Abstract
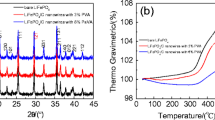
CuV2O6 nanowires were prepared via a simple hydrothermal route using NH4VO3 and Cu(NO3)2 as starting materials. The structures and electrochemical properties of CuV2O6 nanowires were characterized by means of X-ray diffraction(XRD), scanning electron microscopy(SEM) and transmission electron microscopy(TEM). The results show that the CuV2O6 nanowires are about 100 nm in width and single crystalline grown along [001] direction. CuV2O6 nanowires delivered a high initial discharge capacity of 435 and 351 mA·h/g at current densities of 50 and 100 mA·h/g, respectively. The electrochemical kinetics of the CuV2O6 nanowires was also investigated by means of electrochemical impedance spectroscopy(EIS) and the poor rate performance was observed, which may be attributed to the low ion diffusion coefficient of the CuV2O6 nanowires.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Aidoudi F. H., Aldous D. W., Goff R. J., Slawin A. M. Z., Attfield J. P., Morris R. E., Lightfoot P., Nat. Chem., 2011, 3, 801
Nakano M., Shibuya K., Okuyama D., Hatano T., Ono S., Kawasaki M., Iwasa Y., Tokura Y., Nature, 2012, 487(7408), 459
Marley P. M., Banerjee S., Inorg. Chem., 2012, 51(9), 5264
Devi S. S., Muthukumaran B., Krishnamoorthy P., Ionics, 2014, 20(12), 1783
Li M. L., Yang X., Wang C. Z., Chen N., Hu F., Bie X. F., Wei Y. J., Du F., Chen G., J. Mater. Chem. A, 2015, 3, 586
Zhu K., Yan X., Zhang Y. Q., Wang Y. H., Su A. Y., Bie X. F., Zhang D., Du F., Wang C. Z., Chen G., Wei Y. J., Chem. Plus. Chem., 2014, 79, 447
Wei Y. J., Ryu C. W., Chen G., Kim K. B., Electrochem. Sol. Stat. Lett., 2006, 9(11), A487
Pan A. Q., Zhang J. G., Cao G. Z., Liang S. Q., Wang C. M., Nie Z. M., Arey B. W., Xu W., Liu D. W., Xiao J., Li G. S., Liu J., J. Mater. Chem., 2011, 21, 10077
Zhang S. Y., Li W. Y., Li C. S., Chen J., J. Phys. Chem. B, 2006, 110, 24855
Qiu C. G., Liu L.N., Du F., Yang X., Wang C. Z., Chen G., Wei Y. J., Chem. Res. Chinese Universities, 2015, 31(2), 270
Zhang Y., Pan Y., Liu J., Wang G. L., Cao D. X., Chem. Res. Chinese Universities, 2015, 31(1), 117
Wei Y. J., Nam K. W., Chen G., Ryu C. W., Kim K. B., Sol. Stat. Ion., 2005, 176, 2243
Cheng F. Y., Chen J., J. Phys. Chem., 2011, 21, 9841
Ma H., Zhang S. Y., Ji W. Q., Tao Z. L., Chen J., J. Am. Chem. Soc., 2008, 130, 5361
Cao X. Y., Xie J. G., Zhan H., Zhou Y. H., Mater. Chem. Phys., 2006, 98(1), 71
Cao J. Q., Wang X. Y., Tang A. P., Wang X., Wang Y., Wu W., J. Alloy. Comp., 2009, 479, 875
Shaju K. M., Rao G. V. S., Chowdari B. V. R., Electrochim. Acta, 2003, 48, 2691
Liao X. Z., Ma Z. F., Gong Q., He Y. S., Pei L., Zeng L. J., Electrochem. Commun., 2008, 10, 691
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Basic Research Program of China(No.2015CB251103), the National Natural Science Foundation of China(No.51472104) and the Development Program of Science and Technology of Jilin Province, China(No.20140101093JC).
Rights and permissions
About this article
Cite this article
Hu, F., Li, M., Wei, Y. et al. Synthesis and electrochemical properties of highly crystallized CuV2O6 nanowires. Chem. Res. Chin. Univ. 31, 708–711 (2015). https://doi.org/10.1007/s40242-015-5136-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-015-5136-9