Abstract
Background
Recent clinical trials of anti-Aβ monoclonal antibodies (mAbs) in the treatment of early Alzheimer’s disease (AD) have produced encouraging cognitive and clinical results. The purpose of this network meta-analysis (NMA) was to compare and rank mAb drugs according to their efficacy and safety.
Methods
PubMed, Embase, Web of Science, and the Cochrane Library were searched for randomized controlled trials testing various mAbs for the treatment of cognitive decline in patients with AD, up to March 31, 2023. R software (version 4.2.3) along with JAGS and STATA software (version 15.0) were used for statistical analysis. Odds ratio (OR) for binary variables, mean difference (MD) for continuous variables, and their 95% confidence intervals (CI) were utilized to estimate treatment effects and rank probabilities for each mAb in terms of safety and efficacy outcomes. We calculated the surface under the cumulative ranking area (SUCRA) to evaluate each mAb, with higher SUCRA values indicating better efficacy or lower likelihood of adverse events.
Results
Thirty-three randomized controlled trials with a total of 21,087 patients were included in the current NMA, involving eight different mAbs. SUCRA values showed that aducanumab (87.01% and 99.37%, respectively) was the most likely to achieve the best therapeutic effect based on the changes of Mini-Mental State Examination (MMSE) and Clinical Dementia Rating scale Sum of Boxes (CDR-SB) scores. Donanemab (88.50% and 99.00%, respectively) performed better than other therapies for Alzheimer's Disease Assessment Scale–cognitive subscale (ADAS-cog) and Positron Emission Tomography-Standardized Uptake Value ratio (PET-SUVr). Lecanemab (87.24%) may be the most promising way to slow down the decrease of Alzheimer's Disease Cooperative Study–Activities of Daily Living (ADCS-ADL) score. In the analysis of the incidence of adverse events (subjects with any treatment-emergent adverse event), gantenerumab (89.12%) had the least potential for adverse events, while lecanemab (0.79%) may cause more adverse events. Solanezumab (95.75% and 80.38%, respectively) had the lowest incidence of amyloid-related imaging abnormalities characterized by edema and effusion (ARIA-E) and by cerebral microhemorrhages (ARIA-H) of the included immunotherapies. While SUCRA values provided a comprehensive measure of treatment efficacy, the inherent statistical uncertainty required careful analysis in clinical application.
Conclusion
Despite immunotherapies significantly increasing the risks of adverse events and ARIA, the data suggest that mAbs can effectively improve the cognitive function of patients with mild and moderate AD. According to the NMA, aducanumab was the most likely to achieve significant improvements in different cognitive and clinical assessments (statistically improved MMSE and CDR-SB), followed by donanemab (statistically improved ADAS-Cog, and PET-SUVr) and lecanemab (statistically improved ADCS-ADL).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Among individual drugs, aducanumab was the most likely to achieve significant improvements in different cognitive and clinical assessments (statistically improved MMSE and CDR-SB) followed by donanemab (statistically improved ADAS-Cog, and PET-SUVr) and lecanemab (statistically improved ADCS-ADL). |
These treatments also increase the risk of adverse events, such as amyloid-related imaging abnormalities. Thus, their use requires careful consideration and further research. |
1 Introduction
Alzheimer's disease (AD) is characterized by relentlessly progressive neurodegeneration resulting in symptoms that affect language and motivation, and cause behavioral and cognitive disorders that gradually lead to patient mortality [1, 2]. It is the most common type of dementia, accounting for 60–70% of cases [3]. It is estimated that the number of patients diagnosed with AD will increase to 65.7 million by 2030 and 115.4 million by 2050, which has attracted worldwide attention [4, 5].
New diagnostic methods, including advanced neuroimaging technology and biochemical biomarkers, are steadily becoming available as accurate methods for detecting AD. However, there is no effective treatment, and prevention is still the priority [6]. Therefore, it is essential to understand the underlying pathology of AD and develop disease-modifying therapies that not only improve symptoms but also alter the disease course.
AD is characterized by three neuropathological features including extracellular amyloid beta-peptide (Aβ) plaques, intraneuronal neurofibrillary tangles (NFT) composed of hyper-phosphorylated tau protein, and loss of synapses as well as neurons, but the exact mechanisms by which they ultimately cause cognitive deficits are still unclear [1, 7].
The treatment scheme for AD remains limited. The established US Food and Drug Administration (FDA) approved pharmacological interventions, such as donepezil, galantamine, rivastigmine, and memantine, have only modest and transient effects, and do not stop the progression of dementia [8]. The amyloid cascade hypothesis is the most widely accepted hypothesis so far, which proposes that the imbalance between the deposition of neurotoxic Aβ protein and the production and elimination of Aβ triggers a series of pathological changes, including amyloid plaques, neurofibrillary tangles, neuronal dysfunction, and dementia [9, 10]. In the past two decades, strategies targeted at the amyloid cascade hypothesis have included drugs that focus on reducing amyloid-β production (β-secretase 1 inhibitor or α-secretase modulator) or increasing amyloid-β clearance (anti-amyloid-β antibodies or active immunotherapy) [11]. However, unfortunately, due to adverse effects and a lack of therapeutic effect, most compounds have been unsuccessful in phase II/III trials [12, 13]. Recent studies have provided evidence that different therapeutic strategies for clearing brain Aβ may be effective for altering disease course, and monoclonal antibodies (mAbs) against Aβ have attracted more and more attention.
Mounting evidence suggests that mAbs demonstrate potential in improving outcomes for the treatment of patients at early stages of AD. For example, bapineuzumab was the first N-terminus-directed anti-Aβ antibody tested in humans, reducing Aβ level, but demonstrating little improvement in cognition for AD patients [14]. Subsequently, several anti-Aβ mAb drugs were tested in clinical trials. With the recent approval of lecanemab, the first mAb for Alzheimer’s disease, via the accelerated approval pathway in the US, attention now moves to understanding the efficacy and safety profile of this and the other mAbs. Previous paired meta-analysis had evaluated efficacy and safety of all types of immunotherapy drugs against Aβ within a narrow observation range [15]. Therefore, the objective of this network meta-analysis (NMA) was to compare and rank mAbs according to their efficacy and safety in patients with possible or probable AD, in order to provide additional evidence for clinical decision makers to choose the most appropriate treatment.
2 Methods
The study protocol was prospectively registered with the International Prospective Register for Systematic Reviews (CRD42023416047). This NMA was performed following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) [16] and Cochrane guidelines [17].
2.1 Literature Search
In this NMA, randomized controlled trials investigating the treatment of AD with anti-Aβ mAbs were identified through PubMed, Embase, Cochrane Library, and Web of Science with a search deadline of March 31, 2023. The reference lists of pertinent meta-analyses, reviews, pooled analyses, and included trials were carefully reviewed manually in order to identify any additional studies using all relevant combinations of the following search terms: ‘aducanumab’, ‘bapineuzumab’, ‘crenezumab’, ‘donanemab’, ‘gantenerumab’, ‘lecanemab’, ‘ponezumab’ ‘solanezumab’, and ‘Alzheimer Disease’, see Supplementary Material S1 in the electronic supplementary material (ESM) for the specific search strategy.
2.2 Selection Criteria
Upon initial screening, we included published randomized controlled trials (RCTs) that recruited patients diagnosed with mild cognitive impairment (MCI) or AD at any disease stage, in order to compare the efficacy and safety of all mAbs (aducanumab, bapineuzumab, crenezumab, donanemab, gantenerumab, lecanemab, ponezumab, and solanezumab) in this target population.
Reported outcomes included at least one of the following outcome indicators: Mini-Mental State Examination (MMSE), Clinical Dementia Rating scale–Sum of Boxes (CDR-SB), Alzheimer's Disease Assessment Scale–cognitive subscale (ADAS-cog), Alzheimer's Disease Cooperative Study–Activities of Daily Living (ADCS-ADL), standardized uptake value ratio of amyloid positron emission tomography (PET-SUVr), adverse events (AEs; subjects with any treatment-emergent AE [TEAE]); and amyloid-related imaging abnormalities characterized by edema and effusion (ARIA-E) or cerebral microhemorrhages (ARIA-H).
The exclusion criteria are (i) non-RCT research, such as meeting abstracts, agreements, letters, case reports, systematic reviews, and animal experiments; (ii) unavailability of full text or unavailability of usable data; and (iii) studies not including patients with AD. In the case of multiple publications based on the same RCT, we only included articles with the richest information and the largest sample source. Articles were included for review only after a consensus was reached among all three investigators.
2.3 Data Extraction
To ensure accuracy of the data collected, two evaluators independently extracted the relevant data from the retrieved literature, and any conflict was resolved through discussion between the first two evaluators or adjudication by a third author. The following data were extracted from each study: the author, publication year, clinical trial number, country, sample size, sex, age, severity of AD, MMSE baseline, ApoE allele carrying status, follow-up, and results. At the same time, we recorded the incidence of adverse events such as ARIA-E and ARIA-H, so as to evaluate the safety of anti-Aβ mAbs.
2.4 Risk of Bias
Literature quality assessment of the included studies was performed by two independent reviewers through the risk-of-bias analysis assessment tool provided by the Cochrane Handbook for Systematic Reviews of Interventions 5.4.0 [17], and any discrepancies were resolved by consensus. The evaluation included random sequence generation (selective bias), distribution concealment (selective bias), method of blinding implementers and participants (implementation bias), method of blinding result evaluation (detection bias), data result integrity (loss bias), selective report of research results (reporting bias) and other biases.
2.5 Data Analysis
The Bayesian network meta-data analysis was performed using R software (version 4.2.3) along with JAGS software and STATA software (version 15.0). We used mean differences (MD) from baseline with 95% confidence intervals (CI) to calculate the continuous data, such as the change from baseline of MMSE, as reported. As for the binary data, including AE, ARIA-E and ARIA-H, the odds ratio (OR) analysis of 95% CI is calculated. If there was no raw data (e.g., without standard deviations [SDs], only p values or ranges are reported), SDs and 95% CIs provided in the publication were calculated using established methods for estimation [18]. The use of a random effects model or fixed effects model depended on the statistical value of each result heterogeneity. If there was significant heterogeneity, I2 > 50, a random effects model was carried out, if I2 ≤ 50, a fixed effects model was carried out.
In order to verify the consistency between direct comparisons and indirect comparisons, we used the node splitting method through the design-by-treatment interaction model in the closed loop in the evidence network [19]. A p value higher than 0.05 (p > 0.05) indicates that there is no significant inconsistency between direct and indirect comparisons, on the contrary, the difference is statistically significant [20].
We calculated the surface under the cumulative ranking area (SUCRA) to rank the charts and the priority of the different interventions, in which the larger the SUCRA value, the better the efficacy or safety [21]. For the results of each trial, the potential retrieval bias was assessed using funnel plots to represent individual studies against some measure of each study's size or precision.
3 Results
3.1 Literature Search Results
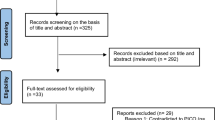
We found 1058 articles from PubMed, Embase, Cochrane Library and Web of Science, of which 400 studies were discarded because of duplication. We excluded a further 609 articles after reviewing the titles and abstracts. Finally, after reviewing the full text of 51 papers, 33 studies were found to fulfil the inclusion criteria. A PRISMA flowchart illustrating the specific screening process is shown in Fig. 1.
3.2 Eligible Studies and Patient Characteristics
A total of 33 randomized controlled trials [14, 22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53] were included, including 17,686 patients who received eight different mAbs (aducanumab, bapineuzumab, crenezumab, donanemab, gantenerumab, lecanemab, ponezumab, and solanezumab), and placebo was the most common comparison. Of the 33 included trials, 32 were two-group trials and one was a three-group clinical trial. See Table 1 for the baseline characteristics and design features of trials included in this NMA.
3.3 Risk of Bias
The overall risk included in the bias assessment of the studies is low to moderate. All the RCTs used the random number acquisition method, but only 19 papers provided a detailed description. Consideration should be given to the lack of information on the randomization process and allocation procedure, which were judged to have ambiguous risk of bias. One study was considered to have a high risk of bias in terms of "incomplete result data" [28]. Almost all studies are funded by large pharmaceutical companies. The risk-of-bias assessment of the included literature is shown in Fig. 2.
3.4 Network Diagram and Testing for Inconsistency
The network diagram of qualified comparisons of all result measurements is shown in Figs. 3 and 4, which includes nine interventions. RCTs are connected by a network diagram composed of points and lines. The dots represent different interventions, and their sizes represent the number of participants in each intervention. The straight lines indicate that there is direct comparison evidence between two interventions, and the thickness of the lines indicates the number of studies directly compared between the two interventions. In this study, there was a closed loop of indirect evidence and the node-split model was used to test the inconsistency. When using the node splitting method to evaluate MMSE and CDR-SB analysis, there is no obvious inconsistencies between direct evidence and indirect evidence (see Table 2 and Supplementary table S10 in the ESM).
Network graphs of randomized controlled trials comparing the efficacy of monoclonal antibodies (mAbs) in the treatment of Alzheimer’s disease: a Mini-Mental State Examination (MMSE), b Clinical Dementia Rating scale–Sum of Boxes (CDR-SB), c Alzheimer's Disease Assessment Scale–cognitive subscale (ADAS-cog), d Alzheimer's Disease Cooperative Study–Activities of Daily Living (ADCS-ADL), e standardized uptake value ratio of amyloid positron emission tomography (PET-SUVr)
4 Network Analysis of Outcomes
4.1 Change in MMSE
In the evaluation of the change in MMSE from baseline, 11 studies were included with a total of 9897 patients, with higher scores indicating better mental performance. There were seven interventions including aducanumab, bapineuzumab, crenezumab, donanemab, gantenerumab, placebo, and solanezumab. In terms of MMSE score, aducanumab was significantly more effective than placebo (MD 0.93, 95% CI 0.39–1.41), as shown in Supplementary table S2 (see ESM). No significant differences were found among the other meta-analysis pairs. The results of the ranking according to SUCRA values were that aducanumab (87.01%) ranked first in MMSE score change, followed by solanezumab (64.03%) and donanemab (63.80%) (Table 3). The cumulative probability showed that aducanumab was associated with the greatest benefit on MMSE score, as shown in Fig. 5a.
Rank of the cumulative probabilities for basic parameters: a Mini-Mental State Examination (MMSE), b Clinical Dementia Rating scale–Sum of Boxes (CDR-SB), c Alzheimer's Disease Assessment Scale–cognitive subscale (ADAS-cog), d Alzheimer's Disease Cooperative Study–Activities of Daily Living (ADCS-ADL), e standardized uptake value ratio of amyloid positron emission tomography (PET-SUVr)
4.2 Change in CDR-SB
In the evaluation of the CDR-SB, 14 RCTs were included with a total of 13,835 patients. The higher the score, the greater the damage. There were eight interventions: aducanumab, bapineuzumab, crenezumab, donanemab, gantenerumab, lecanemab, placebo and solanezumab. A total of 28 direct or indirect comparisons were generated by the NMA, which revealed that aducanumab (MD −0.58, 95% CI − 0.69 to − 0.48), lecanemab (MD − 0.34, 95% CI − 0.49 to − 0.18), and solanezumab (MD −0.34, 95% CI − 0.35 to − 0.33) had significantly lower CDR-SB scores compared with placebo, indicating a statistically significant difference (Supplementary table S3, see ESM). According to the SUCRA values, aducanumab (99.37%) had the highest likelihood of being the best treatment for decreasing CDR-SB score, followed by solanezumab (74.69%), whereas donanemab (5.38%) had the lowest probability (Table 3). The cumulative probability showed that aducanumab is related to the maximum benefit on CDR-SB, as shown in Fig. 5b.
4.3 Change in ADAS-cog
Results for change in ADAS-cog score were obtained from 14 RCTs covering eight treatment regimens and involving 12,252 participants, with higher scores indicating greater impairment. The eight interventions were aducanumab, bapineuzumab, crenezumab, donanemab, gantenerumab, lecanemab, placebo, and solanezumab. A total of 28 direct or indirect comparisons were generated by the NMA, which revealed that aducanumab (MD − 0.82, 95% CI − 1.35 to − 0.29), donanemab (MD − 1.78, 95% CI − 3.52 to − 0.01), gantenerumab (MD − 0.94, 95% CI − 1.35 to − 0.53), lecanemab (MD − 1.09, 95% CI − 1.69 to − 0.49) and solanezumab (MD − 0.9, 95% CI − 1.63 to − 0.17) were associated with a significant decrease in ADAS-cog compared with placebo (Supplementary table S4, see ESM). According to SUCRA curve analysis, donanemab (88.5%) was the most effective in terms of ADAS-cog score, and crenezumab (16.17%) was the least effective of the treatments examined (Table 3). The cumulative probability showed that donanemab was associated with the greatest benefit on ADAS-cog, as shown in Fig. 5c.
4.4 Change in ADCS-ADL
Seven of the included articles including a total of 8931 participants reported the change in ADCS-ADL score of six interventions: aducanumab, crenezumab, donanemab, lecanemab, placebo, and solanezumab, with a lower score meaning greater functional loss. A total of 15 direct or indirect comparisons were generated by the NMA, which revealed that aducanumab (MD 0.91, 95% CI 0.16–1.66), lecanemab (MD 2.00, 95% CI 0.39–3.59), and solanezumab (MD 1.35, 95% CI 1.31–1.39) were more effective than placebo according to ADAS-cog (Supplementary table S5, see ESM). The results of the ranking according to SUCRA values were that lecanemab (87.24%) exhibited the highest efficiency on ADCS-ADL, whereas crenezumab (9.83%) exhibited the lowest efficiency (Table 3). The cumulative probability showed that lecanemab was associated with the greatest benefit on ADCS-ADL, as shown in Fig. 5d.
4.5 Change in Amyloid PET SUVr
Nine articles providing data on the changes of amyloid protein PET SUVr from baseline, including a total of 1095 participants in eight interventions (aducanumab, bapineuzumab, crenezumab, donanemab, gantenerumab, lecanemab, placebo, and solanezumab). At first glance, one can conclude that aducanumab (MD − 0.17, 95% CI − 0.27 to − 0.07) and donanemab (MD − 0.37, 95% CI − 0.57 to − 0.17) were more effective than placebo. On the contrary, solanezumab (MD 0.55, 95% CI 0.14–0.95) was associated with a significant increase in the risk of abnormal imaging of amyloid PET (Supplementary table S6, see ESM). In the analysis of the curative effect of SUCRA, donanemab scored the highest in SUCRA (99.00%) in the amyloid protein PET SUVr, ranking first, while solanezumab (0.6%) performed worst (Table 3). The cumulative probability showed that donanemab was associated with the greatest benefit on amyloid PET SUVr, as shown in Fig. 5e.
4.6 Adverse Events (Subjects With Any TEAE)
Discontinuation rates for adverse events were reported in 19 trials, involving 12,867 patients with nine treatments (aducanumab, bapineuzumab, crenezumab, donanemab, gantenerumab, lecanemab, ponezumab, placebo, and solanezumab). Relative to the other types of mAbs, bapineuzumab (OR 1.3; 95% CI 1.05–1.6) and lecanemab (OR 2.16; 95% CI 1.83–2.55) can increase the significant risk of adverse events during treatment compared with placebo. However, regarding gantenerumab (OR 0.61; 95% CI 0.38–0.97), there was a significantly reduced risk for adverse events (Supplementary table S7, see ESM) and lecanemab (OR 2.16; 95% CI 1.83–2.55) exhibited the lowest safety (Table 3). The results of the ranking according to SUCRA values were that gantenerumab (89.12%) exhibited the highest safety, followed by ponezumab (84.91%), solanezumab (56.43%), placebo (56.02%), aducanumab (55.34%), donanemab (47.85%), crenezumab (36.65%), bapineuzumab (22.89%), and lecanemab (0.79%) (Table 3). The cumulative probability showed that gantenerumab was associated with minimal adverse effects, as shown in Fig. 6a.
4.7 ARIA-E
In total, 16 studies with 18,300 patients were included in the analysis of the events with ARIA-E, involving eight treatments—aducanumab, bapineuzumab, crenezumab, donanemab, gantenerumab, lecanemab, placebo, and solanezumab. The incidence of ARIA-E associated with mAbs against Aβ was significantly higher in bapineuzumab (OR 30.29; 95% CI 19.88–48.89), aducanumab (OR 15.09; 95% CI 11.51–20.20), gantenerumab (OR 14.78; 95% CI 6.36–43.88), donanemab (OR 12.26; 95% CI 6.58–25.19), and lecanemab (OR 8.32; 95% CI 5.50–13.15) compared with placebo in the treatment of AD (Supplementary table S8, see ESM). According to SUCRA curve analysis, placebo (95.75%) exhibited the lowest risk of ARIA-E, followed by solanezumab (82.67%), and bapineuzumab (2.92%) exhibited the highest risk of ARIA-E (Table 3). The cumulative probability showed that solanezumab was associated with the least ARIA-E adverse effects except placebo, as shown in Fig. 6b.
4.8 ARIA-H
Fourteen articles were included in the analysis for the events of ARIA-H, involving 10,565 patients and eight interventions—aducanumab, crenezumab, donanemab, gantenerumab, lecanemab, ponezumab, placebo, and solanezumab. The NMA revealed that a higher risk of ARIA-H in aducanumab (OR 4.44; 95% CI 3.75–5.28), donanemab (OR 5.77; 95% CI 3.39–10.26), gantenerumab (OR 1.81; 95% CI 1.32–2.49), and lecanemab (OR 2.12; 95% CI 1.60–2.83) was statistically different compared with placebo in the treatment of AD (Supplementary table S9, see ESM). According to SUCRA curve analysis, solanezumab (89.26%) exhibited the lowest risk of ARIA-H, followed by placebo (80.38%), and donanemab (4.93%) exhibited the highest risk of ARIA-H (Table 3). The cumulative probability showed that solanezumab was associated with the lowest ARIA-H adverse effects, as shown in Fig. 6c.
4.9 Publication Bias
Funnel plots are used to show the publication bias of the evaluation scales, including MMSE, CDR-SB, ADAS-cog, ADCS-ADL, the change of amyloid PET-SUVr, adverse events, ARIA-E, and ARIA-H. In this study, NMA may have a potential publication bias, as shown in Fig. 7.
Funnel plots for the efficacy and safety outcomes: a Mini-Mental State Examination (MMSE), b Clinical Dementia Rating scale–Sum of Boxes (CDR-SB), c Alzheimer's Disease Assessment Scale–cognitive subscale (ADAS-cog), d Alzheimer's Disease Cooperative Study–Activities of Daily Living (ADCS-ADL), e standardized uptake value ratio of amyloid positron emission tomography (PET-SUVr), f adverse events (AE), and g amyloid-related imaging abnormalities characterized by edema and effusion (ARIA-E) and h cerebral microhemorrhages (ARIA-H)
5 Discussion
A total of 21,087 subjects from 33 studies were included in the network analysis in which the efficacy and safety of mAbs against Aβ in the treatment of mild or moderate AD were investigated. The results of a pairwise meta-analysis found that aducanumab exerted better efficacy compared with other interventions in improvements of MMSE and CDR-SB scores, with SUCRA values of 87.01% and 99.37%, respectively. Donanemab, with respective SUCRA values of 88.50% and 99.00%, showed superior performance over other therapies in improving ADAS-cog scores and amyloid PET-SUVr. Additionally, lecanemab, with a SUCRA value of 87.24%, may be the most beneficial option for slowing down the decline in ADCS-ADL scores. However, our findings also highlight the increased risk of adverse events associated with these treatments, notably ARIA. Apart from solanezumab, all other mAb therapies increased the risk of ARIA. Meanwhile, compared with the placebo group, the application of gantenerumab, ponezumab, and solanezumab did not statistically increase the risk of AEs. In interpreting SUCRA rankings, it's crucial to consider their statistical uncertainties and dependence on the quality of included studies; these rankings are relative and sensitive to methodological variations in NMA.
Considering the efficacy profile, we showed the changes in MMSE, ADAS-cog, CDR-SB, ADCS-ADL, and amyloid PET SUVr. Although the NMA was not ideal given the lack of head-to-head studies among the interventions (i.e., only against placebo; a star-shaped network), except for a three-arm experiment, interesting results were found. According to the ranking probabilities, aducanumab performed most effectively, followed by solanezumab, donanemab, and bapineuzumab, and gantenerumab was superior to crenezumab regarding MMSE scores. When we chose the improvement of CDR-SB score as the efficacy outcome, aducanumab was still ranked ahead of other anti-Aβ mAbs. In 2017, a previous meta-analysis of a total of 17 clinical studies revealed that aducanumab exerted the most beneficial effects, followed by solanezumab, but bapineuzumab, crenezumab, and gantenerumab had no effect on the improvement of clinical outcomes [54]. A previous NMA included five interventions, involving 5141 patients; it was reported that aducanumab and solanezumab were significantly superior to placebo in MMSE [55]. These two NMAs reported similar comparative results regarding aducanumab and solanezumab. Aducanumab is a whole human IgG 1 mAb isolated from elderly donors with cognitive health as the first disease-modifying therapy (DMT) for AD [56]. Aducanumab binds to amyloid plaques and high molecular weight amyloid oligomers, and it also stimulates microglia to clear Aβ in a dose- and time-dependent manner accompanied by a slowing of cognitive impairment based on MMSE and CDR-SB [57]. The original statement discusses the approval of aducanumab and lecanemab based on their ability to reduce Aβ rather than their impact on cognitive functions. Previous analysis revealed that, despite statistically significant results, the effect of these medications on cognitive performance is relatively minor and lacks clinical significance [54, 58, 59]. In addition, a study of 196 patients with AD showed a similar result, that is, aducanumab reduced Aβ plaques, and it slowly decreased clinical indicators of patients [60]. Similarly, when assessed by ADAS-Cog, another cognitive subscale, the cognitive improvement with donanemab was more obvious than with other mAbs. ADAS-Cog was designed as a standard tool in key clinical trials related to attention, concentration, nonverbal memory and practice, which is used to evaluate the cognitive aspects of AD [61, 62]. Only a phase II trial of donazumab was involved in our NMA, and Mintun et al. considered no definitive conclusions could be drawn regarding the difference between groups in the change in ADAS-Cog score [31]. At the same time, lecanemab, aducanumab, and solanezumab also improved ADAS-Cog scores, which had been confirmed in another meta-analysis [63, 64]. Like most measures of functioning used in AD, ADCS-ADL was developed as an interview-based, informant-reported measure of level of independence in specific tasks to assess basic as well as instrumental living skills [65]. In our study, lecanemab achieved better results on ADCS-ADL from the perspective of SUCRA rankings, followed by solanezumab, and donanemab was superior to aducanumab. A recent systematic review and meta-analysis of anti-amyloid-β mAbs in phase III clinical trials indicated that aducanumab and lecanemab significantly improved ADCS-ADL-MCI scores, consistent with our findings [66]. Lecanemab is a humanized IgG1 of the mouse mAb158, defined as the second FDA-approved anti-amyloid drug to treat AD after aducanumab, and supposedly the only one that shows a statistically significant clinical benefit [47, 67]. A multicenter, double-blind, phase III trial showed that after 18 months, according to the ADCS-MCI-ADL evaluation, compared with placebo, lecanemab delayed the progress of AD by 37%, and the gap was increasing [34]. The amyloid cascade hypothesis postulates that the amyloid precursor protein (APP) is broken down to form Aβ, and the imbalance between Aβ generation and Aβ clearance results in an abnormal accumulation of amyloid-β plaques in various regions of the brain [68, 69]. As for the data of amyloid PET-SUVr, results from our NMA showed donanemab had the highest SUCRA value in reducing the amyloid burden, followed by gantenerumab and aducanumab; other mAbs were inferior to placebo. Among these antibody molecules, donanemab was very effective in reducing florbetapir amyloid PET signal and amyloid load but depended on percent change [30, 31]. Patients with higher levels of Aβ at baseline experienced a more obvious reduction, while those with lower initial Aβ levels might achieve complete clearance of amyloid. However, according to the results of a previous meta-analysis, cognitive improvement was not found across the donanemab phase I/II trials, measured by MMSE, ADAS-Cog, and CDR-SB scores [70]. Research indicated that mAbs used in AD treatments, such as aducanumab, gantenerumab, and solanezumab, have different specificities for various forms of amyloid-β. They can influence the effectiveness at various stages or under varying conditions of amyloid-β accumulation, which might explain the variations in their therapeutic outcomes [71, 72]. Overall, the efficacy of these mAbs in improving cognitive function among patients with mild or moderate AD has been observed. At the same time, their efficacy showed considerable variation, with each mAb exhibiting a distinct profile of efficacy and associated risks for potential adverse effects.
In a safety assessment, amyloid-related imaging abnormalities (ARIA), ARIA-E, and ARIA-H, were selected for comparison between immunotherapy and placebo. Drawing on the findings of recent studies, the risk-to-benefit ratio of anti-Aβ mAbs in Alzheimer's disease continues to be unfavorable [58, 63, 73]. Several articles suggest that amyloid-β, as a barrier protein, has a unique sealant which can prevent immune-mediated brain tissue damage, and thus assume the main anti-pathogenic function. Therefore, the risk of amyloid-β deficiency can lead to a series of complications, such as downstream bleeding and edema, which should be paid attention to [74]. In addition, the risk of ARIA increased with the dose of the antibodies, suggesting a relationship between the efficacy of amyloid clearance and the imaging abnormality. In the current NMA, compared with the placebo group, most of the mAbs significantly increase the risk of ARIA-E and ARIA-H. According to the ranking results, the best alternatives were gantenerumab and solanezumab, which showed the lowest rates of adverse events (subjects with any TEAE) and ARIAs. Jeremic et al. suggested that solanezumab was the only drug that did not increase ARIA-E risk [66]. Recent studies revealed that the lack of specific targeting Aβ oligomers or blocking their toxicity is a serious defect in immune therapeutic approaches: solanezumab, ponezumab, and lecanemab are targeted to soluble Aβ; bapineuzumab and crenezumab recognize all three forms of Aβ; and aducanumab, donanemab, and gantenerumab bind AβOs and fibrils. In SCarlet RoAD, at doses of 105 and 225 mg every 4 weeks, gantenerumab was chosen to minimize the risk of any adverse events incidence [40]. However, compared with the positive results of the phase Ib trial for aducanumab [24], it shows that the dose of gantenerumab may be too low to obtain substantial clinical benefit. Because anti-amyloid treatments have been associated with ARIA with edema or hemorrhage and raised some safety concerns [75], we compared immunotherapies versus placebo. Solanezumab that also recognizes soluble, monomeric amyloid has showed the least risk of ARIA-E and ARIA-H incidence among immunotherapies. The incidence of ARIAs was 0.9% and 0.4% for solanezumab and placebo, respectively, indicating that solanezumab did not induce ARIAs, thus displaying its great tolerability and safety [38]. Lecanemab ranked lowest in terms of AEs in our study, but in clinical trials of lecanemab, the incidence of brain swelling (referred to as ARIA-E) was 12.6%, whereas in the clinical trials of donanemab, this rate was 24% [25, 34].
Our study, the latest and largest evidence-based NMA, for the first time evaluated early Alzheimer's disease subjects treated with anti-Aβ mAbs. All randomized controlled trials in our study are considered to be of high quality. However, our research has some limitations. First of all, the number of head-to-head studies of mAbs is extremely limited, which might lead to mostly indirect estimates for all the comparisons in our NMA and affect the precision of the results. Secondly, we compare the changes in cognitive function mainly through clinical measurement scales. Each research variable is different in key biomarkers, so it is impossible to compare. Thirdly, we counted all the overall outcomes of the results rather than divide the evidence base by dose and mode of each drug administration, and the follow-up time ranged from 6 months to 4 years. Finally, meta-analysis data were sourced only from published scientific literature, and some negative results and non-statistical data are difficult to publish. Therefore, there is publication bias. In general, these results show that these anti-Aβ mAbs could prevent cognitive decline of patients with early AD but the curative effect on the symptoms of functional, behavioral, and systemic change in patients with severe AD is questionable. In fact, some results are difficult to evaluate today because the tools for manipulating non-cognitive results are immature [76]. We only paid attention to anti-Aβ mAbs, and more directions need to be considered in the future treatment of Alzheimer's disease, including gene therapy, immunotherapy, peptide dynamics, metal creators, and probiotics. According to the NMA of mAbs in existing research, its clinical benefits in the treatment of AD have been observed, but whether it is really worth popularizing these in clinic needs further discussion.
6 Conclusion
NMA provides effective evidence for evaluating the efficacy and safety of anti-Aβ mAbs in the treatment of AD. From the perspective of SUCRA rankings, aducanumab may be the best choice for MMSE and CDR-SB cognitive improvement. Donanemab is thought to be potentially more effective than other interventions in ADAS-cog and amyloid PET-SUVr, and lecanemab may be the most beneficial to ADCS-ADL. In terms of safety, immunotherapies significantly increase the risks of adverse events and ARIA; gantenerumab and solanezumab might be safer than other mAbs included. Due to statistical uncertainties in SUCRA rankings, specific analysis is needed when applying them in clinical practice.
References
Serrano-Pozo A, Frosch MP, Masliah E, Hyman BT. Neuropathological alterations in Alzheimer disease. Cold Spring Harb Perspect Med. 2011;1(1): a006189.
Neary D, Snowden JS, Gustafson L, Passant U, Stuss D, Black S, et al. Frontotemporal lobar degeneration: a consensus on clinical diagnostic criteria. Neurology. 1998;51(6):1546–54.
Dementia 2023 [updated 15 March 2023]. https://www.who.int/news-room/fact-sheets/detail/dementia.
Viscogliosi G, Marigliano V. Alzheimer’s disease: how far have we progressed? Lessons learned from diabetes mellitus, metabolic syndrome, and inflammation. J Am Geriatr Soc. 2013;61(5):845–6.
Judge D, Roberts J, Khandker RK, Ambegaonkar B, Black CM. Physician practice patterns associated with diagnostic evaluation of patients with suspected mild cognitive impairment and Alzheimer’s disease. Int J Alzheimer’s Dis. 2019;2019:4942562.
Briggs R, Kennelly SP, O’Neill D. Drug treatments in Alzheimer’s disease. Clin Med (Lond). 2016;16(3):247–53.
2022 Alzheimer's disease facts and figures. Alzheimer’s Dementia. 2022;18(4):700–89.
Ferreira-Vieira TH, Guimaraes IM, Silva FR, Ribeiro FM. Alzheimer’s disease: targeting the cholinergic system. Curr Neuropharmacol. 2016;14(1):101–15.
Hardy J, Selkoe DJ. The amyloid hypothesis of Alzheimer’s disease: progress and problems on the road to therapeutics. Science. 2002;297(5580):353–6.
Hardy JA, Higgins GA. Alzheimer’s disease: the amyloid cascade hypothesis. Science. 1992;256(5054):184–5.
Behl T, Kaur I, Fratila O, Brata R, Bungau S. Exploring the potential of therapeutic agents targeted towards mitigating the events associated with amyloid-β cascade in Alzheimer’s disease. Int J Mol Sci. 2020;21(20):7443.
Grossberg GT, Tong G, Burke AD, Tariot PN. Present algorithms and future treatments for Alzheimer’s disease. J Alzheimer’s Dis JAD. 2019;67(4):1157–71.
Pinheiro L, Faustino C. therapeutic strategies targeting amyloid-β in Alzheimer’s disease. Curr Alzheimer Res. 2019;16(5):418–52.
Salloway S, Sperling R, Gilman S, Fox NC, Blennow K, Raskind M, et al. A phase 2 multiple ascending dose trial of bapineuzumab in mild to moderate Alzheimer disease. Neurology. 2009;73(24):2061–70.
Lacorte E, Ancidoni A, Zaccaria V, Remoli G, Tariciotti L, Bellomo G, et al. Safety and efficacy of monoclonal antibodies for Alzheimer’s disease: a systematic review and meta-analysis of published and unpublished clinical trials. J Alzheimers Dis. 2022;87(1):101–29.
Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6(7): e1000097.
Cumpston M, Li T, Page MJ, Chandler J, Welch VA, Higgins JP, et al. Updated guidance for trusted systematic reviews: a new edition of the Cochrane Handbook for Systematic Reviews of Interventions. Cochrane Database Syst Rev. 2019;10:Ed000142.
Wan X, Wang W, Liu J, Tong T. Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med Res Methodol. 2014;14:135.
Higgins JP, Jackson D, Barrett JK, Lu G, Ades AE, White IR. Consistency and inconsistency in network meta-analysis: concepts and models for multi-arm studies. Res Synth Methods. 2012;3(2):98–110.
Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002;21(11):1539–58.
Shim S, Yoon BH, Shin IS, Bae JM. Network meta-analysis: application and practice using Stata. Epidemiol Health. 2017;39: e2017047.
Cummings JL, Cohen S, van Dyck CH, Brody M, Curtis C, Cho W, et al. ABBY: a phase 2 randomized trial of crenezumab in mild to moderate Alzheimer disease. Neurology. 2018;90(21):e1889–97.
Salloway S, Honigberg LA, Cho W, Ward M, Friesenhahn M, Brunstein F, et al. Amyloid positron emission tomography and cerebrospinal fluid results from a crenezumab anti-amyloid-beta antibody double-blind, placebo-controlled, randomized phase II study in mild-to-moderate Alzheimer’s disease (BLAZE). Alzheimer’s Res Ther. 2018;10(1):96.
Sevigny J, Chiao P, Bussière T, Weinreb PH, Williams L, Maier M, et al. The antibody aducanumab reduces Aβ plaques in Alzheimer’s disease. Nature. 2016;537(7618):50–6.
Shcherbinin S, Evans CD, Lu M, Andersen SW, Pontecorvo MJ, Willis BA, et al. Association of amyloid reduction after donanemab treatment with tau pathology and clinical outcomes: the TRAILBLAZER-ALZ randomized clinical trial. JAMA Neurol. 2022;79(10):1015–24.
Vandenberghe R, Rinne JO, Boada M, Katayama S, Scheltens P, Vellas B, et al. Bapineuzumab for mild to moderate Alzheimer’s disease in two global, randomized, phase 3 trials. Alzheimer’s Res Ther. 2016;8(1):18.
Rinne JO, Brooks DJ, Rossor MN, Fox NC, Bullock R, Klunk WE, et al. C-11-PiB PET assessment of change in fibrillar amyloid-beta load in patients with Alzheimer’s disease treated with bapineuzumab: a phase 2, double-blind, placebo-controlled, ascending-dose study. Lancet Neurol. 2010;9(4):363–72.
Brashear HR, Ketter N, Bogert J, Di J, Salloway SP, Sperling R. Clinical evaluation of amyloid-related imaging abnormalities in bapineuzumab phase III studies. J Alzheimer’s Dis JAD. 2018;66(4):1409–24.
Nakamura T, Fujikoshi S, Funai J, Matsumura T. Disease-modifying effect of solanezumab evaluated by delayed-start analysis in a Japanese subpopulation with mild Alzheimer’s disease. Neurol Clin Neurosci. 2017;5(5):141–9.
Lowe SL, Duggan Evans C, Shcherbinin S, Cheng YJ, Willis BA, Gueorguieva I, et al. Donanemab (LY3002813) phase 1b study in Alzheimer’s disease: rapid and sustained reduction of brain amyloid measured by florbetapir F18 imaging. J Prev Alzheimer’s Dis. 2021;8(4):414–24.
Mintun MA, Lo AC, Duggan Evans C, Wessels AM, Ardayfio PA, Andersen SW, et al. Donanemab in early alzheimer’s Disease. N Engl J Med. 2021;384(18):1691–704.
Ostrowitzki S, Bittner T, Sink KM, Mackey H, Rabe C, Honig LS, et al. Evaluating the safety and efficacy of crenezumab vs placebo in adults with early Alzheimer disease two phase 3 randomized placebo-controlled trials. JAMA Neurol. 2022;79(11):1113–21.
Ferrero J, Williams L, Stella H, Leitermann K, Mikulskis A, O’Gorman J, et al. First-in-human, double-blind, placebo-controlled, single-dose escalation study of aducanumab (BIIB037) in mild-to-moderate Alzheimer’s disease. Alzheimer’s Dementia (New York, N Y). 2016;2(3):169–76.
van Dyck CH, Swanson CJ, Aisen P, Bateman RJ, Chen C, Gee M, et al. Lecanemab in early Alzheimer’s disease. N Engl J Med. 2023;388(1):9–21.
Ostrowitzki S, Deptula D, Thurfjell L, Barkhof F, Bohrmann B, Brooks DJ, et al. Mechanism of amyloid removal in patients with Alzheimer disease treated with gantenerumab. Arch Neurol. 2012;69(2):198–207.
Landen JW, Cohen S, Billing CB Jr, Cronenberger C, Styren S, Burstein AH, et al. Multiple-dose ponezumab for mild-to-moderate Alzheimer’s disease: safety and efficacy. Alzheimer’s Dementia (New York, N Y). 2017;3(3):339–47.
Lu M, Brashear HR. Pharmacokinetics, pharmacodynamics, and safety of subcutaneous bapineuzumab: a single-ascending-dose study in patients with mild to moderate Alzheimer disease. Clin Pharmacol Drug Dev. 2019;8(3):326–35.
Doody RS, Thomas RG, Farlow M, Iwatsubo T, Vellas B, Joffe S, et al. Phase 3 trials of solanezumab for mild-to-moderate Alzheimer’s disease. N Engl J Med. 2014;370(4):311–21.
Brody M, Liu E, Di J, Lu M, Margolin RA, Werth JL, et al. A phase II, randomized, double-blind, placebo-controlled study of safety, pharmacokinetics, and biomarker results of subcutaneous bapineuzumab in patients with mild to moderate Alzheimer’s disease. J Alzheimer’s Dis JAD. 2016;54(4):1509–19.
Ostrowitzki S, Lasser RA, Dorflinger E, Scheltens P, Barkhof F, Nikolcheva T, et al. A phase III randomized trial of gantenerumab in prodromal Alzheimer’s disease. Alzheimer’s Res Ther. 2017;9(1):95.
Landen JW, Andreasen N, Cronenberger CL, Schwartz PF, Börjesson-Hanson A, Östlund H, et al. Ponezumab in mild-to-moderate Alzheimer’s disease: randomized phase II PET-PIB study. Alzheimer’s Dementia (New York, N Y). 2017;3(3):393–401.
Swanson CJ, Zhang Y, Dhadda S, Wang J, Kaplow J, Lai RYK, et al. A randomized, double-blind, phase 2b proof-of-concept clinical trial in early Alzheimer’s disease with lecanemab, an anti-A beta protofibril antibody. Alzheimers Res Ther. 2021;13(1):80.
Farlow M, Arnold SE, van Dyck CH, Aisen PS, Snider BJ, Porsteinsson AP, et al. Safety and biomarker effects of solanezumab in patients with Alzheimer’s disease. Alzheimers Dement. 2012;8(4):261–71.
Arai H, Umemura K, Ichimiya Y, Iseki E, Eto K, Miyakawa K, et al. Safety and pharmacokinetics of bapineuzumab in a single ascending-dose study in Japanese patients with mild to moderate Alzheimer’s disease. Geriatr Gerontol Int. 2016;16(5):644–50.
Miyoshi I, Fujimoto Y, Yamada M, Abe S, Zhao Q, Cronenberger C, et al. Safety and pharmacokinetics of PF-04360365 following a single-dose intravenous infusion in Japanese subjects with mild-to-moderate Alzheimer’s disease: a multicenter, randomized, double-blind, placebo-controlled, dose-escalation study. Int J Clin Pharmacol Ther. 2013;51(12):911–23.
Landen JW, Zhao Q, Cohen S, Borrie M, Woodward M, Billing CB, et al. Safety and pharmacology of a single intravenous dose of ponezumab in subjects with mild-to-moderate Alzheimer disease: a phase I, randomized, placebo-controlled, double-blind, dose-escalation study. Clin Neuropharmacol. 2013;36(1):14–23.
Logovinsky V, Satlin A, Lai R, Swanson C, Kaplow J, Osswald G, et al. Safety and tolerability of BAN2401—a clinical study in Alzheimer's disease with a protofibril selective Abeta antibody. Alzheimer's Res Ther. 2016;8(1) (no pagination).
Guthrie H, Honig LS, Lin H, Sink KM, Blondeau K, Quartino A, et al. Safety, tolerability, and pharmacokinetics of crenezumab in patients with mild-to-moderate Alzheimer’s disease treated with escalating doses for up to 133 weeks. J Alzheimer’s Dis JAD. 2020;76(3):967–79.
Black RS, Sperling RA, Safirstein B, Motter RN, Pallay A, Nichols A, et al. A single ascending dose study of bapineuzumab in patients with Alzheimer disease. Alzheimer Dis Assoc Disord. 2010;24(2):198–203.
Salloway S, Farlow M, McDade E, Clifford DB, Wang G, Llibre-Guerra JJ, et al. A trial of gantenerumab or solanezumab in dominantly inherited Alzheimer’s disease. Nat Med. 2021;27(7):1187–96.
Honig LS, Vellas B, Woodward M, Boada M, Bullock R, Borrie M, et al. Trial of solanezumab for mild dementia due to Alzheimer’s disease. N Engl J Med. 2018;378(4):321–30.
Salloway S, Sperling R, Fox NC, Blennow K, Klunk W, Raskind M, et al. Two phase 3 trials of bapineuzumab in mild-to-moderate Alzheimer’s disease. N Engl J Med. 2014;370(4):322–33.
Haeberlein SB, Aisen PS, Barkhof F, Chalkias S, Chen T, Cohen S, et al. Two randomized phase 3 studies of aducanumab in early Alzheimer’s disease. J Prev Alzheimer’s Dis. 2022;9(2):197–210.
Avgerinos KI, Ferrucci L, Kapogiannis D. Effects of monoclonal antibodies against amyloid-β on clinical and biomarker outcomes and adverse event risks: a systematic review and meta-analysis of phase III RCTs in Alzheimer’s disease. Ageing Res Rev. 2021;68: 101339.
Lombardo I, Ramaswamy G, Fogel I, Mo Y, Friedhoff L, Bruinsma B. A summary of baseline efficacy characteristics from the mindset study: a global phase 3 study of intepirdine (RVT-101) in subjects with mild to moderate Alzheimer’s disease. Alzheimers Dement. 2017;13(7):P936.
Dhillon S. Aducanumab: first approval. Drugs. 2021;81(12):1437–43.
Linse S, Scheidt T, Bernfur K, Vendruscolo M, Dobson CM, Cohen SIA, et al. Kinetic fingerprints differentiate the mechanisms of action of anti-Aβ antibodies. Nat Struct Mol Biol. 2020;27(12):1125–33.
Imbimbo BP, Ippati S, Watling M, Imbimbo C. Role of monomeric amyloid-β in cognitive performance in Alzheimer’s disease: insights from clinical trials with secretase inhibitors and monoclonal antibodies. Pharmacol Res. 2023;187: 106631.
Richard E, den Brok MGHE, van Gool WA. Bayes analysis supports null hypothesis of anti-amyloid beta therapy in Alzheimer’s disease. Alzheimers Dementia. 2021;17(6):1051–5.
Haeberlein SB, O’Gorman J, Chiao P, Bussière T, von Rosenstiel P, Tian Y, et al. Clinical development of aducanumab, an anti-Aβ human monoclonal antibody being investigated for the treatment of early Alzheimer’s disease. J Prev Alzheimer’s Dis. 2017;4(4):255–63.
Mohs RC, Knopman D, Petersen RC, Ferris SH, Ernesto C, Grundman M, et al. Development of cognitive instruments for use in clinical trials of antidementia drugs: additions to the Alzheimer’s Disease Assessment Scale that broaden its scope. The Alzheimer’s Disease Cooperative Study. Alzheimer Dis Assoc Disord. 1997;11(Suppl 2):S13-21.
Doraiswamy PM, Kaiser L, Bieber F, Garman RL. The Alzheimer’s Disease Assessment Scale: evaluation of psychometric properties and patterns of cognitive decline in multicenter clinical trials of mild to moderate Alzheimer’s disease. Alzheimer Dis Assoc Disord. 2001;15(4):174–83.
Avgerinos KI, Ferrucci L, Kapogiannis D. Effects of monoclonal antibodies against amyloid-beta on clinical and biomarker outcomes and adverse event risks: a systematic review and meta-analysis of phase III RCTs in Alzheimer’s disease. Ageing Res Rev. 2021;68: 101339.
Mo JJ, Li JY, Yang Z, Liu Z, Feng JS. Efficacy and safety of anti-amyloid-β immunotherapy for Alzheimer’s disease: a systematic review and network meta-analysis. Ann Clin Transl Neurol. 2017;4(12):931–42.
Galasko D, Bennett D, Sano M, Ernesto C, Thomas R, Grundman M, et al. An inventory to assess activities of daily living for clinical trials in Alzheimer’s disease. The Alzheimer’s Disease Cooperative Study. Alzheimer Dis Assoc Disord. 1997;11(Suppl 2):S33–9.
Jeremic D, Navarro-López JD, Jiménez-Díaz L. Efficacy and safety of anti-amyloid-β monoclonal antibodies in current Alzheimer’s disease phase III clinical trials: a systematic review and interactive web app-based meta-analysis. Ageing Res Rev. 2023;90: 102012.
Barthel H. Amyloid imaging-based food and drug administration approval of lecanemab to treat Alzheimer disease-what lasts long finally becomes good? J Nucl Med. 2023;64(4):503–4.
Fulop T, Witkowski JM, Bourgade K, Khalil A, Zerif E, Larbi A, et al. Can an infection hypothesis explain the beta amyloid hypothesis of Alzheimer’s disease? Front Aging Neurosci. 2018;10:224.
Multhaup G, Huber O, Buée L, Galas MC. Amyloid precursor protein (APP) metabolites APP intracellular fragment (AICD), Aβ42, and Tau in nuclear roles. J Biol Chem. 2015;290(39):23515–22.
Rashad A, Rasool A, Shaheryar M, Sarfraz A, Sarfraz Z, Robles-Velasco K, et al. Donanemab for Alzheimer’s disease: a systematic review of clinical trials. Healthcare (Basel, Switzerland). 2022;11(1):32.
Rabinovici GD, La Joie R. Amyloid-targeting monoclonal antibodies for Alzheimer disease. JAMA. 2023;330(6):507–9.
Salemme S, Ancidoni A, Locuratolo N, Piscopo P, Lacorte E, Canevelli M, et al. Advances in amyloid-targeting monoclonal antibodies for Alzheimer’s disease: clinical and public health issues. Expert Rev Neurother. 2023;23(12):1113–29.
Villain N, Planche V, Levy R. High-clearance anti-amyloid immunotherapies in Alzheimer’s disease. Part 1: meta-analysis and review of efficacy and safety data, and medico-economical aspects. Revue Neurol. 2022;178(10):1011–30.
Atwood CS, Perry G. Playing Russian roulette with Alzheimer’s disease patients: do the cognitive benefits of lecanemab outweigh the risk of edema, stroke and encephalitis? J Alzheimer’s Dis JAD. 2023;92(3):799–801.
Sperling RA, Jack CR Jr, Black SE, Frosch MP, Greenberg SM, Hyman BT, et al. Amyloid-related imaging abnormalities in amyloid-modifying therapeutic trials: recommendations from the Alzheimer’s Association Research Roundtable Workgroup. Alzheimer’s Dementia. 2011;7(4):367–85.
Cotta Ramusino M, Perini G, Altomare D, Barbarino P, Weidner W, Salvini Porro G, et al. Outcomes of clinical utility in amyloid-PET studies: state of art and future perspectives. Eur J Nucl Med Mol Imaging. 2021;48(7):2157–68.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was supported by the Shenyang Science and Technology Program [grant number 20-205-4-090].
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Data availability statement
The original contributions presented in the study are included in the article/supplementary material; further inquiries can be directed to the corresponding author.
Author contributions
All authors contributed to the study conception and design. YQ: subject design and writing—original draft. YC, MY and JG: investigation. YM: conceptualization, supervision, and funding acquisition—reviewing and editing. All authors contributed to the article and approved the submitted version.
Consent for publication
Not applicable.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Code availability
Not applicable.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Qiao, Y., Gu, J., Yu, M. et al. Comparative Efficacy and Safety of Monoclonal Antibodies for Cognitive Decline in Patients with Alzheimer’s Disease: A Systematic Review and Network Meta-Analysis. CNS Drugs 38, 169–192 (2024). https://doi.org/10.1007/s40263-024-01067-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40263-024-01067-2