Abstract
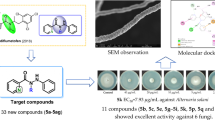
To discover novel strobilurins analogues with good and broad spectrum activity, a series of novel 3,4-dihydro-2H-1,2,4-benzothiadiazine 1,1-dioxides-based strobilurins was designed, synthesized, and tested against various phytopathogenic fungi. Compounds 7b, 7c, and 7k exhibited substantial and broad spectrum antifungal activities against the tested phytopathogenic fungi, especially compound 7b, which showed 100%, 80%, 90%, and 90% antifungal activity(in virto) against Erysiphe graminis(E. graminis), Puccinia sorghi Schw.(P. sorghi Schw.), Colletotrichum lagenarium(C. lagenarium), and Pseudoperonospora cubensis(P. cubensis) at 300 µg/mL, respectively, better or comparable to the positive control azoxystrobin. Moreover, compound 7b exhibited 85% greenhouse inhibition activity(in vivo) against E. graminis even at 0.2 µg/mL, equal to azoxystrobin(90%) and trifloxystrobin(90%). Meanwhile, compound 7b against P. cubensis displayed 70% and 55% greenhouse inhibition activity(in vivo) at 1.56 and 0.2 µg/mL, respectively, much better than those of azoxystrobin and trifloxystrobin(both 0% at 1.56 and 0.2 µg/mL). Therefore, compound 7b could be considered as the most promising fungicidal candidate for further study. Furthermore, based on the effective concentration(EC50) against C. arachidicola, the built CoMSIA model provided the useful reference for the further structural optimization design.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Price C. L., Parker J. E., Warrilow A. G., Kelly D. E., Kelly S. L., Pest Manage. Sci., 2015, 71(8), 1054
Wang X., Ma J., Li X., Zhao X., Lin Z., Chen J., Shao Z., IEEE Trans. Biomed. Eng., 2014, 62(1), 80
Thomas A., Neufeld K. N., Seebold K. W., Braun C. A., Schwarz M. R., Ojiambo P. S., Plant Dis., 2018, 102(8), 1619
Gisi U., Sierotzki H., Eur. J. Plant Pathol., 2008, 122(1), 157
Howell C. C., Semple K. T., Bending G. D., Chemosphere, 2014, 95, 370
Bai Y. L., Shandong Pesticide News, 2018, 4, 24
Becker W. F., Jagow G. V., Anke T., Steglich W., Febs. Lett., 1981, 132(2), 329
Utture S. C., Banerjee K., Dasgupta S., Patil S. H., Jadhav M. R., Wagh S. S., Kolekar S. S., Anuse M. A., Adsule P. G., J. Agric. Food Chem., 2011, 59(14), 7866
Liu X. Y., Chen X., Wang H. Y., Yang T. L., Ye Q. F., Ding X. C., J. Agric. Food Chem., 2014, 62(15), 3343
Bartlett W., Clough J. M., Godwin J. R., Hall A. A., Hamer M., Parr-Dobrzanski B., Pest Manage. Sci., 2002, 58(7), 649
Liu P., Wang H., Zhou Y., Meng Q., Si N., Hao J. J., Liu X., Pestic. Biochem. Phys., 2014, 112, 19
Flampouri E., Mavrikou S., Mouzaki-Paxinou A. C., Kintzios S., Biochem. Pharmacol., 2016, 113, 97
Chen L., Zhao B., Fan Z. J., Hu M. X., Li Q., Hu W. H., Li J. W., Zhang J. L., J. Agric. Food Chem., 2019, 67(45), 12357
Kamal A., Reddy K. S., Ahmed S. K., Khan M. N. A., Rakesh K. S., Yadav J. S., Arora S. K., Bioorg. Med. Chem., 2006, 14(3), 650
Harrouche K., Lahouel A., Belghobsi M., Pirotte B., Khelili S., Can. J. Chem., 2019, 97(12), 824
Kamal A., Khan M. N. A., Reddy K. S., Ahmed S. K., Kumar M. S., Juvekar A., Sen S., Zingde S., Bioorg. Med. Chem. Lett., 2007, 17(19), 5345
Chen X., Zhu C., Guo F., Qiu X. W., Yang Y. C., Zhang S. Z., He M. L., Parveen S., Jing C. J., Li Y., Ma B., J. Med. Chem., 2010, 53(23), 8330
Drapier T., Geubelle P., Bouckaer C., Nielsen L., Laulumaa S., Goffin E., Dilly S., Francotte P., Hanson J., Pochet L., Kastrup J. S., Pirotte B., J. Med. Chem., 2018, 61(12), 5279
Saiz-Urra L., Gonzalez M. P., Collado I. G., Hernandez-Galan R., J. Mol. Graphics Modell., 2007, 25(5), 680
Liu J. B., Li F. Y., Wang Y. H., Zhang H. X., Li Y. X., Li Z. M., Med. Chem. Res., 2020, 29, 495
Li F. Y., Wang Y. H., Liu J. B., Li Y. X., Li Z. M., Bioorg. Med. Chem., 2019, 27(5), 769
He F., Shi J., Wang Y., Wang S., Chen J., Gan X. H., Song B. A., Hu D. Y., J. Agric. Food Chem., 2019, 67(31), 8459
Chen Z., Xu W., Liu K., Yang S., Fan H., Bhadury P. S., Huang D. Y., Zhang Y., Molecules, 2010, 15(12), 9046
Sudhamani H., Thaslim B. S. K., Muni C. R. S., Sreedhar B., Adam S., Naga R. C., Res. Chem. Intermed., 2016, 42(10), 7471
Asundaria S. T., Pannecouque C., De Clercq E., Patel K. C. Pharm. Chem. J., 2014, 48(4), 260
Abramovitch R. A., Chellathurai T., Holcomb W. D., Mcmaster I. T., Vanderpool D. P., J. Org. Chem., 1977, 42(17), 2920
Cheng L., Zhang R. R., Wu H. K., Liu X. H., Xu T. M., Front. Chem. Sci. Eng., 2019, 13(2), 369
Liu J. B., Li F. Y., Wang Y. H., Zhang H. X., Dong J. Y., Sun P. W., Li Y. X., Li Z. M., Chin. Chem. Lett., 2019, 30(3), 668
Xie Y. Q., Huang Y. B., Liu J. S., Ye L. Y., Che L. M., Tu S., Liu C. L., Pest Manage. Sci., 2015, 71(3), 404
Liu X. H., Xu X. Y., Tan C. X., Weng J. X., Xin J. H., Chen J., Pest Manage. Sci., 2015, 71(2), 292
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supported by the Scientific Project of Tianjin Municipal Education Commission, China(No.2018KJ008).
Supporting information
40242_2020_160_MOESM1_ESM.pdf
Design and Synthesis of Novel 3,4-Dihydro-2H-1,2,4-benzothiadiazine 1,1-Dioxides-based Strobilurins as Potent Fungicide Candidates
Rights and permissions
About this article
Cite this article
Li, F., Gong, J., Liu, J. et al. Design and Synthesis of Novel 3,4-Dihydro-2H-1,2,4-benzothiadiazine 1,1-Dioxides-based Strobilurins as Potent Fungicide Candidates. Chem. Res. Chin. Univ. 36, 1190–1195 (2020). https://doi.org/10.1007/s40242-020-0160-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-020-0160-9