Abstract
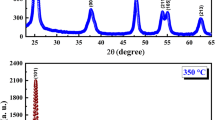
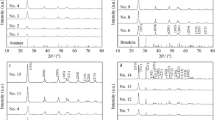
A facile approach was developed to prepare highly dispersed TiO2 nanoparticles with selected phase. The crystallization phase of the nanoparticles can be easily tuned from anatase to rutile by the dosage of hydrochloric acid in the reaction system. The crystallite size of the as-prepared anatase TiO2 nanoparticles was ca. 3.2 nm with high dispersion. A transparent TiO2 colloid was obtained by dispersing the as-prepared anatase TiO2 nanoparticles in deionized water without any organic additives added. The concentration of TiO2-H2O colloid can be as high as 1600 g/L. The optical transmittance of TiO2-H2O colloid with a low concentration was nearly 100% in the visible region. Furthermore, anatase TiO2 nanoparticles(TiO2-NPs) showed superior photocatalytic performance compared to rutile TiO2-NPs.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Buso D., Post M., Cantalini C., Mulvaney P., Martucci A., Adv. Funct. Mater., 2008, 18(23), 3843
Han F., Kambala V. S. R., Srinivasan M., Rajarathnam D., Naidu R., Appl. Catal. A: Gen., 2009, 359(1/2), 25
Nikawa T., Naya S., Tada H., J. Colloid Interface Sci., 2015, 456, 161
Yang C., Fan H., Xi Y., Chen J., Li Z., Appl. Surf. Sci., 2008, 254(9), 2685
Feng X. J., Shankar K., Varghese O. K., Paulose M., Latempa T. J., Grimes C. A., Nano Lett., 2008, 8(11), 3781
Crepaldi E. L., Soler-Illia G. J. D. A., Grosso D., Cagnol F., Ribot F., Sanchez C., J. Am. Chem. Soc., 2003, 125(32), 9770
Li H. X., Bian Z. F., Zhu J., Zhang D. Q., Li G. S., Huo Y. N., Li H., Lu Y. F., J. Am. Chem. Soc., 2007, 129(27), 8406
Nishiyama N., Fujiwara Y., Adachi K., Inumaru K., Yamazaki S., Appl. Catal. B: Environ., 2015, 176, 347
Feng X., Zhai J., Jiang L., Angew. Chem. Int. Ed., 2005, 44(32), 5115
Li Y., Sasaki T., Shimizu Y., Koshizaki N., Small, 2008, 4(12), 2286
Lai Y., Tang Y., Gong J., Gong D., Chi L., Lin C., Chen Z., J. Mater. Chem., 2012, 22(15), 7420
Li G. S., Li L. P., Boerio-Goates J., Woodfield B. F., J. Am. Chem. Soc., 2005, 127(24), 8659
Bokhimi X., Morales A., Ortiz E., Lopez T., Gomez R., Navarrete J., J. Sol-Gel Sci. Techn., 2004, 29(1), 31
Li J. G., Ishigaki T., Sun X. D., J. Phys. Chem. C, 2007, 111(13), 4969
Li W., Ni C., Lin H., Huang C. P., Shah S. I., J. Appl. Phys., 2004, 96(11), 6663
Cozzoli P. D., Kornowski A., Weller H., J. Am. Chem. Soc., 2003, 125(47), 14539
Li N., Liu G., Zhen C., Li F., Zhang L. L., Cheng H. M., Adv. Funct. Mater., 2011, 21(9), 1717
Wen P. C., Cai C., Zhong H., Hao L. Y., Xu X., J. Mater. Sci., 2015, 50(18), 5944
Lou X. W., Archer L. A., Adv. Mater., 2008, 20(10), 1853
Eiden-Assmann S., Widoniak J., Maret G., Chem. Mater., 2004, 16(1), 6
Yang H. G., Liu G., Qiao S. Z., Sun C. H., Jin Y. G., Smith S. C., Zou J., Cheng H. M., Lu G. Q., J. Am. Chem. Soc., 2009, 131(11), 4078
Li G. H., Gray K. A., Chem. Mater., 2007, 19(5), 1143
Hu Y., Yuan C. W., J. Cryst. Growth, 2005, 274(3/4), 563
Serrano D. P., Calleja G., Sanz R., Pizarro P., J. Mater. Chem., 2007, 17(12), 1178
Wu N. L., Wang S. Y., Rusakova I. A., Science, 1999, 285(5432), 1375
Yoshitake H., Sugihara T., Tatsumi T., Chem. Mater., 2002, 14(3), 1023
Alamgir, Khan W., Ahmad S., Naqvi A. H., Mater. Lett., 2014, 133, 28
Charbonneau C., Holliman P. J., Davies M. L., Watson T. M., Worsley D. A., J. Colloid Interface Sci., 2015, 442, 110
Ohya T., Nakayama A., Ban T., Ohya Y., Takahashi Y., Chem. Mater., 2002, 14(7), 3082
Thompson T. L., Yates J. T., Chem. Rev., 2006, 106(10), 4428
Yan X. M., Pan D. Y., Li Z., Liu Y. Y., Zhang J. C., Xu G., Wu M. H., Mater. Lett., 2010, 64(16), 1833
Sreethawong T., Suzuki Y., Yoshikawa S., Catal. Commun., 2005, 6(2), 119
Spurr R. A., Myers H., Anal. Chem., 1957, 29(5), 760
Li H., Duan X., Liu G., Jia X., Liu X., Mater. Lett., 2008, 62(24), 4035
Zhou J., Zhao G., Song B., Han G., CrystEngComm, 2011, 13(7), 2294
Cheng H., Ma J., Zhao Z., Qi L., Chem. Mater., 1995, 7(4), 663
Oaki Y., Anzai T., Imai H., Adv. Funct. Mater., 2010, 20(23), 4127
Oaki Y., Nakamura K., Imai H., Chem-Eur. J., 2012, 18(10), 2825
Brus L. E., J. Chem. Phys., 1984, 80(9), 4403
Ma Y., Wang X. L., Jia Y. S., Chen X. B., Han H. X., Li C., Chem. Rev., 2014, 114(19), 9987
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China(Nos.21071058, 21371066, 21301067).
Rights and permissions
About this article
Cite this article
Liu, X., Chen, Y., Jiao, S. et al. One step preparation of highly dispersed TiO2 nanoparticles. Chem. Res. Chin. Univ. 31, 688–692 (2015). https://doi.org/10.1007/s40242-015-5300-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-015-5300-2