuthem HJ, van Delden JJ. Experiences of guardians of psychogeriatric patients regarding decision not to treat. Ned Tijdschr Geneeskd. 2000;144(8):378–81.
van der Steen JT, van Soest-Poortvliet MC, Hallie-Heierman M, et al. Factors associated with initiation of advance care planning in dementia: a systematic review. J Alzheimers Dis. 2014;40(3):743–57.
Ainslie N, Beisecker AE. Changes in decisions by elderly persons based on treatment description. Arch Intern Med. 1994;154(19):2225–33.
Marcus EL, Clarfield AM, Moses AE. Ethical issues relating to the use of antimicrobial therapy in older adults. Clin Infect Dis. 2001;33(10):1697–705.
Brody H, Campbell ML, Faber-Langendoen K, et al. Withdrawing intensive life-sustaining treatment—recommendations for compassionate clinical management. N Engl J Med. 1997;336(9):652–7.
Wanzer SH, Adelstein SJ, Cranford RE, et al. The physician’s responsibility toward hopelessly ill patients. N Engl J Med. 1984;310(15):955–9.
Parsons C, Hughes CM, Passmore AP, et al. Withholding, discontinuing and withdrawing medications in dementia patients at the end of life: a neglected problem in the disadvantaged dying? Drugs Aging. 2010;27(6):435–49.
Chen LK, Chou YC, Hsu PS, et al. Antibiotic prescription for fever episodes in hospice patients. Support Care Cancer. 2002;10(7):538–41.
Stiel S, Krumm N, Pestinger M, et al. Antibiotics in palliative medicine–results from a prospective epidemiological investigation from the HOPE survey. Support Care Cancer. 2012;20(2):325–33.
van der Steen JT, Radbruch L, Hertogh CM, et al. White paper defining optimal palliative care in older people with dementia: a Delphi study and recommendations from the European Association for Palliative Care. Palliat Med. 2014;28(3):197–209.
van der Steen JT, Lane P, Kowall NW, et al. Antibiotics and mortality in patients with lower respiratory infection and advanced dementia. J Am Med Dir Assoc. 2012;13(2):156–61.
van der Steen JT, Pasman HR, Ribbe MW, et al. Discomfort in dementia patients dying from pneumonia and its relief by antibiotics. Scand J Infect Dis. 2009;41(2):143–51.
Schwaber MJ, Carmeli Y. Antibiotic therapy in the demented elderly population: redefining the ethical dilemma. Arch Intern Med. 2008;168(4):349–50.
Hertogh CM, Ribbe MW. Ethical aspects of medical decision-making in demented patients: a report from the Netherlands. Alzheimer Dis Assoc Disord. 1996;10(1):11–9.
Rozzini R, Trabucchi M. Antibiotic therapy in elderly persons affected by severe dementia. Arch Intern Med. 2008;168(22):2497–8.
Acknowledgements
This study is supported by the Netherlands Organisation for Scientific Research (NWO), the Hague; Innovational Research Incentives Scheme, a career award to JTS (Grant number Vidi 91711339).
Conflict of interest
We declare that there are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material (ESM)
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
van der Maaden, T., Hendriks, S.A., de Vet, H.C.W. et al. Antibiotic Use and Associated Factors in Patients with Dementia: A Systematic Review. Drugs Aging 32, 43–56 (2015). https://doi.org/10.1007/s40266-014-0223-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40266-014-0223-z