Abstract
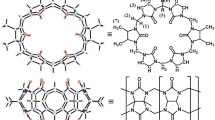
In this paper, 1H NMR spectroscopy, isothermal titration calorimetry, X-ray crystallography and other characterization methods were used to investigate the interaction modes of tetramethyl cucurbit[6]uril(TMeQ[6]) and three benzimidazole derivatives in an aqueous solution-solid state. The results showed that the aromatic ring moieties in the three derivatives all entered the cavity of TMeQ[6] and their substituents were located at the port of TMeQ[6], forming 1:1 host-guest inclusion complexes. The crystal structures showed that the aromatic part of the benzimidazole derivatives interacted with the cavity of TMeQ[6] via hydrogen bond interactions and the N atoms on the benzimidazole ring formed hydrogen bonds with the carbonyl oxygen of TMeQ[6]. The ion-dipole interactions between [ZnCl4]2− and TMeQ[6] formed supramolecular self-assembly entities.

Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Freeman W. A., Mock W. L., Shih N. Y., J. Am. Chem. Soc., 1981, 103, 7367
Kim J., Jung I. S., Kim S. Y., Lee E., Kang J. K., Sakamoto S., Yamaguchi K., Kim K., J. Am. Chem. Soc., 2000, 122, 540
Liu S. M., Zavalij P. Y., Isaacs L., J. Am. Chem. Soc., 2005, 127, 16798
Li Q., Qiu S. C., Zhang J., Chen K., Huang Y., Xiao X., Zhang Y. J., Li F., Zhang Y. Q., Xue S. F., Zhu Q. J., Tao Z., Lindoy L. F., Wei G., Org. Lett., 2016, 18, 4020
Cheng X. J., Liang L. L., Chen K., Ji N. N., Xiao X., Zhang J. X., Zhang Y. Q., Xue S. F., Zhu Q. J., Ni X. L., Tao Z., Angew. Chem. Int. Ed., 2013, 52, 7252
Chen K., Hua Z. Y., Zhao J. L., Redshaw C., Tao Z., Inorg. Chem. Front., 2022, 9, 2753
Yang D., Liu M., Xiao X., Tao Z., Redshaw C., Coordin. Chem. Rev., 2021, 434, 213733
Chen K., Ge W. W., Xu J., Zhang S. M., Li Y. W., Hua Z. Y., Tao Z., J. Mol. Struct., 2019, 1181, 220
Shen F. F., Zhao J. L., Chen K., Hua Z. Y., Chen M. D., Zhang Y. Q., Zhu Q. J., Tao Z., CrystEngComm, 2017, 19, 2464
Ni X. L., Xue S. F., Tao Z., Zhu Q. J., Lindoy L. F., Wei G., Coordin. Chem. Rev., 2015, 287, 89
Wu H., Wang Y., Jones L. O., Liu W. Q., Zhang L., Song B., Chen X. Y., Stern C. L., Schatz G. C., Stoddart J. F., Angew. Chem. Int. Ed., 2021, 60, 17587
Wu H., Jones L. O., Wang Y., Shen D. K., Liu Z. C., Zhang L., Cai K., Jiao Y., Stern C. L., Schatz G. C., Stoddart J. F., ACS Appl. Mater. Interfaces, 2020, 12, 38768
Shan P. H., Tu S. C., Lin R. L., Tao Z., Liu J. X., Xiao X., CrystEngComm, 2017, 19, 2168
Wang C. Z., Zhao W. X., Shen F. F., Zhang Y. Q., Zhu Q. J., Xiao X., Tao Z., CrystEngComm, 2016, 18, 2112
Qu Y. X., Zhou K. Z., Chen K., Zhang Y. Q., Xiao X., Zhou Q. D., Tao Z., Ma P. H., Wei G., Inorg. Chem., 2018, 57, 7412.
Rad N., Sashuk V., Chem. Commun., 2022, 58, 5249
Tang B. H., Zhao J. T., Xu J. F., Zhang X., Chem. Eur. J., 2020, 26, 15446
Zhao Y. J., Xue S. F., Zhu Q. J., Tao Z., Zhang J. X., Wei Z. B., Long L. S., Hu M. L., Xiao H. P., Day A. I., Chin. Sci. Bull., 2004, 49, 1111
Lin R. L., Fang G. S., Sun W. Q., Liu J. X., Scl. Rep., 2016, 6, 39057
Meng Y., Zhao W. W., Zheng J., Jiang D. F., Gao J., Jin Y. M., Ma P. H., RSC Adv., 2021, 11, 3470
Xiao X., Gao Z. Z., Shan C. L., Tao Z., Zhu Q. J., Xue S. F., Liu J. X., Phys. Chem. Chem. Phys., 2015, 17, 8618
Cong H., Tao L. L., Yu Y. H., Tao Z., Yang F., Zhao Y. J., Xue S. F., Lawrance G. A., Wei G., J. Phys. Chem. A, 2007, 111, 2715
Yang B., Xiao X., Zhang Y. Q., Zhu Q. J., Xue S. F., Tao Z., Wei G., RSC Adv., 2014, 4, 44359
Yang L. G., Kan J. L., Wang X., Zhang Y. H., Tao Z., Liu Q. Y., Wang F., Xiao X., Front. Chem., 2018, 6, 289
Huang Y. H., Geng Q. X., Jin X. Y., Cong H., Qiu F., Xu L., Tao Z., Wei G., Sensor. Actuat. B: Chem., 2017, 243, 1102
Akhtar W., Khan M. F., Verma G., Shaquiquzzaman M., Rizvi M. A., Mehdi S. H., Akhter M., Alam M. M., Eur. J. Med. Chem., 2017, 126, 705
Bistrović A., Krstulović L., Stolić I., Drenjančević D., Talapko J., Taylor M. C., Kelly J. M., Bajić M., Raić-Malić S., J. Enzym. Inhib. Med. Chem., 2018, 33, 1323
Arora R. K., Kaur N., Bansal Y., Bansal G., Acta Pharm. Sin. B, 2014, 4, 368
de Oliveira H. C., Rodrigues M. L., Fungal. Biol. Rev., 2021, 37, 27
Tonelli M., Novelli F., Tasso B., Vazzana I., Sparatore A., Boido V., Sparatore F., la Colla P., Sanna G., Giliberti G., Busonera B., Farci P., Ibba C., Loddo R., Bioorgan. Med. Chem., 2014, 22, 4893
Wu L. T., Jiang Z., Shen J. J., Yi H., Zhan Y. C., Sha M. Q., Wang Z., Xue S. T., Li Z. R., Eur. J. Med. Chem., 2016, 114, 328
Ge J. Y., Xue S. F., Zhu Q. J., Tao Z., Zhang J. X., J. Incl. Phenom. Macro., 2007, 58, 63
Zhao Y. J., Pourgholami M. H., Morris D. L., Collins J. G., Day A. I., Org. Biomol. Chem., 2010, 8, 3328
Albdallah S. K., Assaf K. I., Bodoor K., Al-Sakhen N. A., Malhis L. D., Alhmaideen A. I., El-Barghouthi M. I., J. Solution. Chem., 2018, 47, 1768
Wang Q., Guo J. Z., Luo D., Ye M. F., Lin R. L., Sun W. Q., Liu J. X., Phys. Chem. Chem. Phys., 2022, 24, 25930
Zheng L. M., Zhang K., Lin R. L., Chu X. F., Liu J. X., J. Incl. Phenom. Macro., 2020, 96, 125
Lu Y., Yu Z. C., Yang X. N., Dai J. J., Shan P. H., Feng X. H., Tao Z., Redshaw C., Xiao X., Chin. Chem. Lett., 2022, 108040
Qu Y. X., Lin R. L., Zhang Y. Q., Zhou K. Z., Zhou Q. D., Zhu Q. J., Tao Z., Ma P. H., Liu J. X., Wei G., Org. Chem. Front., 2017, 4, 1799
Shi Z. L., Liu X. K., Zhou Y. H., Cong H., Polym. Adv. Technol., 2022, 33, 2498
Zheng J., Ma Y., Yang X. N., Ma P. H., RSC Adv., 2022, 12, 18736
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No.22161010).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no conflicts of interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Yang, N., Dai, X., Ma, Y. et al. Study on the Host-Guest Interactions Between Tetramethyl Cucurbit[6]uril and Benzimidazole Derivatives. Chem. Res. Chin. Univ. 39, 1017–1022 (2023). https://doi.org/10.1007/s40242-023-3078-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-023-3078-1