Abstract
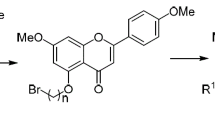
Twenty prenylated flavonoids 1―20 were synthesized by glycoside hydrolysis, dehydrogenation, selective O-methylation, O-prenylation and Claisen rearrangement reaction, from abundant and inexpensive natural flavonoids naringin, hespiredin, quercetin and myricetin. Among them, 1―7, 10―15 and 17―20 are novel compounds, the natural product 3,3′,4′,7-tetramethoxy-8-prenyl-5-hydroxy flavonoid(16) was synthesized in a high yield. Their antiprolirative activities were evaluated in vitro on a panel of three human cancer cell lines(HeLa, HCC1954 and SK-OV-3). The results show that most of the target compounds displayed moderate to potent antiprolirative activities against the three cancer cells with half maximal inhibitory concentration(IC50) values from 0.49 μmol/L to 95.07 μmol/L. Among them, 3′,4′,7-trimethoxyl-5-hydroxyl-8-prenyl flavonoid(12) exhibited the strongest antiprolirative activity against the three cancer cells mentioned above with IC50 values of 0.91―7.08 μmol/L. 3′,7-Dimethoxy-5-O-prenyl flavone(6) and 3′,4′,7-trimethoxy-5-O-prenyl flavone(10) showed selective antiproliferative activity against HCC1954 cells with IC50 value of 0.49 and 5.32 μmol/L, respectively.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Raffa D., Maggio B., Raimondi M. V., Plescia F., Daidone G., Eur. J. Med. Chem., 2017, 142, 213
Verma A. K., Pratap R., Tetrahydron, 2012, 68(41), 8523
Vogel S., Ohmayer S., Brunner G., Heilmann J., Bioorg. Med. Chem., 2008, 16(8), 4286
Lei Q., Zhang S., Liu M., Li J., Zhang X., Long Y., Mol. Divers., 2017, 21(4), 957
Dong X., Liu Y., Yan J., Jiang C., Chen J., Liu T., Hu Y., Bioorg. Med. Chem., 2008, 16(17), 8151
Neves M. P., Cidade H., Pinto M., Silva A. M. S., Gales L., Damas A. M., Lima R.T., Vasconcelos M. H., Nascimento M. S. J., Eur. J. Med. Chem., 2011, 46(6), 2562
Wen Z., Zhang Y., Wang X., Zeng X., Hu Z., Liu Y., Xie Y., Liang G., Zhu J., Luo H., Xu B., Eur. J. Med. Chem., 2017, 133, 227
Basabe P., de Roman M., Marcos I. S., Diez D., Blanco A., Bodero O., Mollinedo F., Eur. J. Med. Chem., 2010, 45(9), 4258
Wang C., Wu P., Shi J. F., Jiang Z. H., Wei X. Y., Eur. J. Med. Chem., 2015, 100, 139
Jung A. H., Jin E. S., Choi J. R., Manh T. H., Kim S. Y., Min S. B., Son K. Y., Ahn R. B., Kim W. B., Sohn S. S., Choi S. J., Arch. Pharm. Res., 2011, 34(12), 2087
Zhang B., Duan D., Ge C., Yao J., Liu Y., Li X., Fang J., J. Med. Chem., 2015, 58(4), 1795
Chen M., Shi L., Tang J., Wang Q. A., Chem. Res. Chinese Universities, 2016, 32(5), 754
Nguyen V. S., Li W., Li Y., Wang Q. A., Med. Chem. Res., 2017, 26, 1585
Nguyen V. S., Shi L., Wang S. C., Wang Q. A., Anti-cancer Agents Med. Chem., 2017, 17(1), 137
Liu J. D., Chen L., Cai S. L., Wang Q. A., Carbohyd. Res., 2012, 357, 41
Wu Z., Cai S. L., Fan W. J., Wang Q. A., Chin. J. Org. Chem., 2012, 32(7), 1296
Ou L., Han S., Ding W., Che Z., Ye Z., Yang H., Zhang G., Lou Y., Chen J., Yu Y., Mol. Divers., 2011, 15(3), 665
Pinhey J. T., Southwell I. A., Aust. J. Chem., 1973, 26(2), 409
Song Y. P., Xin Z. Y., Wan Y. M., Li J. B., Ye B. P., Xue X. W., Eur. J. Med. Chem., 2015, 90, 695
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China(Nos.J1210040, 21173074).
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Vongdeth, K., Ran, L., Yan, L. et al. Synthesis and Activity Evaluation of Novel Prenylated Flavonoids as Antiproliferative Agents. Chem. Res. Chin. Univ. 34, 564–570 (2018). https://doi.org/10.1007/s40242-018-8013-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-018-8013-5